Abstract
Sepsis is one of the most common causes of death among hospitalized patients. Activity of xanthine oxidase (XO), a reactive oxygen species-producing enzyme, is known to be elevated in septic patients. Our aim was to investigate the possible protective role of XO inhibitor, febuxostat (FEB), in a rat model of sepsis-induced liver and kidney injures. Adult male albino rats were divided into four groups (n = 12 each): sham control, sham + FEB, cecal ligation and puncture (CLP), and CLP + FEB groups. FEB (10 mg/kg per os (p.o.)) was given once daily for 2 days and 30 min prior to laparotomy with CLP. CLP was associated with a high mortality rate accompanied by significant liver and kidney injuries indicated by elevated serum alanine aminotransferase, aspartate aminotransferase, urea, and creatinine levels and confirmed by histopathological tissue injury. Moreover, there was an increase in neutrophil gelatinase-associated lipocalin, uric acid, malondialdehyde, and nitric oxide levels and with decreased superoxide dismutase activity and total antioxidant capacity. In addition, CLP caused increased expression of the inflammatory markers tumor necrosis factor alpha, interleukin 1beta protein levels, and nuclear factor kappa B immunoexpression. Finally, CLP operated rats exhibited an upregulation in the apoptotic mediators, caspase 3, and P-C-Jun N-terminal kinases (JNK) proteins. FEB treatment of CLP rats caused a significant improvement and normalization in all measured parameters. Moreover, FEB amerliorates degenerative histopathological changes and improves the overall survival rate. In conclusion, FEB exhibited a protective effect in sepsis-induced liver and kidney injuries most probably through its anti-inflammatory, antioxidant, and antiapoptotic properties and attenuating JNK signaling pathway secondary to its XO enzyme inhibitory activity.
Introduction
Sepsis is a life-threatening condition known as the systemic inflammatory response to infection. Early recognition and diagnosis of sepsis is required to prevent the occurrence of septic shock, which is associated with a mortality rate of 40% or more. 1 Among the most affected organs in septic patients are lung, liver, kidney, heart, and intestine. 2 The mortality rate that is reported as a result of acute liver injury (ALI) and acute kidney injury (AKI) accounts for 90% and 50%, respectively. 3,4
Although sepsis is the most common trigger of ALI and AKI, the underlying mechanisms are not entirely identified. Previous studies have detected that the pathophysiology of ALI and AKI in sepsis is complex and multifactorial, including oxidative stress, inflammatory pathways, endothelial dysfunction, and microcirculatory disorders. 5 –7 The oxidative stress inherent to sepsis has been considered as a direct mechanism of ALI and AKI. 8,9 In sepsis, oxidative stress occurs due to excessive production of reactive oxygen species (ROS) and decrease in endogenous antioxidants. There are many sources of ROS in sepsis including xanthine oxidase (XO) enzyme activation which is responsible for remote organ injury. 10
The enzyme XO stimulates the formation of uric acid with the coproduction of superoxide radicals, which results in activation of neutrophils and their adherence to endothelial cells, which activates the formation of XO in the endothelium, with further production of superoxide radicals. 10
The increase in ROS leads to oxidation of DNA and protein leading to alterations in membrane permeability, modification of protein structure, and function. In addition, oxidative damage to the mitochondrial membrane can occur and cause membrane depolarization and the uncoupling of oxidative phosphorylation with altered cellular respiration. This can lead to mitochondrial damage with activation of c-Jun N-terminal kinases (JNK) pathway. 10
Moreover, activation of JNK by oxidative stress causes mitochondrial release of cytochrome c into the cytoplasm leading to caspase activation and apoptosis with subsequent organ failure. 11 Also, JNK activation in epithelial cells (e.g. in the injured kidney or liver) can recruit and activate leukocyte populations via chemokine and cytokine production. It can directly activate nuclear factor kappa B (NF-κB), which is a major factor in initiating transcription of genes involved in inflammatory responses. 12 Thus, JNK activation has the potential to promote organ injury in sepsis through its pro-apoptotic and pro-inflammatory actions.
Different cytokines are released during sepsis and they are classified as pro-inflammatory and anti-inflammatory agents. Pro-inflammatory cytokines play important roles in the induction of systemic inflammation and development of multiple organ failure in such cases. Tumor necrosis factor alpha (TNF-α) and interleukin 1beta (IL-1β) are essential pro-inflammatory cytokines detected in sepsis. 2
Moreover, NF-κB is highly involved in the pathophysiology of sepsis. 13 Inhibition of NF-κB has been shown to improve survival in an animal model of polymicrobial sepsis. 14
Different pathways are involved in sepsis and the essential role of XO, IL-1β, JNK, and NF-κB directed our attention to study the possible protective effect of XO inhibitor, febuxostat (FEB) in cecal ligation and puncture (CLP)-induced sepsis.
