Abstract
Background:
Doxorubicin (DOX) is used for treatment of many cancer types. Thymoquinone (THQ) is a powerful antioxidant agent used for reducing side effects of several drugs. The aim of this study is to determine possible therapeutic effects of THQ on doxorubicin-induced testicular toxicity in rats.
Methods:
Rats were divided into five groups (n = 8): control, THQ, olive oil, DOX (a single dose of 15 mg/kg intraperitoneally (i.p.) on seventh day of the experiment), and DOX + THQ (10 mg/kg THQ per day and 15 mg/kg DOX i.p. on seventh day). Animals were euthanized, and testis tissues were evaluated histopathologically. Caspase 3 and HSP90 immunostaining were performed to determine the expression levels of these proteins among groups. Terminal deoxynucleotidyl transferase 2′-deoxyuridine, 5′-triphosphate nick-end labeling method was used for evaluation of apoptotic index. Moreover, serum testosterone levels and total antioxidant status (TAS) and total oxidant status (TOS) in testicular tissue were measured by ELISA assay.
Results:
The DOX group had histopathological deterioration compared to the control group. There was an increase in apoptotic index, caspase 3 and HSP90 expressions in the DOX group. While TAS level of the DOX group decreased, TOS level increased when compared with the other groups. Serum testosterone levels in the DOX group decreased compared to the control group. However, there was improvement in testicular tissue in DOX + THQ group compared to the DOX group. There was a decrease in apoptotic index, caspase 3, and HSP90 expressions in DOX + THQ group compared to the DOX group. Testosterone level of DOX + THQ significantly increased compared to the DOX group.
Conclusion:
We suggest that THQ can be used as a protective agent to reduce the toxic effects of DOX.
Introduction
Chemotherapy is an important method in vital diseases, such as cancer, but chemotherapy drugs generally cause notable toxicity. 1 Doxorubicin (DOX) is an anthracycline antibiotic used for treatment of many cancer types. 2 The mechanism of its anticancer activity is referred to prevention in DNA replication by inhibiting the DNA and RNA polymerase and DNA topoisomerase II activities. These restrictions induce the apoptosis in most cell types. 3 However, its usage is restricted because of the harmful side effects on various organs, such as heart, testis, brain, and kidney. 4 –6 The exact mechanism responsible for DOX-induced testicular toxicity is not uncertain, 7 but findings from recent studies have been reported to be cellular apoptosis, lipid peroxidation, and oxidative stress. 8
Natural products play a substantial role for treatment of many diseases. Particularly, herbal medicines have drawn attention in recent years. Thymoquinone, an active component of Nigella sativa seed oil, is used as an antioxidant, 9 anti-inflammatory, 10 and antitumor agent. 11 Therefore, researchers have given these properties an undivided attention to explore its molecular mechanisms and its potential for clinical usage. Its low systemic toxicity and high biological activity make it a hopeful alternative to traditional therapeutic drugs. 12 Nowadays, THQ is considered as a powerful antioxidant in treatment of many damages.
Heat shock proteins (HSPs) are main components of the cellular stress response to decrease injury, accelerate regeneration, and maintain homeostasis. 13 Overexpression of HSPs is induced by many types of cell stress, such as oxidative stress, inflammation, and toxic compounds. 14 As one of the widely known HSPs, HSP90 has an important role in the regulation of the cell’s life process, cell functions, and continuousness of immunity. 15
Apoptosis has a major function in most defense mechanism such as immune reaction triggered when cells are injured by a disease or hazardous agents. 16 To date, researchers have reported that there are two major apoptotic pathways: the intrinsic which is commonly known mitochondrial pathway and the extrinsic or death receptor pathway. The intrinsic and extrinsic reach to same destination—execution pathway. This pathway begins with the activation of caspase 3, which is an initiator of apoptosis and characterized by DNA fragmentation, degeneration of cytoskeletal and nuclear proteins, apoptotic body formation, and production of antigens for receptors of phagocytic cells. 17
In the present study, we aimed to investigate the potential therapeutic effects of THQ on DOX-induced testicular toxicity in rats. For this purpose, a significant testicular damage was induced by DOX in rats and THQ administration was performed to reduce the detrimental effects of DOX. Moreover, histopathological, immunohistochemical, and biochemical changes were evaluated to determine the effects of DOX and THQ.
