Abstract
The present study has three purposes; first evaluating cytotoxicity of (E)-4-chloro-2-((3-ethoxy-2-hydroxybenzylidene)amino)phenol (ACES), second deciphering ACES-mediated cellular death mechanism, and third estimating ACES-mediated alterations in the expressions of mitogen-activated protein kinase (MAPK) pathway-related genes. Neutral red uptake assay, cell cycle analysis, mitochondrial membrane potential (MMP), reactive oxygen species (ROS) measurements, caspase 3/7 and 9 activations, and quantitative reverse transcription-polymerase chain reaction (qRT-PCR) were implemented. IC50 values of ACES-treated five cells were around 4–6 µg/mL. However, Caco-2 and Huh-7 cells were found to be twofold resistant and fivefold sensitive with IC50 values of 11 µg/mL and 0.93 µg/mL, respectively. In this study, it was initially reported that ACES exhibits selective cytotoxicity to Huh-7 cells. In addition, ACES induced apoptosis by nuclear fragmentation, MMP disruption, and intracellular ROS elevation in MCF-7 cells. qRT-PCR experiment indicated the expressions of 30 genes including
Introduction
Chemotherapy is still one of the first preferred methods for cancer treatment, particularly for patients with advanced disease. While chemotherapy may be curative in the early stages, the therapeutic effect of the drug rapidly vanishes with the development of resistance. Cancer cells acquire the resistance by several mechanisms, including but not limited to decrease the drug accumulation, increase the drug export, alter the drug targets or the signal transduction molecules, boost the repair of drug-induced DNA damage, and evade from apoptosis. 1 P38/stress-activated protein kinases (SAPKs) are members of mitogen-activated protein kinase (MAPK) family pathways. There are accumulating data suggesting the p38/SAPK activation as a major mechanism of rapid cell proliferation and tumor cell resistance to cytotoxic drugs including vincristine resistance in gastric cell lines 2 ; trastuzumab resistance in breast cancer patients 3 ; cisplatin, irinotecan, and 5-fluorouracil resistance in colorectal cancer patients 4 ; in vivo unresponsiveness to chemotherapy in non-Hodgkin lymphoma patients 5 ; imatinib resistance in chronic myeloid leukemia cells 6 ; and cisplatin resistance in melanoma cells. 7 Targeted drugs that inhibit the p38/SAPK hold great significance in the management of chemoresistance.
Schiff bases have been explored for various applications, including their utilities of an inhibitor of amyloid-β aggregation, antimicrobial, antiviral, and anticancer activities. 8 Moreover, Schiff bases are suggested as effective anticancer drugs with their substantial cytotoxic activities. 9 –11 Mainly, condensation products of 3-ethoxysalicylaldehyde were effective cytotoxic ligands on HepG2, HCT116, and MCF-7 cells. 12 In addition, antimicrobial effects of 3-ethoxysalicylaldehyde 13 –15 and 2-amino-4-chlorophenol-derived Schiff bases are reported. 16,17 Therefore in this study, it was found worthy of investigating the anticancer activity of a Schiff base, (E)-4-chloro-2-((3-ethoxy-2-hydroxybenzylidene)amino)phenol (ACES), which is a condensation product of 2-amino-4-chlorophenol with 3-ethoxysalicylaldehyde. ACES-meditated cytotoxicity was screened on a panel of normal and cancer cell lines by using the neutral red uptake (NRU) assay. Then a model cell line, MCF-7, was selected for further investigations including cell cycle analysis, mitochondrial membrane potential (MMP) measurements, caspase 3/7 and 9 activations, and intracellular reactive oxygen species (ROS) level measurements which are intending to figure out the mechanism of ACES-triggered cellular death. Alterations in the expressions of MAPK pathway-related genes were measured in the ACES-treated MCF-7 cells.
Materials and method
Cell culture
Five human cancer cell lines, a normal human cell line, and a normal mouse cell line were used in this study. Human cancer cells were A549, human non-small cell lung cancer cell line, obtained from the Institute for Fermentation, Osaka (IFO, Japan) and donated from Professor Dr Tansu Koparal (Eskişehir Technical University, Eskişehir, Turkey); Caco-2, human colon adenocarcinoma cells obtained from the ŞAP Institute (Ankara, Turkey) donated from Professor Dr Hülya Sivas (Eskişehir Technical University, Eskişehir, Turkey); HCT-116, human colon carcinoma cells obtained from the Deutsche SammLung von Mikroorganismen und Zellkulturen (DSMZ, Braunschweig, Germany) donated from Professor Dr Sreeparna Banerjee (Middle East Technical University, Ankara, Turkey); Huh-7, hepatocellular carcinoma cells were donated from Professor Dr Mehmet Öztürk (Dokuz Eylül University, İzmir, Turkey); and MCF-7, human breast adenocarcinoma cells (ATCC, HTB-22) were donated from Professor Dr Tansu Koparal (Eskişehir Technical University, Eskişehir, Turkey). Normal cells, hTERT-HPNE human pancreatic normal epithelial cells (ATCC, CRL-4023) and 3T3-L1 mouse embryonic normal fibroblast cells (ATCC, CL-173) were donated from Professor Dr Tansu Koparal (Eskişehir Technical University, Eskişehir, Turkey). Cells were cultured as a monolayer in Dulbecco’s modified eagle medium (DMEM) (Sigma-Aldrich, Munich, Germany) medium supplemented with 10% fetal bovine serum and 1% penicillin (100 U/mL)-streptomycin (100 μg/mL) in a humidified 5% CO2 atmosphere at 37°C. Cells were detached from the culture flasks with the treatment of 0.25% trypsin/ethylenediaminetetraacetic acid (Invitrogen, Carlsbad, California, USA).
