Abstract
Various studies in rodents have shown that nanoparticles are transferred to the breast milk. Under the present study, lactating Wistar rats were repetitively gavaged 5, 25, and 50 mg/kg bw of zinc oxide nanoparticles (ZnO-NPs) and 50 mg kg−1 bw of bulk zinc oxide (bZnO) for 19 days after parturition. The results showed that ZnO-NPs were absorbed in the small intestine of dams and distributed to the liver. Furthermore, ZnO-NPs were distributed to the intestine and liver of rat pups through dam’s milk. No significant change in body weight was observed in the dams treated with ZnO-NPs or bZnO and their offsprings as compared to the control group. The spleen weight significantly increased in the rat dams treated with 50 mg kg−1 of ZnO-NPs. ZnO-NPs were mostly excreted through feces. The levels of liver cytochrome P450 reductase and serum total antioxidant capacity significantly decreased in the rat dams treated with ZnO-NPs (50 mg kg−1) and their offsprings. The levels of serum cytokines (tumor necrosis factor-alpha and interleukin-1 beta) and liver injury marker enzymes (alanine aminotransferase and aspartate aminotransferase) significantly increased in the rat dams treated with ZnO-NPs (25 and 50 mg kg−1) and their offsprings. The level of immunoglobulin A secretion in the intestinal fluid of rat dams and their offsprings is significantly increased by increasing the dose of ZnO-NPs. Histopathology of intestine and liver of offsprings whose rat dams were treated with ZnO-NPs (50 mg kg−1) showed gross pathological changes. These results provide information for the safety evaluation of ZnO-NPs use during lactation. In conclusion, a dose-dependent postnatal transfer of ZnO-NPs is hazardous to the breastfed offsprings.
Introduction
Nanotechnology possesses an immense potential, thus having several applications in various fields. The products of nanotechnology are more prevalent in the market as compared to their bulk counterparts. Nanomaterials are more reactive than their bulk equivalents due to their high surface to volume ratio. Zinc oxide nanoparticles (ZnO-NPs) have a very high annual production and considered as the third most highly produced inorganic nanoparticles (NPs) in the world. 1 ZnO-NPs have been extensively used in the manufacture of ceramics, photocatalysis, and ultraviolet (UV) filters. 2 Furthermore, ZnO-NPs have a profound place in consumer products such as sunscreen, additives, and packaging agents due to their antimicrobial properties. 3,4 ZnO-NPs are used as juice clearing agents and nano-fertilizers in the food and agriculture industries, respectively. 5 –8 Also, ZnO-NPs have been used as animal feed additives for better absorption of nutritional supplements. 9 –11 However, several studies have demonstrated the detrimental effect of ZnO-NPs on the health of humans and animals. 12 –16 ZnO-NPs can penetrate the cell membrane easily due to their much smaller size and then distributed to various organs by systemic circulation. 17 –20
Several studies have been conducted to demonstrate the prenatal effect of NPs on the offspring through the placenta as well as the breastmilk pathway.
21
–25
For example, Jo et al.
26
found that zinc level significantly increased in the liver and kidneys of pups, indicating a similar toxico-kinetics behavior of ZnO-NPs to that of female adult rats. Respectable numbers of reports are available on the transfer of natural as well as synthetic compounds from the bloodstream of lactating mother to milk and subsequently to the offsprings. Bisphenol A (BPA) concentration in human colostrum was higher than those in the blood sera.
27
BPA levels tend to increase in the milk samples of lactating women who have consumed polyethylene terephthalate bottle drink frequently.
28
Fujimoto et al.
29
demonstrated the potential risk of BPA exposure to Wistar rat’s offsprings through breastfeeding by postnatal exposure to the lactating dams for seven consecutive days. Irmak et al.
30
extensively reviewed the integration of maternal genome into the neonate genome through breast milk. They concluded that the genetic signals may transfer from mother to neonate during breastfeeding through the milk microvesicles. The results of the study of Anderson et al.
31
revealed the appearance of
Very few reports are available on the postnatal transfer of NP to the offsprings through milk. For example, Sumner et al. 33 reported radioactivity in the gastrointestinal (GI) tract and liver of offspring after a single tail-vein injection of radioactive carbon-14 labeled C60 NPs to lactating rats on postnatal day 8 (PND 8). Their study indicated that the NPs were transferred via the breast milk and consequently absorbed in the GI tract of offsprings followed by distribution to the liver. Likewise, Zhang et al. 34 reported the transfer of small (8 nm) and larger (50 nm) titanium dioxide (TiO2) NPs to the offsprings after intravenous injection through the tail vein to lactating mother mice. They concluded that TiO2 NPs induced oxidative stress in the mammary gland of lactating mouse dams which in turn disrupted tight junction of the blood milk barrier. These findings were depicted by the loss of tight junction proteins and shedding mammary alveolar epithelial cells. Recently, Morishita et al. 35 found that intravenous administration of silver (Ag) and gold NPs to lactating mouse dams were distributed to breast milk without producing apparent damage to the mammary gland. Also, Ag NPs were distributed from the breast milk into the brains of breastfed pups after oral administration to the mother.
To our knowledge, no report has examined the effect of ZnO-NPs transfer to breast milk on offsprings after oral postnatal exposure to the dams. The present study aimed to assess the safety of dose-dependent ZnO-NPs in rat offsprings mediated through breast milk.
Materials and methods
Zinc oxide (ZnO)
Zinc oxide (ZnO) dry nanopowder (Cat# RN-PL-ZnO25-25g) imported and marketed by Reinste Nano Ventures Private Limited (New Delhi, India) from PlasmaChem GmbH (Berlin, Germany) was purchased. Bulk zinc oxide (bZnO; Product no. 20298, CAS# 1314-13-2) was purchased from S.D. fine chemicals (Haryana, India). The ZnO-NPs were characterized by a high-resolution transmission electron microscope (HR-TEM; Tecnai G-20, USA). The size and shape of ZnO-NPs were determined by dynamic light scattering (DLS; Malvern Instruments Ltd., Malvern, Worcestershire, UK) and transmission electron microscopy (TEM). The ZnO-NPs were then weighed and reconstituted in distilled water to the desired dose. After reconstitution, NPs were sonicated (Branson Sonic Power Company, Danbury, Connecticut, USA) for 10 min before use.
