Abstract
Hydrogen sulfide (H2S) is an endogenously produced gaseous messenger that participates in regulation of cardiovascular functions. This study evaluates the possible protective effect of H2S in cardiovascular dysfunction induced by cecal ligation and puncture (CLP) in rats. After 24 h of induction of CLP, heart rate (HR), mortality, cardiac and inflammation biomarkers (creatine kinase-MB (CK-MB) isozyme, cardiac troponin I (cTnI), C-reactive protein (CRP), and lactate dehydrogenase (LDH)), in vitro vascular reactivity, histopathological examination, and oxidative biomarkers (malondialdehyde (MDA), reduced glutathione (GSH), and superoxide dismutase (SOD)) were determined. CLP induced elevations in HR, mortality, serum CK-MB, cTnI, CRP, and LDH, in addition to impaired aortic contraction to potassium chloride and phenylephrine and relaxation to acetylcholine without affecting sodium nitroprusside responses. Moreover, CLP increased cardiac and aortic MDA and decreased SOD, without affecting GSH and caused a marked subserosal and interstitial inflammation in endocardium. Sodium hydrosulfide, but not the irreversible inhibitor of H2S synthesis
Introduction
Sepsis is a complex dysregulation of inflammation arising when the host is unable to successfully contain an infection. Sepsis has two stages, namely, an early hyperdynamic stage characterized by increased heart rate (HR), cardiac output, vascular resistance, and proinflammatory cytokines and a later hypodynamic stage characterized by decreased HR, cardiac output, and vascular resistance and increased levels of anti-inflammatory cytokines. 1
Complications arising from sepsis include disseminated intravascular coagulation, systemic vascular collapse, and multiple organ failure. 2 Despite extensive research, sepsis and associated multiple organ failure are still the most common causes of death in surgical intensive care units. 3
The cecal ligation and puncture (CLP) model in rodents is the gold standard in sepsis research, a realistic model for the induction of polymicrobial sepsis. 4 CLP model resembles the clinical observations of vascular reactivity and inflammation during polymicrobial peritonitis, bacteremia, and systemic sepsis. 5
Hydrogen sulfide (H2S) has been recently identified as an endogenously produced gaseous messenger that participates in regulation of cardiovascular functions. The production of H2S in mammalian systems has been attributed to two pyridoxal-5′-phosphate-dependent key enzymes, namely, cystathionine β-synthase (CBS) and cystathionine γ-lyase (CSE). The distribution of CBS and/or CSE is tissue specific with CSE, enriched in cardiovascular system. 6
H2S possesses a diverse physiological profile that contributes to its cardioprotective actions. Physiological actions include vasodilatation, opening of adenosine triphosphate (ATP)-sensitive potassium channels (KATP) in vascular smooth muscle cells, 7 inhibiting the chloride channels in a concentration-dependent manner, 8 and closing the L-type calcium channel (LTCC). 9 The antioxidant effects of H2S may be of critical importance for the treatment of cardiovascular disease; for example, H2S reacts with and neutralizes cytotoxic reactive oxygen species (ROS), such as peroxynitrite. 10
The precise role of H2S in inflammation is still unclear, with both pro- and anti-inflammatory roles. H2S plays a potential proinflammatory role in several inflammatory conditions, such as hind paw edema 11 , lipopolysaccharide (LPS)-induced endotoxemia in rodents, 12 and CLP-induced sepsis in rodent animals. 13 In contrast, H2S plays a potential anti-inflammatory role in several inflammatory conditions. For example, it reduces leukocyte infiltration in an air pouch model and decreases carrageenan-induced hind paw edema. 14 Moreover, H2S reduces LPS-stimulated tumor necrosis factor α (TNF-α) and nitric oxide (NO) formation in microglia 15 and upregulation of nuclear factor κB (NF-κB) in RAW264.7 macrophages stimulated with LPS. 16
Since H2S has effects on cardiovascular system, in addition to having both pro- and anti-inflammatory action; therefore, we evaluated its effect on cardiovascular dysfunction induced by CLP model in rats.