Materials and methods
Chemicals
FEB powder was obtained from GlobalNapi Co. (Egypt). Neutrophil gelatinase-associated lipocalin (NGAL) enzyme-linked immunosorbent assay (ELISA) kit (catalog # E-EL-R0662) was obtained from Elabscience Co. (China). TNF-α ELISA kit (catalog # KRC3011), IL1β (catalog # PA5-47480), caspase 3 (catalog # PA1-26426), total JNK (catalog # MAB1387), phosphorylated JNK (catalog # MAB1205), and mouse monoclonal anti-β actin (catalog # MAB8929) antibodies were obtained from Thermo Fisher Scientific Co. (Waltham, MA, USA). Total antioxidant capacity (TAC) kit was obtained from Biodiagnostic Co. (Egypt). Aspartate aminotransferase (AST), alanine aminotransferase (ALT), creatinine, urea, and uric acid kits were obtained from Biomed Co. (Egypt).
Animals and experimental design
The present study was conducted on adult albino male Wistar rats weighing 200–250 g. Rats were obtained from the National Research Center (Giza, Egypt). Rats were fed a standard diet of commercial rat chow and tap water and left to acclimatize to the environment for 2 weeks to adapt to the environment before being divided, randomly, into four groups. The research was performed in accordance with the guide for the Care and Use of Laboratory Animals of the National Institutes of Health. The study protocol was approved by the Committee for the Care and Use of Laboratory Animals of the Faculty of Medicine, Minia University. Approval number is 157:2/2019.
Rats were randomly divided into four groups (n = 6 each). Group I was the sham group. It has underwent laparotomy without CLP. Group II was the sham + FEB group. It was supplemented with FEB (10 mg/kg per os (p.o.) once daily) for 2 days and 30 min prior to laparotomy without CLP. 15 Group III was the CLP group. It has underwent laparotomy with CLP. 16 Group IV was the CLP + FEB group. It was supplemented with FEB (10 mg/kg p.o. once daily) for 2 days and 30 min prior to laparotomy with CLP. Rats were euthanized 24 h after CLP operation, with no prior fasting before euthanization, when tissues and blood samples were collected.
A survival study was conducted in parallel to test the effect of FEB on survival. Twenty-four rats (same weight and sex as previously described) were randomly divided into four experimental groups (n = 6 per group): sham operated, sham operated plus FEB (10 mg/kg) group, untreated septic group, and septic rats treated with FEB (10 mg/kg). The mortality of rats was observed daily for 2 weeks.
The surgical procedures
The animals were anesthetized with ketamine (50 mg/kg body weight intraperitoneally (i.p.)) and xylazine (5 mg/kg body weight i.p.). The abdomen was shaved, and the peritoneum was opened. The cecum was ligated with a 3/0 silk ligature just distal to the ileocecal valve. Two punctures were made with a 22-gauge needle through the cecum distal to the point of ligation. The abdominal incision was then closed with a 4/0 sterile synthetic absorbable suture. The sham-operated group underwent laparotomies, and the rats’ ceca were manipulated but not ligated or perforated. All the animals were given 3 mL/100 g body weight of normal saline subcutaneously at the time of surgery and 6 h afterward for fluid resuscitation. 16
Blood and tissue collection and serum biochemistry
The blood samples were collected from neck jugular veins 24 h after CLP and rats were not fasted before blood collection. Blood samples were centrifuged at 5000 r/min for 10 min for serum collection using centrifuge (Jantezki, T30, Germany). Sera of animals were stored at −80°C for measurements of AST, ALT, creatinine, NGAL, TAC, and uric acid according to manufacturer’s instructions.
The collected samples of blood, liver, and kidney were carefully dissected, washed, and divided into two parts: the left parts of the liver and kidney were used for histopathological examination, while the right parts of the liver and kidney were homogenized and frozen to assess the different biochemical markers.
Determination of hepatic and renal oxidative stress and antioxidant defense parameters
The hepatic and renal malondialdehyde (MDA) is the breakdown product of lipid peroxides. The content of lipid peroxides in liver and kidney was determined using previously described thiobarbituric acid method, 17 which measures the thiobarbituric acid reactive substance concentration that refers to MDA concentration.
Evaluation of the antioxidant activity of liver and kidney was done by measuring the activity of superoxide dismutase (SOD) and TAC. The evaluation of SOD activity depends on the fact that the oxidation of pyrogallol is inhibited by SOD. One unit of SOD is generally defined as the amount of enzyme that inhibits the oxidation of pyrogallol by 50%. The activity of SOD was measured at 420 nm and expressed as U/g tissue. 17 The determination of the antioxidative capacity was performed by the reaction of antioxidants in the sample with a defined amount of exogenously added hydrogen peroxide (H2O2). The antioxidants in the sample eliminated a certain amount of the provided H2O2. The residual H2O2 was determined colorimetrically by an enzymatic reaction which involved the conversion of 3,5, dichloro-2-hydroxybenzensulphonate to a colored product according to the method previously described. 18
Western blot analysis of IL-1β, caspase 3, and JNK pathway
For estimation of IL-1B, caspase 3, total and phosphorylated JNK expression in hepatic and renal tissues, Western blotting was performed as previously described. 19 Briefly, hepatic or renal tissues were homogenized, total protein was estimated and then a volume equivalent to an equal content of total protein from each tissue homogenate was loaded and separated on a 10 % sodium dodecyl sulfate polyacrylamide gel electrophoresis gel. Protein bands were transferred to a nitrocellulose membrane using a semi-dry blotter (Bio-Rad). The blot was subsequently blocked with TBS containing Tween 20 buffer containing 5% skim milk powder for 1 h at room temperature. Then, the blot was probed overnight at 4°C with antibodies for IL-1β, caspase 3, total and phosphorylated JNK or mouse monoclonal anti-β actin antibodies (Thermo Fisher Scientific Co., USA). Subsequently, incubation with alkaline phosphatase-coupled secondary antibody (Sigma–Aldrich Co., St. Louis, MO, USA) was performed for 1 h at room temperature. Blots were finally analyzed using 5-bromo-4-chloro-3-indolyl phosphate/nitro blue tetrazolium colorimetric detection method. Protein bands on the blots were analyzed using Image-J and Prism-6 programs.
