Abstract
Sulfur dioxide (SO2) is a common exogenous atmospheric pollutant. Studies have shown that SO2 can cause vasodilation as a gas signaling molecule, but the specific signaling pathways are not well understood. This study aimed to explore the underlying mechanism behind the effects of SO2 on vasodilation of isolated rat aorta. The results showed that when the dose of SO2 was 30 μM, the vasodilation of endothelium-intact rings was partially suppressed by LY294002 and NG-nitro-
Introduction
Sulfur dioxide (SO2) is a common toxic atmospheric pollutant. 1 At the same time, SO2 is a systemic toxin, which not only has obvious toxic effects on the cardiovascular system 2 but also has toxic effects on the central nervous system, digestive system, respiratory system, immune system, and reproductive system. 3 –5 Studies have shown that SO2 and its derivatives have a negative inotropic effect on rat hearts. 6 In addition to exogenous inhalation of SO2, it can be synthesized in humans and mammals and is an endogenous compound. 7,8 Studies have shown that mammals can synthesize SO2 endogenously through a variety of pathways, and the normal metabolism of the organism’s own sulfur-containing amino acids and drugs can produce SO2. 7,9 It was found that hydrogen sulfide can also be converted into thiosulfate by oxidation reaction and then sulfite or SO2 can be produced by reductase catalysis. 10 In addition, endogenous SO2 can also be produced in cardiovascular tissues. 11 A previous study showed that the physiological concentration of endogenous SO2 in normal rat thoracic aorta is 110.34 ± 35.22 μM. 12 In recent years, the research on the biological effects of SO2 mainly focused on the pathophysiological and physiological effects of endogenous SO2, including the decrease of blood pressure in rats, the relaxation of vascular rings, the decrease of cardiac contractility, and the regulation of pulmonary endothelial cell remodeling. 13 –15 Moreover, endogenous SO2 acts as a gas signaling molecule that plays a very important role in regulating vasodilation. 16 Previous studies in our group have shown that the physiological and low concentrations of SO2 caused by relaxation are related to big-conductance Ca2+-activated K+ (BKCa) channels, and the high concentration of SO2-induced relaxation may be related to L-type calcium (L-Ca2+) channel and ATP-sensitive K+ (KATP) channels. 16 Many ion channels and signaling pathways play a role in SO2-induced vasodilation, and the complex mechanisms behind it require further research.
The phosphoinositide 3-kinase (PI3K)-Akt pathway is linked to a variety of intracellular growth factors and toxic damage activation and is an important signal transduction pathway in cells. 17 There are multiple effector molecules downstream of PI3K and Akt is at the center of this pathway transmitting information initiated by PI3K; PI3K regulates cell survival by activating Akt to phosphorylate multiple downstream targets. 18 Endothelial nitric oxide synthase (eNOS) plays an important role in the synthesis of nitric oxide (NO). eNOS is essential for the regulation of vascular wall structure and function and participates in the pathophysiological process of cardiovascular disease. 19 Furthermore, it has been established that Akt can directly phosphorylate eNOS, thereby increasing its binding to intracellular calmodulin, activating eNOS, and promoting NO release. 20 NO is a free-diffusion signaling molecule in the body that regulates vasodilatation and protects endothelial cells. 21 We hypothesized that SO2 regulation of vasodilation is associated with the PI3K/Akt/eNOS and NO/cyclic guanosine monophosphate (cGMP) signaling pathways.
In this study, we investigated the relaxation effect of SO2 on thoracic aorta and examined the regulation of the PI3K/Akt/eNOS pathway and cytosolic cyclic adenosine monophosphate (cAMP) and cGMP activity in this effect. In addition, we also investigated the changes in caspase-3/-9 enzyme activity to observe whether SO2 would cause certain damage to blood vessels. Elucidating the molecular mechanism of SO2-induced vasodilation will provide an important experimental basis for the pathogenesis of cardiovascular diseases caused by SO2.
Materials and methods
SO2 stock solution preparation
Pure SO2 gas (purity: 99.9%) was obtained from the Beijing AP BAIF Gases Industry Co., Ltd (Beijing, China). High purity SO2 gas was collected with physiological saline for 2 min to prepare a fresh SO2 stock solution. The concentration of SO2 stock solution was measured according to the pararosaniline hydrochloride spectrophotometric method. 22 The dissolution of SO2 in physiological saline is a physical dissolution, which is mainly in the form of SO2 molecules. Therefore, gaseous SO2 can directly act on the isolated rat thoracic aorta through the solution.
