Abstract
Diabetes increases the possibility of germ cell damage, hypogonadism, and male infertility. Diabetic condition negatively impacts zinc (Zn) and selenium (Se) levels in the body. Zn and Se are among the most important trace elements involved in the regulation of redox reaction, antioxidants enzymes activities, and DNA expression in a germ cell. The present study aimed to elucidate the combined effects of Zn and Se treatment on diabetes-induced germ cell damage in male Sprague Dawley rats. Type 1 diabetes was induced by the single intraperitoneal (i.p.) injection of streptozotocin (55 mg/kg). Zn (3 mg/kg, i.p.) and Se (0.5 mg/kg, i.p.) were administered daily for 8 consecutive weeks. All the animals were provided with normal feed and water throughout the study. The effects on germ cell damage were evaluated by body weight, feed-water intake, organ weight, sperm count, motility, sperm head morphology, biochemical analysis, histology, immunohistochemistry, halo assay, germ cell comet assay, testes terminal deoxynucleotidyl transferase deoxyuridine triphosphate nick end-labeling (TUNEL) assay, sperm TUNEL assay, serum protein pattern analysis, and subcellular analysis using transmission electron microscopy. Further, the expressions of nuclear erythroid-derived related factor 2, catalase, glutathione peroxidase 4, and glutathione peroxidase 5 were carried out to ascertain the mechanism of protection. The present results demonstrated that 8 weeks combined treatment of Zn (3 mg/kg, i.p.) and Se (0.5 mg/kg, i.p.) reduced diabetes-induced germ cell damage. This study further highlighted that Zn and Se combination treatment might be a better strategy for the germ cell protection in diabetes and deserve further investigation.
Introduction
Diabetes is a complex metabolic disorder with several etiologies characterized by persistent elevation of blood glucose level and alterations in carbohydrate, fat, and protein metabolism along with complete or relative insulin deficiency. 1 The diabetic condition interferes with the normal functioning and structural organization of different organs including the male reproductive system. 2 Literature indicated that type 1 diabetes disrupted the proper development and functioning of reproductive organs such as testes, epididymis, and prostate of young individuals at reproductive age. 3 Individuals who have type 1 diabetes have been reported with impaired spermatogenesis, decreased sperm production, and sperm quality. 4 Diabetic condition induced metabolic disturbances and increased the production of reactive oxygen species (ROS). 5 ROS disrupted the normal biochemical and cellular processes and induced insufficiencies in antioxidant defense enzymes and vitamin. 6 It has been reported that in diabetic conditions, ROS induced inflammation, apoptosis, and DNA damage due to the downregulation of DNA repair enzymes. 7,8 Further, ROS induced lesions in pyrimidine and purine bases in DNA that can alter various proteins and metabolic enzymes involved in cellular defense mechanisms. 9 A recent report by Illum et al. suggested that DNA methylation-based epigenetic inheritance (intergenerational and transgenerational) happens during the gametogenesis and in the zygote period due to the associated metabolic disease phenotype. 10 It also altered the electrolyte balance by decreasing absorption and increasing excretion of essential trace elements such as zinc (Zn) and selenium (Se). 11 The loss of Zn through urine and from the whole body during diabetic conditions critically affects the physiological process. 12 Plasma Se concentration significantly affected individuals with type 1 diabetes, and supplementation of the same may have future implications in the management of diabetes. 13
Zn plays a crucial role in testicular sperm production and male fertility. 14 Zn is associated with more than 300 biochemical reactions and regulatory functions in the body. 15 Zn is one of the most crucial micronutrients required for homeostasis, physiological functions of testes, and proper regulation of transcription factor nuclear erythroid-derived related factor 2 (Nrf-2). 16 Decreased testosterone level, Leydig cell abnormality, and different testicular pathophysiology are among the critical effects of Zn deficiency during reproductive age. 17 Supplementation of Zn during diabetes plays a beneficial role in maintaining glutathione peroxidase 5 (GPX5) for epididymal sperm maturation in rats. 18 Zn has an essential role in the synthesis, storage, and secretion of insulin from pancreatic β-cells. 11 Hypozincemia during diabetes has a more significant impact on sperm quality, testicular physiology, and reproductive health. 19 Se is a well-known antioxidant involved in several antioxidant processes, catalyzing detoxification enzymes (selenoproteins) activity, crucial for developmental and reproductive physiology of testes, epididymis, and prostate. Se is essential for healthy human and animal health, deficiency of which may lead to a decrease in quality sperm production, sperm maturation in the epididymis, induction of male infertility, and testicular as well as prostate cancers. 20 Se has been proved to be effective in decreasing the chemotherapy-induced oxidative stress and DNA damage in rodents. 21 It has been reported that dietary Se can affect apoptosis of germ cells by regulating the messenger RNA expression and cell cycle-related genes in the testes during spermatogenesis of roosters. 22 It has been reported that many Korean patients with inflammatory bowel disease are having Zn, Se, and vitamin D deficiency in the serum. 23 It has also been reported that individuals with chronic myofascial pain exhibited low concentrations of Zn and Se and are likely to manifest inflammatory conditions. 24 Serum concentrations of Zn, Se, and prolactin are low in critically ill children as detected within 72 h of admission in a critical care unit. 25 Combination treatment of Zn, Se, and vitamin E exhibited a significant effect in comparison to individual effect of Zn, Se, and vitamin E in controlling hyperglycemia in streptozotocin (STZ)-induced diabetic rats. 26 It has been shown that the combination of Zn + Se showed more protective effects than per se Se in valproic acid-induced hepatotoxicity in Wistar rats. 27 Table 1 summarizes the various reports on the protective effect of Zn and Se in different experimental studies alone as well as in combination. Considering the significance of germ cell damage and the increased incidences of diabetes in humans, the present intervention study was undertaken with the combination treatment of Zn and Se to explore the possible ameliorative effects. The results demonstrate the protective nature of Zn and Se against diabetes-induced biochemical changes, oxidative stress, histological and subcellular injury, DNA damages, apoptosis, altered protein expressions of Nrf-2, catalase, glutathione peroxidase 4 (GPX4), and GPX5 in the rat.
Studies carried out in different test systems with Zn, Se and Zn+Se at different doses, routes, duration along with the observations.

Zn: zinc; Se: selenium; SD: Sprague Dawley; i.p.: intraperitoneal; ZnCl2: zinc chloride; i.v.: intravenous; Na2SeO3: sodium selenite; s.c.: subcutaneous; p.o.: per os; b.w.: body weight.