NRU assay and morphological observations
Cytotoxicity of ACES and doxorubicin on a panel of the cell line was assessed by NRU assay as previously described. 18 This method relies on the binding and accumulating of NR dye to the lysosomes of viable cells. The procedure is distinguished from other cytotoxicity tests with its impressive sensitivity. 19 Briefly, 1 × 104 cells were seeded into each well of a 96-well cell culture plate (Thermo Scientific, Germany). After an overnight incubation, cells were treated with twofold serial dilutions (30, 15, 7.5, 3.25, 1.6, 0.8, and 0.4 μg/mL) of ACES. Then the cells were incubated for 72 h in a humidified 5% CO2 atmosphere at 37°C. Alterations in the morphologies of cells were assessed at 48th hour using an inverted microscope (Olympus, BX 50, Center Valley, Pennsylvania, USA). After the incubation period, the medium was removed, and 200 µL of fresh medium containing 50 μg/mL NR dye was added to each well and further incubated for an additional 3 h at 37°C in 5% CO2. The dye medium was poured, and each well was washed with 200 µL phosphate buffer saline (PBS) followed by the addition of 200 µL of dye extraction solution including acetic acid–water–ethanol (1:49:50). The plates were kept for 15 min at room temperature to extract the dye with shaking on a GFL 3012 (Gesellschaft für Labortechnik mbH, Burgwedel, Germany). Absorbance was measured on ELx 808 Ultra Microplate Reader (BioTek, Winooski, Vermont, USA) equipped with a 540 nm filter. The viability was assessed based on a comparison with solvent control cells treated with 0.1% dimethylsulfoxide (DMSO). The IC50 values determined as the ACES concentrations required to inhibit 50% of cell were calculated from a calibration curve by linear regression using Microsoft Excel. 20 DMSO concentration was kept less than 0.1% in all wells of the experiment.
Detection of apoptotic cells by cell cycle analysis
Sub-G0/G1 cells in the cell cycle analysis have long been used for the detection of apoptotic cells.
21
This method is based on the observation that apoptotic cells have reduced or hypodiploid DNA fluorescence in the cell cycle profile analysis.
22
Cell cycle analysis was performed by the analysis of propidium iodide-stained cells in flow cytometry. Briefly, MCF-7 cells (3 × 105 cells/well) were seeded into each well of six-well plates and allowed to attach for 24 h. Then treated with ½ × IC50 and IC50 concentrations of ACES. The assay also included 0.1% DMSO-treated cells presented as a solvent control and ½ × IC50 and IC50 concentrations of doxorubicin-treated cells presented as a positive control. Cells were further incubated in six-well plates for 72 h. Next, cells were trypsinized and suspended in 3 mL cold fluorescence-activated cell sorting (FACS) buffer (1% bovine serum albumin, 0.01% sodium azide in PBS) and then centrifuged at 300 ×
Analysis of MMP
Disruption of MMP is a hallmark of apoptosis. In order to define the MMP status of the cells as polarized or disrupted, a membrane-permeable cationic dye, 5,5′,6,6′-tetrachloro-1,1′,3,3′-tetraethylbenzimidazolylcarbocyanine iodide (JC-1) which forms J-aggregates in the mitochondria of polarized MMP but remains as monomers in the cytoplasm of disrupted MMP can be utilized in flow cytometry. 23 The assay was done with MitoScreen kit (Becton-Dickinson) according to the directions of the manufacturer. Briefly, MCF-7 cells (3 × 105 cells/well) were seeded into each well of six-well plates and allowed to attach for 24 h. Then treated with ¼ × IC50, ½ × IC50, and IC50 concentrations of ACES. The assay also included 0.1% DMSO-treated cells presented as a solvent control and ¼ × IC50, ½ × IC50, and IC50 concentrations of doxorubicin-treated cells presented as a positive control. Then incubated in six-well plates for 72 h. After the incubation period, the cells were stained with JC-1-containing solution for 30 min in CO2 incubator according to the manufacturer’s protocol. Stained cells were measured in a BD Accuri C6 Flow Cytometer (Becton-Dickinson) by acquiring 10,000 cell events per sample.
Luminometric measurement of caspase 3/7 and 9 activations
The activations of caspase 3/7 and 9 in ACES-treated MCF-7 cells were detected using Caspase-Glo 3/7 and Caspase-Glo 9 Assay kits (Promega, Mannheim, Germany) as previously reported. 20 Briefly, MCF-7 cells (5 × 103 cells/well) were seeded into each well of white-walled 96-well plates and allowed to attach for 24 h. Then treated with ½ × IC50, IC50, and 2 × IC50 concentrations of ACES. The assay also included 0.1% DMSO-treated cells presented as a solvent control and ½ × IC50, IC50, and 2 × IC50 concentrations of doxorubicin-treated cells presented as a positive control. Then incubated in 96-well plates for 6 h. Next, the cells were treated with Caspase-Glo 3/7 and 9 substrates. Then the plates were kept on a plate shaker rotating at 300–500 r/min for 30 s. After the incubation at room temperature for 3 h, luminescence was measured using a Synergy HT multi-detection microplate reader (BioTek, Winooski, Vermont, USA). Caspase activity was converted to the percentage of the solvent control.
Fluorometric measurement of ROS
Cytotoxic drugs have been found to increase the intracellular ROS level frequently. In order to measure the ROS level, a cell-permeable peroxynitrite indicator, 2′,7′-dichlorodihydrofluorescein diacetate (H2DCFH-DA) by OxiSelect™ Intracellular ROS Assay Kit (Green Fluorescence), was used with the guidelines of the manufacturer (Cell Biolabs, San Diego, California, USA). Briefly, MCF-7 cells (5 × 103 cells/well) were seeded into each well of black-walled 96-well plates and allowed to attach for 24 h. Then treated with ½ × IC50, IC50, and 2 × IC50 concentrations of ACES. The assay also included 0.1% DMSO-treated cells presented as a solvent control and ½ × IC50, IC50, and 2 × IC50 concentrations of doxorubicin-treated cells presented as a positive control. Then incubated in 96-well plates for 24 h. After the incubation period, the cells were treated with 100 µL 1× DCFH-DA in DMEM solution and further incubated at 37°C for 30 min. Fluorescence was measured using a SpectraMax® M5 Microplate Reader (Molecular Devices, Biberach, Germany) at 480/530 nm.