Histopathology
Liver and kidney specimens were fixed in 10% formalin solution then dehydrated in ascending grades of alcohol (70%, 95%, and 99% 2 min each) and embedded in paraffin, cleared in xylene, and paraffin-embedded. Five-micrometer-thick sections were prepared from different animal groups and stained with hematoxylin and eosin (H&E). The pathologist examined the sections under the light microscope. Hepatic histopathological changes were scored as grade 0 is minimal or no evidence of injury; grade 1 is mild injury characterized by cytoplasmic vacuolization and focal nuclear pyknosis; grade 2 is moderate injury exhibiting cytoplasmic vacuolization but no frank necrosis, sinusoidal dilatation, and congestion with blurring of intercellular borders; grade 3 is moderate to severe injury with areas of coagulative necrosis, cytoplasmic hypereosinophilia, extensive sinusoidal dilatation, and congestion; and grade 4 is severe injury consisting of severe confluent coagulative necrosis and hemorrhage into hepatic cords leading to a loss of tissue architecture. 20 Renal histopathological changes were graded as 0, none; 1, mild; 2, moderate; and 3, severe for the following changes: degeneration of Bowman’s space and glomeruli, degeneration of proximal and distal tubules, vascular congestion, and interstitial edema. 21
Immunohistochemical assessment of liver and kidney NF-κB p65
Five-micrometer-thick sections were prepared from kidney and liver specimens of different animal groups. Sections were deparaffinized in xylene and rehydrated in descending grades of 99% alcohol for 30 s each. Endogenous peroxidase activity was blocked by applying slides to 0.3% H2O2 in methanol for 12 min at room temperature and then rinsed in running tap water for 30 s. Slides were put in microwave (80°C) in citrate buffer (1.92 g citric acid added to 1 L of distilled water and pH was adjusted to 6.0) for 20 min. They were allowed to cool, reach room temperature, and then washed with distilled water. Subsequently, slides were incubated at 4°C overnight with the primary antibody for NF-κB then rinsed well in distilled water and secondary biotinylated antibody was added for each slide for 30 min at room temperature. Slides were incubated for additional 10 min with streptavidin peroxidase and finally with 3,3′-diaminobenzidine (DAB) plus chromogen for 15 min. Slides were then left till brown color appears or until 15 min at room temperature pass. Slides were rinsed with distilled water, sections were counterstained in hematoxylin, rinsed gently in distilled water, dehydrated in ascending grades of alcohols (70%, 95%, and 100% alcohol), then cleared in xylene, mounted using an aqueous-based mounting medium and covered. Negative control slide was treated by phosphate-buffered saline substituting NF-κB. Positive control sections were included as appropriate. 22
Scoring of immunohistochemistry
NF-κB semiquantitative scoring was done by determining immunoreactivity under light microscope with a magnification of 100×. The extent of staining was scored as 0 (0%), 1 (1–25%), 2 (26–50%), 3 (51–75%), and 4 (76–100%) according to the percentage of positively stained cells of NF-κB. 23
Statistical analysis of the data
Results were expressed as means ± SEM. Statistical comparisons were performed by one-way analysis of variance (ANOVA), followed by Tukey–Kramer as a post-ANOVA test. Differences with a p value of <0.05 were considered significant. GraphPad Prism was used for statistical calculations (version 5.01 for Windows, Graph pad Software, San Diego, California, USA).
Results
Effect of FEB on survival rate
We evaluated the rat survival along 2 weeks of observation. Sham control rats exhibited survival by 100%. Twenty-four hours after CLP, 75% of septic rats died, and after 48 h the remaining septic rats died. In contrast, FEB treatment dramatically increased the survival rate by 65% (p < 0.001) compared to CLP group (Figure 1).

Evaluation of survival rate. FEB treatment markedly improved the survival of septic rats. It was administered to rats as 10 mg/kg p.o. once daily for 2 days and 30 min prior to laparotomy with CLP. Mortality was monitored daily for 2 weeks. Data are presented as the survival percentage of animals (n = 6 rats per group). Results are considered significantly different when *p < 0.05: significantly different from sham group; # p < 0.05: significantly different from CLP group. FEB: febuxostat; CLP: cecal ligation and puncture; p.o.: per os.
Protective effect of FEB against sepsis induced ALI
Effect on AST and ALT
Induction of sepsis by CLP caused a significant increase in serum AST and ALT compared to the sham group. Coadministration of FEB caused a significant decrease in their levels compared to the CLP group (Table 1).
