Abstract
The aim of the present study is to investigate whether nicotine augmented the testicular toxicity and angiotensin converting enzyme inhibitor, enalapril, can ameliorate the effects in diabetic rat. Male Sprague Dawley rats were randomized into five groups: control, nicotine, diabetic, Diab + Nico, and Diab + Nico + Enal. Animals were made diabetic by single injection of streptozotocin (55 mg/kg/intraperitoneally). Nicotine dissolved in drinking water at a concentration of 100 µg/ml was given ad libitum and enalapril was given orally at a dose of 10 mg/kg/day for four consecutive weeks. After 4 weeks of treatment, animals were killed and biochemical parameters glucose, glycosylated hemoglobin, cotinine, and the testosterone levels were measured. Testicular toxicity was evaluated using sperm count, sperm comet assay, histology, and immunohistochemical staining of 8-oxo-7,8-dihydro-2′-deoxyguanosine (8-oxo-dG) and the proinflammatory markers (nuclear factor kappa B (NF-κB), cyclooxygenase (COX-2), and tissue necrotic factor alpha (TNF-α)) evaluated by western blotting. Results showed that nicotine did not alter the blood glucose and glycosylated hemoglobin level, significantly decreased the sperm count and increased the sperm DNA damage. These changes were accompanied by significant increases in the 8-oxo-dG, NF-κB, COX-2, and TNF-α expression. Furthermore, the intervention of enalapril in nicotine-treated diabetic rat attenuated the testicular damage and restored sperm count, sperm DNA damage, as well as reduced the expression of NF-κB, COX-2, and TNF-α. These findings clearly suggest that nicotine not only augmented the testicular toxicity in the diabetic rat but also increases the risk of germ cell toxicity effects that were attenuated by enalapril treatment.
Introduction
Cigarette smoking is identified as a risk factor for the progression of reproductive health in diabetic as well as in nondiabetic individuals. One of the pharmacologically active constituents of cigarette is nicotine and its detrimental effects served as an independent predictor of male infertility. 1 Equally important, diabetes is a well-known cause of impaired reproductive dysfunction in both clinical and experimental setups. 2,3 Epidemiological studies suggest that cigarette smoking augmented the risk factor for sexual dysfunction among both type 1 and type 2 diabetic patients 4 –6 and is also associated with worsening of sexual dysfunction in patients with hypertension. Although the existing clinical and epidemiological studies suggest that smoking and its constituent nicotine exacerbates and accelerates the progression of vascular, renal, and cardiac diseases in diabetes, 7 –9 yet there is no strong evidence to suggest that nicotine is an important risk factor for testicular toxicity in diabetes. Studies suggest that nicotine adversely affects male reproductive functions 10,11 based on the mechanism of apoptosis in Leydig cell by a B-cell lymphoma 2-associated X protein-dependent pathway 12 or causes testicular degeneration by inhibiting the steroidogenic acute regulatory (StAR gene) expression. 13 However, mechanisms by which nicotine accelerate the progression of testicular damage are not well understood.
It has been reported that nicotine worsens the severity of nephropathy by significant increases in nicotinamide adenine dinucleotide phosphate (NADPH) oxidase 4, nitro tyrosine, and protein kinase B (Akt) expression in diabetic mice 7 and promotes mesangial cell proliferation and hypertrophy via activation of nonneuronal nicotinic acetylcholine receptors. 14 Nicotine increases the activation of inflammatory mediators and worsens the glomerular injury in a rat model of acute nephritis. 15 In addition to this, nicotine increases the expression of cyclooxygenase (COX-2) through activation of nuclear factor κB (NF-κB) in human umbilical vein endothelial cells. 16 Nicotine also stimulated the synthesis of prostaglandin E2 by the involvement of transcription factor NF-κB and COX-2 in human gingival fibroblasts. 17 Reddy et al. also reported that nicotine not only aggravates diabetic complications but also increased the risk of diabetes. 18,19 Recently, it has been reported that nicotine exacerbates the progression of cataracts via inflammatory mediators in a streptozotocin (STZ)-induced diabetic rat. 20 These findings reinforced the proposal that nicotine leads to the increase in inflammatory markers through different signaling pathways and it worsens diabetic complications. In earlier studies, it was demonstrated that enalapril is an excellent intervention agent for normal as well as for diabetic hypertensive subjects. 21 –24 Munzel and Keaney represent the angiotensin converting enzyme (ACE) inhibitor as a new antioxidant strategy and explained that by decreasing the angiotensin II (Ang II) it limits the stimulation of vascular NADPH oxidase, thereby preventing the increased superoxide flux associated with the activation of the renin angiotensinogen system. 25 We have already reported that enalapril decreased the expression of proinflammatory marker NF-κB and COX-2 in diabetic testes. 26 These results suggest the possible protective role of enalapril as a disease-modifying therapeutic agent against nicotine-induced testicular toxicity in the diabetic rat.