Animals and treatment
Female nulliparous Wistar rats were obtained from the Small Animal House of National Dairy Research Institute (Karnal, Haryana, India) after approval from the Institutional Animal Ethical Committee (IAEC:1705/GO/ac/13/CPCSEA, Approval no. 106/16). The animals were approximately of similar body weight (∼150 g). They were housed in polypropylene cages (two animals per cage) in an air-conditioned room at 24 ± 1°C for acclimatization before the start of experiments for 1 week. Female and male rats were housed in the ratio of 2:1 until the confirmation of conceiving by constantly monitoring vaginal plug formation for at least 4 days. Then, pregnant female rats were separated into five different groups (six animals each). After parturition, the number of offsprings with each dam was normalized to a 6:1 ratio. The extra litters were rehabilitated with nonexperimental lactating dams. As currently there are no standard methodologies available for in vivo toxicity assessment of NPs, the selection of doses was carried out according to the Organisation for Economic Co-operation and Development test guidelines 420 for a fixed-dose procedure to investigate the oral toxicity of any new substance. 36 The maximum dose of ZnO-NPs (50 mg kg−1 bw) was chosen based on the results of oral toxicity studies at low doses and high doses, and the agglomeration of ZnO-NPs was also taken into account. 37 –40 Also, a dose in between 5 mg kg−1 and 50 mg kg−1 was taken to select three different doses. The doses were prepared in distilled water and sonicated with a Branson sonicator for 10 min before gavaging. The dose volume maintained for all the groups was a maximum of 10 ml kg−1 bw. From PND 1, the rat dams received three oral doses of ZnO-NPs (5, 25, and 50 mg kg−1) and bZnO (50 mg kg−1) every morning consecutively for 19 days as described in Figure 1.
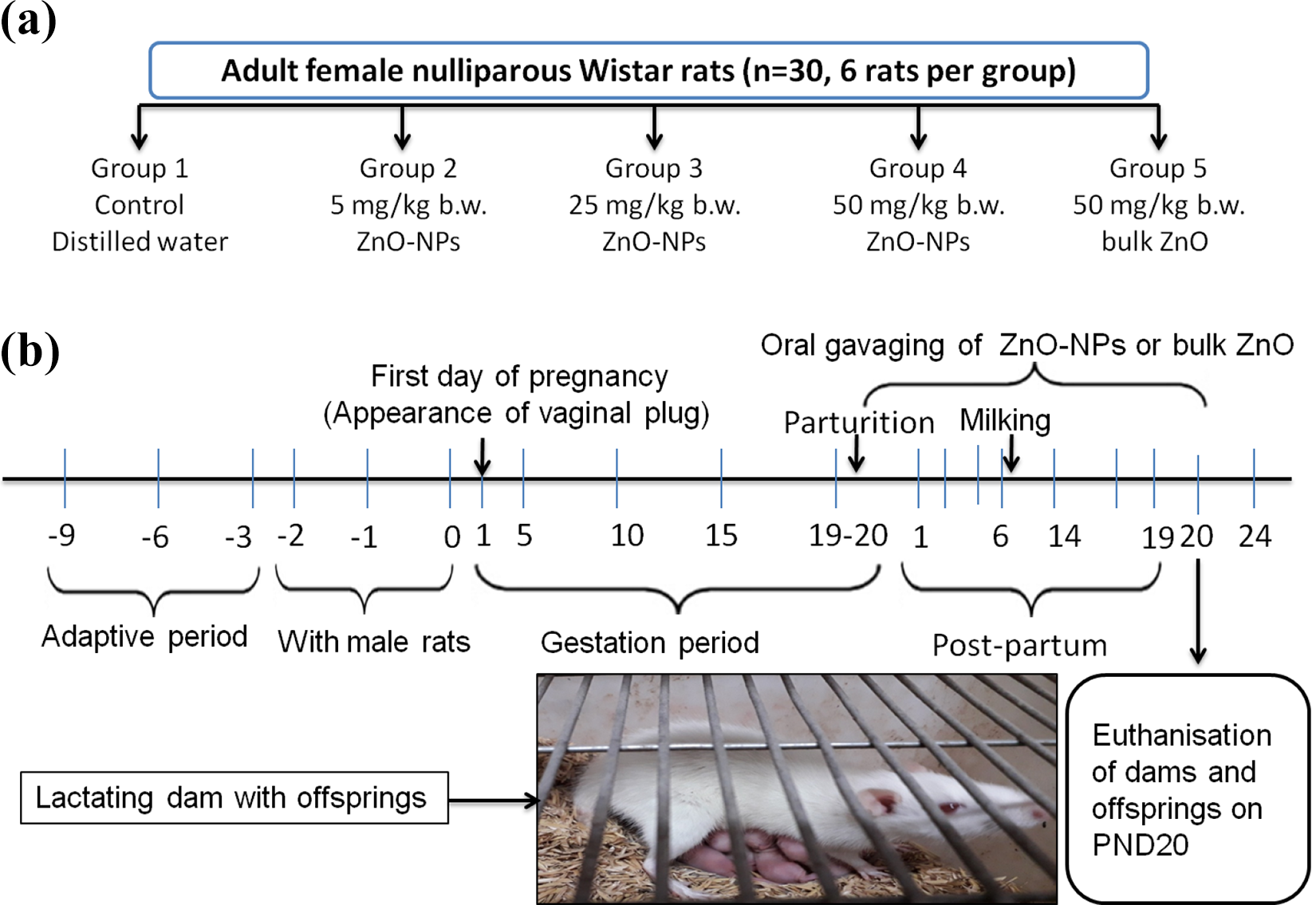
(a) Grouping of animals. (b) Experimental design indicating the period of ZnO-NPs (5, 25, and 50 mg kg−1 bw) and bZnO (50 mg kg−1 bw) treatment to lactating Wistar rats. Dose was calculated according to dam’s body weight. ZnO-NP: zinc oxide nanoparticle; bZnO: bulk zinc oxide; PND: postnatal day.
Collection of milk
Milk was collected from anesthetized Wistar dams using manual kneading of the teat with repetitive bottom-to-top stroking motions. The offsprings were separated from the lactating dams for 4 h. To facilitate milking, the lactating rat received an intraperitoneal injection of 0.1 IU oxytocin after removal from her young ones. Two expressed milk samples were collected from each dam into microcentrifuge tubes and were stored at −20°C (Figure 2). Each sample was analyzed in triplicate for the estimation of zinc content. For unit conversion, 1 ml of breast milk was assumed to weigh 1 g.

(a) Milk collection from an anesthetized Wistar dam by manual kneading of the teat with repetitive bottom-to-top stroking motions and (b) expressed milk was collected in sterile centrifuge tubes and stored at −20°C.
