Abstract
2,3,7,8-Tetrachlorodibenzo-p-dioxin (TCDD) is a ubiquitous environmental contaminant in the environment. The developmental period is more sensitive to TCDD and there is a possibility that maternal exposure to TCDD may affect in adulthood. Adult female Wistar rats were exposed to 0.5, 1.0, and 2.0 µg/kg TCDD during the critical stage of organogenesis, that is, on GD15. The results revealed a significant decrease in indices of reproductive organ weight in adult male rats exposed to prenatal TCDD, and dose-dependent reduction in epididymal sperm reserves, percent motile, and viable sperm with an increase in percent morphological abnormal sperm. Polymerase chain reaction analysis revealed downregulated expression levels of steroidogenic markers such as steroidogenic acute regulatory, cholesterol side-chain cleavage, and 3β- and 17β-hydroxysteroid dehydrogenase (HSDs) in experimental rats. Immunofluorescence sections portrayed reduced distribution of 3β- and 17β-HSD proteins in testes of experimental rats. Furthermore, spermatogenic markers (acid phosphatase, alkaline phosphatase, lactate dehydrogenase, and sorbitol dehydrogenase) were significantly altered in the testes. Serum levels of testosterone, follicle stimulating hormones, and luteinizing hormone were significantly decreased. Testicular levels of hydrogen peroxide and lipid peroxidation were significantly elevated with a decline in superoxide dismutase, catalase, glutathione peroxidase activities, and total thiol levels. Moreover, histological and morphometric examination of testicular cross-sections depicted degenerative changes. Male fertility assessment in adult rats revealed a significant decrease in mating index, fertility index, and mean number of pre- and postimplantations with an increase in pre- and postimplantation losses in rats cohabited with in utero TCDD-exposed adult males. In conclusion, the findings of this study provided clear evidence that maternal exposure to TCDD during the critical stage of development results in suppressed reproductive health in adulthood.
Introduction
In recent years, there have been increased concerns over the deterioration of male reproductive health. The prime reason for this cause might be the inevitable exposure to environmental contaminants. Dioxins are among the priority listed pollutants that cause detrimental effects in male endocrine systems of both humans and wildlife. 1,2 2,3,7,8-Tetrachlorodibenzo-p-dioxin (TCDD) is a well-known toxic congener of dioxins that belongs to the class of polyhalogenated aromatic hydrocarbon chemicals. TCDD is mainly produced as a by-product from the manufacturing of pesticides, bleaching of paper, and motor vehicle emissions. 3,4 A significant amount of TCDD is also released into the environment in combustion activities such as forest fires, burning of household trash, and incineration of household and industrial wastes. 5 Low levels of TCDD are found in water, soil, sediments, air, and in the majority of foods such as meat, fish, shellfish, and dairy products. 5
TCDD is ubiquitous in the environment and its exposure is unavoidable due to its low biodegradable and persistent nature and has a wide range of effects such as wasting syndrome, metabolic derailment, tumorogenicity, carcinogenicity, and endocrine disruption by means of developmental and reproductive toxic effects. TCDD can either be transferred through the placenta during the gestation period and/or through lactation after birth and affects at a later stage of life, that is, adulthood. The lipophilic and low metabolic nature of TCDD makes maternal exposure of TCDD to persist in testis even at postnatal day 120 in animals. 6 TCDD binds to aryl hydrocarbon receptor (AHR), which binds to AHR nuclear translocator in the cytosol and then translocates to the nucleus to induce the expression of aryl hydrocarbon hydroxylases (cytochrome P450 s). 7 Esser et al 8 reported that TCDD produces toxic effects by interfering with the physiological signaling of AHR. In utero exposure to TCDD results in reduction of sperm count with an increase in morphological abnormal sperm in their adulthood. 9 Earlier studies pointed out that the testis is extremely sensitive to TCDD toxicity, which after exposure presented with loss of Sertoli cell contact with neighboring cells and germ cells development, elevated levels of lipid peroxidation, depleted antioxidant defenses, and reduced spermatogenesis. 10,11 Studies also demonstrated that TCDD reduces the Leydig cell number and volume through its effect on testicular steroidogenesis. 12,13 TCDD reduces the expression of steroidogenic markers such as steroidogenic acute regulatory (StAR), cholesterol side-chain cleavage (P450scc), 3β-hydroxysteroid dehydrogenase (HSD), and cyclic adenosine monophosphate (cAMP) levels, thereby inhibiting human chorionic gonadotropin (hCG)-induced testosterone secretion in Leydig cell primary cultures. 14 Sanabria et al. 15 reported that in utero exposure to TCDD results in compromised sperm quality and fertility in next-generation offspring.
Although numerous studies have pointed out the effect of prenatal exposure to TCDD on male reproductive health of offsprings, still there is ambiguity concerning the data related to maternally exposed TCDD and its effect on male reproductive functions at adulthood. 16 –20 So an elaborate study was conducted to investigate the effects of TCDD exposure during the critical period of organogenesis (GD15) on male reproduction using different reproductive endpoints: weights of reproductive organ weights; sperm quantity and quality; serum hormonal (testosterone, follicle stimulating hormones (FSH), and luteinizing hormone (LH)) levels; steroidogenic and spermatogenic markers; oxidative and antioxidative stress markers in testes; histopathology of testes; and fertility parameters. Furthermore, the scope of study is further broadened by evaluating the gene and protein expression of steroidogenic markers.
Materials and methods
Procurement and maintenance of animals
Healthy adult male (220–240 g) and female (180–200 g) Wistar rats were procured from Kunming University Laboratory Animal Centre, China, and allowed to acclimatize for 7 days before being used for actual experimentation. The animals were maintained under well-controlled experimental conditions such as temperature (23 ± 2°C), photoperiod (12 h light:12 h dark), and humidity (50 ± 10%) and provided with ad libitum access to standard rodent feed and water. All study protocols and number of animals used were reviewed and approved by the Institutional Animal Ethical Committee of the First People’s Hospital of Yunnan Province, the Affiliated Hospital of Kunming University of Science and Technology (Ethical Committee Approval number: 20181237).
Chemicals
TCDD was purchased from Sigma Chemical Co., St Louis, Missouri, USA. The other chemicals and reagents used in this study were of analytical grade.