In view of these reports, the present study was designed to elucidate the mechanisms by which nicotine influences testicular toxicity and further to examine the possible role of enalapril in the protection against the effect of nicotine on testicular toxicity of diabetic rat. Interestingly, nicotine did not cause significant changes in the blood glucose and glycosylated hemoglobin level in the control and diabetic rat receiving nicotine, but decreased the concentration of testosterone in plasma and increased the cotinine concentration. The administration of nicotine in diabetic rat leads to significant decreases in sperm count and significant increases in sperm DNA damage and 8-oxo-7,8-dihydro-2′-deoxyguanosine (8-oxo-dG)-positive cells. These changes were accompanied by significant increases in the level of COX-2, NF-κB, and tissue necrotic factor alpha (TNF-α) in diabetic testes exposed to nicotine.
Materials and methods
Animals
All the animal experiments were approved by the Institutional Animal Ethics Committee. Experiments were performed on male Sprague Dawley (SD) rats (250 ± 10 g, 8 weeks) procured from the Central Animal Facility of the institute. The animals were kept at room temperature (22 ± 2°C), with 50 ± 10% humidity and an automatically controlled cycle of 12 h light and 12 h dark. They were fed with standard laboratory animal feed (purchased from a commercial supplier) and provided with free access to water (Aquapure, New Delhi, India). All the animals were acclimatized for 1 week before the initiation of the experimentation, and experiments on animals were performed in accordance with the Committee for the purpose of Control and Supervision of Experimentation on Animal guidelines.
Chemicals
Enalapril maleate, nicotine hydrogen tartrate, STZ, trizma, dithiothreitol, proteinase-K, and SYBR Green I were purchased from Sigma-Aldrich Chemicals (St Louis, Missouri, USA). Dimethyl sulfoxide, normal-melting point agarose (NMA), low-melting point agarose (LMA), triton-X 100, ethylenediaminetetraacetic acid (EDTA), and Hank’s balanced salt solution (HBSS) buffer were obtained from Hi-media Laboratories Ltd (Mumbai, India).
Induction of diabetes
Experimental diabetes was induced by a single dose of STZ (55 mg/kg) dissolved in ice-cold 10 mM citrate buffer, pH 4.4, and administered intraperitoneally immediately after preparation to the SD rats. Age-matched control rat received an equivalent volume of the vehicle. Blood glucose level was measured after 48 h of STZ administration using commercially available spectrophotometric kits (Accurex Biomedical Pvt. Ltd, Boisar, Thane, India). Rats with blood glucose level of ≥250 mg/dl were considered to be diabetic and used in the present study. 27 Blood glucose level was also measured at the termination of the study.
Experimental design and rationale for dose selection
Male SD rats were divided into five groups: control, nicotine, diabetic, Diab + Nico, and Diab + Nico + Enal. The dose of nicotine (100 µg/ml/day) treatment was selected based on the study conducted by Hua and Rodella et al. 7,28 Nicotine at a concentration of 100 µg/ml dissolved in drinking water was given ad libitum to the nicotine only group for four consecutive weeks. Due to more water intake (approximately more than double the volume in comparison to control animals) in the diabetic groups (Diab + Nico and Diab + Nico + Enal), the concentration of nicotine in drinking water was further reduced to 25 µg/ml and was given ad libitum. Finally, the concentration of nicotine was determined from the cotinine analysis in the serum of animals. The nicotine intake as calculated from the consumption of drinking water was in the range of 12–15 mg/kg/day. Furthermore, the dose of enalapril 10 mg/kg body weight was selected based on the earlier studies in which enalapril shows the protective effect in the STZ-induced diabetic model. 29,30
Chemical preparation and animal treatment
Nicotine was freshly prepared in the drinking water and the bottles were wrapped in aluminum foil to prevent any light-induced decomposition of the nicotine since nicotine is light sensitive. Enalapril was freshly prepared in distilled water and given orally at a dose of 10 mg/kg/day for four consecutive weeks. Initially, eight animals were included in the diabetic groups considering the rate of mortality during the progression of diabetes. In the final experiment, only six animals were considered for the analysis. However, in the control group only six animals were kept throughout the conduct of the study.