Preparation of thoracic aorta ring
The method of preparing the rat thoracic aorta ring was conducted as previously described. 16,23 All animal experiments and care processes were approved and performed according to standards of the Animal Management Rule of the Animal Care Committee of Shanxi University.
Eight-week-old male Sprague Dawley rats (200–225 g) from the Academy of Military Medical Sciences (Beijing, China) were sacrificed by decapitation. The aortas were immediately excised and the adhering fat and superficial connective tissue was peeled away. Aortas were then cut transversally into ring-like segments approximately 2 mm long. The rings were rapidly inserted into organ baths with 5 ml of Krebs buffer (pH 7.4, 37°C) and bubbling continuously with mixed gas containing 95% O2 and 5% carbon dioxide (CO2). The preparation of the aortic rings should be done with care so as not to damage the endothelial cells. Rings can lose the endothelial cells by rubbing the intimal surface with forceps. The properties of endothelium cells were determined by the relaxation reactions of the rings against acetylcholine (10−6 M). JZ101 Force Sensor (Beijing, China) connected to MedLab Biological Signal Acquisition System (Nanjing, China) was used to record tension. Each aortic ring was given 1.5 g resting tension and equilibrated for 1 h, replacing the Krebs buffer liquid every 15 min. After achieving equilibrium, rings were treated with 60 mM potassium chloride and bath fluid was changed every 15 min until responses were stable (two times).
Vascular reactivity studies
After the vascular ring was stabilized, norepinephrine (NE, 10−6 M) was treated with the Krebs’ solution to peak the vasoconstriction and acetylcholine (10−6 M) was added to examine the integrity and viability of the vascular endothelium.
24
To explore whether the PI3K/Akt/eNOS pathway is associated with the changes in vascular tension induced by SO2, endothelium-intact and endothelium-denuded rings were preincubated with PI3K inhibitor 2-(4-morpholinyl)-8-phenylchromone (LY294002, 10 μM) or NOS inhibitor NG-nitro-
Tissue sample acquisition and real-time quantitative reverse transcription polymerase chain reaction
Male Sprague Dawley rats, weighing 200–225 g, were sacrificed by decapitation and the aortas were excised immediately. The connective tissue on the surface of vessels was removed and they were then cut into several segments and placed in six-well plates preloaded with Krebs buffer and bubbled with mixed gas containing 95% O2 and 5% CO2. Different doses (30, 300, or 1500 μM) of SO2 were added into the six-well plates and shaken at 37°C for 2 h. The SO2-treated aortic tissue was quickly frozen by liquid nitrogen and placed at −80°C for analysis.
The aortic tissue of rats was homogenized in glassware with TRIzol reagent after freezing. The concentration of RNA and OD260/OD280 ratio (1.8–2.2) was determined using NanoDropTM 2000C (Thermo, Waltham, MA, USA). The synthesis of the first-strand cDNA was in accordance with the manufacturer’s instructions. The quantification of mRNA expressions was determined by real-time polymerase chain reaction on an iQ iCycler (Bio-Rad, Hercules, California, USA). The target gene quantities were calculated by standard curve. Housekeeping gene β-actin was used as an internal control. To amplify specific gene products, the sequences of primers used were: PI3K (NM 005027), sense: 5′-CTTGCCTCCATTCACCACCTCT-3′, antisense: 5′- GCCTCTAATCTTCTCCCTCTCCTTC-3′, β-actin (NM 031144), sense: 5′-CCTATGCCAACACAGTGCTGTCT-3′, antisense: 5′-GCTCAGGAGGAGCAATGATCTTGA-3′.
Western blotting analysis
The protein contents of rat aortic extracts were determined according to the instructions of Beyotime Biotech Company (Shanghai, China) protein detection kit. The target proteins (β-actin, PI3K, Akt, eNOS, p-PI3K, p-Akt, p-eNOS) were separated by gel electrophoresis and transferred to nitrocellulose filter (NC) membranes. The membranes were treated with primary antibodies against PI3K (Cell Signaling Technology, Boston, MA, USA, Cat#: 4257), Akt (Cell Signaling Technology, Cat#: 4691), eNOS (Bioss, China, Cat#: bs-20608R), p-PI3K (p85; Abcam, Cambridge, MA, USA, Cat#: ab182651), p-Akt (Ser473; Cell Signaling Technology, Cat#: 4058), and p-eNOS (Bioss, Cat#: bs-3447R), β-actin (Cell Signaling Technology, Cat#: 4970) at 4°C overnight and incubated with fluorescently labeled secondary antibodies (Qcbio Biotech, Shanghai, China) for 90 min. Subsequent measurement of protein contents was conducted using a LI-COR Odyssey Infrared Fluorescent System (Lincoln, NE, USA).