Materials and methods
Drugs and chemicals
1,1′,3,3′-Tetramethoxypropane, acetylcholine hydrochloride (ACh),
Experimental animals
Male Sprague Dawley rats, weighing 200 ± 20 g with an average age of 6–8 weeks, were purchased from Egyptian Organization for Biological Products and Vaccines, Giza, Egypt. The animal care and experiments described in this study comply with the ethical principles and guidelines for the care and use of laboratory animals adopted by the Research Ethics Committee of Faculty of Pharmacy, Mansoura University, Egypt, which are in accordance with the Principles of Laboratory Animal Care (NIH publication no. 85–23, revised 1985).
Cecal ligation and puncture
Rats were anesthetized with pentobarbital sodium (45 mg/kg, intraperitoneal (i.p.)), and a small midline incision was made through the skin and peritoneum of the abdomen to expose the cecum. The cecal appendage was ligated with Silkam 2-0 thread at half the distance between distal pole and the base of the cecum without occluding the bowel passage and then perforated in two locations with a 16-gauge needle distal to the point of ligation. After this, a small amount of stool was squeezed out through both holes. Finally, the bowel was repositioned; the peritoneum, fasciae, and abdominal musculature were closed by applying simple interrupted sutures with 4/0 silk sutures. The skin was closed using subcuticular running sutures with 4/0 silk sutures. The rats were resuscitated by subcutaneous injection of 1 ml of pre-warmed 0.9% saline solution using a 25-guage needle. Sham-operated animals were used as control.
Experimental protocol
The animals were allocated into six groups each consisting of 8–17 rats. Group 1: sham group, n = 8; group 2: CLP group, n = 15; group 3: NaHS group, n = 8, rats receiving NaHS (0.8 mg/kg, intravenous (i.v.)) 17 ; group 4: PAG group, n = 8, rats receiving the irreversible inhibitor of CSE, PAG (50 mg/kg, i.v.) 18 ; group 5: NaHS + CLP group, n = 17, rats receiving NaHS (0.8 mg/kg, i.v.) 30 min before CLP; and group 6: PAG + CLP group, n = 17, rats receiving PAG (50 mg/kg, i.v.) 30 min before CLP.
NaHS and PAG were injected in a volume of 1 ml/kg of rat. The dose of PAG (50 mg/kg) used in this study did not show nephrotoxicity in rats. 19
After 24 h, all the rats were anesthetized with pentobarbital sodium. Electrocardiogram (ECG) was recorded, and blood samples were collected from the retro-orbital venous plexus to obtain serum and used to measure cardiac and inflammatory biomarkers. Both aorta and heart were excised immediately for in vitro vascular reactivity, histopathological examination, and measurement of oxidative biomarkers.
Electrocardiogram
ECGs were recorded from standard lead II limb leads using a single-channel ECG (model: 501-B III, Fukuda ME Kogyo Co Ltd, Tokyo, Japan). The electrocardiograph was standardized before each tracing to get sensitivity of 2 mV pulse that produces 20 mm height and a speed of 50 mm/s.
In vitro vascular reactivity
The descending thoracic aorta was quickly separated and placed in a cold-oxygenated physiological salt solution (PSS) with the following composition and pH 7.4 (mmol/l): sodium chloride 118, potassium chloride (KCl) 4.7, calcium chloride 2.5, potassium dihydrogen phosphate 1.2, magnesium sulfate heptahydrate 1.2, sodium bicarbonate 25, and glucose 11.1. The vessels were dissected free of fat and connective tissue and cut into rings (2–4 mm).
Aortic rings were mounted between two stainless steel hooks in an organ bath filled with 10 ml of PSS at 37°C and bubbled with a mixture of 95% oxygen and 5% carbon dioxide. Rings were allowed to equilibrate under 1g resting tension for 60 min, during which time the bath solution was replaced every 15 min and the resting tension was readjusted when necessary. Isometric tension generated by the vascular smooth muscle was measured using research-grade isometric transducer (model 72-4493, Harvard Apparatus Ltd, South Natick, Massachusetts, USA) and recorded with a Powerlab unit/400 linked to a PC running Chart Version 4.2 software (ADInstruments Pty Ltd, Australia). After the equilibration period, arterial ring responsiveness was assessed by measuring contraction to 80 mM KCl.