Measurement of body weight and organ weight
At the end of the study, the body weight of all the dams as well as the offsprings was measured. The liver, spleen, and pair of kidney of the dams as well as offsprings were weighed following euthanization. The differences in body weight in the treatment groups were measured and compared with those of the control group. Similarly, various organs of treatment groups were separately measured and compared to those from the control group.
Collection of organs and feces
The liver, intestine, and feces from the dams as well as their offsprings were weighed (∼100 mg) and stored at 4°C for inductively coupled plasma mass spectrometry (ICP-MS). The liver and intestine were collected and stored in neutral buffered formalin for histopathology.
Collection of blood and separation of serum
Blood was collected from the overnight-fasted rat dams and their offsprings through a cardiac puncture in microcentrifuge tubes. For separation of serum, the whole blood was centrifuged at 4000 × g for 20 min. The serum was gently collected and stored in centrifuge tubes at −20°C until further use.
Collection of intestinal fluid
The intestinal tissue (ileum, 2 cm) was flushed with 2 ml of phosphate-buffered saline (pH 7.4) followed by teasing with sterile needles in the same medium to separate the cells and then centrifuged at 2000 × g for 20 min at 4°C. The resultant supernatant (intestinal fluid) was recovered and stored at −80°C until further use for the measurement of secretory immunoglobulin A (IgA).
Measurement of zinc content
The samples of milk, liver, small intestine, and feces were taken to determine the zinc content using ICP-MS. The samples were weighed and digested in a screw cap sample beaker with 2 ml wet-digest solution (75% nitric acid (HNO3):70% perchloric acid = 1:1 volume/volume). Samples were placed on a hot plate overnight at 120°C. The cover was removed and heating was continued at 80°C until the sample digest became clear. Five percent of HNO3 was added to dissolve the sample digest residue and the final volume was brought to 10 ml. The resulting solution was then diluted with 5% HNO3. The total dilution made up to 20 times the original volume and analyzed using ICP-MS (Agilent [Santa Clara, CA, USA] ICP-MS 7900 with Ultra High Matrix Introduction, IIT Delhi). Calibration standards of 10, 20, 50, 100, and 200 ppb were used for method validation.
Analysis of cytochrome P450 (CYP450) reductase assay
Cytochrome P450 (CYP450) reductase was assayed by the method given by Masters et al. 41 using Cytochrome P450 reductase assay kit (Sigma, Catalog no. CY0100). The liver was homogenated in potassium phosphate buffer (300 mM, pH 7.8). The liver homogenate was centrifuged at 12,000 r min−1 for 10 min at 4°C. Furthermore, the supernatant was transferred to a new centrifuge tube and centrifuged at 19,000 r min−1 for 90 min at 4°C. After discarding the supernatant, the pellet was gently washed and resuspended in enzyme assay buffer. All reagents and samples were brought to 25°C before performing the assay. The lysate (30 μl) was mixed with 0.95 ml of cytochrome c working solution (0.45 mg cytochrome c per ml of assay buffer) and 20 µl of cytochrome c oxidase inhibitor solution. Mixed by inversion and started the reaction by addition of 100 µl of nicotinamide adenine dinucleotide phosphate (NADPH) solution. The reduction of cytochrome c is monitored by an increase of cytochrome c absorbance at 550 nm using UVD-3500 double beam UV/visible spectrophotometer (Labomed, Los Angeles, CA, USA). The concentration of reduced cytochrome c was calculated from its molar extinction coefficient (21.1 mM−1 cm−1). The total protein in the homogenate was estimated by the Lowry method, and enzyme units mg−1 protein was calculated accordingly.
Measurement of total equivalent antioxidant capacity (TEAC)
Serum total equivalent antioxidant capacity (TEAC) was measured by using the method of Miller et al. 42 with commercially available kits (Randox Laboratories, Lakewood, California, USA). This method is based on the inhibition by antioxidants of the absorbance of the radical cations of 2, 29-azinobis (3-ethylbenzothiazoline 6-sulfonate) (ABTS) at 600 nm. ABTS radical cations are formed by the incubation of ABTS with metmyoglobin and hydrogen peroxide. The final results are expressed as micromoles of Trolox equivalents per liter.
Measurement of cytokines (tumor necrosis factor-alpha (TNF-α) and interleukin-1 beta (IL-1β))
The concentrations of tumor necrosis factor-alpha (TNF-α) and interleukin-1 beta (IL-1β) in the serum were determined using commercially available enzyme-linked immunosorbent assay (ELISA) kits (eBioscience, San Diego, California, USA). Briefly, each well of a microplate was coated with 100 µl of capture antibody and incubated overnight at 4°C. After washing and blocking with assay dilution buffer, diluted serum or standard was added to the individual wells; then the plates were maintained for 2 h at room temperature. The plates were then washed by washing buffer, biotin-conjugated detecting mouse antibody was added to each well, and they were incubated at room temperature for 1 h. The plates were washed again and further incubated with avidin-horseradish peroxidase (HRP) for 30 min before detection with 3,3′,5,5′-tetramethylbenzidine (TMB) solution. Finally, reactions were stopped by adding 2 M sulfuric acid (H2SO4) and the absorbance at 450 nm was measured with an ELISA reader (Molecular Devices, Sunnyvale, California, USA). The amount of cytokine was calculated from the linear portion of the generated standard curve.
Measurement of serum liver enzymes (alanine aminotransferase (ALT) and aspartate aminotransferase (AST))
The levels of serum liver enzymes including aspartate aminotransferase (AST) and alanine aminotransferase (ALT) were examined by routine colorimetric methods using commercially available kits (Recombigen, India). Liver AST and ALT are the biological markers for liver injury.
Measurement of IgA in the intestinal fluid
Total secretory IgA (sIgA) concentration in the intestinal fluid was determined using quantitative sandwich ELISA kits (eBiosciences). To detect this antibody, plates were coated with 1 µg ml−1 of goat anti-mouse IgA. The samples were diluted 300 times in dilution buffer before adding in the experimental wells followed by the addition of 100 µl of HRP-conjugated goat anti-mouse IgA. Plates were allowed to develop with the TMB substrate and the reaction was finally stopped with 100 µl of 2 M H2SO4. Plates were read at 450 nm in an ELISA reader (Molecular Devices).
Evaluation of histopathological changes
Histopathology of liver and intestine was performed on the offsprings of the control group and the treated groups with ZnO-NPs (50 mg kg−1 bw) and bZnO (50 mg kg−1 bw). The tissues were removed after euthanization and washed with 1% ice-cold saline and fixed in neutral buffered 10% formalin. The tissues were embedded in paraffin blocks and then trimmed and sectioned using a microtome. Paraffin sections of 3 µm thickness were stained with hematoxylin and eosin after mounting on a glass microscope slide. The slides were examined under an inverted light microscope (Olympus, CKX4, Tokyo, Japan) at magnification power (400×).
