Abstract
Background and aim:
Cancer is a fatal and serious disease. Cyclophosphamide (CYC) is a commonly used anticancer drug. Cardiotoxicity and myelotoxicity are life-threatening side effects of CYC treatment. We aimed to evaluate the effect of the xanthine oxidase (XO) inhibitors, allopurinol (ALL) and febuxostat (FEB), on CYC-induced cardio- and hematopoietic toxicity in rats.
Methods:
ALL (100 mg/kg/day) or FEB (10 mg/kg/day) were administered orally to rats in the presence and absence of CYC (200 mg/kg kg i.p. single dose) treatment. Serum creatine kinase-MB creatine kinase myocardial band (CK-MB) and lactate dehydrogenase (LDH) activities were estimated. Complete blood counting (CBC), cardiac and bone marrow XO activity, malondialdehyde level, and superoxide dismutase activity were determined. Cardiac and bone marrow histopathological changes were also evaluated.
Results:
ALL and FEB significantly decreased CK-MB and LDH induced by CYC. Disturbed levels of XO, oxidative stress parameters, and CBC were also corrected by both XO inhibitors tested, with amelioration of cardiac histopathological changes caused by CYC. Treatment with FEB, but not ALL, prior to CYC challenges normalized bone marrow histopathological changes.
Conclusion:
These results suggest that both XO inhibitors tested; ALL and FEB can ameliorate CYC-induced cardiotoxicity. However, only FEB can protect against CYC-induced myelotoxicity, whereas ALL, to the contrary, might aggravate it.
Introduction
Cancer is a fatal and serious disease with worldwide spread. 1 Cyclophosphamide (CYC) is a commonly used anticancer drug. Unfortunately, cardiotoxicity is a common side effect that occurs with CYC as a result of many mechanisms, including oxidative stress process with the production of free radicals. 2 It was also reported that CYC causes bone marrow depression and decreases hematopoietic process. 3 Cardiovascular disease is the second leading cause of long-term morbidity and mortality among cancer survivors. 4 Cardiotoxicity is the occurrence of heart electrophysiological dysfunction or myocardial damage. Cardiotoxicity may be caused by chemotherapy treatment, 4 radiotherapy, 5 adverse effects of heavy metals, 6 or an incorrectly administered drug such as bupivacaine. 7 Myelotoxicity is another side effect of chemo- and radiotherapy which manifested by bone marrow depression in the form of anemia, thrombocytopenia, and neutropenia. 8
Xanthine oxidase (XO) inhibitors are of two kinds: purine analogs and others. Purine analogs include allopurinol (ALL), oxypurinol, and tisopurine. 9 Others include febuxostat (FEB), topiroxostat, and inositols (phytic acid and myoinositol). 10 ALL and FEB act by decreasing the production of uric acid, which has harmful effects on cardiovascular system. 11 These drugs have also been reported to inhibit oxidative stress process and production of free radicals, 12 thus they might have a role in decreasing toxic effects of CYC.
Uric acid has deleterious effects of oxidative stress, so XO inhibition is considered as a therapeutic tool in cardiovascular diseases. This explains to somewhat the role of ALL in the treatment of coronary artery disease, endothelial cell dysfunction, hypertension, myocardial ischemia–reperfusion injury, and chronic heart failure. 13
FEB has a cardioprotective effect following myocardial ischemia reperfusion (IR) via reduction of reactive oxygen species (ROS) generation and mitochondrial apoptosis. It increased significantly the antiapoptotic proteins, Bcl-XL and Bcl-2, and reduced the proapoptotic proteins, Bax and Bak expression, thus ameliorating myocardial injury. 14 FEB also inhibits endothelial-bound XO implicated in vascular ROS production. 15
It is worth mentioning that this is the first study to evaluate the purine versus nonpurine XO inhibitors in ameliorating CYC-induced cardio- and myelotoxicity. So, the aim of the current study is to evaluate the effect of the XO inhibitors, ALL and FEB, on CYC-induced cardio- and hematopoietic toxicity in rats.