Effect of FEB on serum AST and ALT.a

FEB: febuxostat; CLP: cecal ligation and puncture; SEM: standard error of the mean.
a Values represent the mean ± SEM (n = 6).
b Results are considered significantly different when p < 0.05 is significantly different from sham group.
c Results are considered significantly different when p < 0.05 is significantly different from CLP group.
Effect on hepatic histopathology
Histological study of hepatic tissue of sham and sham + FEB groups showed normal lobular architecture, no parenchymal injury, and normal portal tract. CLP group showed features of severe hepatocellular injury with marked loss of architecture, severe periportal inflammation, sinusoidal congestion, and necroinflammatory foci of hepatocytes. CLP + FEB group showed significant decrease in hepatocellular injury induced by CLP with mild periportal inflammation and sinusoidal congestion (Figure 2).

Photomicrograph showing softransverse sections in rat liver. (a and b) Sham and sham + FEB groups, respectively, showed normal architecture of lobules, no parenchymal injury, and normal portal tract. (c) CLP group showed features of severe hepatocellular injury with marked loss of architecture, severe periportal inflammation, sinusoidal congestion, and necroinflammatory foci of hepatocytes. CLP + FEB group showed a significant decrease in hepatocellular injury induced by CLP with mild periportal inflammation and sinusoidal congestion (H&E, ×40). Black arrow refers to portal tract inflammation, blue arrow refers to sinusoidal congestion, yellow arrow refers to hepatocyte degeneration, and green arrow refers to necroinflammatory foci of hepatocytes. FEB: febuxostat; CLP: cecal ligation and puncture; H&E: hematoxylin and eosin.
Grading of histopathological findings in liver
Both sham and sham + FEB groups were graded as grade 0. While CLP group was graded as grade 3 or 4 and CLP + FEB was graded as grade 1 or 2 (Table 2).
Semiquantitative analysis of histopathological score in liver.a

FEB: febuxostat; CLP: cecal ligation and puncture; SEM: standard error of the mean.
a Values represent the mean ± SEM (n = 6).
b Results are considered significantly different when p < 0.05 is significantly different from sham group.
c Results are considered significantly different when p < 0.05 is significantly different from CLP group.
Protective effect of FEB against sepsis-induced AKI
Effect on serum creatinine and NGAL
CLP group showed a significant increase in serum creatinine and NGAL levels compared to the sham group, while the CLP + FEB group showed a significant decrease in their levels compared to the CLP group.
Effect on renal histopathology
Histological study of the rat renal cortical tissue of sham and sham + FEB groups showed normal architecture; numerous renal corpuscles, proximal and distal convoluted tubules and kidney vasculature. CLP group showed marked impaired renal morphology with severe Bowman’s capsule degeneration, proximal and distal tubules degeneration, and interstitial edema. CLP + FEB group showed a significant decrease in CLP-induced renal damage with mild tubular degeneration and mild interstitial edema (Figure 3).

Photomicrograph softransverse sections in kidney. (a and b) Sham and sham + FEB groups, respectively, showed normal architecture; numerous renal corpuscles, PCTs, and distal convoluted tubules and kidney vasculature. (c) CLP group showed marked impaired renal morphology with severe Bowman’s capsule degeneration, PCT and DCT degeneration, and marked interstitial edema. (d) CLP + FEB group showed a significant decrease in CLP-induced renal damage with mild tubular degeneration and mild interstitial edema (H&E, ×40). Red arrow refers to Bowman’s capsule degeneration and glomerular membrane degeneration; yellow arrow refers to PCT and DCT degeneration; black arrow refers to interstitial edema; and blue arrow refers to interstitial congestion. FEB: febuxostat; CLP: cecal ligation and puncture; PCT: proximal convoluted tubule; DCT: distal convoluted tubule; H&E: hematoxylin and eosin.
Grading of histopathological findings in kidney
Both sham and sham + FEB groups were graded as grade 0. While CLP group was graded as grade 3 or 4 and CLP + FEB was graded as grade 1 or 2 (Table 3).
Semiquantitative analysis of histopathological score in kidney.a
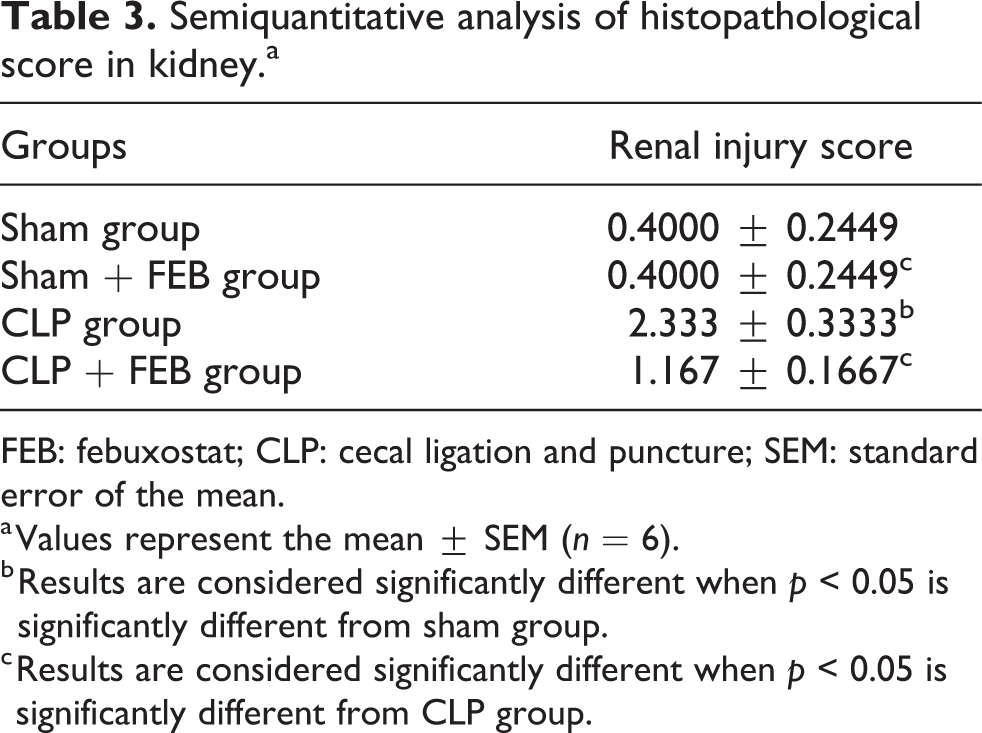
FEB: febuxostat; CLP: cecal ligation and puncture; SEM: standard error of the mean.