Cumulative concentration–response curves (CRCs) were then constructed to PE (10−9–10−5 M). To measure vasorelaxation, rings were first preconstricted with 1 µM PE, and after reaching a steady-state contraction (plateau), cumulative CRCs to ACh (10−9–10−5 M) or to SNP (10−9–10−5 M) were constructed.
Determination of serum CK-MB isozyme activity
Creatine kinase-MB (CK-MB) isozyme activity was determined according to the method described by Wurzburg et al. 20 using a commercial kit (Centronic GmbH, Germany). The method is based on measuring CK activity in the presence of an antibody to the CK-M monomer that does not affect the activity of CK-B subunits. CK-MB activity was measured at wavelength 340 nm and expressed as units per liter.
Determination of serum cTnI concentration
Cardiac troponin I (cTnI) concentration was determined according to the method followed by Larue et al. 21 using a solid-phase, enzyme-labeled chemiluminescent immunometric assay (Immulite 1000 Troponin I, Siemens Medical Solutions Diagnostics, Mountain View, California, USA). cTnI concentrations were expressed as nanograms per milliliter.
Determination of serum CRP
C-reactive protein (CRP) is a recognized acute-phase protein and a very sensitive marker of systemic inflammation and tissue damage. CRP was measured using agglutination test kit Reactivos GPL (Chemelex, Barcelona, Spain), according to the method described by Pepys et al. 22 The level of CRP is expressed as milligrams per liter.
Determination of serum LDH activity
Lactate dehydrogenase (LDH) activity was assessed according to the method described by Henry et al. 23 The experimental procedure works by monitoring the disappearance of the reduced form of nicotinamide dinucleotide, which absorbs at 340 nm. LDH activity was expressed as units per liter.
Determination of cardiac and aortic MDA concentration, reduced GSH, and SOD activity
Heart and aorta were isolated, weighed, and homogenized in phosphate-buffered saline containing heparin (0.16 mg/ml) as 10% (w/v) using Omni-125 handheld homogenizer (Omni international, Tulsa, Oklahoma, USA). The homogenates were centrifuged at 2000g at 4°C for 15 min, and the supernatant was used for fresh assay of oxidative biomarkers malondialdehyde (MDA), GSH, and superoxide dismutase (SOD).
TBA reactive substance was measured as MDA, the end product of lipid peroxidation, according to the method of Ohkawa et al. 24 The absorbance was determined at 532 nm spectrophotometrically and expressed as nanomoles per milligram tissue.
To determine the GSH levels, TCA-deproteinized serum was used to measure nonprotein sulfhydryl compound by the method followed by Ellman, 25 which is based on the reaction of GSH with Ellman’s reagent to give a compound that absorbs at 412 nm and expressed as micromoles per milligram tissue.
SOD activity was measured by the method described by Marklund and Marklund. 26 The degree of inhibition of the autoxidation of pyrogallol at an alkaline pH by SOD was used as a measure of the enzyme activity and expressed as units per milligram tissue.
Histopathological examination
At the end of the experiment, the heart and part of the aorta were rapidly dissected out, washed immediately with saline, and fixed in 10% buffered formalin. The fixed tissues were embedded in paraffin and serial sections (5 μm thick) were cut. Each section was stained with hematoxylin and eosin (H&E). The analyses were performed microscopically (Leica Imaging Systems, Cambridge, UK). The pathologist performing histopathological evaluation was blinded to the treatment assignment of different study groups.
Statistical analysis
Data are expressed as mean ± SEM, where n = number of rats. Statistical analysis was carried out using one-way analysis of variance, followed by Tukey–Kramer multiple comparisons post hoc test. Additionally, Kruskal–Wallis test was used, followed by Dunn’s multiple comparisons post hoc test for nonparametric measurements. The level of significance was set at p < 0.05. Statistical analysis and graphing were performed with GraphPad Prism Version 5.02 (GraphPad Software Inc., San Diego, California, USA).