Materials and methods
Experimental design
The study protocol was accepted by the Erciyes University’s Experimental Animal and Local Ethics’ Committee with number 19/084/2019. In this study, all the animals received human care according to standard guidelines. In the present study, 40 male Wistar albino rats (9 weeks old, weighing 200–250 g) were obtained from Hakan Çetinsaya Experimental and Clinic Research Center, Erciyes University, Kayseri, Turkey, for the experiment. During the experiment, rats were harbored in a 12-h light/12-h dark cycle at room temperature (20–24°C) and environmental humidity. Standard chow and tap water were given to animals ad libitum. In the beginning of the experiment, the rats were randomly divided into five groups as follows: The control group (n = 8) were untreated rats, the olive oil (n = 8) group administered olive oil intraperitoneally (i.p.), the THQ (n = 8) group given 10 mg/kg THQ i.p. (274666-5G, purity ≥ 98%, Sigma–Aldrich Co., St Louis, MO, USA) per day (dissolved in olive oil) throughout the experiment, the DOX (n = 8) group injected with a single dose of 15 mg/kg i.p DOX on seventh day of the experiment and the DOX + THQ (n = 8) group administered 10 mg/kg THQ per day and 15 mg/kg DOX i.p. on seventh day. Experiment continued for 14 days. 18,19
At the end of the experiment, animals were anesthetized with 30 mg/kg ketamine and 4 mg/kg xylazine and they were euthanized after blood samples were collected for serum isolation. Collected serum samples were centrifuged during 10 min at 3000 r/min. After euthanizing, testis tissues were extracted from the animals for the histopathological and immunohistochemical examinations. Serum samples were kept at −80°C for later biochemical assays.
Histopathological evaluation
A histopathological evaluation of the structure of the testicular tissue was performed using routine histological methods. Testis tissues were fixed in 10% formalin solution for 24–48 h, dehydrated with alcohol, cleared with xylene, embedded in paraffin wax, and cut into 5-μm-thick sections. Hematoxylin–eosin were conducted for the evaluation of the histopathological changes in tissue samples. Photographs were taken with a photomicroscope (Olympus BX51; Olympus, Tokyo, Japan) analyzed by the study group.
Testicular tissue structure was examined and evaluated randomly with standard light microscopy by histologists of the study group. Mean seminiferous tubule diameters (MSTDs) were measured in micrometers (Analysis LS Research Program). More than 20 seminiferous tubular sections were chosen for per testis and their histopathological score was evaluated with Johnsen’s testicular biopsy score (JTBS) from 1 to 10, as described previously. In this classification system, all tubular sections in each section of the testicular biopsy are evaluated systematically and each is given a score from 1 to 10. Complete spermatogenesis with many spermatozoa present is evaluated as score 10. 20
Immunohistochemistry
Immunohistochemistry method was used to investigate caspase 3 and HSP90 antibodies in germ cells of testis tissues. Five-micrometer-thick sections were obtained from testis tissues embedded in paraffin blocks. The sections were kept in the oven at 60°C for at least 2 h so that the paraffin melted. The tissues were deparaffinized and rehydrated using xylene and alcohol series. Sections were taken into 0.01 M citrate buffer and heated in microwave oven at 350 W, consequently, antigen retrieval was obtained. Then, sections were kept three times in phosphate-buffered saline (PBS) for 5 min. The sections were kept in 3% (w/v) hydrogen peroxide for 10 min to block endogenous peroxidase activity. After washing again three times in PBS, ultra V block solution was added to the tissues and kept in the tank for 5 min. After then, HSP90-specific polyclonal antibody (1:100) (sc-101494, Santa Cruz Biotechnology, Santa Cruz, California, USA) and caspase 3 polyclonal antibody (1:80) (ab2302, Abcam, Cambridge, UK) were added to the tissues and incubated overnight at 4°C. The following morning, the tissues were washed again three times with PBS and the secondary antibody (TA-125-HDX, Thermo Fisher Scientific, Waltham, Massachusetts, USA) was instilled for10 min at room temperature. After washing with PBS, the immunoreaction was amplified using the streptavidin–avidin–peroxidase complex and the sections were visualized using 3,30-p-diaminobenzidine tetrahydrochloride (TA-060-HDX, Thermo Fisher Scientific) lightly counterstained with Gill hematoxylin. For the final step, increasing alcohol serial concentrations were used to remove water, the sections were then passed through xylene, and finally, they were covered with an entellan. Images were taken using a light microscope. ImageJ program was used to evaluate antibody expressions.