a Values represent the mean ± SEM (n = 6).
b Results are considered significantly different when p < 0.05 is significantly different from sham group.
c Results are considered significantly different when p < 0.05 is significantly different from CLP group.
FEB improves hepatic and renal oxidative and antioxidant defense profile in septic rats
In liver tissues
As shown in Table 4, CLP group produced a significant increase in hepatic tissue MDA and a significant decrease in serum TAC and hepatic SOD activity compared to sham group. On the other hand, CLP + FEB group produced a significant decrease in MDA and a significant increase in TAC and SOD compared to CLP group.
Effect of FEB on hepatic MDA and SOD and serum TAC.a

FEB: febuxostat; CLP: cecal ligation and puncture; SEM: standard error of the mean.
a Values represent the mean ± SEM (n = 6).
b Results are considered significantly different when p < 0.05 is significantly different from sham group.
c Results are considered significantly different when p < 0.05 is significantly different from CLP group.
In kidney tissues
As shown in Table 5, CLP group produced a significant increase in renal tissue MDA and a significant decrease in renal tissue SOD activity compared to sham group. On the other hand, CLP + FEB group produced a significant decrease in MDA and a significant increase in SOD activity compared to CLP group.
Effect of FEB on renal tissue MDA and SOD.a

FEB: febuxostat; CLP: cecal ligation and puncture; SEM: standard error of the mean.
a Values represent the mean ± SEM (n = 6).
b Results are considered significantly different when p < 0.05 is significantly different from sham group.
c Results are considered significantly different when p < 0.05 is significantly different from CLP group.
FEB attenuates inflammation: effect on uric acid, IL-1β, and NF-κB expression in hepatic and renal tissues in septic rats
Effect on serum uric acid level
Induction of sepsis by CLP caused a significant increase in serum uric acid level. While coadministration of FEB caused a significant decrease in its levels (Figure 4).

Semiquantitative analysis of effect of FEB on serum uric acid. CLP group showed a significant increase in serum uric acid level compared to sham group. CLP + FEB group showed a significant decrease in its level compared to CLP group. Values represent the mean ± SEM (n = 6). Results are considered significantly different when *p < 0.05 is significantly different from sham group and # p < 0.05 significantly different from CLP group. FEB: febuxostat; CLP: cecal ligation and puncture; SEM: standard error of the mean.
Effect on IL-1β expression
As shown in Figure 5(a) to (c), the CLP group produced a significant increase in expression of hepatic and renal tissue IL-1β protein compared to the sham group. On the other hand, the CLP + FEB group showed a significant decrease in its expression compared to the CLP group.

Western blot analysis of (a) hepatic IL-1β and (b) renal IL-1β. (c) Semiquantitative analysis of Western blot of hepatic and renal IL-1β. Values represent the mean ± SEM (n = 6). Results are considered significantly different when *p < 0.05 is significantly different from sham group and # p < 0.05 is significantly different from CLP group. IL-1β: interleukin 1beta; FEB: febuxostat; CLP: cecal ligation and puncture.
Effect of FEB on NF-κB expression
The data represented in Figures 6 and 7 reveal the immunohistochemical expression of NF-κB in liver and kidney in the form of brown staining. Its expression was significantly increased in the CLP group compared with the sham group, while the CLP + FEB group showed a significant decrease in its expression compared with the CLP group.

Immunohistochemical expression of NF-κB in rat liver. (a) and (b) Sham and sham + FEB groups, respectively, showed negative expression of NF-κB. (c) CLP group showed more than 70% positively stained cells with NF-κB. (d) CLP + FEB group showed less than 20% positively stained cells in five high power field. NF-κB: nuclear factor kappa B; FEB: febuxostat; CLP: cecal ligation and puncture.

Immunohistochemical expression of NF-κB in rat kidney. (a) and (b) Sham and sham + FEB groups, respectively, showed negative expression of NF-κB. (c) CLP group showed more than 75% positively stained cells with NF-κB. (d) CLP + FEB group showed less than 25% positively stained cells in five high power fields. NF-κB: nuclear factor kappa B; FEB: febuxostat; CLP: cecal ligation and puncture.