Materials and methods
Drugs, kits, and chemicals
ALL and FEB powders were a kind gift from EPICO Co. and Global Napi Co., (Egypt), respectively. CYC vial was purchased from Baxter Oncology GmbH, (Germany). XO activity enzyme-linked immunosorbent assay (ELISA) kit was purchased from Sigma-Aldrich Co. (St Louis, MO, USA). Creatine kinase-MB (CK-MB) and lactate dehydrogenase (LDH) kits were purchased from Spectrum diagnostic, Egypt. All other used chemicals are purchased from their commercial sources.
Animals
Forty-eight adult male Wistar rats, weighing 165−225 g, were purchased from the National Research Center (Giza, Egypt). All experimental designs were in accordance with the ethical standards approved by the faculty board committee of Faculty of Medicine, Minia University, Egypt. Rats were fed a standard diet of commercial rat chow and tap water and were left to acclimatize to the environment for 2 weeks before inclusion in the experiments.
Experimental groups
Rats were divided into six groups (n = 8) and treated according to Table 1. The above-mentioned doses of CYC, 16 ALL, 17 and FEB 18 were selected on the basis of the previously published results and our preliminary studies. The time course of the experiment is shown in Figure 1.
Experimental groups.

ALL: allopurinol; FEB: febuxostat; CYC: cyclophosphamide; CMC: carboxy methylcellulose.
Time course of the experimental procedures.
Samples collection and preparation
At the end of the experimental period, the animals were weighed and then euthanized. Venous blood samples were collected from the jugular vein. Some of the blood samples were supplemented by ethylenediaminetetraacetic acid (EDTA) and prepared for complete blood counting (CBC). The remaining of blood samples were centrifuged at 5000 r/min for 10 min. Sera were collected and stored at −80°C until used for the determination of CK-MB and LDH activities. Hearts were separated and weighted for the estimation of heart/body weight ratio. Heart tissue was homogenized in 10% w/v ice-cold phosphate-buffered saline (PBS) (0.01 M, pH 7.4) and bone marrow was homogenized in 1 ml PBS. The homogenate was centrifuged at 3000 r/min for 20 min and the supernatant was aliquoted to avoid sample thawing and refreezing. Aliquots were stored at −80°C until used for the determination of superoxide dismutase (SOD) and XO activities, as well as malondialdehyde (MDA) level. A slice of heart tissue was fixed in 10% formalin, then embedded in paraffin and was used for cardiac histopathological examination. Slides from bone marrow suspensions were also prepared for bone marrow histopathological examination.
Biochemical analysis
Assessment of serum cardiac enzymes
Serum CK-MB and LDH activities were estimated using CK-MB and LDH kinetic kits according to the manufacturer instructions and results were expressed as U/l (Spectrum diagnostic).
Determination of cardiac and bone marrow oxidative stress parameters
Biochemical oxidative stress markers were determined in cardiac muscle and bone marrow homogenates, where SOD activity and MDA levels were evaluated. Cardiac and bone marrow SOD activity was determined according to the previously described method 19 and results were expressed as U/g tissue. Cardiac and bone marrow MDA levels were determined by biochemical assessment of thiobarbituric acid reacting substance through spectrophotometric measurement of the resulting color at 535 nm 20 and the results were expressed as nmol/g tissue.
Determination of XO activity
Cardiac and bone marrow XO activity was estimated using colorimetric XO ELISA kit according to the manufacturer instructions (Sigma-Aldrich).
Complete blood counting
CBC including red blood cells (RBCs), white blood cells (WBCs), and platelet counting was done using manual cell counting method by hemocytometer. Hemoglobin level estimation was also done via direct spectrophotometric method after the addition of hemoglobin buffer.
Histopathological examination of heart tissues
Cardiac specimens were fixed in 10% formalin, paraffin-embedded, sectioned at 5 μm, stained with hematoxylin and eosin (H&E) for routine histological assessment, and examined under an Olympus light microscope. Histopathological cardiac changes were graded as previously described, 21 so that (A) is blood vessel congestion, (B) is perinuclear vacuolization, (C) is focal necrosis, and (D) is round cell infiltration among degenerated or necrosed cardiac myocytes. Grading for each category was performed using a semiquantitative scale, where 0 was normal, while 1+, 2+, and 3+ represented mild, moderate, and severe abnormalities, respectively. The total cardiac injury score for each heart was calculated as an average of all the component injury scores.