Materials and methods
Animals
The animal experimentation protocol was approved by the Institutional Animal Ethics Committee (IAEC) vide animal protocol approval no. IAEC 16/52 and the experiment on animals was performed based on the Committee for the Purpose of Control and Supervision of Experiments on Animals (CPCSEA) guidelines, Government of India. This experiment was performed on male juvenile Sprague Dawley rats (4 weeks old) procured from Central Animal Facility of NIPER, S.A.S Nagar, Punjab, India. All the procured animals were kept under controlled conditions at room temperature (22 ± 2°C) with 50 ± 10% humidity and 12-h light and 12-h dark cycles. Standard laboratory animal feed and water were provided ad libitum. Animals were acclimatized to the experimental conditions for 1 week before the commencement of the experiment. Animals were euthanized after 8 weeks per the study plan to evaluate various parameters (Figure 1).

Pictorial representation of the experimental study design depicts four different treatment groups, dosing schedule, subsequent time of sampling, and endpoints of evaluation. The different groups are control, Zn + Se (Con.), diabetic, and Dia + (Zn + Se). The Zn + Se treatment was given i.p. to the Zn + Se (Con.) and Dia + (Zn + Se) groups for 8 consecutive weeks (4 weeks before diabetic initiation and 4 weeks after diabetic induction). The diabetes was induced at the end of the fourth week during the study. The experimental animals were sacrificed at the end of the eighth week. Zn: zinc; Se: selenium; i.p.: intraperitoneal.
Chemicals
STZ (CAS# 18883-66-4) was purchased from MP Biomedicals, LLC (BP, Illkirch, France). Zinc sulfate heptahydrate (CAS# 7446-20-0) and sodium selenite (CAS# 10102-18-8), hematoxylin, eosin, ethidium bromide (EtBr), and SYBR green I were purchased from Sigma-Aldrich (St Louis, Missouri, USA). Biochemical reagent kits such as alkaline phosphatase (ALP), blood urea nitrogen (BUN), serum glutamic-pyruvic transaminase (SGPT), serum glutamic oxaloacetic transaminase (SGOT), and albumin were purchased from ERBA Diagnostics (Mannheim, Germany). Testosterone enzyme-linked immunosorbent assay (ELISA) kit was purchased from DBC Diagnostics Biochem (Canada), terminal deoxynucleotidyl transferase deoxyuridine triphosphate nick end-labeling (TUNEL) assay kit from Calbiochem (Inc. San Diego,California, USA), immunohistochemistry (IHC) from Novolink, Leica Biosystems (UK), primary and secondary antibodies from Santa Cruz (Dallas, Texas, USA), and Zn quantification kit from Abcam Inc. (UK) was purchased. All other reagents were purchased from Sigma-Aldrich.
Experimental design
All the animals were randomly divided into four different groups as follows: group 1, normal control (control; n = 5); group 2, Zn + Se control (Zn + Se (Con.); n = 5); group 3, diabetic (diabetic; n = 5); and group 4, diabetic + (Zn + Se) (Dia + (Zn + Se); n = 5). Per se effects of Zn and Se groups were not considered in the present experimental design because of their already reported several beneficial effects against different toxicants induced germ cell damage from our laboratory 16,29 as well as studies conducted by others. 31,44,45 All the groups were kept under the study for 8 weeks with normal feed and water. Type 1 diabetes was induced by a single dose of STZ (55 mg/kg, intraperitoneal (i.p.)) in chilled citrate buffer having pH 4.5. 18 After 48 h, fasting blood glucose levels were measured by the Arkray blood glucose monitoring kit (ARKRAY Factory, Inc., Shiga, Japan). Animals with blood glucose level ≥ 250 mg/dl were considered as diabetic in the present experiment. The control group was provided with standard commercial feed and water for the entire study period. The Zn + Se control was treated with Zn 3 mg/kg per day in the form of zinc sulfate heptahydrate i.p. and Se 0.5 mg/kg per day in the form of sodium selenite i.p. for 8 consecutive weeks. The fourth diabetic + (Zn + Se) group was considered diabetic and treated with Zn 3 mg/kg per day in the form of zinc sulfate heptahydrate i.p. and Se 0.5 mg/kg per day in the form of sodium selenite i.p. for 8 consecutive weeks (4 weeks before diabetic initiation and 4 weeks after diabetic induction). 16,46 Distilled water was used for the preparation of the drug solution. Bodyweight, water intake, feed intake, and survival rate were recorded during treatment. All the animals were euthanized after 24 h of last dose administration. Organ weight and photomicrographs/appearances were recorded at gross necropsy. Plasma, serum, isolated organs, and tissue samples were processed and analyzed for different end points. The schematic experimental design is depicted in Figure 1.
Determination of morphometric parameters
Experimental animals were euthanized 24 h after the last dose was administered, immediately paired testes and epididymis were collected, washed (with chilled phosphate-buffered saline (PBS)/saline), soaked with smooth clothes, and weights (in paired) were measured using weighing machine and expressed in grams. 47
Biochemical parameters
Before euthanization, blood was collected via retro-orbital, and then serum and plasma were separated. Testes and epididymis were dissected out, and fatty material was removed, appropriately washed with chilled PBS, and weighed. Testes were immediately processed for biochemical analyses.
Determination of lipid peroxidation
Testes were rinsed with chilled PBS, minced, and the tissue was homogenized in phosphate buffer (pH 7.4) with ethylenediaminetetraacetic acid (EDTA) and centrifuged at 700 × g for 10 min at 4°C. Then 100 µl of supernatant, 100 µl of 8.1% sodium dodecyl sulfate (SDS), 750 µl of 20% acetic acid, and 750 µl of 0.8% aqueous solution of thiobarbituric acid (TBA) were taken in test tubes as performed previously. 48 The volume was made up to 2 ml with distilled water, mixed thoroughly, and heated at 95°C for 60 min. After cooling, the contents were centrifuged at 10,000 r/min for 10 min at room temperature. The absorbance of the supernatant was taken at 532 nm. 1,1,3,3-Tetra methoxy propane was used as the external standard. Results were expressed as nanomole malondialdehyde (MDA)/mg of the tissue protein.
Determination of GSH level
Reduced glutathione (GSH) was estimated based on the procedure as specified. 49 Then 100 mg testes tissues were rinsed in ice-cold physiological saline, minced, and homogenated in previously prepared 1 ml of phosphate buffer with a pH of 7.4 containing 3 mM EDTA; the equal volume of the separated tissue was homogenated and 5% sulfosalicylic acid were added, vortexed properly, and kept in the ice bath for 30 min. Then the tissue samples were centrifuged, and the supernatant was collected. GSH contents were measured using Ellman’s reagent 5,5-dithiobis (2-nitro-benzoic acid) solution. GSH was used as the standard, and the standard curve was made as a reference for the calculation of GSH levels. Results were expressed in micromoles of GSH per milligram of tissue protein.