Grading of immunohistochemistry of NF-κB
Both sham and sham + FEB groups were graded as grade 0, CLP group was graded as grade 3 or 4, and CLP + FEB group was graded as grade 1 or 2 (Figure 8).

Semiquantitative analysis of immunohistochemical score of NF-κB in rat (a) liver and (b) kidney. Its expression significantly increased in CLP group compared with the sham group. While the CLP + FEB group showed significant decrease in its expression compared with the CLP group. Values represent the mean ± SEM (n = 6). Results are considered significantly different when *p < 0.05 is significantly different from sham group and # p < 0.05 is significantly different from CLP group. NF-κB: nuclear factor kappa B; FEB: febuxostat; CLP: cecal ligation and puncture.
FEB reduces apoptosis: effect on caspase 3 expression in hepatic and renal tissues of septic rats
As shown in Figure 9(a) to (d), the CLP group produced a significant increase in the expression of hepatic and renal caspase 3 compared to the sham group. On the other hand, the CLP + FEB group showed a significant decrease in its expression compared to the CLP group.
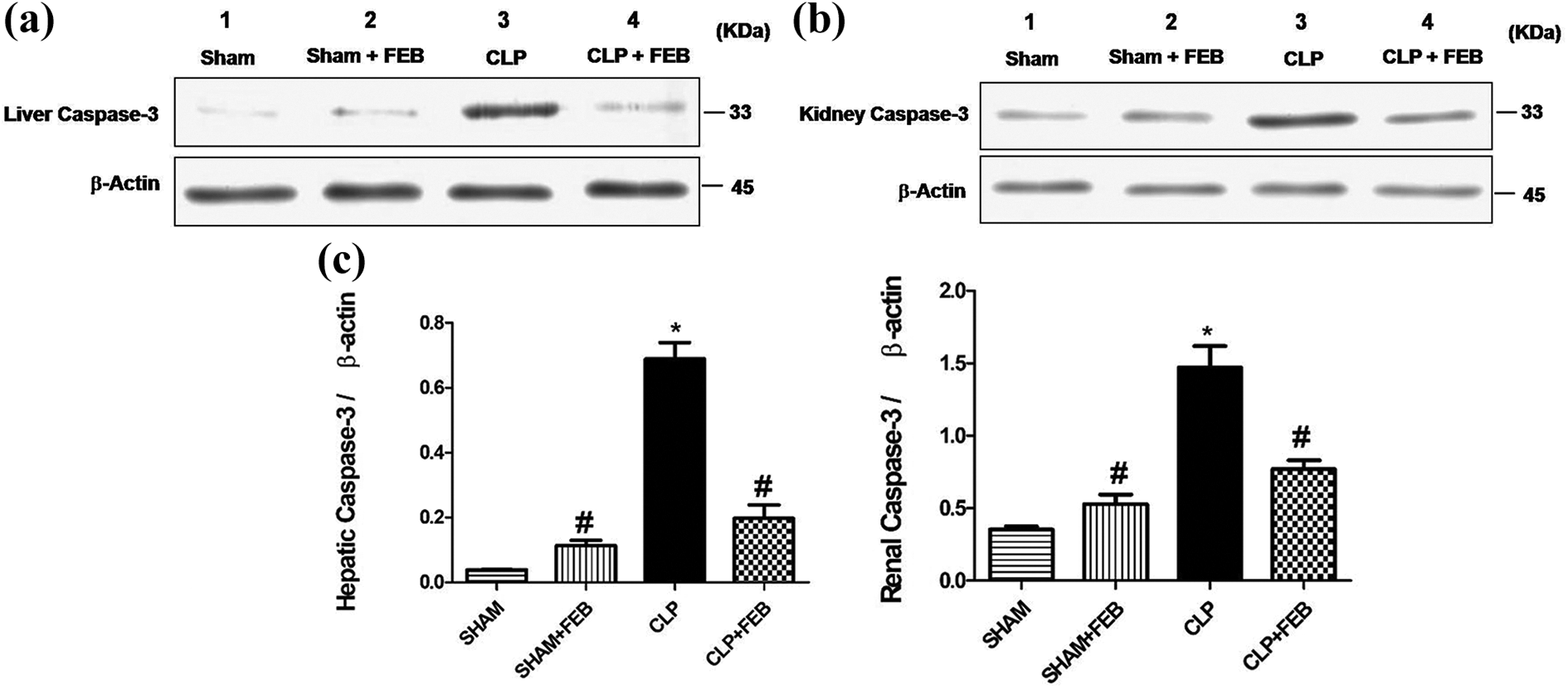
Western blot analysis of (a) hepatic and (b) renal caspase 3. Semiquantitative analysis of Western blot of (c) hepatic and (d) renal caspase 3 showed a significant increase in its expression in the CLP group compared to the sham group. CLP + FEB group showed a significant decrease in its expression compared to the CLP group. Values represent the mean ± SEM (n = 6). Results are considered significantly different when *p < 0.05 is significantly different from sham group and # p < 0.05 significantly different from CLP group. FEB: febuxostat; CLP: cecal ligation and puncture; SEM: standard error of the mean.