Results
Effect of PAG and NaHS on CLP-induced increase in HR of rats
CLP caused a significant (p < 0.05) increase in HR of rats by 30.1 ± 4.5% when compared with the sham group (Table 1). Rats injected with NaHS or PAG showed no significant change in HR when compared with the sham group. Pretreatment with PAG did not significantly affect the increase in HR induced by CLP. Conversely, pretreatment with NaHS significantly prevented CLP-induced elevation in HR, which returned to sham baseline (Table 1).
Effect of PAG and NaHS on HR and mortality changes induced by CLP in rats.a

PAG:
aPAG (50 mg/kg, i.v.) or NaHS (0.8 mg/kg, i.v.) was injected 30 min before induction of CLP. Changes in HR and mortality were calculated 24 h after CLP.
b p < 0.05: significantly different from sham group using one-way ANOVA, followed by Tukey–Kramer multiple comparisons post hoc test.
c p < 0.05: significantly different from sham group using Kruskal–Wallis test, followed by Dunn’s multiple comparisons post hoc test.
d p < 0.05: significantly different from CLP using one-way ANOVA, followed by Tukey–Kramer multiple comparisons post hoc test.
Effect of PAG and NaHS on mortality induced by CLP in rats
CLP caused a significant (p < 0.05) increase in the mortality rate by 47.9% in rats when compared with the sham group (Table 1). Injection of rats with NaHS or PAG did not cause any mortality in rats. Preinjection with PAG before CLP did not significantly affect the mortality rate induced by CLP in rats. However, pretreatment with NaHS decreased the mortality rate to 24.2%, which was not significantly different from the sham group (Table 1).
Effect of PAG and NaHS on CLP-induced increase in cardiac biomarkers in rats
Serum CK-MB and cTnI were measured as specific markers for cardiac dysfunction. CLP caused a significant (p < 0.05, n = 8) increase in CK-MB (Figure 1(a)) and cTnI (Figure 1(b)) by 97.87% and 738.62%, respectively, when compared with the sham group. Neither NaHS nor PAG had a significant effect on normal levels of CK-MB and cTnI of rats. Preinjection of NaHS before CLP caused a significant decrease in CLP-induced elevation of CK-MB and cTnI. Conversely, PAG injection before CLP did not significantly affect CLP-induced elevation in CK-MB and cTnI.

Effect of PAG and NaHS on CLP-induced increase in cardiac, inflammatory, and cytotoxicity biomarkers in rats: PAG (50 mg/kg, i.v.) or NaHS (0.8 mg/kg, i.v.) was injected 30 min before induction of CLP, and serum was collected 24 h after CLP. Data are expressed as mean ± SEM, n = 8. (a) Serum CK-MB; (b) serum cTnI; (c) serum CRP; and (d) serum LDH. *p < 0.05: significantly different from sham group; $p < 0.05: significantly different from CLP group; #p < 0.05: significantly different from PAG + CLP group, respectively, using one-way ANOVA, followed by Tukey–Kramer multiple comparisons post hoc test. PAG:
Effect of PAG and NaHS on CLP-induced increase in inflammatory and cytotoxicity biomarkers in rats
CLP caused a significant (p < 0.05, n = 8) 18-fold increase in CRP (Figure 1(c)) and 2.9-fold increase in LDH (Figure 1(d)) when compared with the sham group. Treatment of rats with NaHS or PAG caused no significant change in CRP and LDH of rats when compared with the sham group. Preinjection with PAG before CLP had no significant effect on CLP-induced increase in CRP and LDH. Conversely, preinjection with NaHS before CLP decreased the CLP-induced elevation in CRP and LDH (Figure 1(c) and (d)).
Effect of PAG and NaHS on CLP-induced changes in cardiac and aortic oxidative biomarkers in rats
CLP caused a significant (p < 0.05, n = 4) increase in cardiac (Figure 2(a)) and aortic (Figure 2(d)) MDA level by 87.9% and 183.6%, respectively, and a significant decrease in cardiac (Figure 2(c)) and aortic (Figure 2(f)) SOD level by 43% and 66.2%, respectively, with no significant change in cardiac (Figure 2(b)) and aortic (Figure 2(e)) GSH level when compared with the sham group. Neither PAG nor NaHS had a significant effect on cardiac and aortic level of MDA, GSH, and SOD when compared with the sham group.