Testosterone measurements
Blood samples (˜0.8 ml) were collected from the orbital plexus of the rats under light ether anesthesia in heparinized microcentrifuge tubes. The plasma was separated by centrifugation (2500g, 5 min) and the concentration of plasma testosterone was estimated by DRG Testosterone ELISA kit (DRG Instruments GmbH, Marburg, Germany) as per the manufacturer’s instruction.
Cotinine measurements
The serum level of cotinine, a metabolite of nicotine, was measured using a Cotinine Direct ELISA kit (CalBiotech, CA, USA) following the manufacturer’s instructions.
Sperm count
The cauda epididymis was removed after killing the animals and placed in a petri dish containing 2–3 ml of HBSS at room temperature. The epididymis was minced into small pieces to allow the sperms to swim out. The sperm suspension thus obtained was centrifuged at 1000 r/min for 5 min. After centrifugation, 1 ml of the supernatant was taken and the epididymal sperm count was determined using Neubauer’s hemocytometer (Lauda-Königshofen, Germany). Data were expressed as the number (no.) of sperms per milligram weight of epididymis.
Histological evaluation
Histological slides were prepared as earlier carried out in our laboratory. 26 The testis and the epididymis were fixed in Bouins’ fixative, dehydrated in increasing concentrations of ethanol and embedded in paraffin. Tissue sections (5 µm) were mounted on glass slides coated with Mayer’s albumin and dried overnight. The sections were then deparaffinized with xylene, rehydrated with alcohol and water. The rehydrated sections were stained using hematoxylin and eosin, mounted with DPX mounting media and examined under a microscope at 20× magnification (Olympus BX51 microscope, Tokyo, Japan). Testis and epididymis sections from each animal were evaluated for structural changes.
Morphometric analysis
For morphometric analysis, the testes sections were stained with periodic acid Schiff reagent to distinguish the spermatogonia, spermatocytes, spermatids, and sertoli cells and the slides were observed under a microscope (Olympus BX51, Tokyo, Japan) and evaluated for (1) average number of seminiferous tubules per focus (148012.6 µm2), (2) the diameter of the seminiferous tubules (in µm), and (3) the average number of spermatogonia, spermatocytes, spermatids, and sertoli cells per tubule. The number of seminiferous tubules per focus was counted at 20× magnification. For seminiferous tubule diameter measurement, 10 most circular seminiferous tubules were identified in each section of the testis using Olympus soft imaging software Cell (Tokyo, Japan) and expressed in micrometer. The number of germ cells and sertoli cells was quantified in 10 tubules/animal. Only sertoli cells exhibiting distinctive morphological features and clear nucleolus were quantified. Since the number of germ cells in tubular section at stages I–VIII is different from those at stages IX–XIV, five tubules at stage I–VIII and five tubules at stage IX–XIV per animal were counted. 31
Sperm comet assay
The sperm comet assay was performed essentially as described with some modifications. 32,33 Sperm sample (5 µl) containing 1–3 × 104 sperm/ml was suspended in 95 μl of 1% (w/v) LMA. From this suspension, 80 μl was applied to the surface of a microscope slide (precoated with 1% NMA) to form a microgel and allowed to set at 4°C for 5 min. Slides were dipped in cell lysis buffer (2.5 M sodium chloride (NaCl), 100 mM EDTA, 10 mM Tris hydrochloric acid (HCl), pH 10.0 containing 1% triton X-100, and 40 mM dithiothreitol) for 24 h at room temperature and protected from light. Following the initial lysis, proteinase K was added to the lysis solution (0.5 mg/ml) and additional lysis was performed at 37°C for 24 h. Following cell lysis, all slides were washed three times with deionized water at 10 min intervals to remove salt and detergent from the microgels. Slides were placed in a horizontal electrophoresis unit and were allowed to equilibrate for 20 min with running buffer (500 mM NaCl, 100 mM Tris HCl, and 1 mM EDTA, pH 9) before electrophoresis (0.60 V/cm, 250 mA) for 30 min. After electrophoresis, the slides were neutralized and the DNA fluorochrome SYBR Green I (1:10,000 dilution) was applied for 1 h. The slides were rinsed briefly with double-distilled water and coverslips were placed before analyzing the image. The fluorescent-labeled DNA was visualized (200×) using an AXIO Imager M1 fluorescence microscope (Carl Zeiss, Gottingen, Germany) and the resulting images were captured on a computer and processed with image analysis software (Metasystem software, Comet Imager V.2.0.0). The parameters for the DNA damage analysis include tail length (TL, in micrometer), tail moment (TM), olive TM (OTM), and percentage of tail DNA (% DNA). Samples were run in duplicate and 50 cells were randomly analyzed per slide for a total of 100 cells per sample. Comet TL is the maximum distance that the damaged DNA migrates from the center of the cell nucleus. The % DNA is the total DNA that migrates from the nucleus into the comet tail. TM is the product of the TL and the % DNA, which gives a more integrated measurement of overall DNA damage in the cell.
