Abstract
In order to assess the effect of arsenic on the male reproductive impairment in mice, 7-week-old animals were exposed to 7.5 mg sodium arsenite (NaAsO2)/kg body weight, during 35 days (one spermatogenic cycle). One group of animals was sacrificed after exposure, while another group received distilled water for an additional period of 35 days, in order to study the spermatoxic effect and the recovery of spermatogenesis. In mice sacrificed after NaAsO2 exposure, a decrease in testis/body weight ratio and reduction of tubular diameter were observed. Both groups of NaAsO2-exposed animals showed remarkable histopathological changes, such as sloughing of immature germ cells. Animals sacrificed after NaAsO2 exposure showed decreased sperm motility, increased abnormal sperm morphology and decreased sperm viability. The effects of NaAsO2 on sperm motility recovered to normal values after one spermatogenic cycle, while increased sperm abnormality was maintained. However, at this period, a decrease in acrosome integrity was detected. Concerning oxidative stress parameters, animals sacrificed after NaAsO2 exposure showed a decreased selenium-dependent glutathione peroxidase activity, which was not detected after the recovery. Conversely, at this period, total glutathione peroxidase activity increased in exposed animals. These results demonstrate the toxic effects of NaAsO2 on mice spermatogenesis and show the lack of recovery after one spermatogenic cycle.
Introduction
Arsenic, in the form of trivalent arsenite (As3+) or pentavalent arsenate (As5+), is a ubiquitous toxic compound naturally occurring in the environment. Arsenical compounds are environmental toxicants that exert multiple effects, on both animals and humans. 1,2 Humans are exposed to arsenic mainly through oral or inhalation route. Oral exposure occurs via consumption of contaminated water and food. Thus, exposure to arsenic compounds may be chronic, 2,3 especially if consumption occurs via drinking water.
Several natural and anthropogenic processes have been implicated in high levels of arsenical compounds reported in various regions of the world. Chronic exposure to arsenic mainly through contaminated drinking water has been reported in many countries, such as Argentina, Bangladesh, India, Mexico, Thailand, Taiwan 4 and USA. 5 This scenario has become an emerging global issue of public health, 6 largely because of the concern with the metalloid’s multisite carcinogenic potential, causing tumors in a variety of human tissues including kidney, liver, skin, lung, bladder, uterus and prostate. 7,8 One of the most studied scenarios of arsenic contamination is present in the Bengal Delta Plain. 9 According to this research paper, the contamination is widespread and is not limited to specific geographical location; the presence of arsenic is reported in thousands of villages, consequently affecting a large number of people (estimated around 200,000 inhabitants). Authors found that the source of arsenic contamination is strictly natural and the quantified levels were variable, evidencing the involvement of a complex geochemical genesis. Variations in arsenic levels were mostly connected to seasonality, but systematically higher amounts were found in hand pumps. Arsenic contamination may not derive only from natural sources. The release of arsenic from aquifers is favored in this region due to specific characteristics, such as adequate redox conditions and the occurrence of organic matter degradation. 10 The work by others 11 showed that the use of fertilizer could result in the presence of significant amounts of arsenic in the ecosystem. Arsenic contamination is not limited to ground water and may also occur due to the use of contaminated irrigation water, as observed by Dahal et al. 12 Anthropogenic sources of arsenic contamination are also noteworthy. The work conducted by Lu et al. 13 showed that roadway dust is an important source of arsenic, and the levels determined in this type of matrix were well above the legal established limits in China. Mining procedures are one of the most important contributing sources for arsenic environmental widespread contamination, as shown by others. 14
The acute toxicity of arsenic is related to its chemical form and oxidation state. In the human adult, the lethal range of inorganic arsenic is estimated at a dose of 1–3 mg As/kg body weight (bw). The LD50 values of several arsenic compounds in mice range from 26 to 48 mg As/kg bw for arsenic trioxide via oral exposure, 8 mg As/kg bw for arsenite compounds and 22 mg As/kg bw for arsenate compounds, both via intramuscular (i.m.) injection. 1
Arsenic can cause other type of effects, besides mortality. The effect of arsenic on male reproductive system is associated with spermatoxicity, leading to inhibition of testicular androgenesis, and reduction of testis and accessory sex organs weights. 15 Arsenic exposure also causes significant decrease in sperm counts and motility, with a simultaneous increase in abnormal sperm. Arsenic exposure has a suppressive influence on spermatogenesis, and exposures to low dosages have been related to testicular cytotoxicity. 16 –18