Statistical analysis
The data were expressed as the average mean with standard error mean. The statistical analysis of the experimental data of quantitative variables was subjected to two-way analysis of variance followed by Bonferroni’s post hoc test (compared each group with the control group) using the Graph pad prism 5, version 5.1 (San Diego, California, USA). All the analysis and comparisons were evaluated at 5% (p < 0.05) level of significance.
Results
Characterization of ZnO-NPs
The characterization of ZnO-NPs obtained from a commercial supplier was performed using TEM and DLS (Figure 3 and Table 1). Particle size determined by HR-TEM was 17.27–31.45 nm. The size distribution analysis of ZnO-NPs by DLS revealed a z-average of 73.62 nm in distilled water. The polydispersity index of 0.416 as given in Table 1 signifies a monodisperse solution with lesser aggregation and agglomeration. In addition, the rings with a dotted pattern in the selected area (electron) diffraction confirm the wide size distribution of ZnO-NPs.

(a) ZnO-NPs crystalline structure and size are shown by the manufacturer. ((b) and (c)) TEM images and corresponding particle size distribution at the different nanometer scale (nm) shown in respective images. (d) DLS micrograph of ZnO-NPs. Size distribution analysis of ZnO-NPs revealed a z-average of 73.62 nm in distilled water. The image (e) shows the SAED pattern of the ZnO-NPs at 101 nm scales and (f) shows particle size at a 10 nm scale. Particle size determined by an HR-TEM was 17.27–31.45 nm. ZnO-NP: zinc oxide nanoparticle; TEM: transmission electron microscopy; DLS: dynamic light scattering; SAED: selected area (electron) diffraction; HR-TEM: high-resolution transmission electron microscope.
ZnO-NP characterization.

ZnO-NP: zinc oxide nanoparticle; PDI: polydispersity index; TEM: transmission electron microscopy; SAED: selected area electron diffraction; DLS: dynamic light scattering.
a According to the manufacturer.
b SAED.
c Using DLS.
Effect of ZnO-NPs on the body and organs weights
The comparison of body and organ weights of the dams and their offsprings are given in Tables 2 and 3. There was no significant difference in the body weight of the rat dams treated with ZnO-NPs (5, 25, 50 mg kg−1) or bZnO (50 mg kg−1) during the lactating period (19 days) and their offsprings. The liver and kidney weight of the treated dams or offsprings did not significantly change as compared to that of the control group. The spleen weight of the dams treated with ZnO-NPs (50 mg kg−1) increased significantly (p < 0.05) as compared to the control group.
Body and organ weight comparison among dams of different groups.a

ZnO-NP: zinc oxide nanoparticle; bZnO: bulk zinc oxide; SEM: standard error mean.
a Data represent the mean ± SEM of six animals and significant (*p < 0.05) compared to control.
Body and organ weight comparison among offsprings of different groups.a

ZnO-NP: zinc oxide nanoparticle; bZnO: bulk zinc oxide; SEM: standard error mean.
a Data represent the mean ± SEM of six animals.
Fate of ZnO-NPs in tissues
The zinc content in the milk, liver, small intestine, and feces was estimated by ICP-MS. The zinc in these organs and milk of the dams increased with an increasing dose of ZnO-NPs (Table 4). The fecal Zn content was increased significantly in a dose-dependent manner. Further, the ZnO-NPs transfer to the suckling pups through the milk of dams. Absorption of ZnO-NPs in the small intestine of offsprings from 25 and 50 mg kg−1 ZnO-NPs group was significantly higher than the control group. A significant level of zinc was distributed to the liver of the offsprings from 50 mg kg−1 ZnO-NPs given dams (Table 5). There was no observed increase in the zinc level of the offspring’s liver from 5 and 25 mg kg−1 ZnO-NPs group as compared to the control.
Levels of zinc in samples of dams after oral gavaging of 5, 25, and 50 mg kg−1 of ZnO-NPs and 50 mg kg−1 of bZnO for 19 days starting from parturition.a
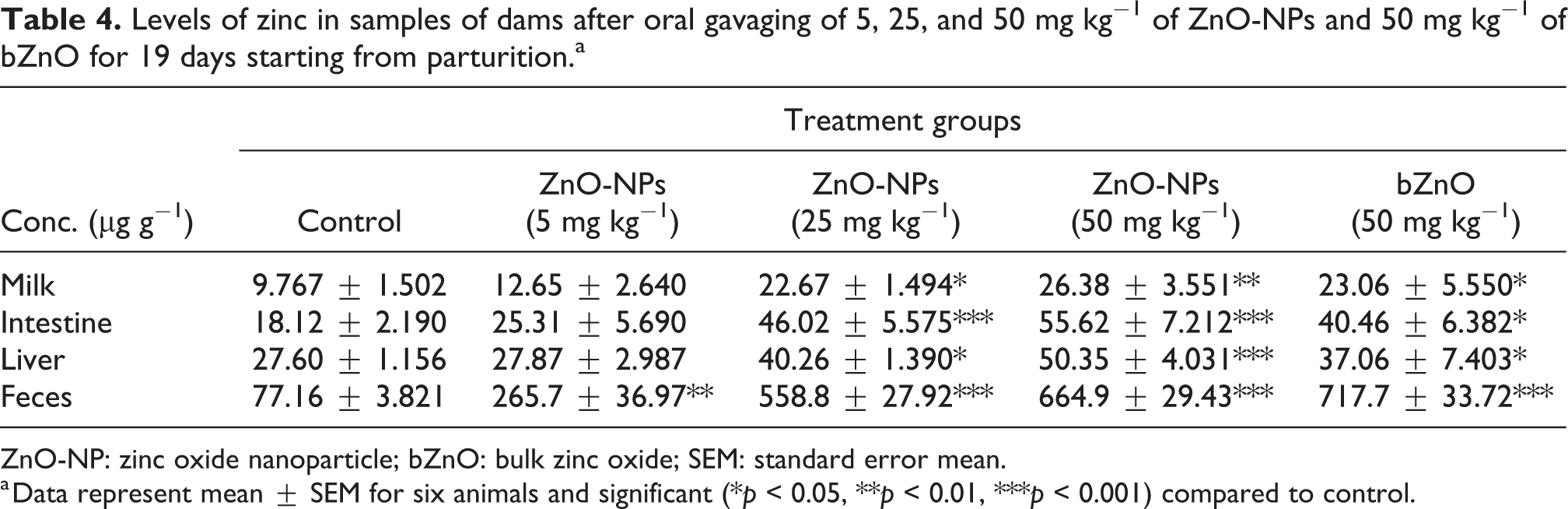
ZnO-NP: zinc oxide nanoparticle; bZnO: bulk zinc oxide; SEM: standard error mean.
a Data represent mean ± SEM for six animals and significant (*p < 0.05, **p < 0.01, ***p < 0.001) compared to control.