ELISA assay
We centrifuged blood samples taken from rats at 10,000 × g at 4°C for 15 min. Total antioxidant status (TAS) and total oxidant status (TOS) were measured in testicular tissue. For this purpose, testis samples stored at −80°C were used. The samples were homogenized on ice and then centrifuged to remove supernatants. One hundred fifty microliters of the supernatant were taken into Eppendorf tubes separately for each group. For analysis, testosterone, TAS, and TOS ELISA kits were used (201-11-5126, DZE201112672, DZE201111669, 96 Wells ELISA kit, Sunred Biological Technology Co., Ltd., Shanghai, China). In standard preparation, standard diluent was added to five tubes. Then, 120 µl of standard solution were added to the first tube and mixed. After then, 120 µl from the previous tube were added to the other four tubes, respectively. Then, 40 µl of 150 µl from the supernatant were added to the samples section. Then, we added 10 µl of testosterone antibody to the samples. We added 50 µl of streptavidin horseradish peroxidase (HRP) to both samples and standard section and then incubated for 60 min at 37°C oven. We washed plate five times and added chromogen solutions A and B. We put it in a 37°C oven for 10 min. Afterward, we added stop solution and measured the optical density under 450-nM wavelength within 10 min.
Apoptosis (TUNEL)
In the damage caused by DOX, apoptotic cells in testicular tissue were demonstrated by terminal deoxynucleotidyl transferase 2′-deoxyuridine, 5′-triphosphate nick-end labeling (TUNEL) method (In Situ Cell Death Detection Kit, Roche Diagnostics GmbH, Mannheim, Germany). Five-micrometer-thick sections were left in a 60°C oven for at least 2 h to melt paraffin. The tissues were deparaffinized and rehydrated using xylene and alcohol series. Sections were kept three times in PBS for 5 min. Sections were taken into sterile urine container with 0.01 M citrate buffer and heated in microwave oven at 350 W, consequently antigen retrieval was obtained. Then, sections were kept three times in PBS for 5 min. Four hundred fifty microliters of the purple-capped label solution and the blue-capped enzyme solution were mixed. We used this mixture for positives. We used 50 µl of solution remaining in the label solution for negatives. These solutions are added to the tissues in the dark and then incubated in a 37°C oven for 1 h. After washing with PBS, it is covered with 4′,6-diamidino-2-phenylindole in the dark and TUNEL-positive cells were calculated using fluorescent microscope.
Statistical analysis
All statistical analyses were carried out using SPSS statistical software (SPSS for Windows, version 24.0, SPSS Inc., Chicago, Illinois, USA) and graphs were drawn using GraphPad Prism 8.0 software. The Kolmogorov–Smirnov test was used to identify normal distribution of the data. In case of normal distribution, quantitative variables were compared using one-way analysis of variance and Tukey’s post hoc test. The data were presented as the mean of normalized data ± standard deviation of mean. The value of p < 0.05 was considered as statistically significant.
Results
Histopathological findings
Body weight in the DOX group decreased significantly compared to the control group (p < 0.005). Testis weight in the DOX group decreased significantly compared to the control group. DOX + THQ group values were significantly higher compared to those of the DOX group (p < 0.05). In the DOX group, seminiferous tubule diameter decreased significantly compared to the control group (p < 0.05). JTBS in the DOX group decreased substantially compared to the control group (p < 0.05). A significant increase was observed in the DOX + THQ group compared to the DOX group (p < 0.05). In other words, THQ administration to DOX-treated rats provided improvement in testicular histological view when compared to the DOX group.
A decline in germinal cells, disorganization in the seminiferous tubule basal membrane and seminiferous epithelium, vacuolization and immature germinal epithelial cells in the lumen were seen in the DOX group. Few spermatozoa were observed in the lumen in the epididymal tissue of the DOX group (Figure 2(d) and (e)). In the DOX + THQ group, although there were a few tubules in which nonmatured germinal epithelial cells were seen in the lumen, the appearance was close to that of the control group (Figure 2(f)). The graphic of animal weight, testes weight, seminiferous tubule diameter, and JTBS is shown in Figure 1.