The main toxicological mechanisms by which arsenic compounds exert deleterious effects have already been documented. Arsenic induces oxidative stress, through the generation of reactive oxygen species (ROS) that cause direct oxidative injuries leading to cell death. 19 High levels of inorganic arsenic inhibit cellular proliferation and induce apoptosis through the generation of hydrogen peroxide (H2O2), consequently to the activation of flavoprotein-dependent superoxide-producing enzymes (ie, nicotinamide adenine dinucleotide phosphate [NADPH] oxidase). It also involves an early decrease of the peroxide potential in the mitochondrial membrane. This event probably acts as a mediator to induce apoptosis through the release of cytochrome c to the cytosol and the activation of caspase 3, leading to DNA fragmentation. 19 H2O2 is one of the ROS secreted by macrophages closely aligned with Leydig cells in the testicular interstitium 20 following arsenic exposure. Cells are normally protected against oxidative damage by multiple enzymatic mechanisms and by antioxidant molecules. The first enzymes to be involved in the oxidative protective mechanism are cytosolic Cu/Zn and mitochondrial Mn superoxide dismutases (SOD); these forms scavenge O2·− by converting it to H2O2, thus preventing cell injury mediated by oxygen radicals, in various circumstances. H2O2 is then metabolized either by peroxisomal catalase or by cytosolic and mitochondrial selenium glutathione peroxidase (Se-GPx). Organic peroxides are reduced by the isoenzymes glutathione-S-transferases (GSTs), which catalyze the reaction between the mentioned compound and the intracellular scavenger glutathione (GSH), in its reduced form. When peroxides are detoxified by different GSH peroxidases (GPx), GSH is converted to oxidized glutathione (GSSG). This dissulfide peptide is subsequently reduced by an NADPH-dependent enzyme, the glutathione reductase (GR), thus maintaining the GSH/GSSG ratio, which is important to maintain the intracellular redox homeostasis. These enzymes work simultaneously, and probably prevent the formation of highly cytotoxic hydroxyradical, 21 the causative agent form of arsenic-derived toxicity. The formation of those hydroxyradical may cause impairment of spermatogenesis, as indicated by previous studies. 20 Thus, the evaluation of sodium arsenite’s deleterious effects on the reproductive features of mice is relevant, the present work being specially focused on pathological changes of testis and epididymis. Possible spermatogenesis recovery, after arsenic-derived injury, was also studied. Since the expected toxicity of arsenic may derive from the impairment of antioxidant defenses, we assessed the effects of enzymatic parameters to understand the toxicological mechanisms involved. To attain this objective, we studied parameters such as testicular histology, sperm functional parameters and the activity of oxidative stress biomarkers, such as activity of enzymes GSTs, total GPx and Se-dependent GPx.
Materials and methods
Chemicals
Sodium arsenite (NaAsO2), 4-(2-hydroxyethyl) piperazine-1-ethanesulphonic acid (HEPES), Coomassie Blue, bovine serum albumin (BSA), tris-HCl, ethylenediaminetetraacetic acid (EDTA), ethylenediaminetetraacetic acid disodium salt (Na2EDTA), 1-chloro-2,4-dinitrobenzene (CDNB), glutathione reductase (GR), nicotinamide adenine dinucleotide phosphate (NADPH), sodium azide (NaN3), hydrogen peroxide (H2O2), cumene hydroperoxide, reduced glutathione (GSH), oxidized glutathione (GSSH), bovine gamma globulin and Triton X-100 were purchased from Sigma (St. Louis, MO, USA). Bradford reagent was acquired from Biorad® (CA, USA).
Animal exposure of arsenic compound
Mice experiments were conducted in agreement with the institutional guidelines for ethics in animal experiments (Rule no. 86/609/CEE–24/11/92). Seven-week-old male ICR-CD1 mice (Charles River Laboratories, L′Arbresle, France), weighing between 30 and 40 g, were housed in stainless steel cages (
Histopathological assessment
The right side testis and epididymis from all groups of animals were fixed by immersion in Bouin’s solution, dehydrated in ethanol series and embedded in paraffin wax. Sections of 5 μm thickness were prepared in a microtome Leitz 1512, (Leitz, Wetzlar, Germany) and stained with haematoxylin and eosin (HE). Thirty testicular and epididymis sections were randomly selected from different slides from each group of animals and analyzed using a light microscope Olympus BX41 (Olympus, Tokyo, Japan).
Measurement of seminiferous tubules diameter
Tubular diameter was measured using Snakes software as described by others. 22 Images were acquired with a camera Olympus Camedia C-5060 connected to a microscope Olympus BX41 (Olympus, Tokyo, Japan), and after loading the image, several points in the tubule contour were picked up as detailed by others. 23 Thirty tubules were randomly selected from three different slides from each group of animals and measured.
Functional parameters of sperm cells
Density
Caudal epididymis were disrupted on capacitating Modified Tyrode medium (MT6) containing 125 mM NaCl, 2.7 mM KCl, 0.5 mM MgCl26H2O, 0.36 mM NaH2PO42H2O, 5.56 mM glucose, 25 mM NaHCO4, 1.8 mM CaCl2, 100 units penicillin and 4 mg/ml BSA 24 and were left for 30 min at 36°C to promote the release of mature sperm cells into the media. Concentration of sperm cells was determined by counting the number of cells on a haemocytometer (Neubauer Improved Chamber) as described by others. 25 The number of sperm cells/ml was calculated by multiplying the mean by 104.