Experimental design and treatment
Initially, female rats were checked for estrus cycle, and the rats proven with normal estrus cycle were further used for experimentation. Pro-estrus stage female rats were kept for mating with adult male rats in a 1:1 ratio during the dark portion of the photoperiod cycle, and the day on which sperm detected in the vaginal smear was considered as day zero of gestation (GD0). The pregnant rats were randomly allocated into four different groups of 10 in each. The rats in groups 2, 3, and 4 received single doses of 0.5, 1, and 2 µg TCDD/kg body weight, respectively, dissolved in corn oil via oral route through oro-gastric tube on GD15 (at the onset of sexual differentiation and the gestational day used in other reported studies), 21 –23 which is considered to be the critical stage of organogenesis and also the beginning of hypothalamus–pituitary–testicular functioning in fetus. 21 –23 While the animals in group 1 received corn oil through the oral route. The rats were allowed to deliver pups; all the litters were culled to six pups (male pups were kept within the litter and females were kept just to maintain equal litter sizes), weaned, and maintained under standard experimental conditions. On postnatal day 90, the control and treated group male rats were used for mating studies. Other reproductive endpoints such as sperm parameters, testicular steroidogenic and spermatogenic markers, serum hormones (testosterone, FSH, and LH), oxidative stress markers, and histology of testes were also evaluated.
Fertility and reproductive performance of F1 male rats
Ninety-day-old rats from both the control (n = 18) and experimental (n = 18) groups were cohabited with normal cyclic female rats in the pro-estrus stage. The presence of copulatory plugs/presence of sperms in the vaginal smears was considered as mating positive and indicated as GD0. The number of corpora lutea and implantations were evaluated in the collected ovaries and uterus, respectively, on GD8. While on GD 18, live and dead fetuses were recorded. From the results obtained, the mating index was calculated as the difference between number of pairings and the number of sperm-positive females (number of sperm-positive females/number of pairings × 100), fertility index was calculated as the difference between the number of sperm-positive females and the number of pregnant females (number of pregnant females/number of sperm-positive females × 100), pre-implantation loss per litter was calculated as the difference between counts of corpora lutea and total implantation sites (number of corpora lutea—number of implantations)/number of corpora lutea × 100), and postimplantation loss per litter was calculated as the difference between total implantation sites and live fetuses (number of implantations—number of live fetuses)/number of implantations × 100).
Necropsy
The rats were fasted overnight, blood collected, weighed, and euthanized by carbon dioxide asphyxiation. Testes, epididymis, vas deferens, seminal vesicles, and ventral prostate were collected and weighed wet to the nearest milligram by clearing off adhering tissues. Tissue somatic indices (TSI) were calculated as follows:
Sperm parameters analysis
The total numbers of sperms were counted by taking cauda epididymis and minced in 5.0 mL of Ham’s F12 medium using Neubauer Chamber as described earlier by Belsey et al. 24 The percentage of motile to non-motile sperms was evaluated microscopically within 5 min following their isolation from cauda epididymis at 37°C. 24 The percentage of live to dead spermatozoa was determined using 1% trypan blue reagent by the method followed by Talbot and Chacon. 25 To analyze the morphologically abnormal sperms, cauda epidydimal sperm suspensions were diluted with 10% normal saline in 1:10 ratio, and smears were prepared on slides and allowed to dry for 90 min to count 200 spermatozoa per animal using a high magnification phase-contrast microscope. 26
Serum hormone levels
To separate serum from blood, the collected blood was kept undisturbed at room temperature for 30 min, centrifuged at 2000 × g for 10 min in a refrigerated centrifuge and stored at −20°C till analysis. The serum levels of testosterone (serial number-H090), LH (serial number-H206), and FSH (serial number-H101) of rats were estimated by enzyme linked immune-sorbent assay (ELISA) kits (Nanjing Jiancheng Bioengineering Institute, Nanjing, China) as per manufacturer’s protocols. Dispense of 10 μL of serum sample and 50 μL of assay diluent were added to pre-coated 96-well ELISA plate and then incubated for 2 h at room temperature. After incubation, the plates were washed five times with wash buffer before addition of 100 μL of peroxidase-conjugated immunoglobulin G (IgG) anti-testosterone or anti-LH or anti-FSH solution into each well and then incubated for 2 h. The plates were washed five times with wash buffer, added with 100 μL of substrate buffer, and incubated for 30 min at room temperature in dark, and the reaction was stopped by the addition of 50 μL of stop solution into each well. Finally, the quantification of testosterone or LH or FSH levels was obtained on a microplate reader at 450 nm. The ELISA kit sensitivity and determination ranges were 0.066 ng/mL and 0.1–25 ng/mL, respectively. The intra-assay coefficients of variation for testosterone, LH, and FSH, were 4.8, 5.4, and 4.1%, respectively. All the samples were run in triplicates and performed at the same time under the same conditions to avoid inter-assay variation.
Testicular steroidogenic and marker enzyme activities
The activities of 3β- and 17β-HSDs in testicular microsomal fraction were performed following the method of Bergmeyer. 27 A total volume of 2 mL reaction mixture contained 100 µmol of sodium pyrophosphate buffer (pH 9.0), 0.5 µmol cofactors (3β-HSD: nicotinamide adenine dinucleotide (NAD); 17β-HSD: nicotinamide adenine dinucleotide phosphate (NADPH)), and 0.08 µmol of substrate (3β-HSD: dehydroepiandrosterone; 17β-HSD: androstenedione) with 100 µL of enzyme source. The absorbance was measured for 3 min with 20-s interval at 340 nm using UV-Visible spectrophotometer. The protein content in the same fraction was determined by Lowry et al. 28 using bovine serum albumin (BSA) as the standard. The enzyme activities of 3β- and 17β-HSD were expressed as nmol NAD converted to NADH per milligram protein per minute and NADPH converted to NADP per milligram protein per minute, respectively.