(a to d) Effect of THQ treatment on (a) animal weight, (b) testes weight, (c) seminiferous tubule diameter, and (d) JTBS in rats treated with DOX. All data are expressed as the mean ± SD (n = 8). Seminiferous tubule diameter and JTBS were reduced significantly (p < 0.001) in the DOX group compared to the control group and other groups. DOX: doxorubicin; THQ: thymoquinone; JTBS: Johnsen’s testicular biopsy score.

(a to f) Light microscopy of testicular tissue stained with H&E in experimental groups. (a) Group I (control group, n = 8), normal testes were observed; (b) group II (olive oil group, n = 8); (c) group III (THQ group, n = 8), normal testicular architecture was observed; (d) group IV (DOX group, n = 8), disorder of seminiferous tubule germinal epithelium; and (e) group V (DOX group, n = 8) desquamation of epithelial cells and formation of the vacuoles in epithelial cells in the lumen were observed; (f) group VI (DOX + THQ group, n = 8) normal testicular architecture was observed. Testicular cross sections were stained with H&E. DOX: doxorubicin; THQ: thymoquinone; H&E: hematoxylin–eosin.
Immunohistochemical findings
Immunohistochemical staining was performed using the avidin–biotin method to determine the testes tissue expressions of caspase 3 and HSP90. Immunohistochemical examinations demonstrated the presence of caspase 3 and HSP90 immunostaining in the germinal cells of the seminiferous tubules. The caspase 3 and HSP90 expressions in the testes of THQ-alone-administrated rats and olive oil group were similar to those in the control group. Especially, caspase 3 and HSP90 immunoreactivities were considerably increased in germinal cells in DOX group. Caspase 3 and HSP90 expressions of DOX + THQ were substantially less compared to those in the DOX group. Figures 3 and 4 show the caspase 3 and HSP90 expressions in experimental groups.

(a to j) Caspase 3 and HSP90 staining of testicular tissues in experimental groups. (a) Group I (control; n = 8), (b) group II (olive oil, n = 8), (c) group III (THQ, n = 8), weak caspase 3 immunostaining in germinal cells in the seminiferous tubules; (d) group IV (DOX, n = 8) caspase 3 expression was increased in the germ cells of seminiferous tubules. (e) Group VI (DOX + THQ, n = 8) caspase 3 expression was substantially less compared to those in the DOX group. Similarly, (f) group I (control; n = 8), (g) group II (olive oil, n = 8), (h) group III (THQ, n = 8), weak HSP90 immunostaining in germinal cells in the seminiferous tubules; (i) group IV (DOX, n = 8) HSP90 expression was increased in the germ cells of seminiferous tubules; and (j) group VI (DOX + THQ, n = 8) HSP90 expression was substantially less compared to those in the DOX group. DOX: doxorubicin; THQ: thymoquinone; HSP90: heat shock protein 90.
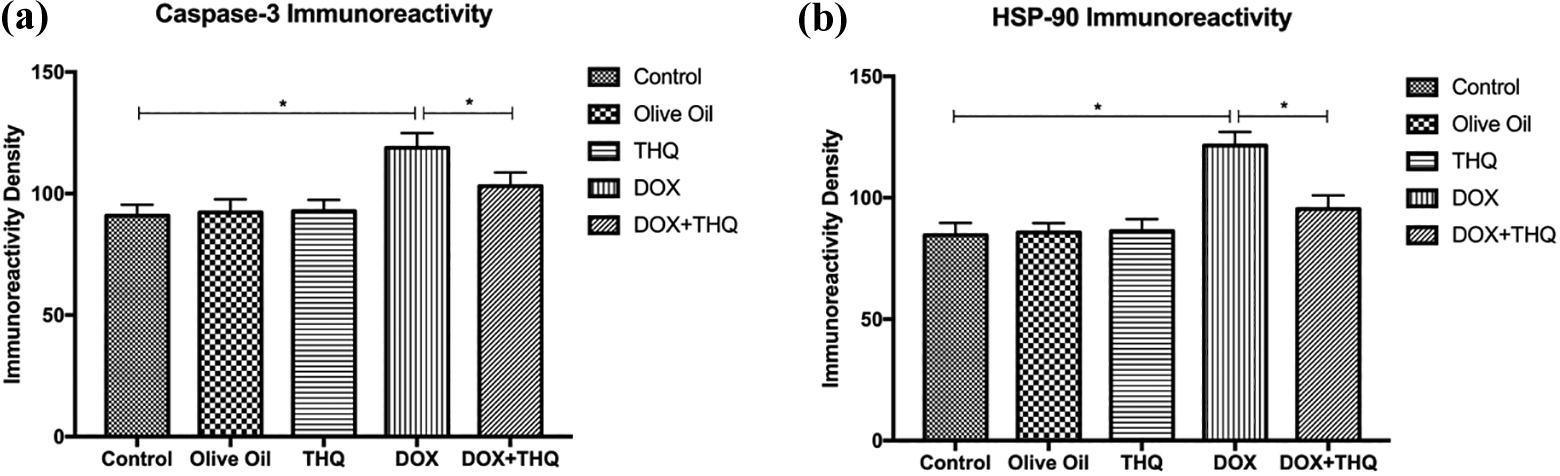
Results of (a) caspase 3 and (b) HSP90 immunoreactivity measurements and statistical analysis of them in experimental groups. Graphs show that caspase 3 and HSP90 expressions are statistically significant in the DOX group when compared to control, olive oil, and THQ groups. Asterisk (*) shows the statistically significance among experimental groups. DOX: doxorubicin; THQ: thymoquinone; HSP90: heat shock protein 90.