Determination of catalase
Catalase activity assay was carried out as described previously. 50 Briefly, 0.2 ml tissue homogenate (kept in ice) was taken with 1 ml of 65 µM hydrogen peroxide (H2O2) in 6.0 mM phosphate buffer (pH 7.4) for 60 s. Control reaction was prepared with no enzyme (blank 1) and no enzyme/no substrate (blank 2). Both the samples and control reactions were stopped by adding 1 ml of 32.4 mM ammonium molybdate. Absorbance was taken at 405 nm by a spectrophotometer. Catalase activity was expressed in kilounits per liter.
Determination of ALP, BUN, SGPT, SGOT, albumin, and total protein
ALP, BUN, SGPT, SGOT, albumin, and total protein were estimated according to the manufacturer’s instructions (ERBA Diagnostics).
Determination of testosterone level
Serum testosterone levels were estimated by commercially available ELISA kit according to the manufacturer’s instructions (DBC Diagnostics Biochem).
Determination of serum Zn and testicular Zn and Se level
Serum Zn level was estimated using the Zn quantification kit (fluorometric, ab176725, Abcam) at Ex/Em 485/525 using a multi-well plate fluorescent reader per the manufacturer’s instructions and as described previously. 51 The relative contents of Zn were expressed as references to the standard curve, and results were expressed accordingly. Testicular Zn estimations were carried out by APHA 3125 B method at Punjab Technological Incubator, Mohali, Government of Punjab, India. 52 Briefly, frozen testes tissues were brought to room temperature, and 100 mg of tissue sample was weighed. Testes tissues were immersed with nitric acid and hydrogen peroxide in test tubes and heated to 100°C for proper digestion of the same. The samples appeared transparent after complete digestion, and the final sample solution was made up to 10 ml with double-distilled water.
Determination of sperm count, motility, and head morphology
After the animals were euthanized, cauda epididymis were collected and placed in a petri plate containing 1 ml of Hanks’ balanced salt solution (HBSS) medium at room temperature as described previously. 18 Briefly, the cauda epididymis was cut into pieces and allowed the sperms to swim out. The suspensions were collected and centrifuged at 1000 r/min for 3 min at room temperature. The samples were diluted per requirements; the number of sperms and the number of motile sperms were quantified by using a hemocytometer. Results were expressed as the number of sperm per milliliter of solution and the number of motile sperm per milliliter of solution taken, respectively. For sperm head morphology, 0.5 ml of each sample containing the sperm were added with an equal volume of 2% eosin and allowed 1 h for the proper stain of sperm. Approximately 2–3 drops of the stained solution were taken, made smear, air-dried, and fixed with absolute methanol (high-performance liquid chromatography grade). Smear slides were washed with distilled water three times each for 5 min and allowed to air dry. Approximately 200 sperms per animal were investigated to determine the morphological abnormalities under the oil immersion method (100×, Olympus microscope, Tokyo, Japan). Sperm head morphology scoring was done and classified as percent normal, quasi-normal, and grossly abnormal, as mentioned by Gatimel et al. 53 Sperm missing superior sensitive part and/or posterolateral area of the acrosome was considered as quasi-normal heads. Sperm with collapsed/crashed, triangular, and spherical heads with extremely deformed acrosome caps and nuclei were estimated as grossly abnormal sperm head.
Quantification of histological parameters
Testes and epididymis were collected after euthanization, washed with chilled saline, soaked by cloth, and kept in 10% formal saline. After 48 h, formal saline was replaced with fresh and kept for 1 day. Tissues were dehydrated in increasing concentrations of ethanol (70%, 80%, 90%, and 100%), kept overnight in 100% ethanol with gentle shaking. Next day morning, tissues were processed further with mixed ethanol and xylene (1:1), 100% xylene, liquid paraffin, and finally embedded in paraffin. Tissue sections of 5 µm thick were taken by semi-automated microtome (LEICA RM 2145; Leica Biosystems, Watzlar Germany), mounted on glass slides coated with Mayer’s albumin, and dried overnight in a hot air oven. Tissue sections were deparaffinized with xylene and rehydrated with alcohol and water. The rehydrated sections were stained with hematoxylin and eosin, mounted with dibutylphthalate polysterene xylene mounting media, dried in a hot air oven, and examined under the microscope at different magnifications. Testes sections were evaluated for structural changes as compared to control, and the number of seminiferous tubules per unit area was counted for all the groups. The total of 20 images per animal (total animals in each group, n = 5) was taken into consideration and compared. Further, Johnson’s score was evaluated by considering the criteria of calculation such that 1 stands for no seminiferous epithelial cells and tubular sclerosis and 10 indicates full spermatogenesis. 47,54
Evaluation of DNA damage by halo assay
It has been reported that the nuclear halo assay is a simplified, rapid, reliable, and inexpensive method to detect and quantify the DNA damage breakage induced by ionizing radiation and chemical agents. 55 The present halo assay was performed as described previously 56 ; briefly, a small piece of testis was placed in 1 ml microcentrifuge tube containing cold HBSS 800 µl, 20 mM EDTA (100 µl), and 10% dimethyl sulfoxide (100 µl). The tissue was chopped into small pieces and passed through a syringe to make a uniform single-cell suspension. The suspension was centrifuged at 1000 r/min for 1 min, 5 µl of this was suspended in 95 µl of 0.5% low melting point agarose and layered on slides (frosted precoated with 1% normal melting point agarose), covered with cover glass, formed a microgel, and was placed on a glass slab at 4°C for 5 min. Cover glasses were removed, brought to room temperature; and another layer of 1% LMPA 100 µl was added, the cover glass was placed over it and kept at 4°C for 5–10 min. After removing the cover glass, the slides were dipped in freshly prepared lysis solution (2.5 M sodium chloride (NaCl), 2 mM EDTA, 10 mM Tris pH 10.0, and 1% triton X-100) for 2 h at 4°C. After lysis, the slides were incubated with 0.3 N sodium hydroxide (NaOH) (alkaline medium) for 20 min. Then the slides were washed with chilled distilled water and stained with EtBr. Randomly 100 cells were quantified per slide under the microscope and quantified by ImageJ software, Java 1.8.0_60 (64-bit), NIH, USA. 57 The damaged cells were classified as mild, moderate, and extensive.