Determination of total NOS, inducible NOS, constitutive NOS, and NO
Rat aortic homogenate was centrifuged, and the supernatant was collected. The level of total NOS (tNOS), inducible NOS (iNOS), constitutive NOS (cNOS), and NO in the supernatant was determined using commercial detection kits in accordance with the manufacturer’s instructions (Jiancheng Biological Company, Nanjing, China).
Assay for cAMP and cGMP activity
The rat aortic tissue was homogenized and then centrifuged for 10 min with a rotational speed of 3000 r min−1. The levels of cAMP and cGMP in the rat aortas were obtained using the enzyme linked immune sorbent assay kits (Westang Company, Shanghai, China) according to the manufacturer’s instructions.
Measurement of caspase-3 and caspase-9 enzymatic activity
Aortic tissue was homogenized to determine the activity of caspase-3 and -9. Precipitate was removed by centrifugation and the supernatant was collected to measure the caspase-3 and -9 enzymatic activity. The protein concentrations of the supernatants were estimated using Coomassie light blue according to the methods described by Bradford. 26 The activity of caspase-3 and caspase-9 was evaluated in a microplate reader using the caspase activity assay kit (Beyotime Biotech) according to the kit instructions.
Statistical analysis
One-way analysis of variance was used to determine the difference between groups, followed by a Tukey’s post hoc test. All values are presented as mean ± standard deviation (SD). A p value <0.05 was considered statistically significant.
Results
Aortic responses to SO2 in the presence of LY294002 or L-NAME
Preincubation of the LY294002 or L-NAME in the presence of the endothelium significantly reduced the aortic ring tension caused by SO2 at 30 μM (Figure 1(a)). In the endothelium-denuded groups, the vasodilation effect did not change aortic ring tension induced by SO2 after application of the inhibitor (Figure 1(b)).

Inhibitory effects of LY294002 and L-NAME on the SO2-induced vasodilatation in the endothelium-intact (a) or endothelium-denuded (b) rat aortas. The rat aortas were pretreated for 10 min by LY294002 (10 μM) and
Effects of SO2 on the expressions of PI3K of aortic rings
The level of PI3K mRNA in rat aortas treated with SO2 (30 μM) was significantly higher than that of the control group (Figure 2(a)). The expression of PI3K protein in rat aortas was similar to that of mRNA. The protein expression of PI3K significantly increased when treated with 30 μM SO2 (Figure 2(b)). Conversely, 300 μM of SO2 significantly decreased the PI3K mRNA and protein level in rat aortas (Figure 2(a)).

Effects of SO2 on mRNA and protein levels of PI3K (a and b) in the isolated rat aortas. Data are shown as mean ± SD (n = 6). *p < 0.05 versus control. SO2: sulfur dioxide; PI3K: phosphoinositide 3-kinase; SD: standard deviation.
Effects of SO2 on the expressions of p-Akt and p-eNOS of aortic rings
Figure 3 shows 30 μM SO2 resulted in increased phosphorylation of Akt and eNOS. However, rat aorta treated with 1500 μM SO2 showed a decrease in p-Akt expression compared with that of the control group (Figure 3(b)). Rat aorta treated with 300 μM SO2 showed a decrease in p-eNOS expression compared with that of the control group (Figure 3(c)).
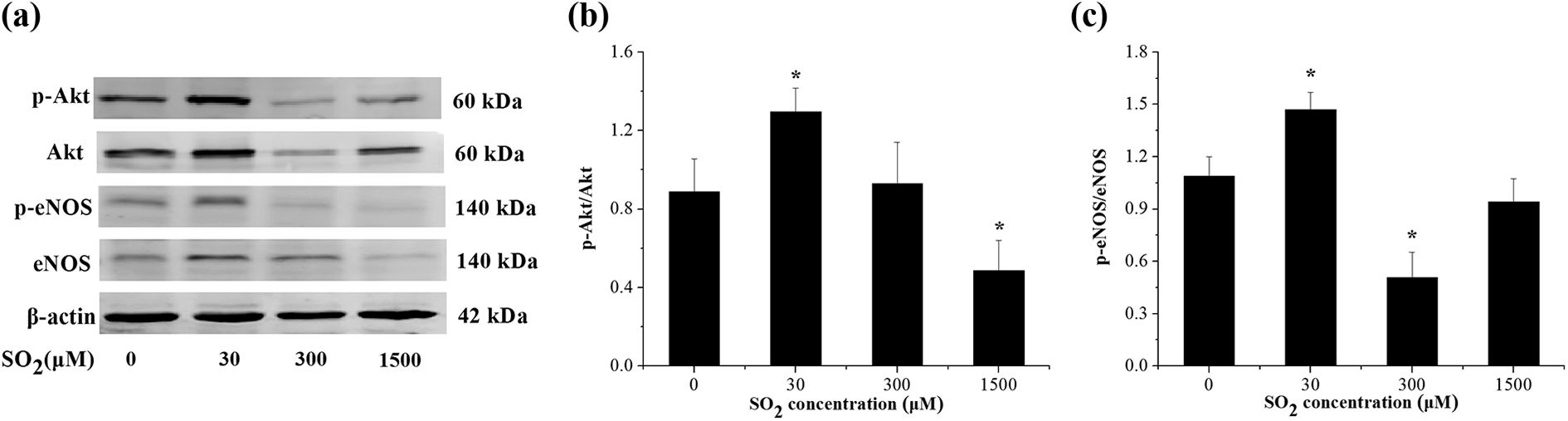
Effects of SO2 on protein levels of p-Akt and p-eNOS in the isolated rat aortas (a). The relative expression of p-Akt (b) and p-eNOS (c) were calculated and were shown, respectively. Data are shown as mean ± SD (n = 6). *p < 0.05 versus control. SO2: sulfur dioxide; eNOS: endothelial nitric oxide synthase; SD: standard deviation.