Data scoring and photomicrographs of comet assay
The fluorescence-labeled DNA was visualized (200×) using an automated AXIO Imager M1 fluorescence microscope (Carl Zeiss), and the images were captured with image analysis software (Comet Imager V.2.0.0, Metasystem). Duplicate slides were prepared for each treatment and were independently coded and scored without knowledge of the code. The parameters for the DNA damage analysis include TL (in µm) TM, OTM, and % DNA.
Immunohistochemistry of 8-oxo-dG
Paraffin-embedded testes were cut into 5-µm-thick sections, placed on poly-
Estimation of protein expressions by western blotting
For western blot analysis, proteins were transferred onto nitrocellulose membrane and performed using the anti-NF-κB (rabbit polyclonal 1:500, Santa Cruz, CA, USA), anti-COX-2 (rabbit polyclonal 1:500, Santa Cruz), anti-TNF-α (goat polyclonal 1:500, Santa Cruz), antiactin (rabbit polyclonal 1:2500, Santa Cruz), and horseradish peroxidase-conjugated secondary antibodies (antirabbit and antigoat, Santa Cruz). Proteins were detected using enhanced chemiluminescence and quantified with the help of software Imagequant TL (Imagequant 350, GE Healthcare, Hong Kong, China).
Statistical analysis
Results were shown as mean ± SEM for each group. Statistical analysis was performed using Jandel Sigma Stat (Version 3.5) statistical software (San Rafael, CA, USA). For multiple comparisons, one-way analysis of variance (ANOVA) was used. In case ANOVA showed significant differences, post hoc analysis was performed with Tukey’s test. A p < 0.05 was considered to be statistically significant.
Results
Effect of nicotine on the body and organ weight, glucose, glycosylated hemoglobin, cotinine and testosterone level in STZ-induced diabetic rat and the intervention of enalapril
The changes in final body weights were not significant when diabetic group was compared with the Diab + Nico and Diab + Nico + Enal groups; however, body weight significantly reduced in the diabetic, Diab + Nico, and Diab + Nico + Enal groups when compared with the control group. There was significant reduction in the paired testes and epididymis weight in diabetic group as compared to Diab + Nico group, but not up to the significant level (Table 1). The levels of blood glucose (in mg/dl) and glycosylated hemoglobin (in %) were significantly elevated in diabetic, Diab + Nico, and Diab + Nico + Enal groups as compared to the control group and the results showed that treatment with nicotine did not alter the blood glucose and glycosylated hemoglobin. The cotinine level was found to be approximately >150 ng/ml in nicotine-treated rat and it was >200 ng/ml in Diab + Nico and Diab + Nico + Enal groups (Table 1). The cotinine le-vel was more in Diab + Nico and Diab + Nico + Enal groups as compared to per se nicotine group, even if the concentration of nicotine was adjusted according to the more water-intake by the animals in diabetic group. There were significant decrease in the testosterone level in both diabetic and Diab + Nico groups compared to control group. However, there was no significant difference in diabetic versus Diab + Nico (Table 1). Enalapril increased the testosterone level but not up to the significant level when compared with the Diab + Nico group.