Expression of MAPK pathway-related genes in the ACES-treated MCF-7 cells
The expression pattern of MAPK pathway-related genes of ACES-treated MCF-7 cells was investigated through the quantitative reverse transcription-polymerase chain reaction (qRT-PCR). Briefly, MCF-7 cells (3 × 105 cells/well) were seeded into each well of six-well plates and allowed to attach for 24 h. Then treated with the IC50 concentration of ACES. The assay also included 0.1% DMSO-treated cells presented as a solvent control. Then incubated in six-well plates for 72 h. After the incubation period, cells were trypsinized, and total RNA was isolated using RNeasy mini kit (Qiagen, Hilden, Germany) according to the guidelines of the manufacturer. The quantity and purity of isolated RNA were assessed by analyzing the raw absorbance value at 260 nm and the ratios of 260/280, 260/230 obtained from NanoDrop (Thermo, Waltham, Massachusetts, USA) device. After that, 500 ng total RNA was converted to cDNA with RT2 First Strand kit (Qiagen) according to the manufacturer’s guidelines. Quantitative PCR amplification was performed with the use of RT
2 SYBR Green Master Mix (Qiagen, Hilden, Germany) and PCR Array (MAP Kinase Signaling Pathway PCR Array, PAHS-061Z; Qiagen) on a real-time PCR platform (Mx3005P, Agilent, Santa Clara, California, USA). The PCR Array includes five internal control genes (
Statistical analysis
Three independent biological replicates were carried out for the experiments, except for the qRT-PCR, which was conducted with two replicates. Statistical analyses and graphing were performed using GraphPad Prism 6 (GraphPad, San Diego, California, USA). One-way analysis of variance with Dunnett’s post hoc test was used for differential analyses, and
Results
ACES exhibits selective toxicity to Huh-7 cells
The cytotoxicity of the ACES was evaluated by the NRU assay, and the IC50 values are specified in Table 1. For the estimation of selectivity indexes SI1 and SI2, the IC50 value of hTERT-HPNE human pancreatic normal epithelial cells and the IC50 value 3T3-L1 mouse embryonic normal fibroblast cells were divided by the IC50 values of cancer cells, respectively. As it is seen in Table 1, the IC50 values and selectivity indexes of ACES were between 0.93 and 11.6 µg/mL and 0.43 and 5.73 µg/mL, respectively. In addition, the IC50 values of ACES were around 5 µg/mL in five of seven tested cells (71%). However, a colon cancer cell line, Caco-2, was found to be twofold resistant to ACES-induced cytotoxicity. On the other hand, a liver cancer cell line, Huh-7, was found to be fivefold sensitive to ACES-induced cytotoxicity. In other words, ACES displayed nonselective cytotoxicity to the lung, colon, and breast cancer cells, whereas it was found to exhibit more than fivefold selective cytotoxicity toward a hepatocellular carcinoma cell line, Huh-7, with the SI1 and SI2 values of 5.73 and 5.33, respectively. The IC50 value of doxorubicin, which was evaluated by NRU experiments in our laboratory in MCF-7 cells, was previously reported as 0.19 µg/mL. 24 Based on these promising results for the selective cytotoxicity on the hepatocellular carcinoma cells, it was decided to implement further experiments to figure out the ACES-induced possible death mechanism.
Selectivity indexesa (SI1 and SI2) and IC50 values (µg/mL) of ACES on cancer and normal cell lines were evaluated by the NRU assay.
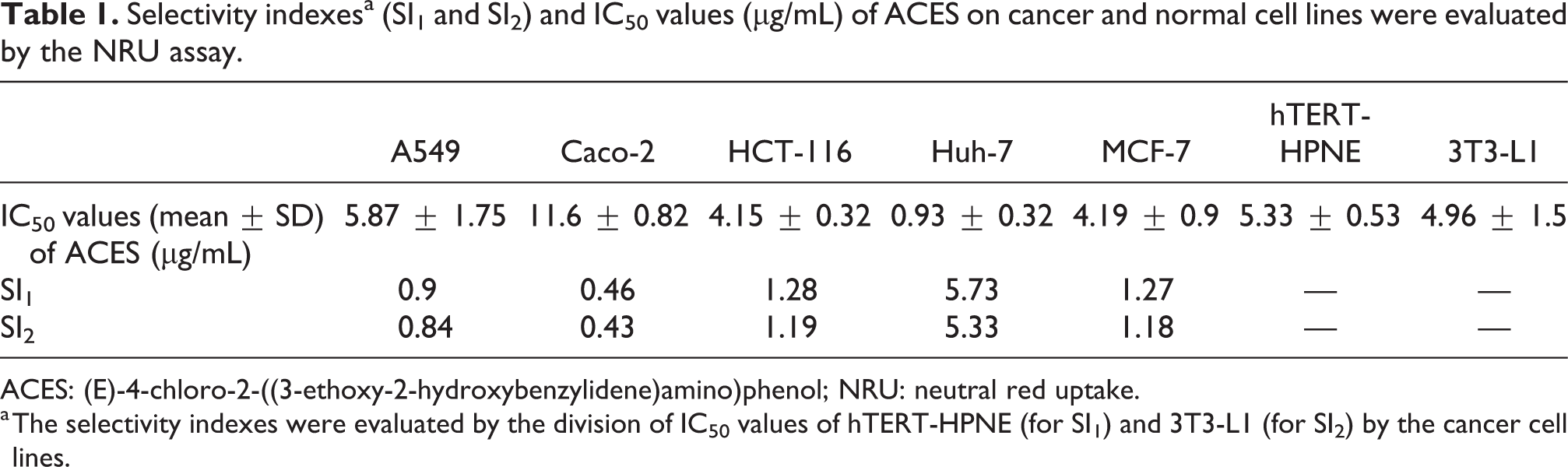
ACES: (E)-4-chloro-2-((3-ethoxy-2-hydroxybenzylidene)amino)phenol; NRU: neutral red uptake.
a The selectivity indexes were evaluated by the division of IC50 values of hTERT-HPNE (for SI1) and 3T3-L1 (for SI2) by the cancer cell lines.