Effect of SO2 on tNOS, iNOS, and cNOS activity and NO content in isolated rat aorta
The effect of SO2 on tNOS, iNOS, and cNOS activity and NO levels in the aortic rings can be seen in Figure 4. SO2 enhanced the activity of tNOS, iNOS, and cNOS in a concentration-dependent pattern. The activity of tNOS, iNOS, and cNOS treated with 300 μM or 1500 μM SO2 was significantly higher than the control group (Figure 4(a) to (c)). The NO levels were significantly increased by SO2 in a concentration-dependent pattern compared with the control group (p < 0.05) (Figure 4(d)).

Effects of SO2 on tNOS (a), iNOS (b), cNOS (c) activity, and NO content (d) in the isolated rat aortas. Data are shown as mean ± SD (n = 6). *p < 0.05 versus control. SO2: sulfur dioxide; tNOS: total nitric oxide synthase; iNOS: inducible nitric oxide synthase; cNOS: constitutive nitric oxide synthase; NO: nitric oxide; SD: standard deviation.
Effect of SO2 on cAMP and cGMP activation in isolated rat aorta
Figure 5(a) shows that SO2 did not induce a statistically significant difference in cAMP. SO2 increased the level of cGMP following a bell-shaped dose–response relationship. At concentrations of 300 μM or 1500 μM, the level of cGMP increased significantly compared to the control group, with the maximum effect observed at 300 μM (Figure 5(b)).

Effects of SO2 on the contents of cAMP (a) and cGMP (b) in the isolated rat aortas. Data are shown as mean ± SD (n = 6). *p < 0.05 versus control. SO2: sulfur dioxide; cAMP: cyclic adenosine monophosphate; cGMP: cyclic guanosine monophosphate; SD: standard deviation.
Effect of SO2 on caspase-3 and caspase-9 activation in isolated rat aorta
As can be seen in Figure 6, 1500 μM of SO2 induced caspase-3 and caspase-9 activity significantly compared to the control group.

Effects of SO2 on the levels of caspase-3 (a) and caspase-9 (b) in the isolated rat aortas. Data are shown as mean ± SD (n = 6). *p < 0.05 versus control. SO2: sulfur dioxide; SD: standard deviation.
Discussion
SO2 is an endogenous gas that acts as a relaxation factor for vascular tissue. 16 Previous studies by our group have shown that SO2 causes a dose-dependent vasodilation in the range of 1–2000 μM. 27 When the concentration of SO2 is less than 450 μM, the resulting vasodilation is endothelium-dependent and this relaxation is associated with BKCa channels. 16 When the concentration of SO2 is greater than 500 μM, the vasodilation is non-endothelium-dependent, and this relaxation is associated with L-Ca2+ channels and KATP channels. 16 The treatment dose in the current study was based on the half-maximum effective concentration (EC50); The EC50 of SO2 causing vasodilation was 1247.38 ± 98.32 μM. 16 Therefore, we chose 30, 300, and 1500 μM SO2 as the dose in this study.
It is well documented that the regulation of vascular tone involves the PI3K/Akt/eNOS cascade. 28 –30 PI3K can activate Akt and phosphorylate eNOS, leading to NO production and ultimately vascular relaxation. 28 To explore whether the PI3K/Akt/eNOS pathway is related to the changes in vascular tension induced by SO2, rat aortic rings were preincubated with LY294002 or L-NAME. The results in the endothelium-intact group showed that the vasodilation effects of SO2 were partially suppressed by the preincubation in LY294002 or L-NAME. These observations suggest that PI3K/Akt/eNOS cascade is involved in the vasodilation caused by SO2 in the aorta.