Histopathological examination of bone marrow
Both femur bones were dissected and the marrow cells were flushed out with 1 ml PBS and pipetted several times. The cell suspension was centrifuged for 5 min and then the supernatant was discarded. The cell pellet was resuspended and a drop of the suspension was spread on a clean glass slide, dried overnight, refreshed with concentrated Leishman stain for 3 min, and counterstained with diluted Leishman stain (1:6) for 15 min. Then slides were examined under a light microscope at 40× magnification.
Statistical analysis of the data
GraphPad Prism was used for statistical analysis (version 6 for Windows, GraphPad Software, San Diego, California, USA; www.graphpad.com). Results were expressed as means ± standard error of the mean. Results were analyzed by one-way analysis of variance followed by Tukey’s postanalysis test. Differences with p value <0.05 were considered significant.
Results
Effect on serum cardiac enzymes
Single intraperitoneal injection of CYC (200 mg/kg) caused a significant increase in serum levels of both CK-MB and LDH as compared with the control group. Meanwhile, the administration of either ALL or FEB with CYC significantly decreased CK-MB and LDH levels as compared to CYC-challenged group (Table 2).
Effect of ALL and FEB on serum CK-MB and LDH levels in CYC-challenged and unchallenged rats.a
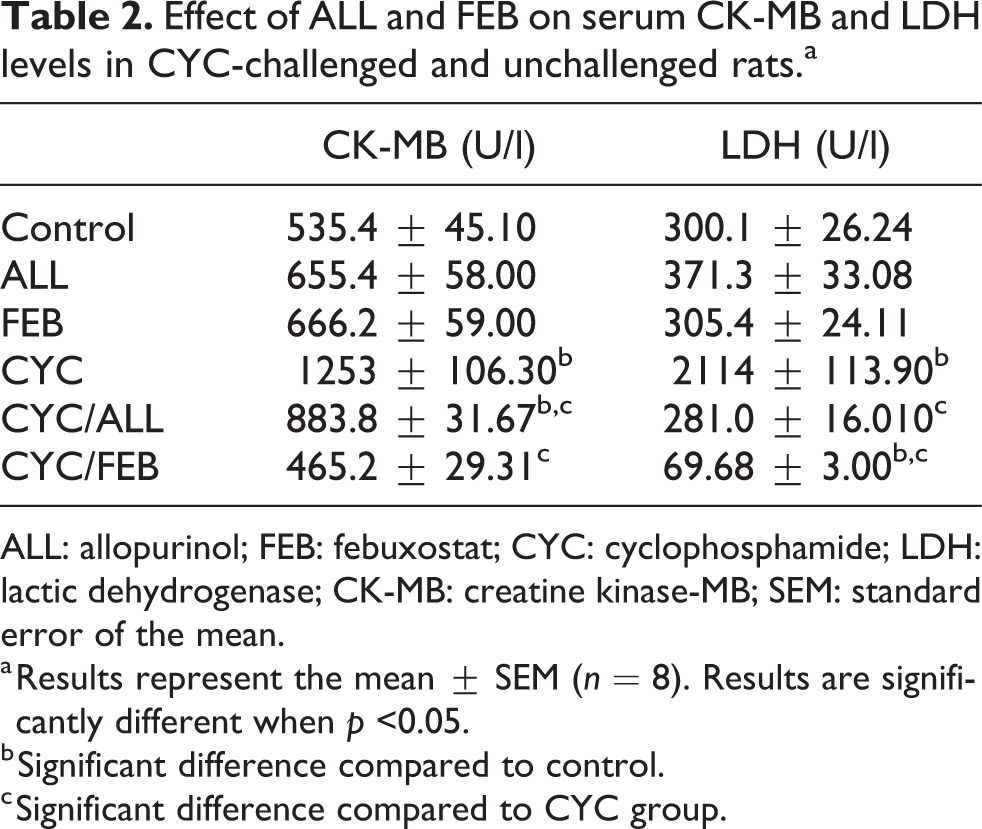
ALL: allopurinol; FEB: febuxostat; CYC: cyclophosphamide; LDH: lactic dehydrogenase; CK-MB: creatine kinase-MB; SEM: standard error of the mean.
a Results represent the mean ± SEM (n = 8). Results are significantly different when p <0.05.
b Significant difference compared to control.
c Significant difference compared to CYC group.