Testicular spermatogenic marker enzyme activities
Ten percent total testicular homogenate was prepared in potassium phosphate buffer and then centrifuged at 10,000 × g for 10 min at 4°C and the obtained supernatant was used as an enzyme source to estimate the activities of alkaline phosphatase (ALP; serial number-A059-2 -1), acid phosphatase (ACP; serial number-A060-2-2), lactate dehydrogenase (LDH; serial number-A020-2 -1), and sorbitol dehydrogenase (SDH). The activities were determined using diagnostic reagent kits (Nanjing Jiancheng Bioengineering Institute, China). To estimate ACP activity, a mixture of supernatant and buffer (p-nitrophenol phosphate (PNPP) in acetate buffer) was incubated for 30 min at 37°C. The reaction was stopped by addition of 0.1 mol sodium hydroxide (NaOH) provided by the kit. The assay was based on the hydrolysis of PNPP and formation of PNP measured at 420 nm. The ALP and ACP activities were quantified in terms of µg of PNP liberated/milligram tissue/hour from a standard curve. To estimate ALP activity, a mixture of supernatant and buffer (p-nitrophenol phosphate (PNPP) in Tris buffer) was incubated for 30 min at 37°C. The reaction was stopped by addition of 0.1 mol NaOH provided by the kit. The assay was based on the hydrolysis of PNPP and formation of p-nitrophenyl (PNP) measured at 420 nm. The LDH and SDH assays were based on the interconversion of lactate and pyruvate and fructose and sorbitol, respectively. The NADH is oxidized to NAD during the reduction reaction of either pyruvate or fructose. The oxidation of NADH is directly proportional to decrease in absorbance at 340 nm. This decrease in absorbance is a measure of LDH or SDH activity. One unit of LDH or SDH activity is equal to 0.1 μM of substrate transformed/min at pH 7.6 at 25°C. The LDH and SDH activities were measured as units/milligram of protein. The protein content in the supernatant fraction was determined by Lowry et al. 28 using BSA as the standard.
Gene expressions analysis
To estimate the mRNA expression levels of StAR protein, P450scc, and 3β- and 17β-HSDs, total testicular tissue RNA was extracted using a total RNA Extraction Kit (Sangon Biotech Co., Ltd, Shanghai, China) as per manufacturer’s protocol by real-time polymerase chain reaction (RT-PCR). The purity of the total RNA was based on optical density (OD) values at 260/280 nm. The band intensity of 18 S and 28 S ribosomal subunits as a measure of RNA integrity number (RIN) was quantified by densitometric analysis using Image J software (NIH, Bethesda, MD, USA; version 1.42q). The OD values at wavelength 260/280 nm and RIN values were around 1.8–2.2 and 7.8–8.1, respectively. The reverse transcription of cDNA from RNA was performed using cDNA synthesis kit (Sangon Biotech Co., Ltd). The cDNA samples were subjected to PCR analyses using SYBR® Premix Ex Taq™ with the help of Applied Biosystems 7900 Fast RT-PCR System (Applied Biosystems, Stadt, California, USA). RT-PCR data were calculated using the gene expression formula 2−ΔΔCt . All values were normalized by β-actin. The primers used in the study were commercially synthesized by Sangon Biotech Institute Co., Ltd. The PCR primer pairs used in the present study are as follows.
The primer sequences of the target genes are as follows:
F:5′TTGGGCATACTCAACAACCA3′, R:5′ATGACACCGCTTTGCTCAG3′;
F:5′AGGTGTAGCTCAGGACTTCA3′, R:5′AGGAGGCTATAAAGGACACC3′;
F:5′TTGGTGCAGGAGAAAGAAC3′, R:5′CCGCAAGTATCATGACAGA3′;
F:5′TTCTGCAAGGCTTTACCAGG3′, R:5′ACAAACTCATCGGCGGTCTT3′;
F:5′GCCATGTACGTAGCCATCCA3′, R:5′GAACCGCTCATTGCCGATAG3′;
Testicular oxidative stress markers
The oxidative stress makers such as levels of hydrogen peroxide (H2O2; Abcam, Cambridge, UK: ab102500), lactoperoxidase (LPO; Abcam: ab233471), total thiols (Abcam: ab112158) and activity levels of superoxide dismutase (SOD; Abcam: ab65354), catalase (CAT; Abcam: ab83464), and glutathione peroxidase (GPx; Abcam: ab102530) were estimated in 10% testicular homogenate supernatant samples using commercially available kits.
H2O2 levels in samples were estimated by dispensing 50 µL of either standard (0, 1, 2, 3, 4, and 5 nmol/well) or sample into each well in duplicates and added 50 µL of reaction mixture (46 µL of assay buffer, 2 µL of oxiRed probe and 2 µL of horseradish peroxidase (HRP; dissolve in 220 μL Assay Buffer)) and then incubated for 10 min at room temperature in dark and measure OD at 570 nm.
Malondialdehyde (MDA) contents in the samples as a measure of lipid peroxidation were determined by taking different concentrations (0, 6.25, 12.5, 25, 50, 100, and 200 µmoles) of MDA standards and test samples. Dispense of 10 µL of MDA color reagent was provided by the kit into each well and incubated for 20 min and then 40 µL of reaction solution provided by kit was added and incubated for 40 min and then absorbance read at 695 nm.
The total thiols in the samples were estimated by dispensing 50 µL of reaction mixture (100× thiol green indicator stock solution (100 µL of DMSO into vial of thiol green indicator) into 5 mL of assay buffer) and added 50 µL of either glutathione (GSH) standard (0–10 µM) or test samples into each well and incubated for 30 min and then the fluorescence intensity was monitored at Ex/Em = 490/520 nm.
SOD activity in the test samples was estimated by adding 200 µL of WST working solution (dilute 1 mL WST solution with 19 mL of SOD assay buffer) into each well and then added 20 µL of dilution buffer to blanks 2 and 3. After dispensing 20 µL of enzyme working solution (dilute 15 µL with 2.5 mL of dilution buffer) to each sample well and blank 1, the contents were mixed and incubated at 37°C for 20 min and OD measured at 450 nm.
CAT activity in the samples was estimated by preparing H2O2 standard dilutions (0, 2, 4, 6, 8, and 10 nmol/well) in 90 µL and then added 10 µL of stop solution. Samples (5 µL of test sample supernatant diluted to 78 µL), standards of different concentrations (100 µL), and sample high control (HC) (5 µL of test sample supernatant diluted to 78 µL) were taken. The activity in HC is stopped by adding 10 µL of stop solution and incubated at 25oC for 5 min; 12 µL of 1 mM H2O2 solution was added into each sample and sample HC and incubated at 25oC for 30 min; 10 µL of stop solution was added to each sample. After that, 50 µL of developer mix (assay buffer—46 µL, oxired probe—2 µL, HRP solution—2 µL) was added to each well and incubated at 25oC for 10 min. After incubation, absorbance was measured at 570 nm.