Levels of zinc in samples of offsprings after oral gavaging of 5, 25, and 50 mg kg−1 of ZnO-NPs and 50 mg kg−1 of bZnO to dams for 19 days starting from parturition.a

ZnO-NP: zinc oxide nanoparticle; bZnO: bulk zinc oxide; SEM: standard error mean.
a Data represent mean ± SEM for six animals and significant (*p < 0.05, **p < 0.01, ***p < 0.001) compared to control.
Effect of ZnO-NPs on the CYP450 reductase in the liver tissues
The NADPH-dependent CYP450 reductase significantly decreased in the liver of the rat dams treated with ZnO-NPs (25 and 50 mg kg−1 bw) as compared to the control group. Also, the level of hepatic CYP450 reductase decreased significantly in the offsprings of the rat dams treated with the highest dose of ZnO-NPs (50 mg kg−1 bw) as shown in Figure 4.

CYP450 reductase activity level in liver among different groups after repeated oral gavaging of 5, 25, and 50 mg kg−1 bw of ZnO-NPs and 50 mg kg−1 bw of bZnO to Wistar rat dams for 19 days. Values are mean ± SEM (n = 6) and significantly different (*p < 0.05, **p < 0.01, ***p < 0.001) compared to control. CYP450: cytochrome P450; ZnO-NP: zinc oxide nanoparticle; bZnO: bulk zinc oxide; SEM: standard error mean.
Effect of ZnO-NPs on serum total antioxidant capacity (TAC)
As shown in Figure 5, serum total antioxidant capacity (TAC) decreased significantly in the dams treated with 25 and 50 mg kg−1 of ZnO-NPs by 22.77% (p < 0.005) and 31.88% (p < 0.001), respectively. It also decreased significantly in the offsprings of the dams treated with 50 mg kg−1 of ZnO-NPs.

Comparison of TAC level in serum among different groups after repeated oral gavaging of 5, 25, and 50 mg kg−1 bw of ZnO-NPs and 50 mg kg−1 bw of bZnO to Wistar rat dams for 19 days. Values are mean ± SEM (n = 6) and significantly different (*p < 0.05, **p < 0.01, ***p < 0.001) compared to control. TAC: total antioxidant capacity; ZnO-NP: zinc oxide nanoparticle; bZnO: bulk zinc oxide; SEM: standard error mean.
Effect of ZnO-NPs on the serum TNF-α level
Figure 6 shows the increase in TNF-α in the dams and their offsprings with the increasing dose of ZnO-NPs. The TNF-α level is an indicator of the immunological status of individual well-being. Serum levels of TNF-α increased significantly in the treated dams with three doses of ZnO-NPs (5, 25, and 50 mg kg−1) as compared to the rat dams of the control group. The level of TNF-α was significantly increased in the offsprings from the rat dams treated with ZnO-NPs (25 and 50 mg kg−1). In the case of the offsprings and the dams treated with bZnO, the TNF-α level did not change compared to the control group.
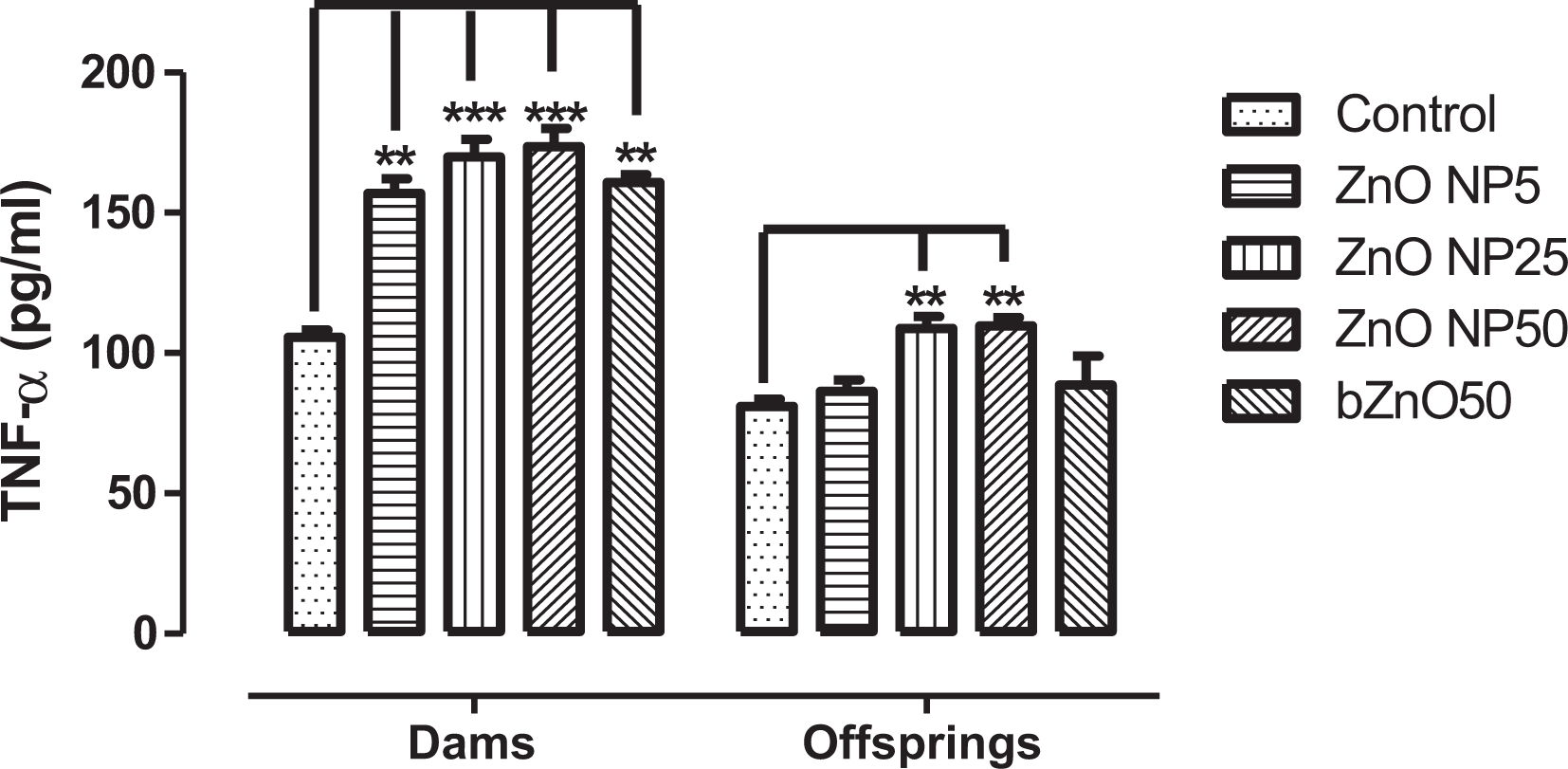
TNF-α level in serum among different groups after repeated oral gavaging of 5, 25, and 50 mg kg−1 bw of ZnO-NPs and 50 mg kg−1 bw of bZnO to Wistar rat dams for 19 days. Values are mean ± SEM (n = 6) and significantly different (**p < 0.01, ***p < 0.001) compared to control. TNF-α: tumor necrosis factor-alpha; ZnO-NP: zinc oxide nanoparticle; bZnO: bulk zinc oxide; SEM: standard error mean.