Effect of nicotine and intervention of enalapril treatment on body weight and organ weight, blood glucose, glycosylated hemoglobin, cotinine, and testosterone levels in STZ-induced diabetic rat.a
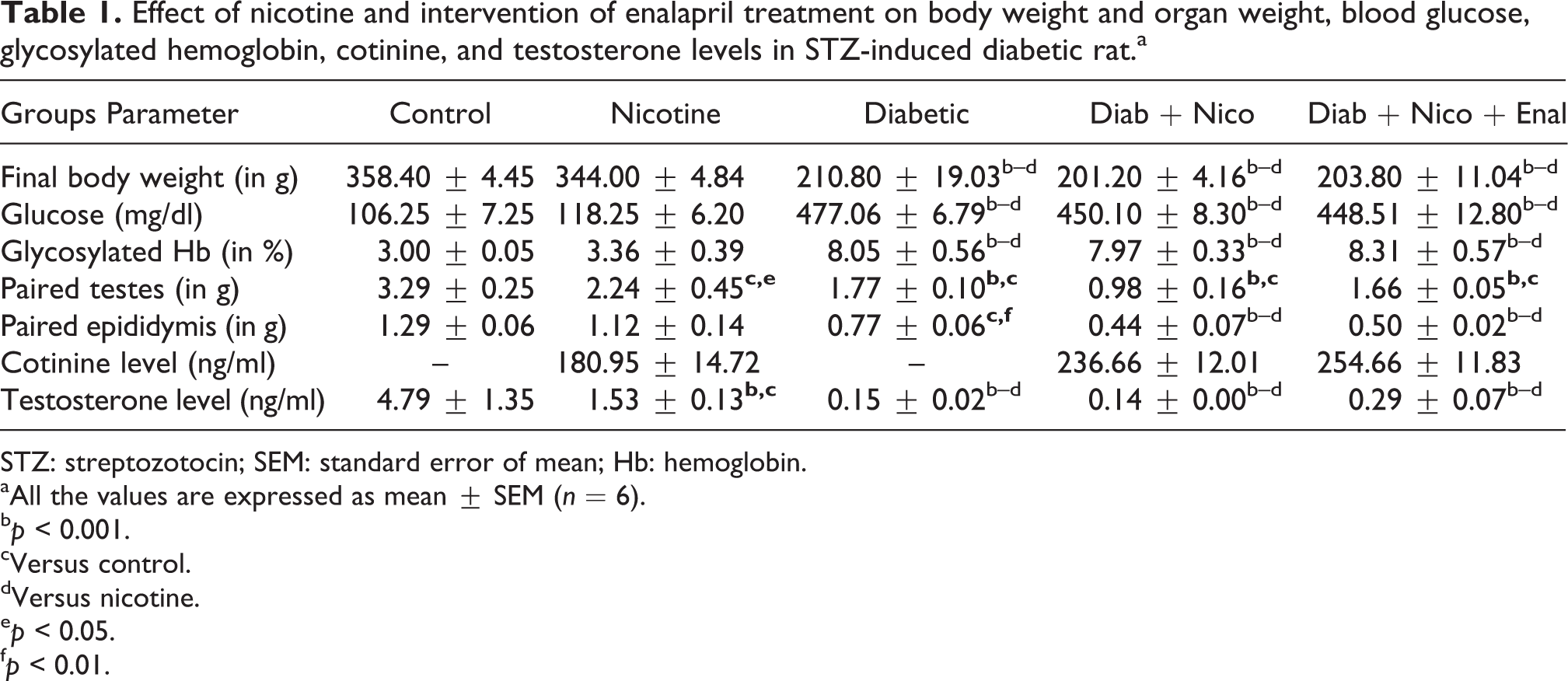
STZ: streptozotocin; SEM: standard error of mean; Hb: hemoglobin.
aAll the values are expressed as mean ± SEM (n = 6).
b p < 0.001.
cVersus control.
dVersus nicotine.
e p < 0.05.
f p < 0.01.
Effect of nicotine and intervention of enalapril treatment on sperm count
Statistically significant decrease in the sperm count was observed in Diab + Nico group as compared to diabetic group. It clearly indicated that nicotine further decreased the sperm count in diabetic testis. Furthermore, intervention of enalapril treatment in Diab + Nico group significantly increased the sperm count (Figure 1).

Histogram showing the effects of nicotine and intervention of enalapril in Diab + Nico sperm count. All the values are expressed as mean ± SEM (n = 6), ***p < 0.001 and **p < 0.01, ‘a’ versus control, ‘b’ versus diabetic, and ‘c’ versus Diab + Nico. SEM: standard error of mean.