GPx activity in the samples was estimated by preparing NADPH standard (reconstitute with 500 µL of dH2O to get a 40-mM NADPH), glutathione reductase (GR; diluted with 220 µL of assay buffer), GSH (reconstituted with 220 µL of assay buffer), cumene hydroperoxide (diluted with 1.25 mL of assay buffer), and GPX positive control (reconstituted with 100 µL of assay buffer). Standard curve was prepared with 0, 20, 40, 60, 80, and 100 nmol/well. Plate was set for standard (100 µL), samples (50 µL), positive control (50 µL), and reagent control wells (50 µL) in duplicates; 40 µL of reaction mixture was added (assay buffer—33 µL, 40 mM NADPH solution—3 µL, GR solution—2 µL, and GSH solution—2 µL) to sample, positive control, and reagent control wells and then the plate was incubated for 15 min at room temperature. After incubation, OD was measured at 340 nm. 10 µL of cumene hydroperoxide solution was added to the sample, positive control, and reagent control wells. OD was measured at 340 nm before and after incubation of plate at 25oC for 5 min.
The levels of H2O2, LPO, and total thiols and activity levels of SOD, CAT, and GPx were measured as nmol of H2O2 generated/min/mg protein, µmol of MDA formed/min/mg protein, nmol/mg protein, units/mg protein/min, H2O2 decomposed/s/mg protein, and GSH oxidized/min/mg protein.
Histology and immunofluorescence
After dissection, testicular tissues were collected from both the control and experimental rats and then immediately fixed in 4% paraformaldehyde. The fixed tissues were properly trimmed by clearing off adhering tissues, processed in ascending concentrations of alcohols, cleared off in xylene, embedded in paraffin, sliced into 5-µm thick sections, and then stained with hematoxylin and eosin stain for histology. The sections were visualized under Olympus phase contrast microscope (Tokyo, Japan) and photomicrographs were captured.
The transverse sections of testes from both the control and experimental rats were also used to evaluate different morphometric parameters such as seminiferous tubular diameter, tubular epithelial thickness, total tubular length, and abnormal seminiferous tubular percentage. Total tubular length and abnormal tubular percentage were evaluated in 15 different fields of each animal using Olympus phase contrast microscope (Tokyo, Japan). Twenty round or nearly round seminiferous tubules were randomly chosen from each section and evaluated seminiferous tubular diameter (both longest and shortest for each tubule) and epithelial thickness with the help of Image Pro Plus program associated with an Olympus phase contrast microscope. 29
For immunofluorescence analysis, deparaffinized and rehydrated sections were heated with 10-mM sodium citrate buffer (pH 6.0) for antigen retrieval with the help of a microwave. The sections were blocked with BSA and were incubated overnight at 4°C with primary antibodies (3β- (ab65156) and 17β-HSDs (ab97971) (Abcam; 1:100)). Once this incubation was over, the sections were incubated with IgG-fluorochrome-conjugated secondary antibody FITC (Santa Cruz Biotechnology, Santa Cruz, California, USA) for 1 h at room temperature. The slides were rinsed three times with phosphate-buffered saline and were mounted with Ultracruz mounting medium and counterstained with 4′,6-diamidino-2-phenylindole to visualize the nuclei. Finally, the sections were viewed under an Olympus phase contrast microscope (Tokyo, Japan).
Statistical analysis
The results were analyzed statistically using one-way analysis of variance followed by the Tukey post hoc test using SPSS version 16.0. The results were expressed as mean ± SD and significance was considered at p < 0.05.
Results
Clinical signs of toxicity
The dams present in all the groups showed neither toxic symptoms nor dead throughout the perinatal period. None of the maternal animals have shown aggressive behaviors or no pregnant animal was excluded from the study and no noticeable signs of toxicity were observed in any of the pups delivered by either control or experimental rats. Furthermore, the results of the present study did not reveal any effect on the sex ratio of the pups (Table 1) nor was there a change in gross malformations.
Effect of maternal exposure to TCDD on sex ratio of pups.

TCDD: 2,3,7,8-tetrachlorodibenzo-p-dioxin.
Reproductive performance
After completion of the experimental period, male rats from both the control and experimental groups cohabited with normal cyclic female rats to check male reproductive efficiency (Table 2). All the rats irrespective of the group exhibited sexual desire as soon as the females were introduced into the cage. Dose-dependent decrease in mating and fertility indices were observed in 0.5, 1, and 2 µg/kg TCDD group of rats compared to the control group of rats. The number of corpora lutea was comparable between the different groups. However, there was a significant decrease in the mean number of implantations in females cohabited with males exposed during the gestational period to 0.5 (9.52 ± 0.23), 1 (7.58 ± 0.38), and 2 (7.27 ± 0.27) µg/kg TCDD compared to the mean number of implantations (13.12 ± 0.42) in females mated with control males. The mean number of live fetuses present in the control, 0.5, 1, and 2 µg/kg TCDD group of rats were 12.82 ± 0.84, 7.38 ± 0.36, 5.54 ± 0.67, and 5.12 ± 0.53, indicating dose-dependent decrease in fertility indices of the experimental rats compared with the control rats. The results showed significantly increased pre- and postimplantation losses in females cohabited with males exposed gestationally to TCDD in comparison with females mated with control males. The resorptions were observed in females mated with 1 and 2 µg/kg TCDD-exposed rats.