Effect on oxidative stress parameters (SOD and MDA)
CYC produced a significant decrease in the activity of SOD in heart tissue as compared to the control group. Coadministration of either ALL or FEB with CYC succeeded in significantly increasing SOD activity as compared with CYC intoxicated group. In the bone marrow, groups receiving CYC alone or in combination with either ALL or FEB showed no significant changes in SOD activity as compared with the control group (Table 3).
Effect of CYC, ALL, FEB on SOD activity and MDA level in heart and bone marrow in rat.a

SOD: superoxide dismutatse; MDA: malondialdehyde; ALL: allopurinol; FEB: febuxostat; CYC: cyclophosphamide; SEM: standard error of the mean.
a Results represent the mean ± SEM (n = 8).
b Significant difference from control group.
c Significant difference from CYC group.
d Significant difference from CYC/ALL combination group (p < 0.05).
Cardiac and bone MDA were significantly increased in CYC-intoxicated group compared to control. Treatment with ALL or FEB significantly attenuated the effect of CYC on cardiac MDA. However, only FEB, but not ALL, coadministration with CYC significantly decreased bone marrow MDA compared to CYC alone. CYC/FEB group showed significantly lower bone marrow MDA than CYC/ALL group (Table 3).
Effect on cardiac and bone marrow XO activity
The activity of XO was increased in CYC-intoxicated group in comparison to the control group. This increase was significantly decreased with the coadministration of CYC with either ALL or FEB in comparison to the group treated with CYC alone. On the other hand, bone marrow XO activity in groups treated with single treatment of ALL, FEB, or CYC, as well as those treated with combined CYC/FEB, was significantly increased as compared with the control group. However, XO activity was significantly decreased in CYC/ALL combination treatment group in comparison to the activity seen in the group treated with CYC alone (Figure 2).

Effect of CYC, ALL, and FEB on (a) cardiac and (b) BM XO activity in the rat. ALL: allopurinol; FEB: febuxostat; CYC: cyclophosphamide; BM: bone marrow; XO: xanthine oxidase. Results represent the mean ± SEM. (n = 8). aSignificant difference from control group; bsignificant difference from CYC group; and csignificant difference from CYC/ALL combination group (p < 0.05).
Effect on blood elements
In the current study, the administration of CYC significantly decreased hemoglobin level. Interestingly, the administration of concomitant CYC together with FEB, but not ALL, significantly increased hemoglobin compared to CYC alone. For RBCs, single treatment with ALL, FEB, or CYC did not significantly affect the RBC count compared to control. On the other hand, the combination of CYC/ALL, but not CYC/FEB, significantly decreased RBC count compared to control. For WBC count, FEB did not affect WBC count compared to control. On the other hand, groups treated with either ALL or CYC alone as well as the combination of CYC/ALL and CYC/FEB significantly decreased WBC count compared to control. Concerning platelets, all tested groups showed a significant decrease in platelet count compared to control (Table 4).