Effect of nicotine and intervention of enalapril treatment on testis and epididymis histology
Light microscopic investigations of testes histology clearly showed that nicotine treatment in both nicotine per se and diabetic group not only led to morphological alterations such as disorganization, degeneration, and reduction in the size of the seminiferous tubules but also reduced the spermatogenic cell mass with clear space from the basal lamina. Furthermore, enalapril intervention restored the same in Diab + Nico group. In epididymis, Diab + Nico treatment led to marked constriction of the lumen and decreased the number of sperms in comparison with diabetic group. Intervention with enalapril increased the number of sperms in the lumen of the epididymis of these groups (Figure 2).

Representative photomicrographs of epididymis and testes sections stained with hematoxylin and eosin. In diabetic group, there was decreased number of sperms in epididymis and vacuolization in the testis. In Diab + Nico group, nicotine further decreased the sperms and distorted the testicular structure in epididymis and testes sections, respectively. The symbol “#” indicates the decreased number of sperms and degeneration of germ cells in the seminiferous tubule in Diab + Nico group in epididymis and testes sections, respectively, “*” indicates the restoration by enalapril treatment (magnification: ×20).
Effect of nicotine and intervention of enalapril treatment on testicular histomorphology
In Diab + Nico group, there was significant reduction (128.97 ± 12.27 vs. 304.47 ± 0.56) in diameter (in µm) of seminiferous tubules, increase in number of seminiferous tubules (26.73 ± 0.53 vs. 15.73 ± 1.94) as compared to diabetic group. Moreover, there was de-crease in the number of spermatogonia (6.18 ± 0.13 vs. 9.52 ± 0.28), spermatocytes (34.54 ± 0.56 vs. 23.88 ± 0.61), spermatids (34.06 ± 0.78 vs. 36.08 ± 0.77), and sertoli cells (7.94 ± 0.48 vs. 9.60 ± 0.50) in Diab + Nico group as compared to diabetic group. Furthermore, the intervention of enalapril led to significant restoration in the testicular morphometry in Diab + Nico group (Figure 3).

Scattered plots showing the effect of nicotine exacerbation and intervention of enalapril treatment on the morphometry of different spermatogenic cells and number of seminiferous tubules in Diab + Nico group. All values are expressed in mean ± SEM (n = 6), ***p < 0.001, **p < 0.01, and *p < 0.05. ‘a’ versus control, ‘b’ versus diabetic, and ‘c’ versus Diab + Nico.
Effect of nicotine and intervention of enalapril treatment on diabetes-induced sperm DNA damage
Significant increase in sperm DNA damage (namely, TL (in micrometers), TM, OTM, and % DNA) was observed in the Diab + Nico group as compared to diabetic group. It clearly depicts that nicotine exacerbates the sperm DNA damage in Diab + Nico group. Interestingly, enalapril treatment significantly reduced the DNA damage in Diab + Nico group (Figure 4(a) and (b)).

(a) Effect of nicotine on testicular toxicity and intervention of enalapril on sperm DNA damage as revealed by comet assay parameters in Diab + Nico group. All values are expressed in mean ± SEM (n = 6), ***p < 0.001, **p < 0.01, and *p < 0.05. ‘a’ versus control, ‘b’ versus diabetic, and ‘c’ versus Diab + Nico. (b) Photomicrographs showing the DNA migration pattern of sperm nuclei in single cell gel electrophoresis (comet) assay stained with SYBR Green-I. The symbols “+”and “−” represent anode and cathode, respectively, during electrophoresis. (A) Sperm nuclei from control and (B) sperm nuclei from Diab + Nico treatment.
Effect of nicotine and intervention enalapril treatment on 8-oxo-dG, COX-2, NF-κB, and TNF-α
High intensity of immunostaining of 8-oxo-dG was observed in the testis of Diab + Nico group as compared to the diabetic group. The frequency of 8-oxo-dG-positive nuclei was quantified in the testes. Furthermore, significant increase in the percentage of 8-oxo-dG-positive cells was found in Diab + Nico group as compared to the diabetic group and was attenuated by enalapril treatment (Figure 5(a) and (b)). Results of western blotting showed significant increase in the upregulation of COX-2, NF-κB, and TNF-α expression in Diab + Nico group as compared to diabetic group. Moreover, the expressions of these inflammatory markers were found to be significantly increased in per se nicotine group as compared to the control. These results revealed that nicotine treatment per se and in the diabetic group increased the inflammation process as evident by the increased expression of inflammatory markers. Furthermore, enalapril intervention significantly decreased the expression of NF-κB, COX-2, and TNF-α in Diab + Nico group (Figure 6(a) to (c)).