Effect of maternal exposure to TCDD on reproductive performance in adult male rats.a
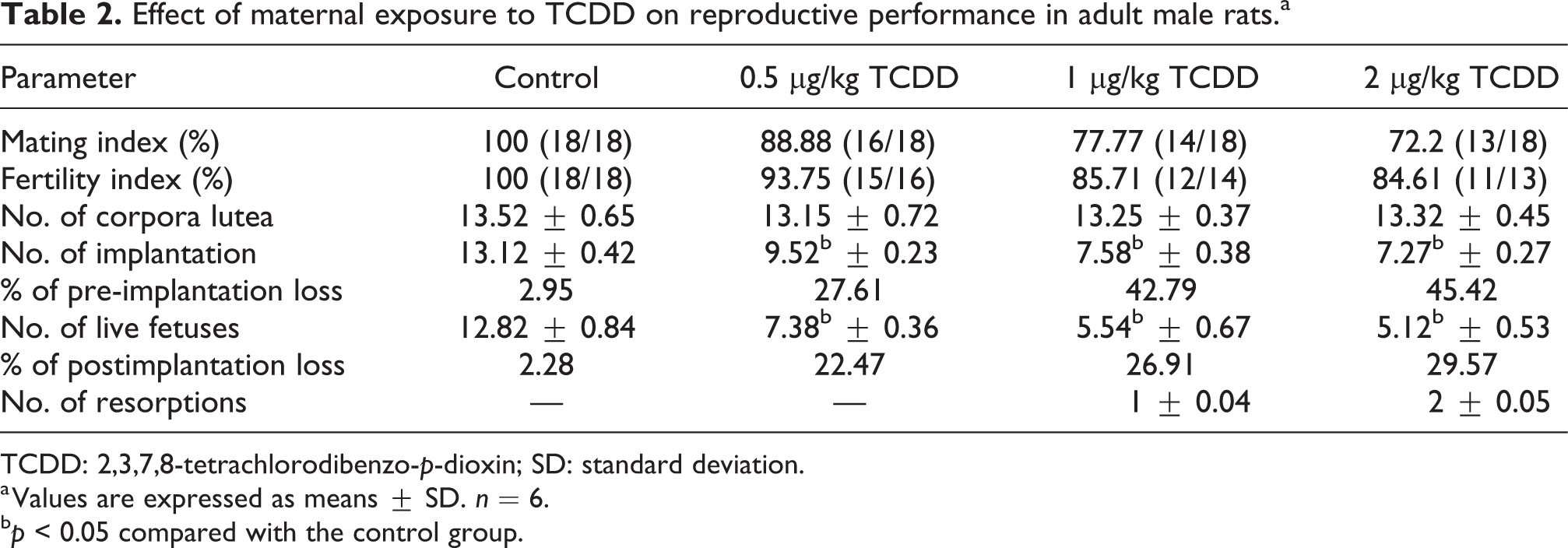
TCDD: 2,3,7,8-tetrachlorodibenzo-p-dioxin; SD: standard deviation.
a Values are expressed as means ± SD. n = 6.
b p < 0.05 compared with the control group.
Body and organ weight
No significant change was observed in feed and water intake of the experimental group of rats compared with control rats (data not provided). The final body weight was comparable in all groups of adult rats (Figure 1(a)); however, the absolute and relative organ weights of testes (Figure 1(b) and (c)), epididymis (Figure 1(d) and (e)), seminal vesicles (Figure 1(h) and (i)), and absolute organ weight of ventral prostate (Figure 1(f)) were significantly decreased in rats exposed to 0.5, 1, and 2 µg/kg TCDD during the gestational period.

Effect of maternal exposure to TCDD on body weight and reproductive organ (absolute and relative) weights in adult male rats: (a) body weight; (b) testes weight; (c) testes TSI; (d) epididymis weight; (e) epididymis TSI; (f) ventral prostate; (g) ventral prostate TSI; (h) full seminal vesicle weight; and (i) full seminal vesicle weight TSI. Values are expressed as means ± SD. n = 6; *p < 0.05 compared with the control group. TCDD: 2,3,7,8-tetrachlorodibenzo-p-dioxin; TSI: tissue somatic indices; SD: standard deviation.
Spermatology
The sperm count (Figure 2(a)), percent viable (Figure 2(b)), percent motile (Figure 2(c)), and percent abnormal morphological sperms (Figure 2(d)) in the control and prenatal TCDD-exposed rats are depicted in Figure 2. The results revealed a significant decrease in the mean epididymal sperm count and mean percentage of viable sperm in 1 and 2 µg/kg TCDD-administered rats. The mean percentage of motile sperm was significantly decreased with a significantly increased mean total abnormal sperm percentage in rats exposed to 0.5, 1, and 2 µg/kg TCDD during the gestation period compared with the control rats.

Effect of maternal exposure to TCDD on epididymal sperm parameters in adult male rats: (a) sperm count; (b) sperm viability; (c) sperm motility; and (d) total abnormal sperm. Values are expressed as means ± SD. n = 6; *p < 0.05 compared with the control group. TCDD: 2,3,7,8-tetrachlorodibenzo-p-dioxin; SD: standard deviation.
Testicular steroidogenic markers and serum hormonal levels
The enzyme activity levels of 3β- (Figure 3(a)) and 17β-HSDs (Figure 3(b)) were found to be significantly decreased in testes of adult rats exposed in utero to 0.5, 1, and 2 µg/kg TCDD when compared with the respective control rats. Furthermore, the immunofluorescence sections showed reduced distribution of 3β-HSD (Figure 4(a)) and 17β-HSD (Figure 4(b)) proteins in testes of adult rats which were exposed to 0.5, 1, and 2 µg/kg TCDD during their prenatal period.

Effect of maternal exposure to TCDD on testicular steroidogenic (3β- and 17β-HSDs) enzyme activities in adult male rats: (a) 3β-HSD and (b) 17β-HSD. Values are expressed as means ± SD. n = 6; *p < 0.05 compared with the control group. TCDD: 2,3,7,8-tetrachlorodibenzo-p-dioxin; HSD: hydroxysteroid dehydrogenase; SD: standard deviation.

Effect of maternal exposure to TCDD on protein distribution of 3β- and 17β-HSDs in testes of adult male rats. Immunofluorescence expression of (a) 3β-HSD and (b) 17β-HSD. Magnification: 400×; scale bar: 100 µm. White arrow indicates the expression of target protein. TCDD: 2,3,7,8-tetrachlorodibenzo-p-dioxin; HSD: hydroxysteroid dehydrogenase.
The results also revealed a significant decrease in serum levels of testosterone (Figure 5(a)), FSH (Figure 5(b)), and LH (Figure 5(c)) in adult rats exposed to 0.5, 1, and 2µg/kg TCDD on gestational day 15 when compared with the respective control rats.

Effect of maternal exposure to TCDD on serum hormonal levels in adult male rats: (a) testosterone; (b) FSH; and (c) LH. Values are expressed as means ± SD. n = 6; *p < 0.05 compared with the control group. TCDD: 2,3,7,8-tetrachlorodibenzo-p-dioxin; FSH: follicle stimulating hormones; LH: luteinizing hormone; SD: standard deviation.
Testicular oxidant and antioxidant status
The testicular production of H2O2 (Figure 6(a)) and the contents of testicular MDA (Figure 6(b)) were significantly elevated in testes of rats after exposure to 0.5, 1, and 2 µg/kg TCDD during the early stages of development when compared with the same levels in the respective control rats indicating generation of reactive oxygen species (ROS) and oxidative stress in testes of TCDD-exposed rats. Conversely, in utero exposure to 0.5, 1, and 2 µg/kg TCDD to rats resulted in a significant diminution in the testicular antioxidant status, as revealed by the significant decrease in the activities of SOD (Figure 6(c)), CAT (Figure 6(d)), GPx (Figure 6(e)), and total thiols (Figure 6(f)) compared with those of control rats.