Biochemical findings
Exposure of DOX substantially decreased the plasma testosterone level in comparison with the control group (Figure 5), suggesting the inhibitory role of DOX on testicular androgenesis. There was no statistically significant difference between the DOX and DOX + THQ groups.

(a to c) Biochemical results obtained by ELISA assay in experimental groups. Graphs show measurements of (a) TAS and (b) TOS levels in testicular tissues and (c) serum testosterone levels and statistical analysis of the measurements. Asterisk (*) shows the statistical significance between experimental groups. TAS: total antioxidant status; TOS: total oxidant status.
Serum testosterone levels were reduced significantly (p < 0.001) in DOX when compared to the control group. There was no statistically significant difference between DOX and DOX + THQ groups. TAS levels were lower in the DOX group compared to the other groups (p < 0.05). However, TOS levels were significantly higher in the DOX group than in the other groups. There was no statistically significance in TAS and TOS level between the other groups excluding DOX group (p > 0.05; Figure 5).
Apoptotic findings
TUNEL staining was performed to determine apoptotic cells in testes tissue (Figure 6). The apoptotic cells in the testis of control group, olive oil group, and THQ group were found 0.37 ± 0.63, 0.37 ± 0.66, and 0.39 ± 0.82, respectively. There was no statistically significance among this group. The increase in the apoptotic cell number in DOX group was found 3.76 ± 1.78 and was statistically significant when compared to the control group (p < 0.0001). In DOX + THQ group, there was a decrease in TUNEL-positive cells and the apoptotic cell number (0.39 ± 0.82). The decrease in the apoptotic cell number was statistically different in DOX + THQ when compared to the DOX group (p < 0.0001).

(a to f) TUNEL staining of testicular tissue was observed. (a) Group I (control, n = 8); (b) group II (olive oil, n = 8); (c) group III (THQ, n = 8), normal testes were observed; (d) group IV (DOX, n = 8); (e) group V (DOX, n = 8) TUNEL-positive cells (arrow) were mainly observed in germ cells of testis; and (f) group VI (DOX + THQ, n = 8) decrease in TUNEL-positive cells was observed. TUNEL: terminal deoxynucleotidyl transferase 2′-deoxyuridine, 5′-triphosphate nick-end labeling; DOX: doxorubicin; THQ: thymoquinone.