ACES induces cytoplasmic vacuolization in MCF-7 cells
Cytoplasmic vacuolization was observed in the MCF-7 cells that were treated with the ACES at the IC50 concentration for 48 h. On the other hand, cytoplasmic vacuolization was not observed in control cells (Figure 1).

Morphology of MCF-7 cells. (a) Control group DMSO-treated cells and (b) test group ACES-treated cells at the concentration of IC50 for 48 h. Arrows indicate cytoplasmic vacuolization induced by ACES leading to cellular death. DMSO: dimethylsulfoxide; ACES: (E)-4-chloro-2-((3-ethoxy-2-hydroxybenzylidene)amino)phenol.
ACES triggers apoptosis in MCF-7 cells
Apoptotic cell population in which fragmented and degraded DNA with the activities of caspase enzymes can be quantitatively measured as sub-G0/G1 peaks in cell cycle analysis. To clarify whether the ACES-triggered cell death is apoptotic or not, cell cycle analysis was performed in DMSO, doxorubicin, and ACES-treated and propidium iodide-stained MCF-7 cells. It was observed that sub-G0/G1 cell ratio was 6% in 0.1% DMSO-treated control cells. However, it was increased to 43% and 75% in the ½ × IC50 and IC50 of doxorubicin-treated cells, respectively. Similarly, it was also increased to 43% and 68% in the ½ × IC50 and IC50 of ACES-treated cells, respectively. These results indicated that nuclear fragmentation takes part in both ACES- and doxorubicin-triggered cellular deaths identically. It was also demonstrated that G0/G1 cell ratio was at 60%, 15%, 6%, 18%, and 8% in DMSO, ½ × IC50 and IC50 of doxorubicin, and ½ × IC50 and IC50 of ACES-treated cells, respectively. In addition, S cell ratio was at 15%, 4%, 2%, 4%, and 2% in DMSO, ½ × IC50 and IC50 of doxorubicin, and ½ × IC50 and IC50 of ACES-treated cells, respectively. Additionally, G2/M cell ratio was at 13%, 7%, 6%, 7%, and 5% in DMSO, ½ × IC50 and IC50 of doxorubicin, and ½ × IC50 and IC50 of ACES-treated cells, respectively (Figure 2).

Analysis of cell cycle distribution in DMSO, doxorubicin, and ACES-treated MCF-7 cells. (a) Representative cytographs of the cell cycle distribution in MCF-7 cells treated with the indicated concentrations of ACES. (b) Bar graphs of cell cycle distribution in MCF-7 cells treated with ACES and doxorubicin. The results from three independent experiments are presented as mean ± SEM. Statistical comparisons were carried out through ANOVA followed by Dunnett’s post hoc (*
The voluminous of healthy cells in the cell cultures will exhibit a polarized MMP, whereas in apoptotic cells the disruption of the potential across the mitochondrial membrane is accompanied by the release of cytochrome c and cellular death. 25 To strengthen the observation that ACES-triggered cellular death is apoptotic, the MMP status of DMSO, doxorubicin, and ACES-treated and JC-1-stained MCF-7 cells was measured by flow cytometry. It was observed that the ratio of cells that bear JC-1 aggregates in the mitochondria was 76% in DMSO-treated control cells. However, it was decreased to 49%, 45%, and 41% in the ¼ × IC50, ½ × IC50, and IC50 of doxorubicin-treated cells, respectively. Similarly, it was also decreased to 67%, 59%, and 43% in the ¼ × IC50, ½ × IC50, and IC50 of ACES-treated cells, respectively (Figure 3(b)). JC-1 monomers in the cytoplasm were 9% in DMSO-treated control cells. However, it was increased to 37%, 42%, and 46% in the ¼ × IC50, ½ × IC50, and IC50 of doxorubicin-treated cells, respectively. Similarly, it was also increased to 25%, 33%, and 43% in the ¼ × IC50, ½ × IC50, and IC50 of ACES-treated cells, respectively. In addition, the ratio of JC-1 aggregates per JC-1 monomers was 7.3 in control cells. However, it was increased to 2.6, 1.8, and 1 in the ¼ × IC50, ½ × IC50, and IC50 of ACES-treated cells, respectively (Figure 3(b)). These results indicated that MMP disruption also takes part in both ACES- and doxorubicin-triggered cellular deaths identically.