Evaluation of DNA damage by comet assay
Comet assay is a versatile, relatively simple, and sensitive gel electrophoresis-based method used to measure DNA damage in single cells. The comet assay was performed as described previously with some modifications. 54,58 A single-cell suspension of 5 µl from each sample was mixed with 95 µl of 0.5% LMPA and layered on the precoated frosted slide (1% NMPA). The cover glass was placed to form a uniform single-cell layer (kept at 4°C for 5 min). Over the slides, after removing the cover glass, 100 µl of 1% LMPA was added and covered with a cover glass. After allowing for 5–10 min at 4°C, the slides were then dipped in lysis solution (2.5 M NaCl, 2 mM EDTA, 10 mM Tris hydrochloric acid buffer pH 10.0, 1% Triton X-100, and 10% DMSO) at 4°C for 24 h. After lysis, the slides were washed three times with cold distilled water, placed in horizontal electrophoresis comet tank (CSL-COM 20; Cleaver Scientific, Rugby, UK), and DNA was allowed to unwind for 20 min in alkaline electrophoresis solution containing 300 mM NaOH and 1 mM EDTA (pH >13.0). The cell suspension was allowed to the electric field at 0.6 V/cm, 300 mA for 30 min. After electrophoresis, DNA was neutralized with the neutralizing buffer. Slides were washed with cold distilled water and stained with SYBR green 1 (1:10,000 dilutions). Slides were rinsed with distilled water and cover glasses were placed over it and then kept in a humidified chamber. Nuclear DNAs were visualized using the AXIO Imager M1 fluorescence microscope (Carl Zeiss, Oberkochen, Germany). Images were captured and scored with comet imager V.2.0.0 software. Two slides were prepared for each treatment and scored randomly blind selection with coded slides. Results were expressed in terms of tail length (TL) in µm of migrated DNA, olive tail movement in arbitrary unit, % tail DNA in the comet tail, and tail moment (TM) 31 in arbitrary unit. Damaged parts of the gel, debris, edges, overlapped, giant, and comet without distinct head were not considered for the analysis.
Determination of apoptotic cells in testes and sperm by TUNEL assay
TUNEL assay was performed to determine the DNA fragmentation according to the manufacturer’s instructions (Calbiochem, Oncogene research product). 59 TUNEL positive cells and total cells were observed under a fluorescent microscope (Carl Zeiss). The TUNEL positive cells were expressed as a percentage of total cells.
Evaluation of protein expression by IHC
IHC evaluation was carried out as per the manufacturer’s instructions (Novolink polymer detection kit, Leica Biosystems, Germany) and as described previously.
60
Paraffin-embedded tissues of testes of all the groups were cut into 5 µ thick sections, placed on precoated poly-
Evaluation of serum protein distribution pattern by gel electrophoresis
Serum protein electrophoretic behavior was determined as described by Pant et al. with some modifications. 61 Briefly, vertical polyacrylamide gel electrophoresis at pH 8.6 with Mini power pack (Bio-Rad Laboratories, Hercules, California, USA) at 120 V at room temperature.
Evaluation of protein expression by Western blot analyses
Western blot analyses were performed as described previously. 62 Briefly, protein samples were resolved on 8–14% sodium dodecyl sulfate-polyacrylamide gel electrophoresis, transferred to nitrocellulose membrane (Pall Corporation, Port Washington, New York, USA) and analyzed with antibodies against Nrf-2, catalase, GPX4, GPX5, and β-actin rabbit polyclonal immunoglobulin G. Then incubated with horseradish peroxidase-conjugated secondary antibody and visualized by luminal reagent (Santa Cruz Biotechnology, Inc.). Images were captured by X-ray film (care stream, Kodak films, Rochester, New York, USA) methods. The protein quantification was performed by ImageJ software.
Subcellular evaluation of testes by TEM analyses
Fresh and frozen testes tissues were thawed and fixed with 2.5% glutaraldehyde and further cut into small pieces of size 1 mm3 as described previously. 63 The sections were transferred to fresh 2.5% glutaraldehyde for 2 h with gentle shaking and then kept in overnight at 4°C. The next day, the slices were washed in PBS (pH 7.2) for 10 min each three times. The sections were fixed in 1% osmium tetroxide for 2 h on the shaker. Then the tissues were washed with PBS (pH 7.2) for 10 min each three times. The tissues were dehydrated with gradient ethanol in increasing order. After dehydration, the specimens were embedded with the gradient increasing order of ethanol and resin as 3:1, 2:2, 1:3, 100% and 100% for 10 min each and transferred to fresh 100% resin and kept in an oven at 45°C for 12 h and polymerized at 65°C for next 24 h. The resin-embedded blocks were cut into ultrathin sections using ultramicrotome and stained with uranyl acetate. The images were captured with the help of a transmission electron microscope (TEM; model TF-20; FEI, Hillsboro, Oregon, USA) in NIPER, S.A.S Nagar, Punjab, India.
Statistical analyses
All the results were expressed as mean ± standard error of the mean for each group. Statistical analyses were performed using Graph Pad Prism 6 statistical software. Significance of difference among multiple groups was evaluated by using one-way analyses of variance, and post hoc analyses were performed with Tukey’s test. The value of p ≤ 0.05 was considered statistically significant.
Results
Observations during treatment
The bodyweight of rats in control and Zn + Se groups was increased in the course of the experiment and at the end of the treatment. The diabetic group body weight significantly (p < 0.001) decreased as compared to the control group. Treatment with Zn and Se significantly (p < 0.05) restored the bodyweight at the end of treatment as compared to the diabetic group (Figure 2(a), Supplementary Figure 1A). The percent survival of the animal during the entire course of treatment is depicted in Supplementary Figure 1B.