Discussion
DOX, an anthracycline antibiotic, has high antitumor efficacy against different types of cancer. 21 In spite of DOX’s potent antineoplastic activity in variety of cancer, its clinical use is hampered due to adverse side effects, such as cardiotoxicity, 22 nephrotoxicity, 23 hepatotoxicity, 24 and gonadotoxicity. 25 In addition, the DOX leads to substantial decreases in reproductive organ weights and their structural deterioration. 26,27 In this study, a significant attenuation in the body weight and testicular weight of DOX-exposed animals is shown. This may be due to parenchymal atrophy in the seminiferous tubules and spermatogenic depredations in testes. DOX induces a significant decrease in size of the seminiferous tubule, degeneration in spermatocytes, spermatogonia and seminiferous tubules, decreased seminiferous epithelial layers, and less number of germ cells. 28 Our study showed that DOX led to decrease in size of the seminiferous tubule (to 262.2 µm from 331.5 μm), number of the seminiferous tubules, vacuolation and degeneration in spermatogonia, spermatocytes, irregular seminiferous tubules, and reduction in the number of germ cells. We aimed to investigate the mechanism of adverse effects of DOX on the testes of rats. Also, this study showed that MSTD and seminiferous tubule of rats in DOX group substantially reduced. Apoptosis has an important role in the development of DOX damage in many tissues. 29,30 DOX causes apoptosis in the germ line stem cells of juvenile rat testes. 31 Caspases play a role at proteolytic activity and are able to cleave proteins at aspartic acid residues, although particular caspases have distinctive specificities including recognition of neighboring amino acids. Once caspases are initially activated, there appears to be an irreversible commitment toward cell death. To date, initiators (caspase-2, -8, -9, and -10), effectors or executioners (caspases 3, 6, and 7) and inflammatory caspases (caspase-1, -4, and -5) have been identified. 32,33 Caspase 3 is accepted to be the most significant of the executioner caspases and is activated by any of the initiator caspases (caspase 8, caspase 9, or caspase 10). Caspase 3 also causes cytoskeletal reorganization and disintegration of the cell into apoptotic bodies. Immunohistochemical staining was used to evaluate caspase 3 levels. In present study, DOX group showed an important increase in caspase 3 expression compared to the other groups.
HSPs, one of the intracellular chaperones, exhibit cell protective properties. HSPS are synthesized after changes in the cellular environment. For instance, abrupt changes in temperature alter the level of reactive oxygen metabolites and glucose levels. 34,35
Mammalian HSPs are divided into four major classes and HSP90 is one of them. 36 In this study, an important increase in HSP90 expression was found in germinal cells. HSP90, which functions together with HSP70, has been demonstrated to be include in cell-cycle regulation, signal transduction, and apoptotic pathways. 37 Also, another study reported that di-(2-ethylhexyl) phthalate administration led to substantial raises in the gene expression levels of HSP90 in rat testes. 38 In this study, we found that DOX group showed a significant increase in HSP90 expression compared to the other groups.
Since DOX is known to be a proapoptotic agent, the apoptotic cells in the testes are demonstrated by TUNEL staining. Several studies have reported increase in the rate of apoptosis in testicular tissues in the DOX group. 8,27 In this study, we showed that the number of apoptotic cells significantly increased in the DOX group compared to the control group.
Increased levels of reactive oxygen species caused by DOX or its direct effects induce the damages in Leydig cells and significant decline in testosterone level. 28 Serum testosterone levels were evaluated studying ELISA method. We found reduced serum testosterone level in DOX-administered group.
It has been reported that oxidative stress plays a crucial role in the testicular damage induced by DOX. The main mechanism of DOX involvement in oxidative damage is attributed to its ability to interact with cellular macromolecules, 39 and this was the reason behind choosing DOX to target rapidly dividing cells, disrupting cell growth, mitotic activity, and differentiation. 40 Many studies have reported that the levels of TOS in testicular tissue increased in chemotherapeutic administered groups. 41 In our study, we found that TOS levels significantly increased in DOX group, but TAS levels are statistically lower in DOX group compared to the control group.
THQ, with its anti-carcinogenic, anti-inflammatory, and antioxidant properties, has attracted the attention of researchers to explore its molecular mechanisms and use in inflammatory diseases. Nowadays, THQ is used as a protective antioxidant in many damages. It has been reported that THQ ameliorates different models of testicular tissue damage as lead, 42 cadmium, 18 and torsion-induced oxidative injury. 43 In another study, it has been reported that THQ attenuates testicular tissue inflammation induced by sodium nitrite. 44 In this study, the THQ-treated animals showed a reformed histological view in DOX groups and the number of apoptotic cells was more in the DOX group compared to those pretreated with THQ.
Our study showed that THQ clearly inhibited histopathological damage caused by DOX in the testes and ameliorated the MSTD, JTBS, and serum testosterone levels in testicular tissues. THQ is a protective agent that reduces the negative effects of DOX. It is known as antiapoptotic and antioxidant agent without negative effects. In our study, we found that THQ reduces apoptosis, oxidative stress, and testicular damages caused by DOX.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study has been supported by a grant from the Research and Technology Department of Erciyes University.