(a) Immunohistochemistry of 8-oxo-dG level in testes of rat (Magnification: ×40). Arrow indicates the dark brown nuclei showing positive signals of 8-oxo-dG in Diab + Nico group, while the light stains nuclei showing weak or no positive signal. (b) The quantitative estimation showed a significant increase in percentage of 8-oxo-dG-positive cells in Diab + Nico group as compared to diabetic group and further decreased by enalapril treatment. All values are expressed in mean ± SEM (n = 6), ***p < 0.001, ‘a’ versus control, ‘b’ versus diabetic and ‘c’ versus Diab + Nico. 8-oxo-dG: 8-oxo-7,8-dihydro-2′-deoxyguanosine; SEM: standard error of mean.

Effect of nicotine on the expression of COX-2, NF-κB, and TNF-α in testes and intervention of enalapril in Diab + Nico group. The representative western blots showing the expression of (a) COX-2, (b) NF-κB, and (c) TNF-α. It showed that nicotine significantly increased the proinflammatory markers in diabetic rat testes and enalapril attenuates the same. The data were normalized with β-actin expression and expressed in mean ± SEM of three individual experiments. ***p < 0.001 and **p < 0.01, ‘a’ versus control, ‘b’ versus diabetic and ‘c’ versus Diab + Nico. SEM: standard error of mean; NF-κB: nuclear factor kappa B; COX-2: cyclooxygenase; TNF-α: tissue necrotic factor alpha.
Discussion
The present study provides the evidence that nicotine augmented the testicular damage and intervention of ACE inhibitor enalapril ameliorated the testicular toxicity in STZ-induced diabetic rat. To confirm the nicotine exposure, the stable metabolite of nicotine, cotinine, was estimated and significant increase was observed in both control and diabetic animals receiving nicotine. The present results indicate that diabetic animals exhibited more cotinine level as compared to normal and this might be due to the differences in the water intake, body metabolism, and the excretion profile from the body over time. It has been reported that the mean cotinine level in the serum of active smokers, who smoked more than 10 cigarettes/day was found to be 301.2 ng/ml, 35 and in the present study, the cotinine exposure in the different groups was found to be in the range of 180.95–234.66 ng/ml, which is a good indicator for the risk assessment purposes. However, nicotine exposure in the diabetic rat did not alter the blood glucose and glycosylated hemoglobin level. 7 Results indicate that nicotine per se significantly reduced the testes and epididymis weight, further decrease in the organ weights was observed in diabetic rat receiving nicotine treatment and the intervention of enalapril improved the same, which was not statistically significant. It has already been reported that nicotine reduced the weight of testis and epididymis due to the unavailability of pituitary gonadotropins, which are essential for the initiation and completion of spermatogenesis and steroidogenesis. 36 Furthermore, Scarano et al. showed that the decrease in the reproductive organ weight in diabetic animals was associated with decrease in the testosterone level, 37 and the present result was in conformity with the same. Intervention of enalapril increased the testosterone level, but not up to the significant level.