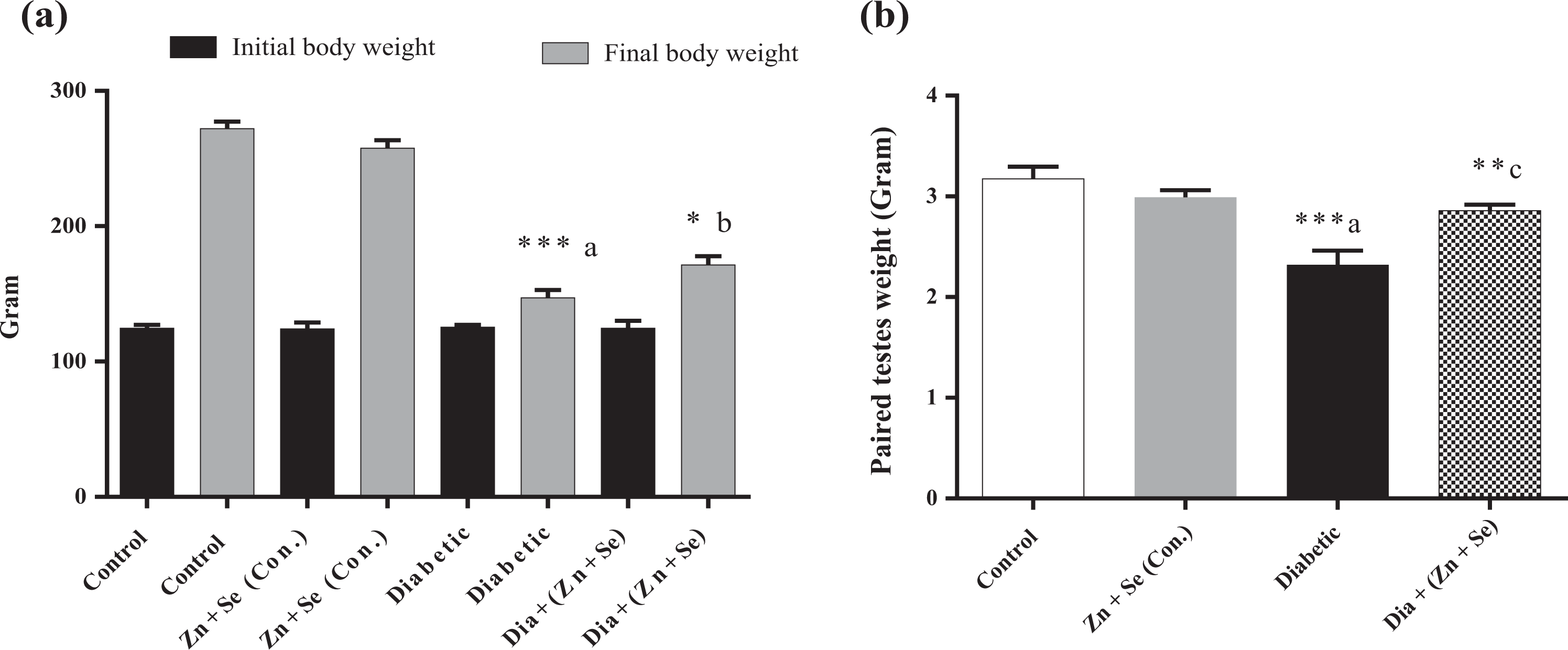
Histogram (a) depicts the effect of Zn + Se treatment on the body weight at the initial and final (at the end of eighth week), and (b) paired testes weight were recorded. All the values are shown as mean ± SEM (n = 5). *p < 0.05, **p < 0.01, ***p < 0.001, inset (a) versus control and inset (c) versus diabetic. Zn: zinc; Se: selenium; SEM: standard error of the mean.
Effect of Zn and Se on feed and water intake
The feed intake in the diabetic group was significantly (p < 0.001) decreased as compared to control, and diabetic animals treated with Zn + Se restored the same at the end of eighth week of treatment (Supplementary Figure 1C). Interestingly, high water intake, which is usually observed in diabetic animals, could not be significantly restored in diabetic animals treated with Zn + Se at the end of the eighth week of treatment (Supplementary Figure 1D).
Observations during gross necropsy
Effect of Zn and Se on testes and epididymis
The testes and epididymis weights were significantly (p < 0.001) decreased in the diabetic group. However, the treatment of Zn + Se significantly (p < 0.01) restored the same (Figure 2(b) and Supplementary Figure 1E).
Observations after gross necropsy
Effect of Zn and Se on biochemical parameters
The final fasting blood glucose levels in the diabetic group significantly (p < 0.001) increased as compared to control, and diabetic animals treated with Zn + Se showed significant (p < 0.001) decrease in the blood glucose levels at the end of 8 weeks of treatment (Figure 3(a)). The Zn level in the serum of diabetic animals was significantly (p < 0.05) decreased, which is significantly (p < 0.05) restored with treatment at the end of 8 weeks (Supplementary Figure 2A). Further, the testicular Zn levels decreased in diabetic animals and Zn + Se treatment increased the same, but the testicular Se level decreased significantly (p < 0.01), and treatment with Zn + Se increased the Se levels in a significant (p < 0.05) way (Supplementary Figure 2B and C). The serum testosterone level was decreased significantly (p < 0.01) in the diabetic group, while diabetic animals treated with Zn + Se restored the same but statistically insignificant (Figure 3(b)). The MDA level was increased in the diabetic group as compared to control, and treatment with Zn + Se decreased the same (Figure 3(c)). The levels of glutathione were decreased in the diabetic group as compared to the control and treatment of Zn + Se did not restore the same (Figure 3(d)). SGPT levels were significantly (p < 0.001) increased diabetic animals as compared to control, while Zn + Se treatment restored the same, which was statistically insignificant (Figure 3(e)), SGOT levels significantly (p < 0.05) increased in the diabetic group as compared to the control group and treatment with Zn + Se to diabetic animals significantly (p < 0.01) reduced the SGOT (Figure 3(f)). Similarly, the BUN levels significantly (p < 0.05) increased in diabetic animals as compared to the control group, but treatment with Zn + Se decreased the same but statistically insignificant (Figure 3(g)). Further, ALP levels were significantly (p < 0.001) increased in the diabetic group, while treatment of Zn and Se significantly (p < 0.01) decreased the same (Figure 3(h)). The antioxidant enzyme catalase decreased in diabetic animals, which was increased with Zn + Se treatment (Figure 3(i)).

Effect of Zn + Se treatment on biochemical parameters (a) initial and final fasting blood glucose, (b) serum testosterone levels, (c) MDA, (d) GSH, (e) SGPT, (f) SGOT, (g) BUN, (h) ALP, and (i) catalase activity. All the values are expressed as mean ± SEM (n = 5). *p < 0.05, **p < 0.01, ***p < 0.001, inset (a) versus control and inset (c) versus diabetic. Zn: zinc; Se: selenium; MDA: malondialdehyde; GSH: reduced glutathione; SGPT: serum glutamic-pyruvic transaminase; SGOT: serum glutamic oxaloacetic transaminase; BUN: blood urea nitrogen; ALP: alkaline phosphatase; SEM: standard error of the mean.
Effect of Zn and Se on sperm number, morphology, and motility
The cauda sperm count has significantly (p < 0.001) decreased in diabetic animals, and Zn + Se treatment increased the same in a significant (p < 0.05) manner. The number and the motility of the sperm significantly (p < 0.001) decreased in the diabetic group, while Zn + Se treatment to diabetic animals increased both sperm motility and sperm count (Figure 4(a) to (c)). Further, the diabetic group has significant (p < 0.001) abnormal sperm count, Zn + Se treatment significantly (p < 0.001) reduced the abnormal sperm count as compared to diabetic animals (Figure 4(d)).