Analysis of MMP status in DMSO, doxorubicin, and ACES-treated and JC-1-stained MCF-7 cells. (a) Representative cytographs of the MMP status in MCF-7 cells treated with the indicated concentrations of ACES. (b) Bar graphs of J-aggregates, J-monomers, and J-aggregates/monomers ratio in MCF-7 cells treated with ACES and doxorubicin. The results from three independent experiments are presented as mean ± SEM. Statistical comparisons were carried out through ANOVA followed by Dunnett’s post hoc (*
It has been long known that while some cytotoxic drugs may induce caspase activities initially than disrupt the MMP, others may disrupt the MMP initially than induce caspase activities. In order to define whether activation of the caspases occurs initially, the activations of caspase 3/7 and 9 were measured on MCF-7 cells which were treated with DMSO, doxorubicin, and ACES for 6 h. It was observed that doxorubicin in which the concentration was 2 × IC50 significantly induced caspase 3/7 activation. In addition, there was also an increase of caspase 9 activations in tested concentrations of doxorubicin; however, these increments were not statistically different from the control group. On the other hand, neither caspase 3/7 nor caspase 9 activations were induced by the ACES treatments (Figure 4). These observations clearly demonstrate that the early activation of caspase 3/7 is a difference between doxorubicin- and ACES-triggered apoptosis. While early caspase 3/7 activation takes part in doxorubicin-triggered apoptosis, it was not the case for the ACES-triggered apoptosis.
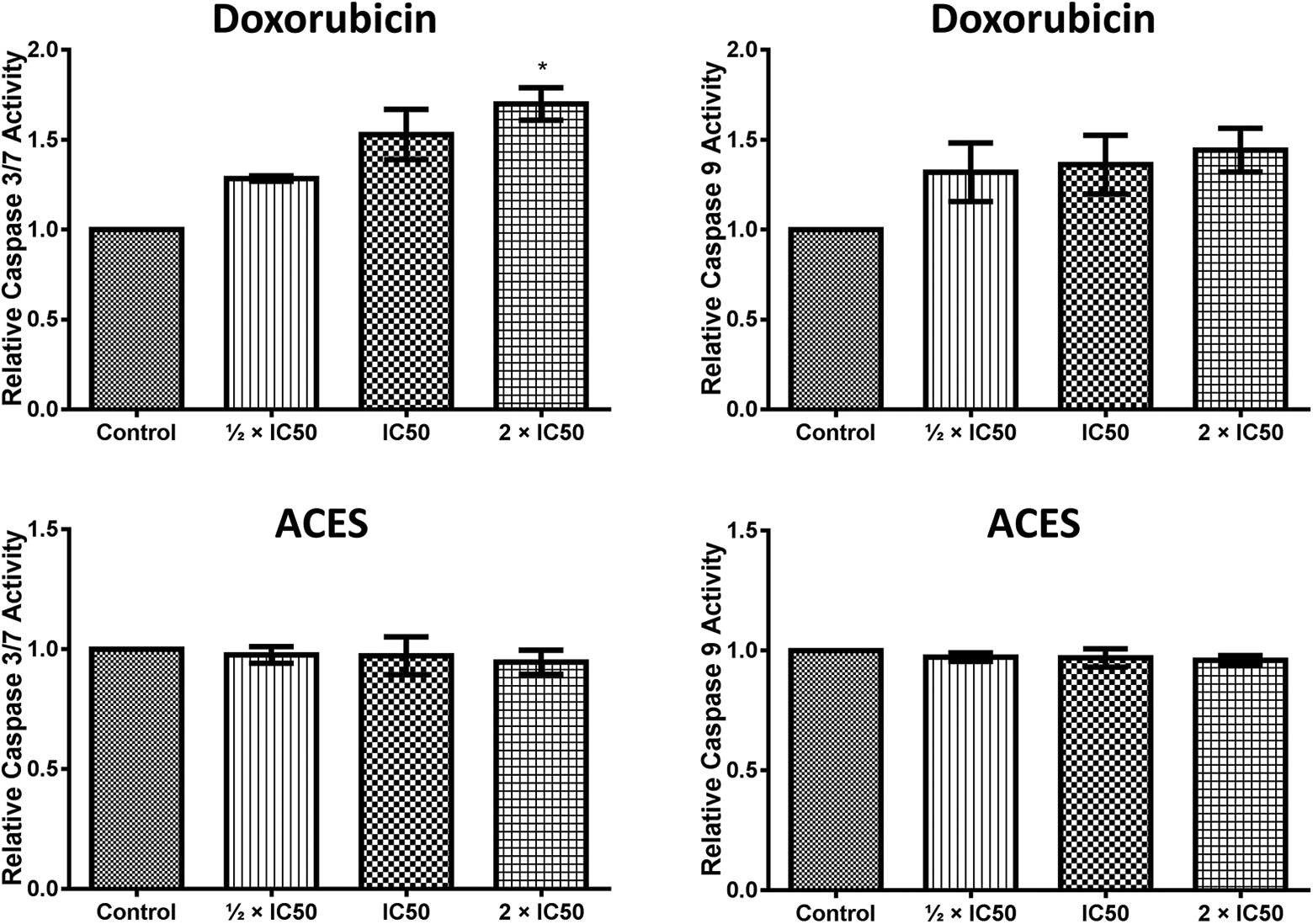
Caspase 3/7 and 9 activations in the early stages of both doxorubicin- and ACES-triggered apoptosis in MCF-7 cells. The results from three independent experiments are presented as mean ± SEM. Statistical comparisons were carried out through ANOVA followed by Dunnett’s post hoc (*
Intracellular ROS usually originates from the metabolism in the mitochondria, endoplasmic reticulum (ER), and membrane. Tumor cells differ from normal cells with their adaptation to survive under hypoxic conditions that increase the intracellular ROS levels. 26 In general, elevated ROS level results in oxidative stress then leads to cellular death with the accumulation of damaged biomolecules such as nucleic acids, lipids, and proteins. Conversely, if the ROS level is under a specific quantity, cancer cells can use the ROS increment to promote tumorigenesis by activating nuclear factor kappa B, phosphoinositide 3-kinase, hypoxia-inducible factors, and MAPK pathways. 27 In addition, the ROS scavenging driven by the antioxidant system is also working for the benefits of a cancer cell to survive under the oxidative stress. Therefore, it is obligatory for a specific cytotoxic drug to prevail both antioxidant system and tumor-promoting pathways to activate the cell death mechanism. In order to define whether the ACES-induced cellular death has arisen from the elevated intracellular ROS level, it was measured in the MCF-7 cells which were treated with DMSO, doxorubicin, and ACES for 24 h. The results indicated that all tested concentrations of doxorubicin groups (½ × IC50, IC50, and 2 × IC50) exhibited significantly elevated intracellular ROS levels from the DMSO-treated control group. On the other hand, only 2 × IC50 of ACES-treated group displayed a statistically significant elevation of intracellular ROS level from the DMSO-treated control group. There are also some expansions in the mean values of ROS quantities in other groups (½ × IC50, IC50) of ACES-treated cells. However, those were not statistically significant (Figure 5). To conclude, both doxorubicin and ACES elevate the intracellular ROS level and lead to cellular death, but the doxorubicin-mediated ROS elevation is much higher than ACES.