Nicotine significantly decreased the sperm count and further exacerbation was found in the diabetic group, which was significantly restored by enalapril treatment. Yamamoto et al. reported that nicotine treatment led to significant decrease in the sperm count in rat. 38 Recently, it has been reported that concurrent administration of alcohol and nicotine exaggerates the adverse effects of nicotine on the sperm count. 39 Furthermore, decrease in sperm count in nicotine-treated diabetic group may be due to the lower concentration of testosterone as evident from the present experiment. Histological analysis revealed that nicotine deteriorated the testicular and epididymal structure in diabetic rat and significant protection was observed with enalapril treatment. In testis, seminiferous tubules were disorganized and alienated from each other with loss of normal architecture. In epididymis, there was a decrease in the sperm count as evident from histological analysis. It was well corroborated by Ricci et al. that abnormal cytoarchitecture and altered spermatogenesis were possibly due to the effect of oxidative stress on Leydig cell function by which it lowers the testosterone level in STZ-induced diabetic rat. 40 Furthermore, histomorphometric analysis of the testes indicated that nicotine treatment deteriorated the spermatogenesis as evident by significant decrease in the seminiferous tubule diameter, number of spermatids, spermatogonia, spermatocytes, and sertoli cells as compared to diabetic rat. Intervention of enalapril significantly restored the histomorphometric changes as compared to Diab + Nico group. To ensure the role of increased oxidative stress in the induction of oxidative DNA damage, quantification of 8-oxo-dG level was immunohistochemically determined. Nicotine significantly increased the percentage of 8-oxo-dG-positive cells in the testis of diabetic rat, which was subsequently attenuated by the intervention of enalapril. It has already been reported that the level of 8-oxo-dG in the sperm DNA was found to be significantly higher than that of nonsmokers, 41 while no data have been reported for further increase in 8-oxo-dG level by nicotine in diabetic testes. The total DNA damage was evaluated using sperm comet assay. The sperm comet assay detects DNA damage in the sperm and showed that nicotine exacerbated the sperm DNA damage in diabetic rat as evident from the significant increase in the TL, TM, OTM, and %DNA in comet tail. This substantiates that DNA damage plays an important role and nicotine further exacerbated the testicular dysfunction in diabetic rat. It has been reported that oxidative stress affects the integrity of sperm chromatin and causes elevated frequencies of single and double-stranded DNA breaks. 42 Furthermore, it has also been reported that nicotine perturbs the plasma membrane integrity and induces the DNA damage. 10 Demirhan et al. showed that nicotine led to increased frequencies of chromosome translocations and DNA strand breaks in smokers. 43 Moreover, smoking damages the chromatin structure and produce endogenous DNA strand breaks in human sperm. 44 The possible explanation for the further exacerbation of sperm DNA damage in diabetic group receiving nicotine treatment might be due to the direct cytotoxic effect on the spermatozoa by damaging the DNA. It has been reported that nicotine and its metabolite cotinine can easily cross the blood–testes barrier. 45 Enalapril do not readily cross the blood–testis barrier as reported. 46 However, the possibility cannot be ruled out as in diabetic condition the blood–testis barrier gets disrupted due to the uneven distribution of occludin in the portions of seminiferous tubules, including the tight junctions. 40 Furthermore, enalapril intervention ameliorates the sperm DNA damage in Diab + Nico group. Consistent with this, it is noteworthy to mention that captopril, another ACE inhibitor, attenuates the nicotine-induced endothelial dysfunction in both in vivo and in vitro models. 47
Furthermore, western blot analysis showed that nicotine significantly increased the levels of NF-κB, COX-2, and TNF-α expression in diabetic testis, which was significantly decreased by the enalapril treatment. It has been reported that administration of nicotine significantly increased COX-2 expression and led to the activation of inflammatory mediators in the rat model of acute nephritis. 15 Furthermore, Lau et al. reported that chronic nicotine exposure increased the NF-κB expression in a hyperlipidemic low-density lipoprotein receptor (–/–) mouse model. 48 Barr et al. also reported that nicotine activates the NF-κB pathway through the reactive oxygen species generation and oxidative stress in rat mesencephalic cells. 49 It has already been reported that in diabetic condition, Ang II level is increased, which attributes a critical role in the induction of inflammation in somatic cells. 50,51 Moreover, Ang II plays a vital role in the reproductive function and in the germinal cells, it mitigated the effects by the local Ang II system. 52 Furthermore, it has been reported that nicotine enhances Ang II in vascular smooth muscle cell and fibroblast in vitro. 53 So it can apprehend that this mechanism might be responsible toward the exaggerated systemic effect of nicotine under the hyperglycemic condition, which can ultimately perturb the gonadal function. Furthermore, it can be emphasized that the ameliorative effect against nicotine induced germinal cell toxicity under hyperglycemic condition may be due to the systemic ACE inhibition and the reduction in oxidative stress as well as the modulation of testosterone level. Our earlier findings showed that enalapril significantly reduces the testicular DNA damage in diabetes. 26 In the present study, it has been reported that there was an additive effect of nicotine and diabetes on testicular DNA damage in STZ-induced diabetic rat and enalapril intervention ameliorates the same. More studies are needed at the molecular level to decipher the exact role of enalapril in the protection of testicular toxicity using animal models.
Footnotes
Funding
This work was financially supported by National Institute of Pharmaceutical Education and Research (NIPER), S.A.S. Nagar, Mohali, Punjab, India; also the Council of Scientific and Industrial Research (CSIR) awarded the Senior Research Fellowship (SRF) to complete the ongoing project (111368/2K11/1).
Conflict of Interest
The authors declared no conflict of interest.