Effect of ZnO-NPs on the serum IL-1β level
There was a significant increase in the level of IL-1β in the dams treated with 50 mg kg−1 bw of ZnO-NPs (Figure 7). Also, the IL-1β levels significantly increased in the serum of offsprings from the dams treated with 50 mg kg−1 bw of ZnO-NPs consecutively for 19 days just after parturition.

IL-1β level in serum among different groups after repeated oral gavaging of 5, 25, and 50 mg kg−1 bw of ZnO-NPs and 50 mg kg−1 bw of bZnO to Wistar rat dams for 19 days. Values are mean ± SEM (n = 6) and significantly different (*p < 0.05, **p < 0.01) compared to control. IL-1β: interleukin-1 beta; ZnO-NP: zinc oxide nanoparticle; bZnO: bulk zinc oxide; SEM: standard error mean.
Effect of ZnO-NPs on the serum liver enzyme levels
The serum levels of AST and ALT from the dams as well as their offsprings increased significantly with the increasing dose of ZnO-NPs to the dams (Figure 8). No significant increase was observed in the rat dams treated with bZnO and their offspring compared to their corresponding control groups.

ALT and AST level among different groups after repeated oral gavaging of 5, 25, and 50 mg kg−1 bw of ZnO-NPs and 50 mg kg−1 bw of bZnO to Wistar rat dams for 19 days. Values are mean ± SEM (n = 6) and significantly different (*p < 0.05, **p < 0.01, ***p < 0.001) compared to control. ALT: alanine aminotransferase; AST: aspartate aminotransferase; ZnO-NP: zinc oxide nanoparticle; bZnO: bulk zinc oxide; SEM: standard error mean.
Effect of ZnO-NPs on the IgA
The IgA levels in the intestinal fluid were determined by sandwich ELISA. The level of IgA in the intestinal fluid was significantly increased in the rat dams treated with ZnO-NPs (5, 25, and 50 mg kg−1) or bZnO (50 mg kg−1) during the lactating period. The IgA levels in the intestinal fluid of offsprings whose dams were treated with 50 mg kg−1 ZnO-NPs increased significantly as compared to the control group offsprings (Figure 9).

IgA levels in intestinal fluid among different groups after repeated oral gavaging of 55, 25, and 50 mg kg−1 bw of ZnO-NPs and 50 mg kg−1 bw of bZnO to Wistar rat dams for 19 days. Values are mean ± SEM (n = 6) and significantly different (**p < 0.01, ***p < 0.001) compared to control. IgA: immunoglobulin A; ZnO-NP: zinc oxide nanoparticle; bZnO: bulk zinc oxide; SEM: standard error mean.
Histopathology change in liver and intestine of offspring
Normal histological structures were observed in the liver and small intestine of the control offspring rats (Figures 10 and 11(a)). Visible hepatocellular necrosis and collapsed central vein were observed in the liver sections of offspring whose dams were treated with ZnO-NPs (50 mg kg−1), as shown in Figure 10(b). However, there was no noticeable change in the liver and intestinal sections of offspring whose rat dams were treated with bZnO (50 mg kg−1), as shown in Figures 10 and 11(c). The intestinal villi and lamina propria were disrupted in the offsprings from the dams orally gavaged 50 mg kg−1 of ZnO-NPs consecutively for 19 days (Figure 11(b)).
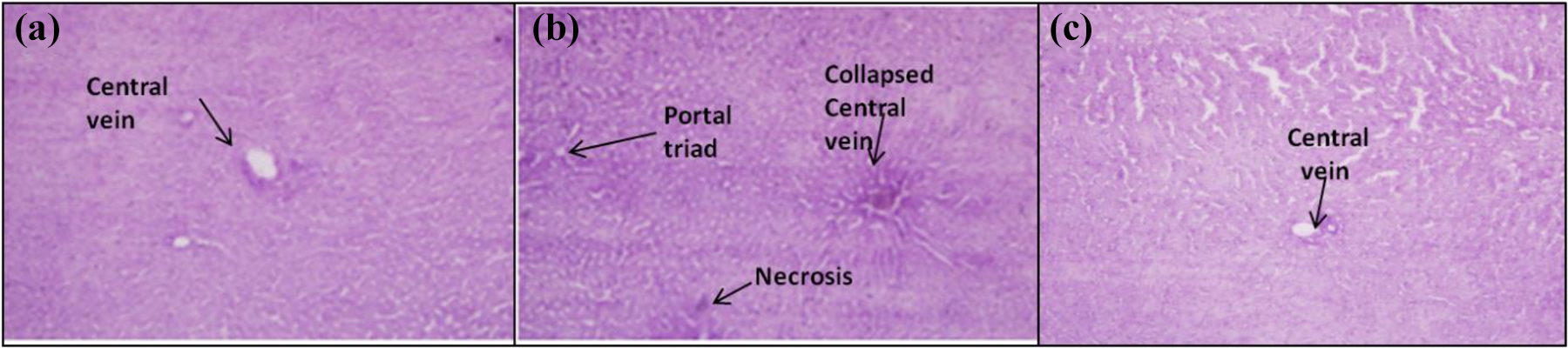
Representative histopathological images of Wistar rat offspring liver: (a) control, (b) 50 mg kg−1 bw of ZnO-NPs, and (c) 50 mg kg−1 bw of bZnO after 19 days consecutive oral gavaging to dams. H&E was used for histological staining (×400). ZnO-NP: zinc oxide nanoparticle; bZnO: bulk zinc oxide; H&E: hematoxylin and eosin.