Representative photomicrographs (×100) of sperm head abnormality (a). Histogram (b) illustrating the percent sperm motility, (c) sperm count and (d) sperms expressed in percent and abnormality classified as normal, quasinormal, and grossly abnormal. All the values are shown as mean ± SEM (n = 5). *p < 0.05, **p < 0.01, ***p < 0.001, inset (a) versus control and inset (c) versus diabetic. SEM: standard error of the mean.
Effect of Zn and Se on histology of testes and epididymis
The histology of testes tissue in diabetic animals revealed seminiferous tubule atrophy, Leydig cell abnormality, perturbations in spermatogenic cells, Sertoli cell abnormalities, endothelial cell swelling, germ cell shedding, and the reduced layer of mature cells (Figure 5). The epididymal histology of caput and cauda revealed several damages, such as disruption of cells in the membrane, change in the density of sperms, and interstitial abnormalities (Supplementary Figure 3A and B). The number of seminiferous tubules significantly (p < 0.001) increased in the diabetic group. Zn + Se treatment resumed the normal seminiferous tubule morphology, as observed in the control group (Figure 5(b)). Furthermore, Johnson’s score significantly (p < 0.001) decreased in the diabetic group. Zn + Se treatment resumed to the normalcy as observed in the control group (Figure 5(c)).

Effect of diabetes and diabetes treated with Zn + Se on the histological perturbations in testes. (a) Representative photomicrographs (×40) of testes histology of all the groups. Zn + Se showed the protective effect to resume the cell number, epithelial lining, testicular cells damages, Sertoli cell damage, Leydig cell damage, damage at the junctions of seminiferous tubules, shrinkage of testes, altered spermatogenesis in the diabetic group, however fewer damages in the diabetic animals treated with Zn + Se. Histogram (b) depicts the effect of diabetes on the number of seminiferous tubules per unit area, and diabetic animals treated with Zn + Se, (c) graph represents Johnson’s score. All the values are shown as mean ± SEM (n = 5). ***p < 0.001, inset (a) versus control and inset (c) versus diabetic. Zn: zinc; Se: selenium; SEM: standard error of the mean.
Effect of Zn and Se on the expression of 8-oxo-2′-deoxyguanosine (8-oxodG) and LC3B
The immunoexpression of 8-oxo-2′-deoxyguanosine (8-oxodG) and LC3B were significantly increased in the diabetic group (p < 0.001), whereas Zn + Se treatment reduced the same in a significant (p < 0.01) manner (Figure 6(a) to (d)).
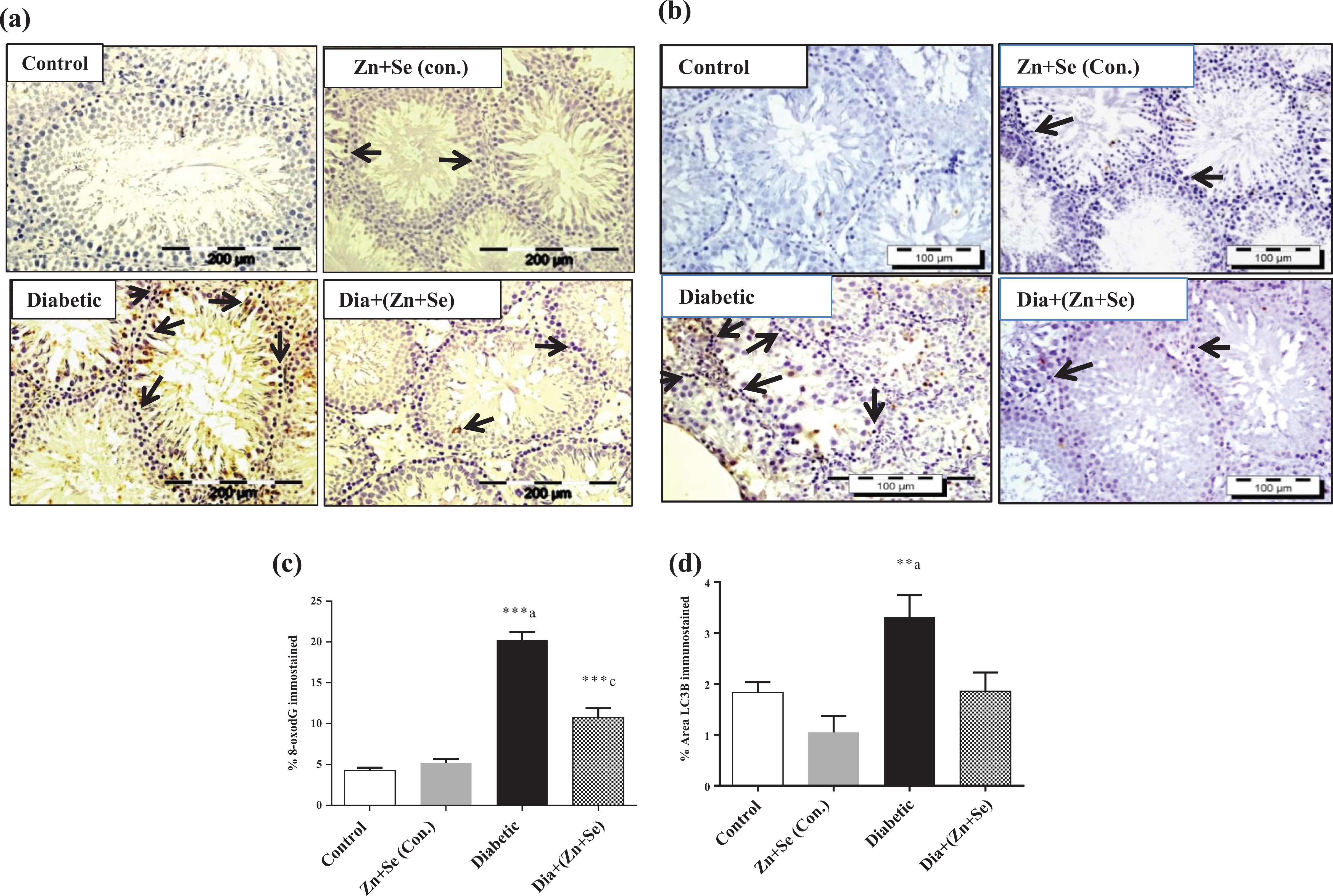
Effect of diabetes and diabetes treated with Zn + Se on (a) 8-oxodG, (b) LC3B protein expression in testes, histogram (c) depicted the quantification of (c) 8-oxodG and (d) LC3B. The positive cells are indicated with the presence of an arrow in different groups. All the values are shown as mean ± SEM (n = 3). **p < 0.01 and ***p < 0.001, inset (a) versus control and inset (c) versus diabetic. Zn: zinc; Se: selenium; 8-oxodG: 8-oxo-2′-deoxyguanosine; LC3B: microtubule-associated protein 1A/1B light chain 3B; SEM: standard error of the mean.