FEB attenuation of ALI and AKI in septic rats was mediated by suppressing JNK signaling pathway
As shown in Figures 10 and 11, induction of sepsis by CLP produced a significant increase in the expression of hepatic and renal JNK compared to the sham group. On the other hand, the CLP + FEB group showed a significant decrease in its expression compared to the CLP group.
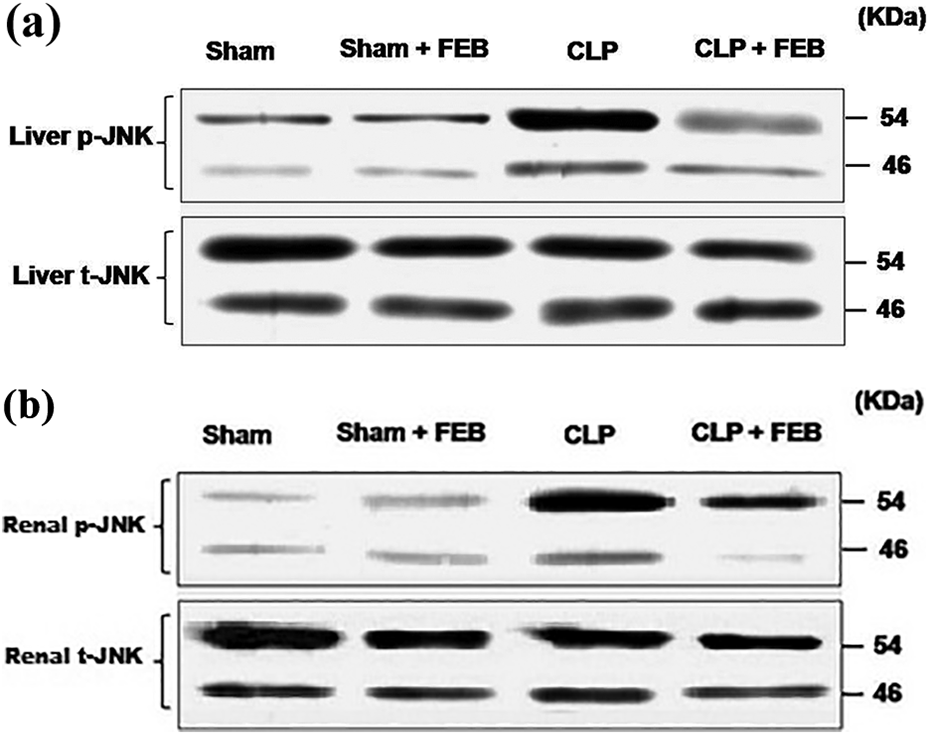
Western blot analysis of hepatic (a) and renal (b) JNK.

Statistical analysis of Western blot of (a) hepatic and (b) renal JNK. Semiquantitative analysis of Western blot of hepatic and renal JNK showed a significant increase in its expression in the CLP group compared to sham group. The CLP + FEB group showed a significant decrease in its expression compared to the CLP group. Values represent the mean ± SEM (n = 6). Results are considered significantly different when *p < 0.05 is significantly different from the sham group and # p < 0.05 is significantly different from CLP group.
Discussion
Sepsis remains a serious worldwide medical problem associated with high mortality rates. Although several experimental models replicate to a various extent the clinical changes and alterations of laboratory parameters observed in sepsis, 24 –26 in our study we chose CLP model, which is one of the perfect models to illustrate these changes due to its greatest compatibility with clinical settings.
The pathophysiological mechanisms of organ dysfunction observed in sepsis are believed to be related to systemic inflammatory response, tissue hypoperfusion associated with hypotension, and disseminated intravascular coagulation. 27 The most affected organs in septic patients are lung, liver, kidney, heart, and intestine. In present study, we aimed to evaluate the apoptotic and inflammatory changes associated with experimental induced sepsis using CLP model and their effects on ALI and AKI in septic rats.
The major finding in this study is that FEB could attenuate liver and kidney injuries in a rat model of sepsis induced by CLP as we found that its administration dramatically increased survival rate of septic rats, decreased oxidative stress, and increased the antioxidant enzyme capacity probably through inhibition of XO enzyme. Moreover, FEB decreased the production of inflammatory mediators such as IL-1β, NF-κB, and the production of apoptotic factors such as caspase 3. Finally, FEB suppressed the JNK signaling pathway activation, a crucial mechanism in mediating tissue inflammation and apoptosis.
XO is an enzyme that plays an essential role in the pathogenesis of various organ dysfunctions in sepsis because of its strong ability to generate ROS resulting in multiple organ damage. Therefore, we demonstrated in the current study the possible protective role of FEB, which is a selective XO inhibitor in sepsis.