Effect of PAG and NaHS on CLP-induced changes in cardiac and aortic oxidative biomarkers in rats. PAG (50 mg/kg, i.v.) or NaHS (0.8 mg/kg, i.v.) was injected 30 min before induction of CLP, and heart and aorta were collected 24 h after CLP. Data are expressed as mean ± SEM, n = 4. (a) and (d) MDA; (b) and (e) reduced GSH; (c) and (f) SOD. *p < 0.05: significantly different from sham group; $p < 0.05: significantly different from CLP group, respectively, using one-way ANOVA, followed by Tukey–Kramer multiple comparisons post hoc test. PAG:
Preinjection with PAG before CLP in rats had no significant effect on CLP-induced increase in cardiac and aortic MDA level and CLP-induced decrease in cardiac and aortic SOD level. Conversely, pretreatment with NaHS before CLP caused a significant reduction in CLP-induced increase in cardiac and aortic MDA, without significantly affecting the decrease in cardiac and aortic SOD level.
Effect of PAG and NaHS on CLP-induced changes in histopathological examination of heart and aorta in rats
Histopathological examination of the heart using H&E stain in sham rats revealed normal endocardium and myocardium. Similarly, rats injected with NaHS and PAG revealed no abnormalities and normal heart muscle (Figure 3). CLP caused a marked subserosal and interstitial inflammation in endocardium. In addition, preinjection with PAG before CLP rats showed atrophic changes and inflammation. Conversely, preinjection of rats with NaHS before CLP showed normal heart myocardium, with no evidence of inflammation (Figure 3).

Effect of PAG and NaHS on CLP-induced changes in cardiac histopathological examination in rats. PAG (50 mg/kg, i.v.) or NaHS (0.8 mg/kg, i.v.) was injected 30 min before induction of CLP, and heart was collected 24 h after CLP. Histopathological examination of the heart using H&E (×100) stain, n = 3. Sham, NaHS, and PAG rats revealed normal heart muscle. CLP caused a marked subserosal and interstitial inflammation in endocardium. Preinjection with PAG before CLP in rats showed atrophic changes and inflammation. Rats pretreated with NaHS before CLP showed normal heart myocardium, with no evidence of inflammation. PAG:
In contrast, histopathological examination of the aorta using H&E stain in all groups showed normal aortic wall (data not shown).
Effect of PAG and NaHS on CLP-induced change in rat aortic vasoconstriction
Aorta from CLP group produced a significant reduction in KCl- and PE-induced maximal contractions (Figure 4(a) and (b)) by 52.9% and 47.1%, respectively, when compared with the sham group. Aorta from rats injected with either NaHS or PAG caused no significant change in KCl- and PE-induced contractions when compared with the sham group.
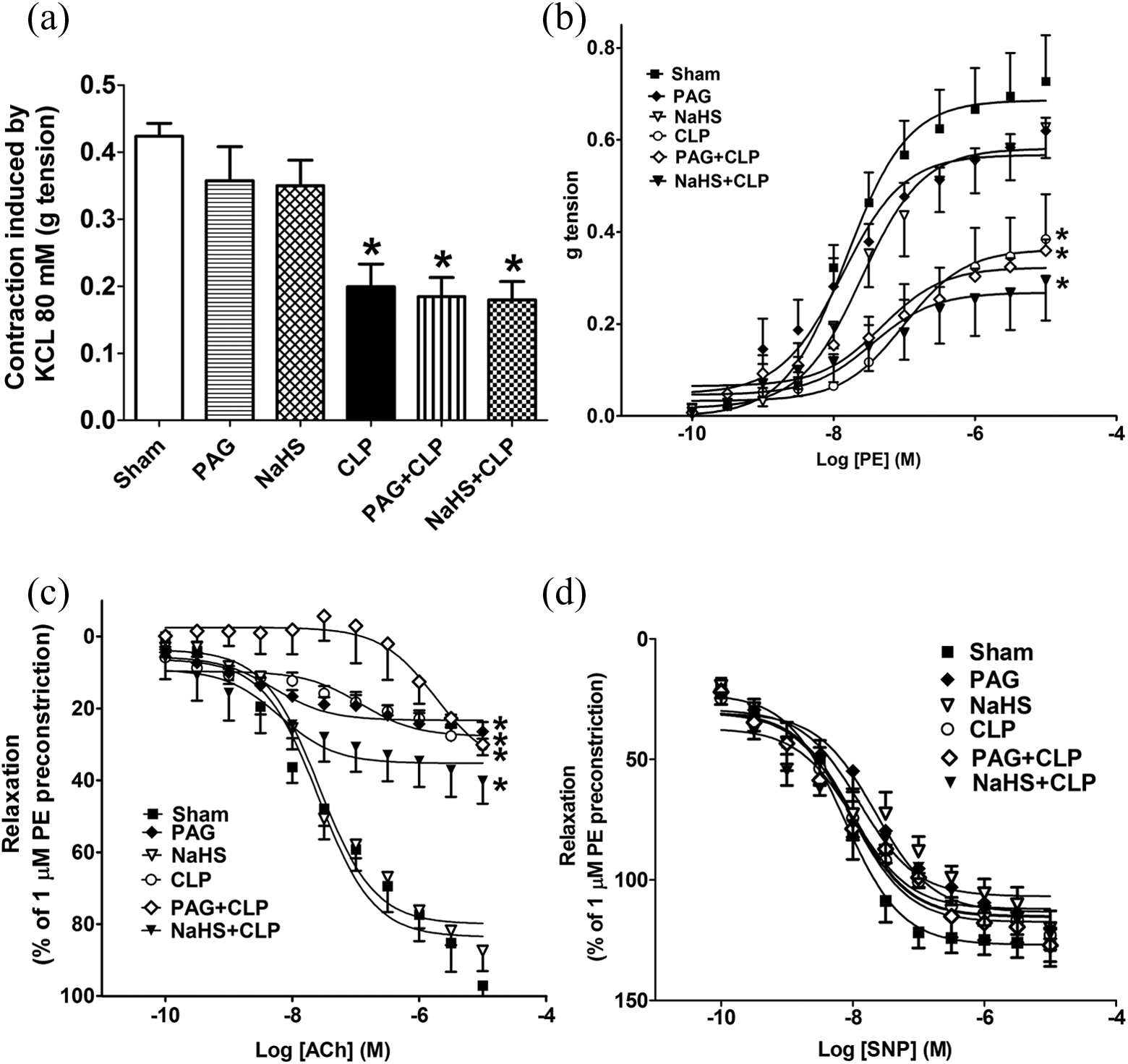
Effect of PAG and NaHS on CLP-induced changes in rat aortic vasoconstriction and vasorelaxation: PAG (50 mg/kg, i.v.) or NaHS (0.8 mg/kg, i.v.) was injected 30 min before induction of CLP, and aorta was isolated 24 h after CLP. Data are expressed as mean ± SEM, n = 6. (a) KCl-induced contraction; (b) PE-induced contraction; (c) ACh-induced relaxation; (d) SNP-induced relaxation.
Preinjection with either PAG or NaHS before CLP had no significant effect on CLP-induced decrease in the contraction of rat aortic rings to KCl or PE (Figure 4(a) and (b)).
Effect of PAG and NaHS on CLP-induced change in rat aortic vasorelaxation
ACh and SNP caused a concentration-dependent relaxation in the precontracted aortic rings. In the aortic rings of CLP group, the ACh-induced relaxation responses were significantly lower than sham group by 68.8% (Figure 4(c)).
Injection of rats with NaHS caused no significant change in relaxation responses of ACh when compared with the sham aortic rings. Conversely, treatment of rats with PAG caused a significant decrease in maximal ACh relaxation responses by 72.7% when compared with sham aortic rings (Figure 4(c)). Pretreatment with either PAG or NaHS before CLP caused no significant effect on CLP-induced decrease in ACh-induced relaxation responses (Figure 4(c)).
SNP-induced relaxation was not affected in all groups (Figure 4(d)).