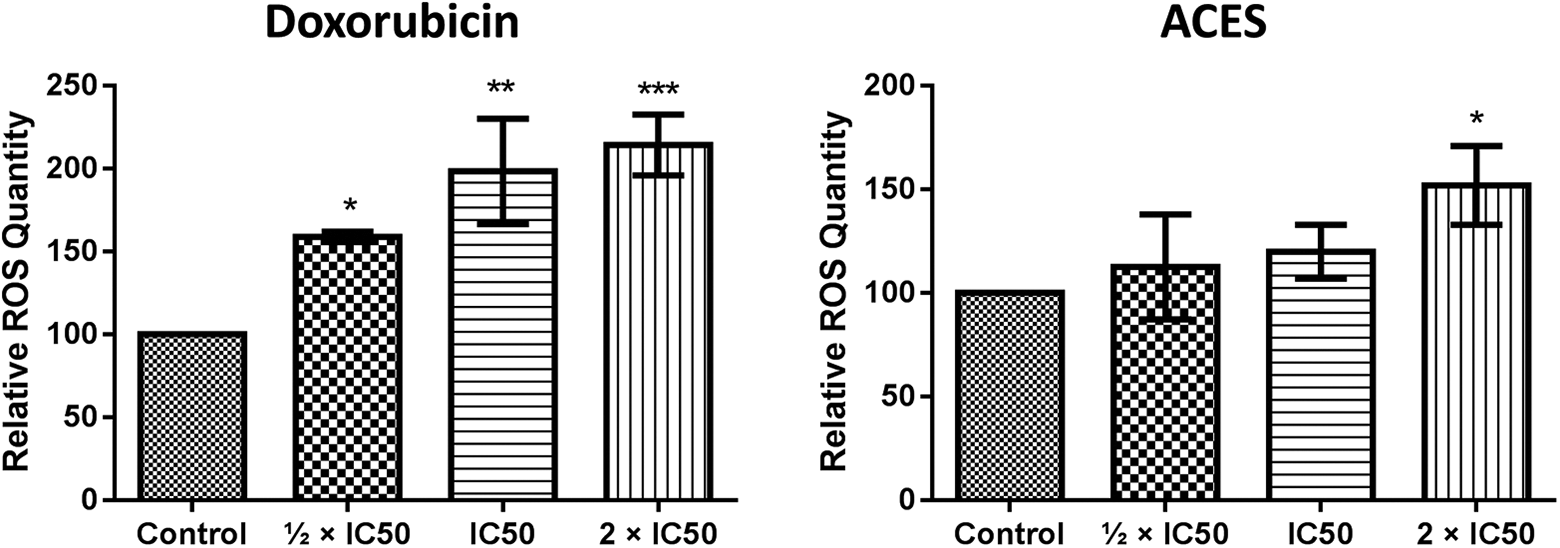
Intracellular ROS levels of doxorubicin- and ACES-treated MCF-7 cells. The results from three independent experiments are presented as mean ± SEM. Statistical comparisons were carried out through ANOVA followed by Dunnett’s post hoc (*
ACES inhibits the expressions of MAPK pathway-related 30 genes in MCF-7 cells
It has been long known that chemotherapeutic agents, including taxol, etoposide, and ceramide, can both trigger apoptosis and MAPK pathway in MCF-7 cells.
28
In addition, putative mechanisms for ROS-mediated activation of MAPK pathways are clearly reviewed in the literature.
29
Therefore, whether the activation of the MAPK pathway occurs with the ACES-mediated ROS elevation is investigated by the determination of expressions of MAPK pathway-related 84 mRNAs in the 72nd hour of ACES-treated MCF-7 cells. There were no significant upregulations in the expressions of tested genes, whereas 20 genes consisting of
The expressions of MAPK pathway-related 30 genes in ACES-treated MCF-7 cells.