Effect of maternal exposure to TCDD on oxidant and antioxidant parameters in testes of adult male rats: (a) H2O2 production; (b) LPO; (c) SOD; (d) CAT; (e) GPx; and (f) total thiols. Values are expressed as means ± SD. n = 6; *p < 0.05 compared with the control group. TCDD: 2,3,7,8-tetrachlorodibenzo-p-dioxin; H2O2: hydrogen peroxide; LPO: lactoperoxidase; SOD: superoxide dismutase; CAT: catalase; GPx: glutathione peroxidase; SD: standard deviation.
Testicular spermatogenic markers
The activities of spermatogenic markers such as ACP (Figure 7(a)), ALP (Figure 7(b)), LDH (Figure 7(c)), and SDH after exposure to TCDD during their critical period of development are depicted in Figure 7. Except for a nonsignificant increase in ACP activity in 0.5 µg TCDD/kg body weight-treated rats, the activities of ACP, ALP, and LDH were significantly increased with a significant decrease in SDH activity in testes of prenatal 0.5, 1, and 2 µg TCDD/kg-exposed rats when compared to the same activities in the respective control rats.
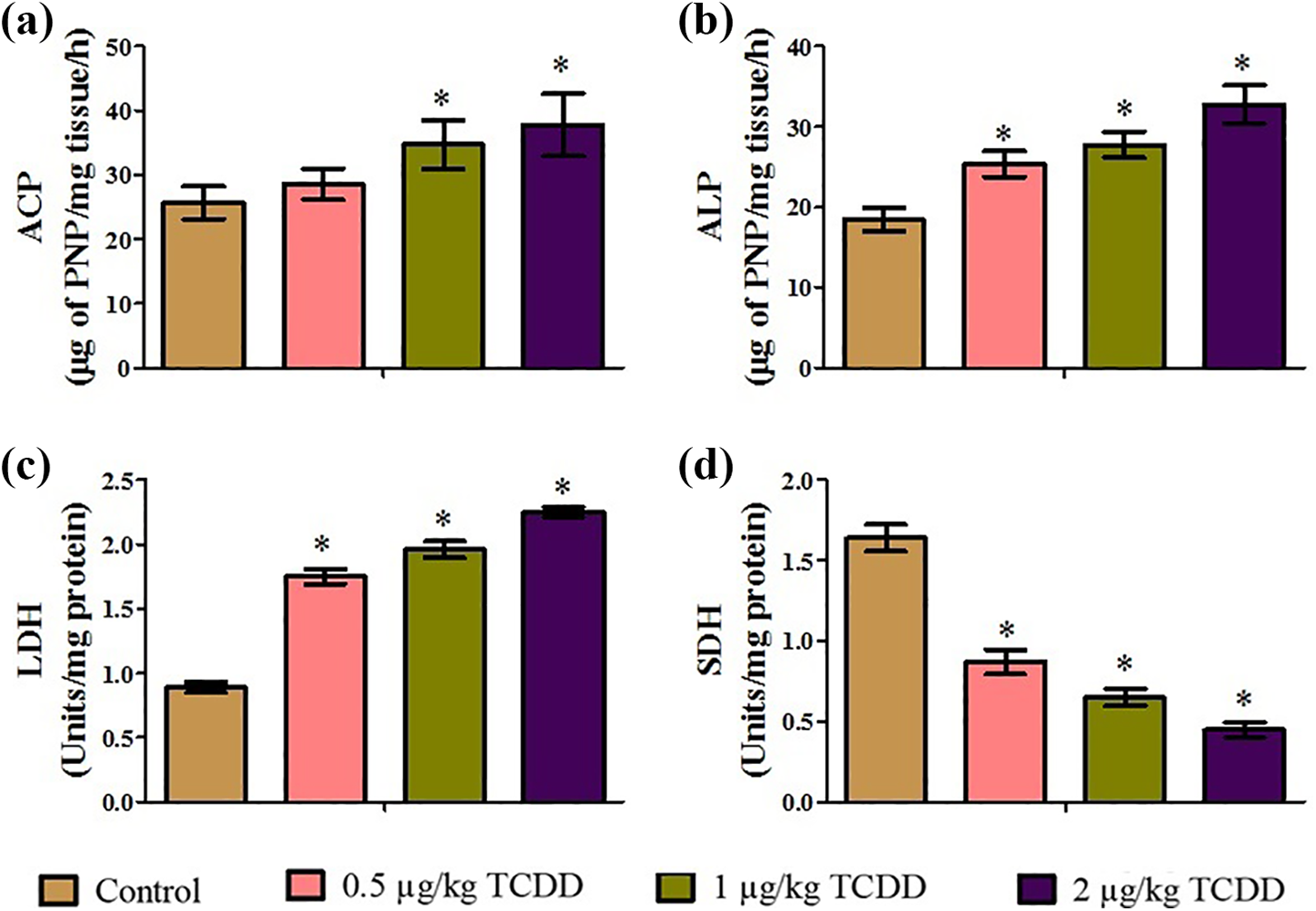
Effect of maternal exposure to TCDD on spermatogenic in testes of adult male rats: (a) ACP; (b) ALP; (c) LDH; and (d) SDH. Values are expressed as means ± SD. n = 6; *p < 0.05 compared with the control group. TCDD: 2,3,7,8-tetrachlorodibenzo-p-dioxin; ACP: acid phosphatase; ALP: alkaline phosphatase; LDH: lactate dehydrogenase; SDH: sorbitol dehydrogenase; SD: standard deviation.
Steriodogenic and StAR mRNA levels
The relative mRNA expression levels of StAR (Figure 8(a)), P450scc (Figure 8(b)), 3β-HSD (Figure 8(c)), and 17β-HSD (Figure 8(d)) were dose-dependently decreased in gestational 0.5, 1, and 2 µg/kg TCDD-treated rats compared to the corresponding expression levels in the control rats.