The PI3K/Akt signaling pathway is associated with cardiac and vascular health. 29,31 In our study, the mRNA and protein levels of PI3K were significantly increased by 30 μM SO2 (Figure 2). PI3K can phosphorylate the Ser473 site of downstream Akt, which leads to activation of the kinase and mediates signaling processes associated with cell migration and growth. 18 It is also involved in endothelium-dependent relaxation. 30 Zhao et al.’s study revealed that in a myocardial ischemia–reperfusion injury animal model, pretreatment with SO2 has a protective effect and this effect was related to PI3K/Akt signaling pathway. 32 In addition, eNOS is a downstream regulator of PI3K and the serine/threonine protein kinase Akt. 33 As a rate-limiting enzyme for the synthesis of NO, eNOS plays a key role in maintaining cardiovascular physiological functions. 34 –36 Furthermore, eNOS leads to NO production by activating PI3K/Akt-mediated phosphorylation. 37 Alterations to eNOS activity can lead to reduce NO synthesis or activity, induction of oxidative stress, increased vascular resistance, and endothelial dysfunction. 38,20 Results from this study show that SO2 (30 μM) significantly upregulated the levels of p-Akt and p-eNOS and increased the release of NO (Figure 3(b), 3(c), and Figure 4(d)). This suggests that the mechanism of the SO2-induced vasodilatation may be influenced by the PI3K/Akt/eNOS pathway. The findings also support the hypothesis that SO2 causes vasodilation by activating the PI3K/Akt/eNOS and NO/cGMP pathway. The results showed that SO2 (300 or 1500 μM) caused a decrease in the expression of PI3K, p-Akt, and p-eNOS. However, specific vasodilation mechanisms require further investigation.
The vascular endothelium plays an important role in maintaining intravascular environmental stability and vascular function by synthesizing and releasing vasoactive substances and cytokines.
39
–41
Normal vascular endothelium has various physiological functions such as regulating vascular tone, maintaining the balance of the body’s coagulation and fibrinolysis system, and regulating the growth and antioxidation of vascular smooth muscle.
42
NO is a free-diffusion signaling molecule in the body that regulates vasodilatation, inhibits platelet aggregation and leukocyte adhesion, and protects endothelial cells.
43
It has been reported that endothelium-derived NO is synthesized by endothelial cells under the action of NOS using
Caspase is an important factor in apoptosis and is involved in the network of apoptotic mechanisms. 46 It has been shown to be associated with central nervous system damage, skin damage and other visceral injuries, and inflammatory responses. 47 –49 In this article, we speculate that high concentrations of SO2 may cause damage to the aorta, so we measured the activity of caspase-3 and caspase-9 in 30, 300, and 1500 μM SO2-treated rat aortic tissues. The results showed that SO2 significantly increased the levels of caspase-3 and caspase-9 compared to the control group at a concentration of 1500 μM. These data suggest that SO2 may induce tissue damage through the cysteine pathway, and specific sites of action needs further study.
In conclusion, results suggest that SO2 promotes vasodilation via the PI3K/Akt/eNOS and NO/cGMP pathway. Additionally, high concentrations of SO2 have a detrimental effect on rat aorta. A greater understanding of the impacts of SO2 on cardiovascular relaxation and the underlying mechanisms would further inform the etiologies of cardiovascular disease caused by SO2.
In addition, our research also has certain limitations. Results indicate that vasodilation caused by SO2 at high doses was related to the NO/cGMP signaling pathway; however, our previous studies also found that it was involved in L-Ca2+ channels and KATP channels. 16 We cannot judge which one plays the main role. The relationship between them is unclear and needs further research. Furthermore, our research object was isolated aortic blood vessels from rats. If we can conduct research on human blood vessels in the next step, this will be of great significance.
Supplemental material
Supplementary_material - Sulfur dioxide induces vascular relaxation through PI3K/Akt/eNOS and NO/cGMP signaling pathways in rats
Supplementary_material for Sulfur dioxide induces vascular relaxation through PI3K/Akt/eNOS and NO/cGMP signaling pathways in rats by Q Zhang, W Lyu, M Yu and Y Niu in Human & Experimental Toxicology
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China [Grant Numbers 21777090 and 21107064], by the Natural Science Foundation of Shanxi Province [Grant Number 2016011082], and by Program for the Outstanding Innovative Teams of Higher Learning Institutions of Shanxi.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