Effect of CYC, ALL, FEB, and their combination on blood elements in different rat groups.a
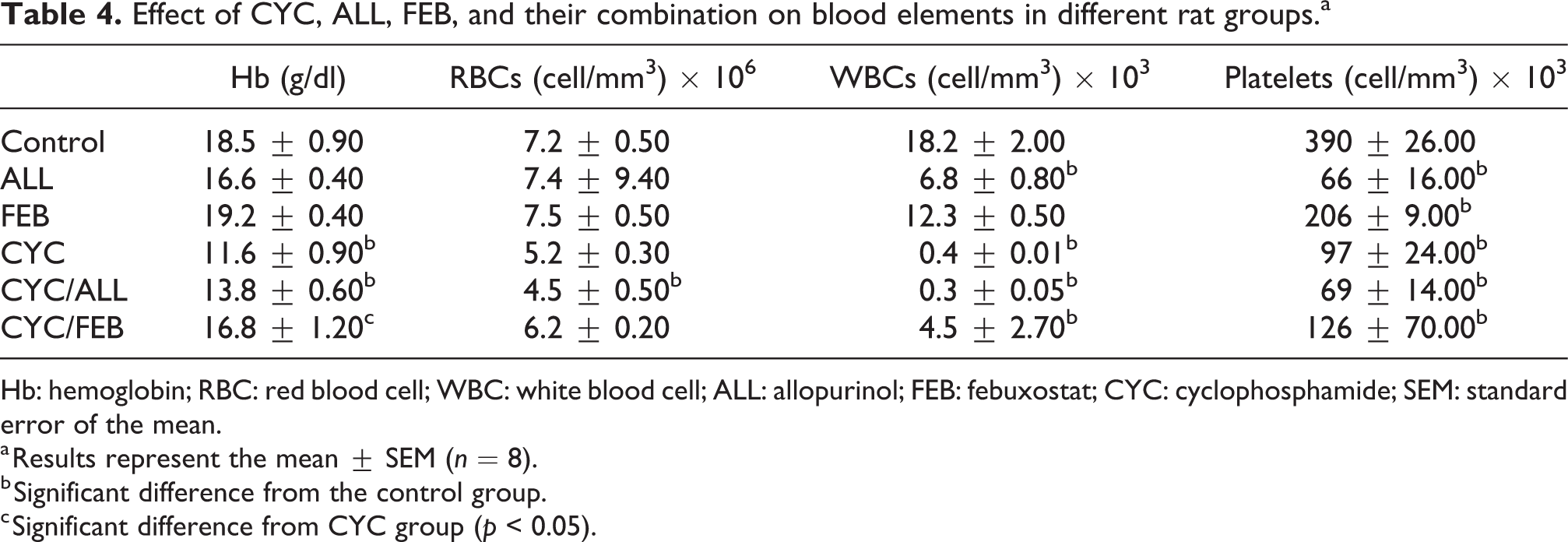
Hb: hemoglobin; RBC: red blood cell; WBC: white blood cell; ALL: allopurinol; FEB: febuxostat; CYC: cyclophosphamide; SEM: standard error of the mean.
a Results represent the mean ± SEM (n = 8).
b Significant difference from the control group.
c Significant difference from CYC group (p < 0.05).
Histopathological changes
Evaluation of cardiac histopathological changes
Sections of rat cardiac tissue stained with H&E showed normal histological pattern in normal control, ALL, and FEB groups (Figure 3(a), (b), and (c), respectively), as also indicated by scoring of different histopathological changes (Table 5). In CYC-treated group, however, microscopic pictures showed several cardiac pathological defects, including diffuse hemorrhagic necrosis, capillary congestion, capillary endothelial damage, perinuclear vacuolization, myocardial degenerations, and infiltration by acute inflammatory cells (Figure 3(d)). These findings were significantly improved in ALL- and FEB-treated groups to nearly normal pattern (Figure 3(e) and (f), respectively).

Effect of CYC (200 mg/kg i.p.), ALL (100 mg/kg/day orally), and FEB (20 mg/kg/day orally) on cardiac histopathological pattern. Sections were stained with H&E, where (a) control, (b) ALL, (c) FEB, showing normal cardiac histological pattern, (d) CYC showing myocardial degeneration (white star), and the inset showing capillary congestion (black stars), prinuclear vacuolization (black arrow), and capillary endothelial damage (white arrow), (e) CYC/ALL, and (f) CYC/FEB both showing relatively better histological picture than CYC (×100). Scoring of histopathological changes is indicated in Table 4. ALL: allopurinol; FEB: febuxostat; CYC: cyclophosphamide; H&E: hematoxylin and eosin.
Effects of CYC, ALL, and FEB on scoring of cardiac histopathological changes in rat.a

ALL: allopurinol; FEB: febuxostat; CYC: cyclophosphamide; SEM: standard error of the mean.
a Results of scoring different histopathological changes are calculated as percent of normal cells and are represented as mean ± SEM (n = 8).
b Significant difference from the control group.
c Significant difference from CYC group (p < 0.05).