Sepsis is a major cause of AKI with high mortality rate. 28 In our study, AKI was evidenced by a significant increase in creatinine and NGAL levels. Also in renal histopathology analysis, we found impaired renal morphology with severe Bowman’s capsule degeneration, proximal and distal tubules degeneration, and interstitial edema. Coadministration of FEB caused a significant improvement of these renal parameters. This was in accordance with a previous study which demonstrated that FEB suppressed renal injury via reduced oxidative stress in a rat model of renal ischemia–reperfusion injury. 29
Also, CLP induced hepatic injury was detected by a significant increase in liver functional enzymes as AST and ALT and histopathological findings. Coadministration of FEB caused a significant improvement of these hepatic parameters. This was in accordance with a previous study which demonstrated that FEB had protective effect against acetaminophen-induced liver injury. 30
FEB, as a direct inhibitor for XO enzyme, reflects an important role for attenuating oxidative damage as a mechanism of treating sepsis. 31,32
ROS induce the production of MDA, which is a direct product of lipid peroxidation and used as an indicator for the severity of lipid peroxidation. Moreover, ROS induce damage to cellular and mitochondrial membranes leading to apoptosis and necrosis.
33
In antioxidant system, SOD is a ROS scavenger that can eliminate superoxide radicals and prevent ROS attack as it catalyzes the superoxide anion
In present study, we found that MDA significantly increased and SOD significantly decreased in septic rats. Coadministration of FEB caused a significant decrease in MDA and increase in SOD. Our results were in accordance with previous studies. 35,36 Moreover, we provided an evidence for the inhibition of the activity of XO enzyme after administration of FEB, through detecting a significant suppression of the serum levels of the uric acid, a direct end product from XO enzyme activity in the body. 37
Many cytokines, including TNF-α and IL-1β, play an important role in systemic inflammation and development of multiple organ failure in sepsis. In our study, we found that there was an increase in IL-1β protein expression in septic rats. On coadministration of FEB there was a decrease in its expression and these results were in agreement with previous study demonstrating effect of FEB on serum cytokines. 38
XO is involved in free radical generation leading to oxidative stress. Oxidative stress in turn produces mitochondrial impairment, which leads to the release of inflammatory cytokines and subsequent cell death. Subsequently, there is a stimulation of intracellular signaling pathways such as mitogen-activated protein kinase (MAPKs). The components of the MAPKs pathway are known to induce NF-κB, an important inflammatory transcription factor. Therefore, inhibition of XO will suppress MAPKs pathway and subsequently suppress NF-κB. 39
In the present study, we explored hepatic and renal expression of NF-κB, one of the most important transcription factors that directs gene encoding for many pro-inflammatory cytokines, such as IL-1β and IL-6 and is involved in gene expression of sepsis-induced inflammation response and the pathophysiology in ALI and AKI. 40
Inhibition of NF-κB reduces the expression of many pro-inflammatory genes, decreases intravascular coagulation, and prevents microvascular endothelial leakage. 41 Coadministration of FEB caused a significant decrease in NF-κB expression. This was in accordance with a previous study, 42 which reported that FEB was better than allopurinol in suppressing inflammation and apoptosis mediating the MAPK/NF-κBp65/TNF-α pathway in an experimental model of cardiac ischemia-reperfusion injury.
Moreover, we found that in septic rats there was a significant increase in tissue levels of caspase 3 and JNK which play an important role in induction of cell apoptosis as they are directly involved in the mitochondrial pathway of apoptosis. Activation of JNK by oxidative stress leads to mitochondrial release of cytochrome c into the cytoplasm resulting in caspase 3 activation and apoptosis. 11 Coadministration of FEB caused a significant decrease in JNK and caspase 3 levels. In previous study, it has been reported that FEB significantly suppresses lipopolysaccharides-induced monocyte chemoattractant protein 1 production in macrophages. These inhibitory effects were mediated by decreasing ROS formation and activating MAPK phosphatase 1, which leads to dephosphorylation and inactivation of JNK. 43
Our results suggest that protective effects provided by FEB in a rat model of sepsis induced by CLP are due to its antioxidant, anti-inflammatory, and antiapoptotic effects, resulting into attenuating liver and kidney injuries in sepsis. Our proposed mechanisms of FEB protection against sepsis-induced liver and kidney injuries are summarized in Supplementary Figure 1. In brief, FEB, through inhibiting XO enzyme, exerts antioxidant activity that leads to the inhibition of NF-κB activation, which eventually inhibits pro-inflammatory cytokines activation, TNF-α and IL-1β. Furthermore, FEB directly inhibits apoptosis mediator p-JNK, which in turn inhibits caspase 3 production. P-JNK, is also a possible target for inhibition by decreasing NF-κB activation. Both arms end up with decreased organ dysfunction, in particular liver and kidney injuries.
Conclusion
We can conclude that FEB has a protective role in preventing CLP-induced hepatic and renal septic damage depending on XO inhibition, antioxidant, anti-inflammatory, and antiapoptotic effects.
Supplemental material
Supplementary_Figure - Protective effect of febuxostat in sepsis-induced liver and kidney injuries after cecal ligation and puncture with the impact of xanthine oxidase, interleukin 1β, and c-Jun N-terminal kinases
Supplementary_Figure for Protective effect of febuxostat in sepsis-induced liver and kidney injuries after cecal ligation and puncture with the impact of xanthine oxidase, interleukin 1β, and c-Jun N-terminal kinases by YF Ibrahim, RR Fadl, SAE Ibrahim, MF Gayyed, AMA Bayoumi and MMM Refaie in Human & Experimental Toxicology
Footnotes
Acknowledgment
The authors thank GlobalNapi Company for its generous gift of &&&FEB powder.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