Discussion
To our knowledge, this study is the first to show that H2S attenuated CLP-induced cardiac dysfunction. In this study, H2S has a protective effect against CLP-induced cardiac dysfunction as elucidated by decreasing CLP-induced tachycardia, mortality, serum CK-MB, cTnI, CRP, LDH, and cardiac and aortic MDA levels, in addition to improving heart histopathological changes without affecting vascular responses in rats.
Cardiovascular dysfunction has been shown in CLP and other models of sepsis. A previous study has shown that sepsis induced by CLP is associated with increased HR and mortality, in addition to increased cardiac biomarkers, such as CK-MB and cTnI. 27 In this study, the HR, mortality, and serum CK-MB and cTnI of rats were elevated at 24 h after CLP, which confirm the cardiac dysfunction induced by this model. Additionally, CLP caused a marked subserosal and interstitial inflammation in endocardium.
H2S plays important roles in different systems including the cardiovascular system. 28 It also improves neutrophil migration and survival in CLP-induced sepsis via KATP channel activation, even when treatment was delayed 6 h after CLP. 29 The effects of H2S on ion channels may explain its inhibitory effect on the HR presented in our study. H2S had a cardioprotective effect through opening of KATP channel, 7 blocking chloride channels 8 and LTCC. 30 In our previous work, we reported that H2S has a protective effect against aconitine- and barium chloride-induced arrhythmias as elucidated by decreasing dysrhythmia and mortality. 17
Previous studies reported that H2S protects against sepsis model. For example, a short course of infusion of an NaHS attenuates LPS-induced organ injury via stimulation of anti-inflammatory pathways, with no additional protection from prolonged infusion. 31 A short course infusion of GYY4137, a novel H2S-releasing molecule, protects against endotoxic shock in the rats injected with LPS. 32 Additionally, inhaled H2S protects against LPS-induced acute lung injury in mice. 33
In this study, NaHS reduced CLP-induced elevation in HR and mortality. In addition to HR, H2S protected against cardiac damage induced by CLP as shown by the decrease in serum CK-MB and cTnI of rats, and histopathological examination revealed normal heart myocardium, with no evidence of inflammation. Conversely, PAG did not improve the CLP-induced cardiovascular dysfunction. This confirms that increasing H2S, but not inhibiting its synthesis by PAG, has a protective effect against sepsis induced by CLP in rats.
In addition to cardiac dysfunction, sepsis causes vascular dysfunction including vascular smooth muscle and endothelial dysfunction. 34 A previous study has shown that LPS decreased the contractile responses of isolated aorta to KCl and PE, and relaxation responses to ACh, but not to SNP. 35
Our findings in this study indicate that CLP model has a similar effect on vascular system as the LPS model. Both contraction to KCl or PE and relaxation to ACh but not to SNP were impaired. This confirms that sepsis, either induced by LPS or CLP, is associated with vascular dysfunction.
Several mechanisms have been shown to contribute to this vascular dysfunction. For example, activation of K+ channels (KATP and KCa channels), 7 impairment of endothelial nitric oxide synthase (NOS), and excessive NO production through activation of inducible NOS (iNOS), 36 in addition to alterations in calcium mobilization. 37
H2S is reported to mediate vasorelaxation mainly through the opening of KATP channels in the smooth muscle. 7 The vasorelaxant responses of H2S are only observed in organ bath in vitro at NaHS concentrations above 100 μM. 38 The significance of this effect and its mechanisms are yet to be fully understood in vivo. A previous study reported that NaHS (28 μmol/kg, i.p.) increased the relaxation response to ACh of LPS-induced injury in pulmonary artery of rats. 39 However, in our study, we used thoracic aorta in which NaHS and PAG had no significant effect on vascular dysfunction induced by CLP.
The protective effect of H2S on cardiac dysfunction could be attributed to its antioxidant and anti-inflammatory effects. Sepsis has been shown to induce inflammation and oxidative stress characterized by overproduction and release of inflammatory mediators and ROS that are mediated by the activation of inducible transcription factors, such as activating protein-1 and nuclear factor κB (NF-κB). 40 Additionally, sepsis leads to overexpression of iNOS, followed by high-level NO production, which in turn contributes to cardiovascular dysfunction, in part through the generation of cytotoxic peroxynitrite, a product of NO and superoxide. 36
In this study, CLP-induced inflammation and cytotoxicity were manifested by increased serum CRP and LDH. In addition, CLP increased cardiac and aortic content of MDA and decreased SOD, with no significant change in GSH confirming that CLP induces oxidative stress and inflammation. Additionally, injection of NaHS before CLP caused a significant decrease in serum CRP and LDH and reduced the CLP-induced elevation in cardiac and aortic MDA level. Conversely, inhibition of H2S by PAG did not show the same protective effect. These results confirm the anti-inflammatory and antioxidant effect of H2S in the CLP model in this study.