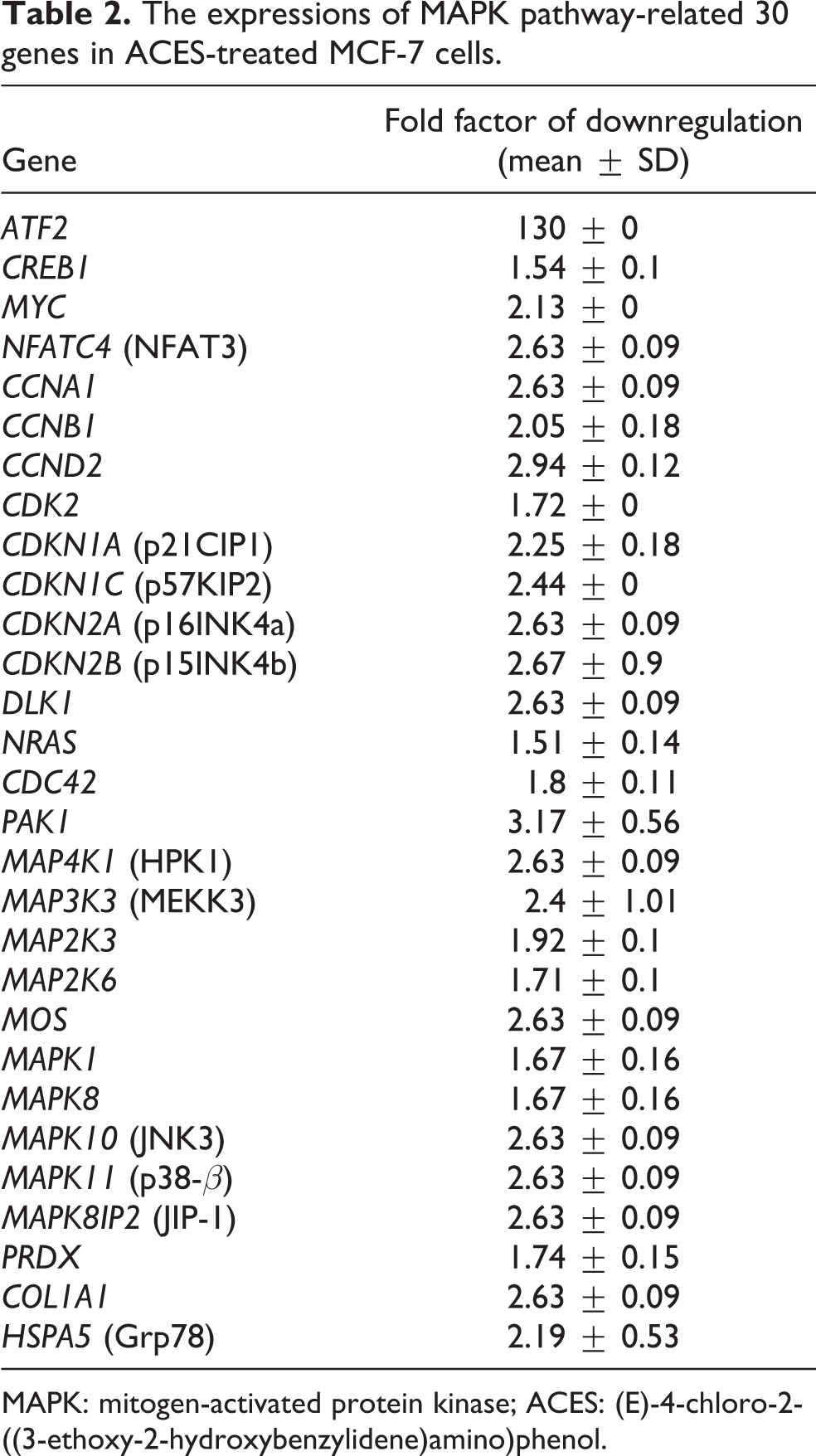
MAPK: mitogen-activated protein kinase; ACES: (E)-4-chloro-2-((3-ethoxy-2-hydroxybenzylidene)amino)phenol.
Discussions
Despite the massive amount of ongoing research, cancer remains as one of the deadly diseases in the world. Although some cytotoxic drugs have been used in cancer therapy, it is essential to discover new drugs to cope with the chemoresistance. In this study, ACES-mediated cellular toxicity, cellular death mechanism, and the expression of genes that code for the elements of the MAPK pathway were investigated. ACES suggested as a new cytotoxic drug candidate based on the observations which are the exhibition of selective cytotoxicity to hepatocellular carcinoma cells, induction of the intrinsic apoptosis, and downregulation of the expressions of the p38-β/SAPK pathway-related genes.
The idea that ACES exhibits selective cytotoxicity to hepatocellular carcinoma cells relies on the observations from the NRU test. In this test, the cytotoxicity of ACES was screened on a cell panel consisting of both cancer and normal cells. The concentration of ACES required to halve the number of cells (IC50) and the effectiveness of ACES for selecting the cancer cells (SI1 and SI2) were estimated. The results indicated that ACES-mediated cytotoxicity is more than fivefold selective to the hepatocellular carcinoma cells with the IC50 of 0.93 µg/mL. Due to the accelerated activities of detoxification enzymes such as cytochrome p450,
Apoptosis is a programmed cell death process characterized by the nuclear fragmentation executed by caspase enzymes. The reduction in the frequency of apoptosis in the cells which are of the genomic instability has long been speculated by the researchers as the generation of malignant cells that resist for surviving.
31
In addition, inducing the apoptosis by the application of anticancer drugs such as doxorubicin, cisplatin, taxol, etoposide, and ceramide is one of the most common approaches in chemotherapy. However, many cancer cells acquire the resistance to drugs with the disrupted balance of proapoptotic and antiapoptotic proteins, reduced caspase function, and impaired death receptor signaling.
32
As a natural result of this resistance, the drugs used by cancer patients lose therapeutic effect; therefore, clinical oncologists need new chemicals to overcome the chemoresistance.