Assessment of bone marrow histopathological changes
Slides of bone marrow tissues from the control group showed normal histological pattern, with densely packed cellular distribution, showing normal erythropoiesis and myeloid maturation (Figure 4(a)). Myeloid/erythroid ratio was 0.6:1 and megakaryocytes were present. On the other hand, smears of bone marrow tissue in rats treated with ALL alone showed mild hypocellularity, with a slight decrease in erythropoiesis and mild decrease in megakaryocytes (Figure 4(b)). This group also showed normal presence of monocytes with the decrease in lymphocytes and neutrophils. Smears of FEB group showed normocellular bone marrow, with almost normal erythropoiesis and myeloid maturation. Interestingly, bone marrow cells were crowded and large, showing signs of stimulation of bone marrow (Figure 4(c)).

Effects of cyclophosphamide, allopurinol, and febuxostat on bone marrow histopathological pattern. Bone marrow slides stained with Leishman’s stain. (a) control, (b) allopurinol, (c) febuxostat, (d) cyclophosphamide, (e) cyclophosphamide/allopurinol, and (f) cyclophosphamide/febuxostat (×50).
In the group treated with CYC alone, aplastic bone marrow exhibiting with frequent empty spaces was noted. Bone marrow was markedly hypocellular, with the deterioration of the myeloid tissue more than the erythroid tissue, where myeloid/erythroid ratio was 0.1:1. In addition, megakaryocytes could not be identified (Figure 4(d)). Concomitant treatment with both CYC and ALL caused less deterioration in bone marrow histopathological pattern compared to CYC group but still retained some features similar to the group treated with CYC (Figure 4(e)). On the other hand, the group treated with both CYC and FEB showed normal cellularity of bone marrow, where erythropoiesis, myeloid maturation, and megakaryocytes were almost normal (Figure 4(f)).
Discussion
Our data showed that CYC-induced cardiotoxicity was manifested by a significant elevation in serum CK-MB and LDH activities. It was reported that the cardiotoxic effect of CYC caused an increase in serum CK-MB and LDH through direct myocardial endothelial damage and destruction of myocardial cells. 22 As a result, these enzymes are released into the blood and serve as the main diagnostic parameters for different cardiac abnormalities, 23 such as myocardial infarction. 24 Our data were in agreement with the previous studies. 25 In the current study, both FEB and ALL showed cardioprotective activities with significantly decreased CK-MB and LDH activities. In accordance with the current results, FEB was reported to ameliorate doxorubicin 18 and IR-induced cardiotoxicity 14 with subsequent decrease in cardiac enzymes. Also, ALL was reported to decrease cardiac enzymes as a result of its amelioration of isoproterenol-induced cardiac injury in rats. 26
In this study, CYC-induced cardiotoxicity was confirmed by significant histopathological changes of cardiac injury, which is in agreement with previous studies. 22,27 Coadministration of either FBS or ALL with CYC significantly improved the histopathological changes, which is in line with a study conducted on the cardioprotective effect of FEB and ALL on ischemic cardiac damage in rats. 28
The current results showed that CYC-induced bone marrow toxicity was manifested by severe leukopenia and anemia without significant thrombocytopenia. CYC-induced myelotoxicity, in our present study, was confirmed by significant histopathological changes in the form of marked hypocellular bone marrow, myeloid deterioration more than erythroid tissue, and absence of megakaryocytes in comparison to the control group, which showed normal histological patterns with normocellular bone marrow. This result was previously supported by a study reported that bone marrow depression was developed after CYC injection in mice. 29 Moreover, changes in blood elements were found in patients with breast cancer treated with CYC. 30 The hematopoietic adverse effects of CYC were reported to be mainly due to genotoxicity. 31 Moreover, CYC has a dose-dependent effect on myeloid cell proliferation and, as it is an alkylating agent, it induces double-stranded breaks with an arrest of cell division and apoptosis of the rapidly proliferating hematopoietic cells. 32
In the present study, ALL alone administration mildly suppressed bone marrow, as evident by histopathological bone marrow picture with significantly decreased WBCs and platelet count. As expected, coadministration of ALL with CYC fails to ameliorate the effect of CYC on peripheral blood elements, despite mildly improving bone marrow smear pattern. This finding is in agreement with a study 33 reported that leucopenia and/or thrombocytopenia were attributed to ALL in hospitalized patients.