Representative histopathological images of Wistar rat offspring small intestine: (a) control group, (b) 50 mg kg−1 bw ZnO-NPs, and (c) 50 mg kg−1 bw of bZnO after oral gavaging to dams for consecutive 19 days. H&E was used for histological staining (×400). ZnO-NP: zinc oxide nanoparticle; bZnO: bulk zinc oxide; H&E: hematoxylin and eosin.
Discussion
In the present study, the safety assessment of ZnO-NPs (5, 25, and 50 mg kg−1) and bZnO (50 mg kg−1) was investigated after postnatal exposure to the dams for 19 consecutive days. This study was carried out on the rat dams and their offsprings before the weaning period using the following endpoints: (1) body and organ weights; (2) concentration of zinc in milk, liver, small intestine, and feces; (3) CYP450 reductase in liver tissues; (4) TAC in blood serum; (5) cytokine production (TNF-α and IL-1B); (6) liver enzymes in blood serum; (7) IgA level in the intestinal fluid; and (8) histopathology for offspring tissues.
In this study, ZnO-NPs did not show a significant effect on the body weight gain of dams and their offsprings. Also, the body weight difference between the ZnO-NPs- and bZnO-treated dams as well as their offsprings did not change significantly. Further, there was no significant increase in the organ weight among the experimental groups except for the spleen weight of the rat dams treated with ZnO-NPs (50 mg kg−1). This is in agreement with the findings of Kumar et al. 43 who reported a significant increase in spleen weight after oral intubation of 50 mg kg−1 bw ZnO-NPs to Balb/c mice for 28 days consecutively. Indeed, the increased splenic weight may be associated with leukocytosis, extramedullary hematopoiesis. 44,45 The NPs enter the circulation system and then get absorbed in various tissues. Their pharmacokinetic rates including absorption, distribution, metabolism, and excretion vary, depending on the physicochemical properties (size, shape, surface, etc.). 17 –19 Our results showed that ZnO-NPs were absorbed by the small intestine and distributed to the liver and breast milk of dams. ZnO-NPs were absorbed more by the small intestine as compared to the bulk counterpart probably due to its extremely small size. Also, ZnO-NPs from the 25 mg kg−1 dams absorbed in the small intestine almost comparable to that of 50 mg kg−1-treated bZnO. Consequently, ZnO-NPs were transferred to the offsprings of the rat dams treated with ZnO-NPs (50 mg kg−1) via the breast milk. At a dose of 50 mg kg−1, ZnO-NPs transferred higher to the breast milk as compared to bZnO due to difference in size. Similar results were obtained by Zhang et al. using different size NPs. 34 Based on biologically based pharmacokinetic modeling during the lactation, Yoon and Barton 46 suggested that the extent of lactational exposure of the pups was often comparable or even higher than maternal levels for longer half-life compounds. Different NPs have been shown to transfer to offspring after oral administration to the lactating dams. 23,24 Furthermore, Jo et al. 26 showed that a significant amount of ZnO-NPs was distributed to the mammary tissue of the dams as well as to the kidneys of the rat offsprings after oral administration of 500 mg kg−1 bw to the dams. In our case, ZnO-NPs (50 mg kg−1) were mostly distributed to the liver of dams in significantly higher quantities as compared to the bZnO after oral administration for 19 consecutive days. Subacute oral exposure to 300 mg kg−1 of ZnO-NPs for 14 consecutive days showed that NPs were mainly found to be retained in the mouse liver. 40 We found that the milk-mediated distribution of ZnO-NPs (50 mg kg−1) to offspring’s liver was significantly higher than that of a comparable dose of bZnO. Most of the ZnO-NPs given to the dams or transferred to the offsprings were excreted through the feces at the end of our study. Matsumoto et al. 47 suggested that repeated oral gavage of NPs reached the GI tract as agglomerates and were mostly excreted via feces.
The total CYP450 reductase activity decreased significantly by 15.63% and 43.62% after oral gavaging of 25 and 50 mg kg−1 bw of ZnO-NPs to lactating rat dams, respectively. As shown in Figure 4, CYP450 decreased significantly in the rat dams treated with ZnO-NPs (25 and 50 mg kg−1 bw). The total liver CYP450 reductase decreased significantly in offsprings whose dams were treated with ZnO-NPs (50 mg kg−1). The literature review has shown that cytochromes P450 involved in drug metabolism can be inhibited by specific inhibitory potencies of different NPs. 48 Similarly, the activity of liver CYP450 enzymes was significantly lowered when ZnO-NPs were added in rat feed at concentrations above 100 mg kg−1 as compared to the control. 49 Further, the effect of ZnO-NPs on the biochemical parameters in lactating rats and their offsprings was measured. In this study, ZnO-NPs inhibited the oxidative defense system by decreasing the level of serum TAC in the rat dams and their offsprings, especially at the high dose (50 mg kg−1). The serum TAC level in 50 mg kg−1 of bZnO given dams significantly decreased compared to the control but remained higher compared to both 25 and 50 mg kg−1 of ZnO-NPs groups. Also, serum TAC in offsprings from the ZnO-NPs (50 mg kg−1)-treated dams was significantly lower than that of the offsprings from the control and the bZnO-treated groups. It appears that ZnO-NPs have induced the formation of reactive oxygen species (ROS) including hydrogen peroxide, hydroxyl radicals, and superoxide anions. The generated ROS have disrupted intracellular metabolic activities and antioxidant defense system. 50 As mentioned previously, low TAC could be indicative of oxidative stress or increased susceptibility to oxidative damage. 51 Similar results were obtained by Abass et al., 52 who found a significant decrease in serum TAC after oral administration of ZnO-NPs to rats for 4 weeks.