Several studies have shown the antioxidant effect of H2S. Geng and colleagues reported that H2S attenuated the release of cardiac enzymes and decreased lipid peroxidation levels in the heart following isoproterenol-induced myocardial ischemic injury by scavenging hydrogen peroxide and superoxide. 41 Also, H2S elevated the antioxidants, SOD and glutathione peroxidase (GSH-Px), in adriamycin-induced cardiotoxicity in rats. 42 Moreover, NaHS increases the activity of endogenous antioxidants, such as SOD, GSH-Px, and GSH in the lung of mice. 43
In addition to the antioxidant effect of H2S, several studies reported that H2S attenuates the inflammation by preventing NF-κB activation. H2S has been shown to decrease LPS-induced upregulation of NF-κB in RAW264.7 macrophages 16 and TNF-α and NO formation in cultured microglial cells. 15 In addition, NaHS prevented NF-κB activation in gastric epithelial cell from ischemia–reperfusion-induced oxidative stress and inflammatory responses. 44 These antioxidant and anti-inflammatory effects of H2S could explain its protective effect against CLP-induced cardiac dysfunction in this study.
In contrast to the anti-inflammatory effects of H2S, other studies showed that H2S is proinflammatory. Administration of the irreversible inhibitor of CSE, PAG (50 mg/kg, i.p.), to septic mice was reported to significantly decrease plasma H2S levels and to improve the progression of sepsis and survival. 18 Additionally, administration of NaHS (10 mg/kg, i.p.) to mice either alone or in combination with LPS induces marked hypotension, liver, lung, and kidney inflammation, and aggravated multiple organ injury through upregulation of tissue NF-κB, p38, and extracellular signal-regulated kinase 1/2 (ERK1/2) signaling. 45 Moreover, administration of NaHS (10 mg/kg, intraperitoneally, at the time of CLP) in male Swiss mice mediated inflammation in CLP through enhancement of substance P production and activation of the ERK-NF-κB pathway. 18
Our results indicate that H2S is protective in CLP in contrast to other studies. 18,45 A possible explanation for these differences in results may be due to using different species of animals (mice and, in our study, rats), different doses of NaHS (10 mg/kg and in our study 0.8 mg/kg), and different time of injection of NaHS (at time of CLP and, in our study, 30 min before CLP). Additionally, H2S at micromolar concentrations demonstrated cytoprotective effects and upregulated endogenous antioxidant levels. 10 Conversely, higher H2S exposure in millimolar concentration tends to be cytotoxic to the cells due to ROS generation, calcium mobilization, GSH depletion, intracellular iron release as well as induction of mitochondrial cell death pathways. 46
Coletta and Szabo speculate that a moderate increase in H2S production (to a smaller extent and in an early stage of sepsis) may be beneficial and would enhance the host defense, preserve endothelial mitochondria, and ameliorate endothelial function, while excessive H2S production (concomitant with iNOS activation, coupled with oxidative and nitrosative stress, and in the later stage of sepsis) may be deleterious, as it may cause smooth muscle dysfunction, hemodynamic dysfunction, and organ failure. 47
In conclusion, this study showed that H2S has a protective effect against CLP-induced cardiac, but not vascular, dysfunction in rats. Therefore, H2S may represent a promising new pathway for the treatment of sepsis. However, more investigations are required to elucidate its precise mechanism of action.
Footnotes
Acknowledgment
We gratefully acknowledge Dr Ramy A Abdelsalam, Department of Pathology, Faculty of Medicine, Mansoura University, Egypt, for his assistance in the histopathological examination.
Conflict of interest
The authors declared no conflicts of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