In line with these findings, in this study, ALL together with CYC, unfortunately, significantly decreased RBC number, which was not seen in groups treated with either drug alone. This is in line with pure red cell aplasia 34 and aplastic anemia cases that were reported to be associated with ALL therapy in chronic kidney disease patients. 35 Such effect of ALL is probably attributed to mild bone marrow depression resulting from retention of oxipurinol, an effect exaggerated by impaired renal functions. 36 The effect of ALL on bone marrow suppression may also be aggravated by coadministration of other drugs, as 6-mercaptopurine, which was reported to have an effect on granulopoiesis and subsequent myelosuppression at very high ALL dosage. 37
The administration of FEB alone, on the other hand, had no effect on Hb, RBCs, or WBCs. However, FEB significantly decreased platelet count. The group treated with FEB also did not show any bone marrow suppression. On the contrary, bone marrow in this group showed signs of stimulation rather than suppression. When coadministered with CYC, FEB caused improvement of bone marrow pattern and improved Hb level. This may be explained by the previously reported decreasing effect of FEB on the proapoptotic protein caspase-3 while increases Bcl-2, an antiapoptotic protein. 18 These antiapoptotic effects, besides FEB antioxidant effects, can be attributed to an overall improvement of bone marrow against CYC-induced myelotoxicity.
In this study, CYC caused a significant cardiac oxidative stress manifested by decreased cardiac SOD activity and increased cardiac MDA level compared to control. The antioxidant SOD enzyme catalyzes the superoxide anion (O2 ·−), 38 so, decreased SOD activity causes accumulation of O2 ·−, which reacted with membrane lipids and resulted in lipid peroxidation with the increase in its marker MDA and eventually loss of cellular components. 38,39 Our results concerning the effect of CYC on cardiac SOD and MDA are in accordance with the previously published data. 40 Pretreatment of CYC-intoxicated rats with either ALL or FEB restored both cardiac SOD activity and MDA level indicating a protective effect against oxidative stress. Our results concerning ALL pretreatment are supported by a study reported that presurgical oral ALL treatment reduced IR injury as indicated via decreasing ROS expression and increasing SOD activity. 41 Also, it was reported that ALL improves cardiac function following induced myocardial infarction in rats via attenuating the ROS-based injury and decreased cardiac MDA. 14 Similarly, FEB was reported to attenuate the induced regional endothelial dysfunction by reversing SOD activity, improving cardiac MDA, 12 and normalizing cardiac SOD. 18
Interestingly, in the current study, CYC administration did not affect bone marrow SOD activity. Most previous studies testing the effect of CYC on oxidative stress and myelotoxicity depended on measuring oxidative stress markers in the periphery and not in bone marrow tissue itself. 42,43 In addition, most of these studies were performed on mice, not rats. It is possible that this discrepancy is due to laboratory animal strain differences and that rats are more resistant to bone marrow oxidative stress than mice. Still, in the current study, CYC caused a significant increase in bone marrow MDA compared to control. Concomitant treatment with CYC/FEB, but not CYC/ALL, significantly improved bone marrow MDA. To the best of our knowledge, this is the first study to document the effect of FEB on bone marrow MDA. However, it was found that FEB significantly decreased the expression of proinflammatory and proapoptotic proteins and TUNEL positivity. FEB also was reported to increase the antiapoptotic protein. 18 Both anti-inflammatory and antiapoptotic effects may contribute to an overall oxidative stress reduction.