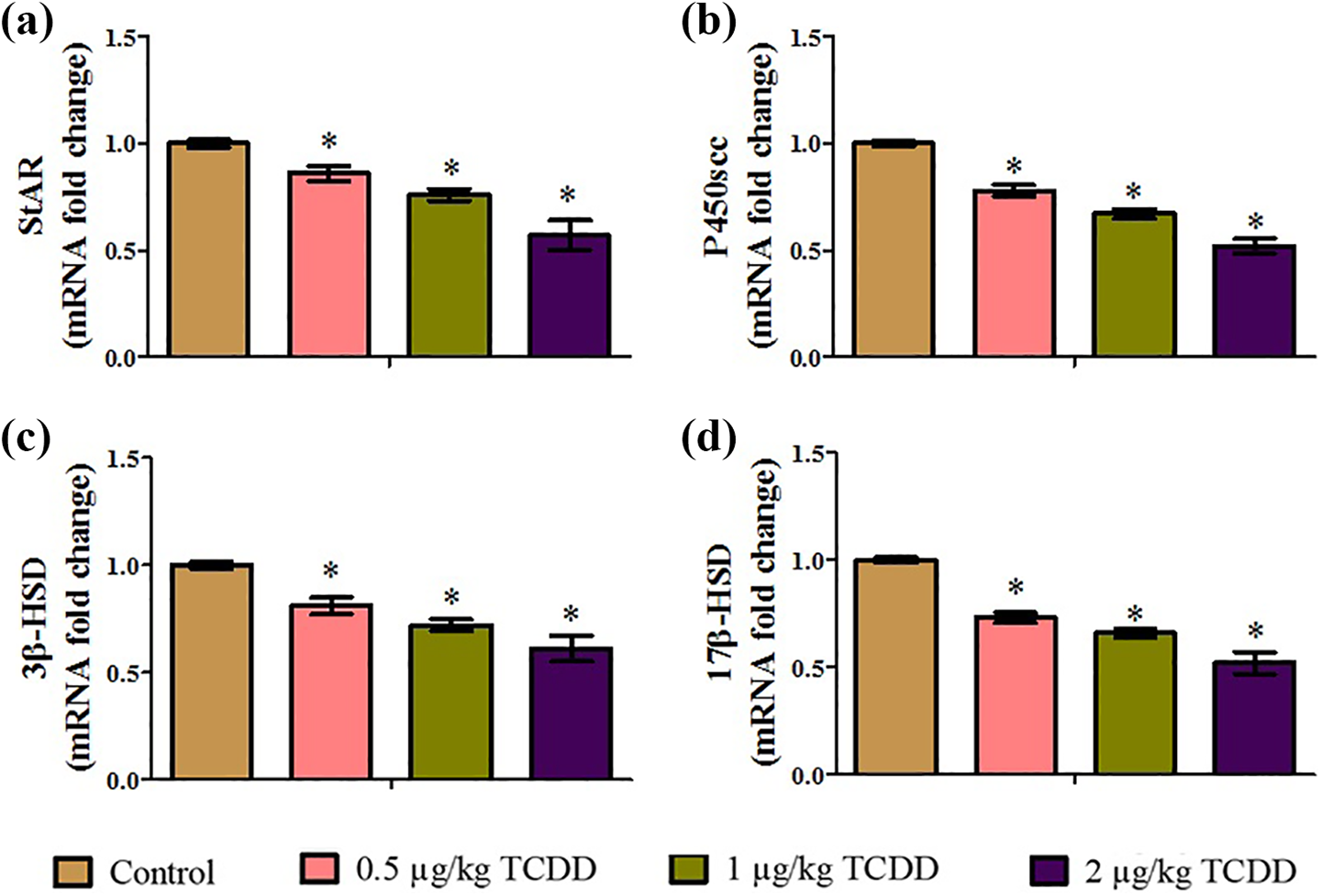
Effect of maternal exposure to TCDD on mRNA levels of steroidogenic genes in testes of adult male rats: (a) StAR; (b) P450scc; (c) 3β-HSD; and (d) 17β-HSD. Values are expressed as means ± SD. n = 6; *p < 0.05 compared with the control group. TCDD: 2,3,7,8-tetrachlorodibenzo-p-dioxin; StAR: steroidogenic acute regulatory; P450scc: cholesterol side-chain cleavage; HSD: hydroxysteroid dehydrogenase; SD: standard deviation.
Testicular histology
The microphotographs of testes in the control and experimental rats are depicted in Figure 9(a). The transverse sections of the control rat testes revealed closely arranged seminiferous tubules, clusters of Leydig cells in interstitium, intact basement membrane, different stages of spermatogenic cells, and lumen with full of sperms. While mild, moderate, and severe degenerative changes were observed in the testicular cross sections of prenatal 0.5, 1, and 2 µg/kg TCDD-exposed rats, respectively.

Effect of maternal exposure to TCDD on histological and morphometric changes in adult testes of control and experimental rats: (a) transverse sections of testes; (b) seminiferous tubular diameter (µm); (c) germinal layer thickness (µm); (d): total tubular length (µm); and (e) abnormal seminiferous tubules (%).Values are expressed as means ± SD. n = 6; *p < 0.05 compared with the control group. Magnification: 400×; scale bar: 100 µm. TCDD: 2,3,7,8-tetrachlorodibenzo-p-dioxin; LC: Leydig cells, ST: seminiferous tubules; SD: standard deviation.
In addition, the evaluation of the testicular morphometric parameters revealed a significant decrease in seminiferous tubular diameter (Figure 9(b)), tubular epithelial thickness (Figure 9(c)), and total tubular length (Figure 9(d)) with a significant increase in the mean percentage of abnormal seminiferous tubules (Figure 9(e)) in experimental rats compared with control rats.
Discussion
Since the developmental period is more sensitive to toxic insults of TCDD, there is a possibility that maternal exposure to TCDD affects at a later stage of life, that is, adulthood. A comprehensive study was performed to assess the possible effects on male reproduction at adulthood in rats exposed to TCDD at the critical stage of organogenesis, that is, GD15. In this study, we used three doses of TCDD (0.5, 1, and 2 µg/kg) to characterize the effects of gestational exposure to TCDD on the male reproductive functions in adult male Wistar rats. The exposure of rats to TCDD on GD15 did not cause any maternal toxicity but caused significant deterioration in reproductive end points such as sperm quantity and quality, steroidogenic and spermatogenic markers, serum hormonal levels, oxidative and antioxidative status, morphology of testes, and male reproductive efficiency in their adulthood.