Effect of Zn and Se on DNA damage
Halo assay results revealed that moderate and extensive types of testicular damage happened in the diabetic group as compared to control and Zn + Se treatment reduced the same (Supplementary Figure 4A and B). From the calculation of the nuclear diffusion factor, it was revealed that significant (p < 0.001) DNA damage was observed in the diabetic group as compared to control and Zn + Se treatment significantly (p < 0.001) reduced the same (Supplementary Figure 4C). The results of comet assay parameters such as percent tail DNA, TL, TM, and olive TM indicated that significant (p < 0.001) DNA damage induced in diabetic animals germ cells and Zn + Se treatment normalized the same (Figure 7(a) to (e)). TUNEL assay results indicated that significant (p < 0.001) number of germ cells and sperm apoptosis was induced in diabetic animals, while Zn + Se treatment decreased the same (Supplementary Figure 5A–D).

Effect of diabetes and diabetes treated with Zn + Se on DNA damage (comet assay) on testes (a), histogram depicts the % tail DNA (b), TL (c), and olive TM (d) and TM (e). All the values are shown as mean ± SEM (n = 4). ***p < 0.001, inset (a) versus control and inset (c) versus diabetic rat. Zn: zinc; Se: selenium; TL: tail length; TM: tail moment; SEM: standard error of the mean.
Effect of Zn and Se on serum electrophoretic behavior
The electrophoretic patterns of serum proteins in the diabetic group were altered in the migration positions and band intensities as compared to control and Zn + Se control groups. Diabetic animals treated with Zn + Se improved these altered protein bands toward the control range (Figure 8).

Effect of diabetes and Zn + Se treatment on the serum gel electrophoretic pattern. Arrow marked indicating the pattern, and bands alterations have been found (n = 4). Zn: zinc; Se: selenium.
Effect of Zn and Se on protein expressions
In the present investigation, testicular Nrf-2 levels were significantly (p < 0.01) decreased in the diabetic group, while Zn + Se treatment attenuated this depletion (Figure 9(a)). Testicular catalase levels were decreased significantly (p < 0.01) in diabetic animals; Zn + Se treatment significantly (p < 0.05) improved the changes (Figure 9(b)). Sperm maturation-specific selenoprotein GPX4 was decreased significantly (p < 0.001) in diabetic rat testes and restored in diabetic animals treated with Zn + Se (Figure 9(c)). Se-independent GPX5 was significantly (p < 0.01) decreased in the diabetic group and restored toward normalcy in diabetic animals treated with Zn + Se (Figure 9(d)).

Effect of diabetes and Zn + Se treatment on expression of protein such as (a) Nrf-2, (b) catalase, (c) GPX4, and (d) GPX5 in testes. All the values are expressed as mean ± SEM (n = 3). *p < 0.05, **p < 0.01 and ***p < 0.001: inset (a) versus control and inset (c) versus diabetic. Zn: zinc; Se: selenium; Nrf-2: nuclear erythroid-derived related factor 2; GPX4: glutathione peroxidase 4; GPX5: glutathione peroxidase 5; SEM: standard error of the mean.
Effect of Zn and Se on the ultrastructural morphology of testes
TEM investigations of diabetic rat testes revealed tubular disarrangements, loss of spermatids membrane integrity, disposition of acrosome and acrosome cap, deformed nuclei, and flagellar microtubule damage as compared to control animals. Diabetic animals treated with Zn + Se ameliorated these changes toward normalcy (Figure 10).

Effect of diabetes and diabetic animals treated with Zn + Se on the ultrastructural morphology of testes. The control and Zn + Se (Con.) showed a typical structure. The arrows indicated the intact as well as the damaged structure of spermatid acrosomes, nuclei, and tail microtubule structures. Abnormal acrosomes, nuclei, and microtubule deformations in the spermatid tail were observed in the testes of diabetic animals. Zn + Se combination treatment restored the altered morphology and resembled towards control animal. Zn: zinc; Se: selenium.
Discussion
In the present investigation, the final body weight of diabetic rats was decreased significantly as compared to control. Zn and Se combination treatment improved diabetes-induced decrease in body weight as observed in the eighth week. It has been reported that dietary Zn supplementation increased the weight gain in children recovering from protein malnutrition. 64 Combination treatment with Zn and Se significantly restored both the weight of the testes and epididymis decreased due to diabetes. Blood glucose levels (≥250 mg/dl) in diabetic rats were significantly decreased by the combination treatment of Zn and Se after 8 weeks of treatment. Treatment with Zn and Se improved serum testosterone, MDA, SGPT, and BUN levels but significantly restored the SGOT, ALP, and catalase levels as compared to diabetic rats. It is worthy to mention that Zn and Se partially alleviate oxidative stress induced by diabetes in rat liver and kidney but protect efficiently when treated in combination. 65 Treatment with Zn and Se provided in drinking water efficiently protects the cadmium-induced testicular damage and reduced sperm motility and antioxidant status. 37 Zn- and Se-protected metal-(loids) induced toxicity and disease manifestations. 66 Frustaci et al. reported that patients receiving Se 300 µg and Zn 13 mg every day for 1 week and every week for 6 months through infusion protected against Zn- and Se-deficient cardiomyopathy. 35 Sperm motility and counts were found significant recovery by Zn- and Se-supplemented rat compared to diabetes. The sperm head morphology depicted extensive structural changes, lack of membrane integrity, membrane damage, and acrosomal cap disposition in the diabetic group, which were ameliorated in Zn- and Se-treated rats. It has also been reported that Zn and Se supplementation improved the semen quality in Barbari bucks. 33 Previously, it has been reported that Zn and Se ameliorated cadmium-induced sperm motility disturbance. 37 It has also been proved that Zn is useful for the maintenance of sperm and semen quality in man. 67 The histopathological investigation revealed extensive testicular damage, structural deformations in the tubular myeloid membrane, luminal contents, and interstitium in the STZ-induced diabetic group, while Zn and Se supplementation restored the same. Epididymis tubular membrane, luminal contents, and interstitium were found damaged in diabetic rats, while Zn and Se supplementation can able to minimize epididymal damage. The individual roles of Se and Zn have been reported beneficial against infertile men on pregnancy rate and epididymal toxicity in the experimental rat. 18,68 Apoptotic cell death (TUNEL positive cells) in testis and caudal sperms were significantly increased in diabetic rats, Zn and Se treatment showed beneficial effects by decreasing diabetes-induced apoptotic cell death. The comet and halo assay results also indicated that significant DNA damage and intervention of Zn and Se reduced these effects. The protective effects of Se supplementation on diabetes-induced germ cell oxidative stress, DNA damage, and cytotoxicity have already been reported. 45 From the above results, it is evident that co-treatment of Zn and Se ameliorated the testicular oxidative stress, DNA damage, and TUNEL positive cells by the involvement of antioxidant mechanisms, which is necessary for cell survival against diabetes-induced germ cell toxicity.