33
MCF-7 cells that have a deletion in the caspase 3 coding
The initial experiment for the evaluation of ACES-mediated cellular death mechanism was the cell cycle analysis. In this analysis, the percentages of cell populations that have the fragmented nuclei were measured as sub-G0/G1 peaks. In the literature, it has been well-documented that doxorubicin increases the sub-G0/G1 population in MCF-7 cells. 36 –38 In this study, based on the observations from the cell cycle analysis, it was suggested that ACES enhances the population of nuclear-fragmented cells, which characterizes the apoptotic cell death identically with doxorubicin. Since the disruption of the MMP is another distinctive character of apoptosis, to confirm the ACES triggered apoptotic cellular death with another experiment, MMP status was measured. In the literature, it has been reported that doxorubicin disrupts MMP in MCF-7 cells. 39 In this study, based on the observations from the measurement of MMP status, it was concluded that ACES enhances the population of disrupted MMP, which further characterizes the apoptotic cell death identically with doxorubicin. These cell cycle and MMP analyses demonstrated that ACES triggers apoptotic cell death. However, in the apoptosis, there is also a long debate among the scientists whether the disruption of MMP activates the caspases 40 or activated caspases disrupt the MMP. 41 Some cytotoxic drugs such as doxorubicin have shown to activate caspase enzymes initially, whereas others disrupt MMP. To clarify whether the activation of the caspase enzymes occurred before the MMP disruption, caspase 3/7 and 9 activations in early stages of ACES- and doxorubicin-triggered apoptosis were measured. Neither caspase 3/7 nor caspase 9 activations were observed in the early stages of ACES-triggered apoptosis. Conversely, caspase 3/7 was found to be activated in the early stages of doxorubicin-triggered apoptosis in MCF-7 cells. Based on this observation, it was concluded that doxorubicin might activate caspase 3/7 enzymes at early times of apoptosis. However, it was not the case in the ACES-triggered apoptosis. Elevated intracellular ROS levels have been suggested to damage the cellular macromolecules, such as nucleic acids, proteins, lipids, and carbohydrates, leading to cell death. It was also an observation that doxorubicin generates elevated ROS production, then damages DNA in MCF-7 cells. 42 In this study, the ROS levels in both ACES- and doxorubicin-triggered apoptosis was measured in MCF-7 cells. The results indicated the elevation of ROS levels both in doxorubicin- and ACES-mediated apoptosis takes place. However, doxorubicin-mediated ROS elevation is twofold higher than ACES. Doxorubicin is widely used as anticancer agent for the treatment of breast, ovary, bladder, and thyroid carcinomas. 43 However, long-term utility of doxorubicin results in the development of heart failure with the cardiotoxic effects. 44 In this study, ACES is observed to be nearly useful as doxorubicin for inducing apoptosis in MCF-7 cells. However, more studies are needed to ensure the safety of ACES in the management of cancer.
Based on the motifs in their activation domains, MAPK proteins are divided into three families as ERKs, JNKs, and p38/SAPKs.
45
While ERK pathway is activated primarily by growth factors, JNKs and p38/SAPKs are activated by stress factors.
46
For the activation of MAPK proteins, the upstream elements, which are MAP3Ks and MAPK2Ks in the signaling cascade, are required to be successively activated. Activated MAPKs further turn on the transcription factors which regulate the expression of the genes that are involved in cell proliferation, differentiation, development, and death.
45
Clinical importance of MAPKs is recently reviewed for cholangiocarcinoma,
47
lung cancer,
48
neuroblastoma,
49
and prostate cancer.
50
High levels of ROS production caused by cytotoxic drugs have been reported to be associated with the activation of MAPK pathways in MCF-7 cells.
28
Since cancer cells can activate the MAPK pathways in response to the elevated intracellular ROS level, then accelerate tumor growth,
51,52
expressions of MAPK pathway-related 84 genes were determined in ACES-mediated apoptosis through the qRT-PCR. The data indicated that there was no upregulation in the expression of tested genes, but 30 genes including
Cell cycle regulators constitute the quarters of the ACES-mediated downregulated genes. These are
ACES-mediated downregulated genes can be dived into three categories based on the involved MAPK pathways.
In mammals, SAPKs are stimulated by stress factors such as cytokines, DNA alkylating agents, ER stress, heat stress, hormones, inflammatory signals, ischemia, osmotic shock, osmostress, oxidative stress, and ultraviolet light.
69
–76
MAPKs in the SAPK signaling cascade are p38 family proteins which are p38-α, p38-β, p38-γ, and p38-δ.
77
These are responsible for the activation of transcription factors. P38-β, which was downregulated with ACES treatment, differs from p38-α with the high affinity to phosphorylate and activate ATF2.
78
ATF2, which was dramatically downregulated with the ACES treatment, regulates the expression of both oncogenic and tumor suppressor genes in response to stress factors.
79
ATF2 forms homodimers or heterodimers with ATF2, BRCA1, CREB1, JDP2, JUN, JUNB, JUND, MAFA, NF1, NFYA, PDX1, POU2F1, and TCF3.
79
–81
These dimers stimulate predominantly oncogenes and infrequently tumor suppressor genes coding for apoptosis regulators, cell adhesion molecules, cell cycle regulators, cytokines, DNA damage proteins, ER stress-response proteins, epithelial-to-mesenchymal transition proteins, extracellular matrix proteins, growth factor receptors, invasion-associated proteins, and transcription factors.
82
–88
In growth plate chondrocytes, ATF2 enhances the apoptosis inhibitor protein, Bcl-2, with the formation of ATF2/CREB1 heterodimers in the promoter of
Future studies can be conducted to further investigate the consequences of ACES treatment on the reversal of p38-β/SAPK-mediated chemoresistance and/or radioresistance in breast cancer. As an illustration, it was suggested that the p38 signaling drives trastuzumab resistance and invasiveness of HER2-overexpressing breast cancer patients. 3 For the reversal of the trastuzumab resistance in breast cancer, researchers can implement ACES within conventional chemotherapy. Moreover, the potential to combine ACES as an inhibitor of p38-β/SAPK signaling pathway with localized radiation in the radiotherapy is considerable. On the other hand, it is obligatory to estimate whether the ACES-included combinatory therapies could act synergistically with conventional chemotherapy and radiotherapy, in addition, to examine any potential interactions optimizing therapeutic outcome.
Footnotes
Acknowledgments
The author is grateful to Professor Dr Ülkü Dilek Uysal and Dr Halil Berber (Eskişehir Technical University, Eskişehir, Turkey) for generously providing (E)-4-chloro-2-((3-ethoxy-2-hydroxybenzylidene)amino)phenol.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