The selected doses did not cause any maternal toxicity as evidenced by their behavioral observations and fecundity. No significant alterations were observed in the body weight gain of adult male experimental rats compared to control rats indicating no overt general toxicity. Whereas the absolute and relative weights of testes and accessory sex organs were significantly reduced in adult male rats exposed to 0.5, 1, and 2 µg/kg TCDD on GD15, indicating that these organs are sensitive targets to toxic insults of TCDD. The decrease in the reproductive organ weights is in consonance with earlier reports. 30,31 The observed decrease in weights of testes and accessory sex organs might be due to reduced bioavailability of testosterone and/or loss of structural integrity. 32 The findings were further supported by degenerative changes in seminiferous tubules with loss of germ cells and spermatogenesis in the testicular cross sections of prenatal TCDD-exposed rats.
The analysis of reproductive endpoints such as sperm count, motility, viability, and its morphology gives valuable information regarding the health status of male reproduction. 33 In this study, exposure of dams to 0.5, 1, and 2 µg/kg TCDD during the gestational period resulted in a significant decrease in the total motile and viable sperm with a significant increase in sperm morphological abnormalities in their adulthood, indicating compromised quantity and quality of sperm. The observations are in line with previous reports, 23,34 indicating the TCDD-mediated toxic effects on sperm quality and functionality.
Testosterone is a male crucial androgen synthesized in Leydig cells of testis that plays a critical role in the development of the male reproductive organs and maintenance of spermatogenesis. The testosterone is synthesized under the influence of many proteins and enzymes: StAR, the mitochondrial membrane protein transports cholesterol from cytoplasm to mitochondria 35 ; the mitochondrial P450scc enzymes takes the role of cleaving cholesterol into pregnenolone 35,36 ; the pregnenolone is transported into the endoplasmic reticulum to synthesize testosterone under the influence of a battery of enzymes such as 3β- and 17β-HSDs. 36 In this study, the gestational exposure of rats to 0.5, 1, and 2 µg/kg TCDD caused a significant decrease in the mRNA expression levels of StAR, P450scc, and 3β- and 17β-HSDs in their adulthood compared to the same mRNA levels in control rats. Furthermore, gestational exposure to TCDD has resulted in a significant decrease in the 3β- and 17β-HSD enzyme activities and protein distribution in testes of rats. The results were well supported by reported observations of reduced expression of steroidogenic marker mRNA levels. 37 The results were further supported by decreased levels of serum testosterone in the current study. The decreased levels of testosterone in this data might be due to the observed effect of TCDD on the steroidogenic machinery (StAR, P450scc, and 3β- and 17β-HSDs), as these are involved in the synthesis of testosterone. 38 The data of the present study also revealed a significant decrease in the serum hormonal levels of FSH and LH. The results corroborated with the earlier reports. 39 Normal levels of FSH and LH are required for spermatogenesis, and reduction in the levels of FSH and LH and subsequent decrease in testosterone levels might be the reason behind the TCDD-induced alterations in spermatogenesis.
The activities of spermatogenic markers such as ACP, ALP, and LDH were significantly elevated with a significant decrease in SDH activity in experimental adult rats exposed to TCDD during the prenatal period. The increased activities of ACP and ALP in experimental rats designate degenerative changes and lytic activity in testicular parenchyma. 40 The observed increase in LDH activity in association with decrease in SDH reflects a deteriorated germinal epithelial layer of seminiferous tubules in testes of experimental rats. The activities of these enzymes are regulated by testosterone and alterations in these enzyme activities might be due to decreased bioavailability and/or production of testosterone in prenatal TCDD-exposed rats. 41,42
The alterations in sperm functions, spermatogenesis, and steroidogenic machinery after TCDD exposure might be due to elevated testicular production of ROS and lipid peroxidation. 43,44 In the present investigation, exposure of rats to 0.5, 1, and 2 µg/kg TCDD during the developmental period resulted in elevated generation of H2O2 and MDA, reflecting the induction of ROS and oxidative stress in adult rat testes. The observed rise in testicular free radicals in TCDD rats might cause damage to the steroidogenic and spermatogenic machinery leading to lipid peroxidation and oxidative damage that progress to their functional inactivation. 45 Testicular cells are equipped with antioxidant defenses to protect the oxidative damage in cells: SOD dismutates the highly toxic superoxide anion radicals into hydrogen peroxide; 40 CAT neutralizes H2O2 into oxygen and water 46 ; and GPx reduces the lipid and hydrogen peroxides using GSH. 47 The reduced activities of SOD, CAT, and GPx in testes of rats that had been exposed to TCDD during critical stages of development, that is, on GD15 may have further increased the generation of ROS and lipid peroxidation, which is in par with earlier observations. 39 Furthermore, the reduction in the level of total thiols in experimental rats is an indication of its dependent functional inactivation. 48 The results indicate that TCDD-induced oxidative stress in the form of elevated levels of ROS and MDA and reduced antioxidant defenses by means of reduction in activities of SOD, CAT, and GPx and levels of total thiols might be responsible for the detrimental effects on the male reproductive functions, supporting the notion that oxidative stress is responsible for the pathophysiology of testes. 48
Another reproductive endpoint tested was to know whether maternal exposure to TCDD during the critical stage of development can have effects on the male offspring’s fertility in their adulthood. In this study, when the normal cyclic females cohabited with male rats that were gestationally exposed to 0.5, 1, and 2 µg/kg TCDD showed a significant effect on the fertility as revealed by a dose-dependent decrease in the mating and fertility indices. Furthermore, the observed decrease in the number of implantations and fetuses and increase in the pre- and postimplantation losses suggests suppressed fertility that might have resulted from reduced quantity and quality of sperm. The results are in line with earlier observations of reduced fertility in their adulthood in male rats exposed to TCDD during the gestational period. However, the present findings warrant in-depth fertility parameters such as sperm chromatin integrity and artificial insemination with a fixed number of cauda epididymal sperms that will actually provide complete information while assessing fertility in experimental rats.
Conclusion
In conclusion, the results of the present study indicate that prenatal exposure to TCDD can have detrimental effects on male reproductive functions such as fertility, steroidogenesis, and spermatogenesis in their adulthood. Particularly, gestational exposure to TCDD resulted in an increased testicular oxidative stress, downregulated steroidogenic and spermatogenic markers, decreased serum testosterone levels, deteriorated testicular architecture with morphometric changes, and suppressed fertility in their adulthood.
Footnotes
Author contributions
XM carried out the experiments, data analysis, manuscript writing, and funding; YD carried out the experimental design and conducted the experiments; LX carried out data analysis and manuscript editing, and ZE contributed to manuscript writing. All the authors read and approved the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.