The IHC of 8-oxodG expression on testes tissue increased during diabetes which is one of the novel markers for the evaluation of oxidative stress-associated DNA damage. The immune positive signal for 8-oxodG was significantly decreased by Zn and Se treatment and indicated the amelioration of oxidative stress and DNA damage. LC3B immune expression was examined and the increased LC3B level during diabetic condition was reduced by Zn and Se supplementation. It has been reported that oxidative stress increased the LC3B level that is responsible for the execution of autophagy, unlike oxidative stress reduced the number of autophagosome formation, and autophagy was inhibited. 69 The protein expression pattern of serum gel electrophoresis confirmed the restoration of diabetes-induced altered protein pattern by Zn and Se treatment. Zn and Se were responsible for the regulation and homeostasis of different cytoprotective proteins against diabetes-induced oxidative stress and DNA damage in testes. 27 The present study findings demonstrated that Zn and Se combination treatment has beneficial effects in diabetes-induced germ cell damage. The present study showed that the Zn and Se combination treatment modulated the plasma Zn level and testicular Zn and Se levels during the diabetic condition. The Zn and Se treatment restored the testicular and epididymis (cauda and caput) structural deformities induced by diabetes. Further, Zn and Se improved sperm quality by decreasing the testicular and sperm apoptotic cell death. Zn and Se combination treatment ameliorated the oxidative stress, DNA damage, cellular changes in the germ, and epididymis of diabetic rats. However, it has been reported that in a randomized, double-blind control study with Zn 30 mg/day and Se 150 µg/day for 4 months study did not restore the lipid profile, SOD, glutathione peroxidase activity, and mineral status in coronary artery disease patient treated with rosuvastatin. 70 TEM results revealed alterations like changes in acrosome structure, disposition of acrosome cap from the acrosomal membrane, deformed nuclei, and disorganization of microtubule in the flagella of diabetic animals. Zn + Se treatment ameliorated these changes in diabetic animals. It has been reported that both Zn and Se deficiency-induced ultrastructural changes in the germ cells of rat and mice. 19,71 Further, it has been reported that sodium selenite supplementation restored the ultrastructural changes induced by monosodium glutamate in the testes of rodents. 71
Protein expression by Western blot analysis revealed that Nrf-2 and catalase levels were decreased significantly in diabetic rat testes. Zn and Se treatment improved these changes significantly in diabetic rats. It has been reported that Zn and Se treatment successfully restored Nrf-2 expression in the germ cell of diabetic animals. 16,18 Further, it has been reported that Nrf-2 knock-out mice disrupted spermatogenesis and epididymal sperm damage by oxidative stress mechanisms. 72 Furthermore, it has been reported that Zn and Se treatment improved the catalase levels and protected germ cell damage induced by cadmium. 37 In the present investigation, GPX4 level reduced significantly in the diabetic group compared to control and diabetic animals treated with Zn + Se restored the same in a significant manner. GPX4 is an abundant selenoenzyme expressed actively in the mitochondria of testes and spermatozoa. The depletion of GPX4 led to abnormal spermatozoa and a significant decrease in GPX4 level and resulted in 30% infertile human suffering with oligoasthenozoospermia. 73 Se-independent GPX5 expression was examined in testes tissues decreased significantly in the diabetic rat as compared to control; the combined treatment of Zn and Se improved the same in a significant manner. It has been reported that GPX5 is expressed in testes and abundant in the epididymis to provide smooth passage for the transit of sperms maintaining antioxidant balance and sperm survival in pig. 24 Zn is one of the vital trace elements and participates from germ cell development to spermeation in humans. 74 So future studies at molecular levels related to Zn dynamics and the role different transporter involved in Zn uptake at different phases of sperm life should be carried out. Both human and animal studies provide many fold evidence regarding the diverse protective roles of Se supplementation. 75 However, well-designed randomized controlled studies are needed to explore the proper supplementation of Se in humans during reproductive age.
Conclusion
Trace elements like Zn and Se are vital for the maintenance and the regulation of reproductive physiology in both male and female. 76 –78 Several publications emphasized the crucial roles of Zn and Se in maintaining the reproductive functions during diabetes. 3,79 –81 From the present study, it is clearly evident that Zn and Se combination treatment protected diabetes-induced germ cell damage in the rat as revealed by several end points of evaluations discussed above. It is also interesting to mention that the role of trace elements in juvenile diabetes mellitus has already been highlighted. 82 So it is highly imperative to further investigate the exact role of combination protection by Zn and Se in maintaining reproductive health during diabetes at both the molecular and cellular levels.
Supplemental material
Supplementary_Data_final - Zinc and selenium combination treatment protected diabetes-induced testicular and epididymal damage in rat
Supplementary_Data_final for Zinc and selenium combination treatment protected diabetes-induced testicular and epididymal damage in rat by C Sahu, DK Dwivedi and GB Jena in Human & Experimental Toxicology
Footnotes
Acknowledgments
The authors would like to acknowledge Mr Vinod Kumar for subcellular image analysis by the transmission electron microscope. The authors would like to acknowledge the inputs of Dr Sabbir Khan (postdoctoral fellow, Department of Neuro-oncology, The University of Texas, MD Anderson Cancer Center, Houston, TX, USA) for correction of the English language as well as improving the scientific clarity of the manuscript.
Author contribution
CS conceived and designed the research hypothesis, conducted experiments, analyzed data and wrote the manuscript. DKD provided technical support and reviewed the manuscript. GBJ conceived the idea, reviewed the manuscript, and carried out administrative aspects of this project. All the authors read and approved the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Department of Science and Technology, Science and Engineering Research Board (DST-SERB EMR/2015/001212/HS), Government of India, for carrying out the above experimentation.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