Cytokines have been regarded as the biomarkers of the NP immunotoxicity. 53 In our study, the levels of the serum cytokines (TNF-α and IL-1β) increased in the serum of rat dams and breastfed offsprings, especially at the high dose of 50 mg kg−1 ZnO-NPs. This is consistent with Abass et al. 52 who reported an increase in both TNF-α and IL-1β of rat serum after oral administration of ZnO-NPs for 4 weeks. The serum TNF-α level of the rat dams treated with 50 mg kg−1 of bZnO increased significantly but was lesser as compared to both 25 and 50 mg kg−1 ZnO-NPs groups. The higher level of cytokines in the ZnO-NPs group compared to the bulk counterpart signifies their more immune-toxic nature due to the high surface to volume ratio. The increased cytokines are in harmony with lowering the levels of antioxidant defense system and CYP450 reductase enzyme in the current investigation. This view is supported by several studies showing that CYP450 enzymes are downregulated during generalized inflammatory CYP450 enzymes. 49,54,55 This observation indicated that ZnO-NPs are involved in the inflammatory responses and the immune-cell differentiation process. As mentioned in Figure 6, all three doses of ZNO-NPs and bZnO caused an increase in the level of TNF-alpha in the rat dams. However, the level of TNF-alpha was increased in the offsprings whose dams were treated with a high dose of ZnO-NP- (50 mg kg−1) versus bZnO-treated dams and their offsprings. In Figure 7, the highest dose of ZnO-NPs (50 mg kg−1) caused an increase in the level of IL-1β in the rat dams and their offsprings. The increase in serum levels of aminotransferases (AST and ALT) was observed in ZnO-NPs-treated rat dams and their offsprings. Further, the AST level in 50 mg kg−1 of bZnO dams increased significantly than control, but it was lower than 25 mg kg−1 of ZnO-NPs-treated group. This is consistent with the findings of Moatamed et al. 56 These data reflected that the ZnO-NPs caused a breakdown of the cell membrane of hepatocytes leading to the release of these enzymes into the bloodstream. Thus, the increase in these enzymes is a traditional marker for liver injury. 47 These observations are similar to those reported previously. For example, 5-day oral subacute exposure to ZnO-NPs (10 mg kg−1) resulted in a remarkable increase in plasma ALT and AST enzymes in male rats. 37 Likewise, Abbasalipourkabir et al. 57 found that 10-day intraperitoneal injection of ZnO-NPs increased the levels of serum ALT and AST at the concentrations above 50 and 150 mg kg−1, respectively, in male rats. The ALT levels significantly increased in male offsprings of albino mice prenatally exposed to 100 mg kg−1 ZnO-NPs for 2 alternate days. 58 Fourteen-day repeated oral administration of ZnO-NPs (5–2000 mg kg−1) induced an increase in the level of serum blood AST and ALT, in an inverse dose-dependent relationship. Thus, the low dose of ZnO-NPs is higher toxic versus high dose, suggesting the importance of particle density rather than conventional mass concentration dose. 38 In this study, subacute oral exposure to ZnO-NPs at a higher dose (50 mg kg−1) resulted in histological lesions in the intestine and liver of breastfed offsprings. Similar histological distortion was observed in the mouse liver exposed to repeated oral treatment with ZnO-NPs (300 mg kg−1) for 14 consecutive days. 40 The injury to the small intestine was supported by the abrupt increase in sIgA of the intestinal fluid of both dams and offsprings. This is in agreement with the findings of Abbasi-Oshaghi et al. 59 who reported the disruption and loosening of intestinal villi after 28-day oral gavaging of ZnO-NPs to rats. In the 50 mg kg−1 of bZnO-treated dams, IgA increased significantly but remained much lower as compared to both 25 and 50 mg kg−1 of ZnO-NPs-treated dams. The histopathological examination depicted the disruption of intestinal lamina propria and collapsed central vein of the liver in the offsprings, revealing the milk-mediated toxicity of ZnO-NPs at a dose of 50 mg kg−1. It was observed that seven of the nine villi counted in ZnO-NPs (50 mg kg−1) group’s offsprings were loosened whereas all nine were intact in the offsprings of bZnO-treated dams. The occurrence of focal necrosis in the case of liver from 50 mg kg−1 ZnO-NPs-treated group is in accordance with earlier report. 38 We found that damage to the liver in 50 mg kg−1 of ZnO-NPs group is co-related by a significant increase in the liver function enzymes (ALT and AST). Likewise, ZnO-NPs caused alteration of CYP450 enzymes that occurred due to damage in the liver. It is consistent with the earlier report of Tang et al. 49 The increase in anti-inflammatory cytokines is associated with the generation of free radicals and damage to the liver tissue. It has been reported that ZnO-NPs induce ROS generation, oxidative DNA damage, and also caused cell death through apoptosis in human liver cells. 40 Furthermore, oxidative stress and ROS production are referred to as the most discussed reason for the toxicity of NPs. 56,60 –62 The increase in anti-inflammatory cytokines from the plasma of Sprague-Dawley rats was also reported after damage to the liver on intranasal administration of ZnO-NPs. 63 Our findings also suggest that histopathological changes in the liver as a result of oxidative stress lead to alteration in various biochemical parameters.
Conclusion
In our study, ZnO-NPs were transferred to the breast milk after repeated oral administration to lactating rats (25 and 50 mg kg−1 bw) for 19 days starting with parturition. ZnO-NPs were distributed to the liver of dams and offsprings after absorption in the small intestine. The damage to the small intestine is evident from an increase in IgA of intestinal fluid. Exposure of ZnO-NPs (25 and 50 mg kg−1 bw) leads to the immune response by increasing serum cytokines and liver enzymes of the dams and their offsprings. The decrease in TAC and CYP450 reductase of treated rat dams (25 and 50 mg kg−1 bw of ZnO-NPs) and offsprings from 50 mg kg−1 of ZnO-NPs group revealed the masking of defense mechanisms by ZnO-NPs. Also, milk-mediated transfer of ZnO-NPs from treated rat (50 mg kg−1 bw) resulted in gross pathological changes to the small intestine and liver of offsprings. The results obtained demonstrated a direct effect of ZnO-NPs on lactating dams and an indirect milk-mediated exposure to offsprings, contributing to an increase in the knowledge of the risk assessment.
Moreover, the exposure limits of ZnO-NPs during lactation also need to be specifically developed to avoid the risk to the mother and offsprings. It is mandatory to address the neurological manifestations of ZnO-NPs on breastfed offsprings to understand the behavioral effect. Further, the study of the size-dependent effect for ZnO-NPs use during lactation will be highly helpful for checking the size constraint in assessing safety to breastfed offsprings.
Footnotes
Acknowledgment
The authors are thankful to the support of the Indian Institute of Technology (IIT), New Delhi (India), for the ICP-MS facility.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was financially supported by the Indian Council of Agriculture Research (ICAR)-National Dairy Research institute, Karnal 132001, Haryana, India. Also, the University Grant Commission, India, provided a contingency grant [Grant No. F1-17.1/2015-16/NFST-2015-17-ST-JAM-507] as financial support to Ahmad Hussain for the research.