In the current study, CYC administration increased cardiac XO enzymatic activity, which was diminished by coadministration of either XO inhibitors tested. It has been previously reported that CYC, in contrast with our study, decreases XO activity in blood. 44 It is possible that this increase in cardiac tissue XO activity by CYC may be a feedback mechanism to oppose CYC cardiac toxicity. The inhibitory effect of both ALL and FEB on cardiac XO enzymatic activity was previously reported after myocardial infarction 45 and in regional endothelial dysfunction. 12 These reports are in line with our results since ALL and FEB only inhibit XO activity in challenged heart tissue. This might be because the level of basal XO in unchallenged cardiac tissues is too low to show further inhibition. XO enzyme is a well-known ROS-inducing enzyme and producer of free radicals, where XO mediates oxidation of hypoxanthine into xanthine then to uric acid, generating O2 ·− and hydrogen peroxide (H2O2). 46 Thus, XO inhibitors decrease oxidative stress and have beneficial effects specifically on the heart, 47 explaining their effect in ameliorating CYC-induced cardiotoxicity in the present study. It is not known whether XO enzyme would act differently in the presence of cancer or not, so further studies are needed to elucidate the pharmacodynamic effect of the currently tested drugs on XO in cancer animal models. However, it was reported that FEB was coadministered with chemotherapy to avoid tumor lysis syndrome of hyperuricemia in cancer patients. 48 It was also reported that FEB inhibits the human adenosine triphosphate (ATP)-binding cassette transporter G2, which is responsible for multidrug resistance of chemotherapeutic agents. 49 ALL was also reported to sensitize the prostate cancer cells to tumor necrosis factor-related apoptosis-inducing ligand. 50
In bone marrow, on the other hand, ALL, FEB, or CYC alone, as well as CYC/FEB combination, but not CYC/ALL significantly increased XO activity. It is difficult to interpret these results due to limited data available in literature about the effect of these drugs on bone marrow XO. In addition, several cellular elements of bone marrow may have contributed to such results. Previous studies, performed on bone marrow XO activity, usually extract one cell type from bone marrow for experimentation, as human myeloid cell line HL-60. 51 It has been reported that the level of XO as well as its enzymatic activity is not uniform in different tissues, 52 which might explain some discrepancies seen in the results of XO inhibition in the current study between the heart and bone marrow.
XO is a well-known source of H2O2 and O2 ·−, which reacts with nitric oxide to forms the peroxynitrite, which can lead to a variety of oxidative and nitrosative injuries to proteins, lipids, and DNA during synthesis of the uric acid. 11,53 ALL was reported to have a direct non-XO inhibition oxidative stress scavenging activitiy. 11
It is worth noticing that the uric acid lowering activity of either ALL or FEB is well documented. 54 However, FEB, the nonpurine analog inhibitor of both the oxidized and reduced forms of XO, was found to be more than 10 to 30 times potent than ALL, the hypoxanthine analog that weakly inhibits the oxidized only form in animal studies. 54 FEB was reported to have more potent and longer-lasting hypouricemic action than ALL, 11 which may explain why the results of ALL were far worse as compared to FEB.
The use of uricouric drugs (e.g. probenecid, sulfinpyrazone) is suspected to be less effective in prevention of CYC-induced toxicities as it manages the hyperuricemia caused by undersecretion of uric acid 54 without managing the oxidative and nitrosative stress caused by unblocked XO. 11 On the other hand, XO inhibitors can manage the hyperuricemia caused by overproduction and undersecretion of uric acid 54 together with preventing and scavenging oxidative and nitrosative stress. 11 However, this hypothesis requires experimental confirmation.
The suspected accumulated xanthine and hypoxanthine in response to XO inhibition have been reported to be more rapidly cleared by the kidneys than uric acid. ALL also reported to increase the reutilization of hypoxanthine for nucleic acid synthesis. 55
The inflammatory and apoptotic pathways might be involved in CYC-ALL or -FEB interaction that might favor one combination over another. A recent study reported that CYC injection resulted in marked cardiac injury, with a profound effect on cardiac oxidative stress markers, overproduction of inflammatory cytokines, and activation of cardiomyocyte apoptosis. 56 So, the inflammatory and apoptotic pathways may be targets for ameliorating CYC-induced toxicities.
Conclusion
Taken together, the present study concluded that ROS and XO enzymatic pathways may largely participate in the mechanism of pathogenesis of cardiac and bone marrow toxicities related to CYC administration. In addition, XO inhibitors, ALL and FEB, can ameliorate CYC-induced cardiotoxicity. However, only FEB can protect against CYC-induced myelotoxicity, whereas ALL, to the contrary, might aggravate it.
Footnotes
Acknowledgments
The authors would like to thank Dr. Azza Hussein Ali Hammoda and Dr. Soha Abdelkawy Abdelwahab, Histology Department, Faculty of Medicine, Minia University, Egypt, for their efficient assistance and great effort throughout the processing stages of this work.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: this research was funded by the Deanship of Scientific Research at Princess Nourah bint Abdulrahman University through the Fast-track Research Funding Program.