Viability
An aliquot of sperm in MT6 medium was transferred onto a slide containing 0.5% eosin solution. For each slide at least 200 sperm cells were counted with a proper counting apparatus, under ×400 magnification. This procedure involved a duplicated analysis of each sperm sample in two different slides. The results were expressed as the average of the living (without dye presence) and dead (red dyed) sperm cells in duplicate slides. This procedure was performed and adapted as described by others. 26
Acrosome integrity
An aliquot of sperm cells in MT6 medium was transferred to a slide and fixed in a solution containing 5% phosphate-buffered saline (PBS) formaldehyde solution during 30 min at 23°C. Fixed spermatozoa were collected by centrifugation at 1000
Morphology
Sperm cells morphology was performed as described by others. 23 An aliquot of sperm suspension in MT6 medium was smeared on a glass slide, dried and fixed with 95% ethanol. Samples were then stained with haematoxylin-eosin and the incidence of spermatozoa abnormalities in head, mid-piece and tail was evaluated by observation of 200 sperm cells under ×1000 magnification. Spermatozoa morphology was classified according to Wyrobek and Bruce. 28
Motility
Sperm motility was assessed by categorization into four groups of motility at 37°C according to others. 26 This parameter was measured using an aliquot of sperm cells in capacitating MT6 medium onto a slide, and the motility of sperm cells was determined by scoring the number of all rapid progressive and slowly progressive spermatozoa, and the nonprogressive and than immotile spermatozoa in the same field. For each slide, about 100 sperm cells were counted and duplicate slides were assessed for each sample. The number of sperm cells in each motility group was divided by the total number of cells, and the average of two slides was assessed according to others. 29
Enzymatic assays
Sample preparation
After killing and tissue removal, the left side testes were frozen in liquid nitrogen. Testicular tissue was then thawed and homogenized on ice-cold phosphate buffer 50 mM, pH 7.0 containing Triton X-100 0.1%, centrifuged at 15,000
Assay for GSTs and GPx activities
The supernatant previously collected was diluted 1:20 in a PBS 0.1 M, pH 6.5. GSTs activity was assessed by the method of other authors, 31 using CDNB as substrate. GSTs catalyze the conjugation of the substrate CDNB with glutathione forming a thioether that can be followed by the increment of absorbance at 340 nm. Results were expressed as nanomoles of thioether produced per minute, per milligram of protein.
For the determination of GPx activity, the supernatant previously collected was diluted 1:20 in a PBS 100 mM, pH 7.0. GPx activity was measured indirectly by monitoring the consumption of NADPH at 340 nm using the H2O2 for the determination of selenium-dependent glutathione (Se–GPx), and cumene hydroperoxide for total GPx activity, according to others. 32 In this assay, the oxidation of NADPH was monitored at 340 nm, when oxidized dimeric glutathione (GSSG) is reduced to monomeric glutathione (GSH) by glutathione reductase (GRed). Enzymatic activity is expressed as nanomoles of NADPH per minute per milligram of protein. The enzymatic activity, using H2O2 (0.255 mM) as substrate, corresponds to selenium-dependent GPx; the enzymatic activity evaluated using cumene hydroperoxide (0.7 mM) as substrate corresponds to selenium-dependent and selenium-independent GPx activities.
Protein quantification
Protein concentration of samples was determined according to the protocol of other authors, 33 in order to express enzymatic activities taking into account the protein content of the analyzed tissues.
Statistical analysis
The statistical analyses of weights, ratio weights, sperm parameters and morphometric results were performed by using a one-way analysis of variance (ANOVA) followed by a multiple comparison Tukey test to compare differences between control and sodium arsenite-treated groups. The
Results
Absolute and relative testis and epididymis weight
No differences in animal’s survival in any of the group animals were observed following NaAsO2 exposure. In all treated groups, the body, testis and epididymis weights were not significantly different from that of the controls (Table 1
). Following 35 days of exposure to 7.5 mg NaAsO2/kg bw (group 3), there was a significant decrease (
Effect of NaAsO2 on testis/body weight ratio, epididymis/body weight ratio and seminiferous tubules diameter of control mice and mice exposed to 7.5 mg NaAsO2/kg bw a

bw: body weight, NaAsO2: sodium arsenite.
a Values represent mean ± S.D.
b Significant difference between the control and NaAsO2-treated samples at
Histopathological assessment
Testis histopathology and tubular diameter
The histopathological examination of testis, stained with haematoxylin and eosin, showed a normal pattern of spermatogenesis for all control animals (groups 1 and 2; Figure 1a and b). On the contrary, animals exposed to 7.5 mg NaAsO2/kg bw during 35 days (group 3) exhibited a significant decrease (

Histological sections of testis following 7.5 mg sodium arsenite (NaAsO2)/kg bw treatment in mice. Representative section from control testis (a) and (b) shows normal spermatogenesis. NaAsO2 treatment after 35 days (group 3) shows disorganization of the testicular seminiferous epithelium (arrow) with sloughing of immature germ cells (*; c). NaAsO2 treatment after 70 days (group 4) shows remarkable epithelium desquamation, sloughing of immature cells and the presence of intraepithelial vacuoles (d). Haematoxylin and eosin (HE), scale bar = 100 μm.
Furthermore, animals treated with 7.5 mg NaAsO2/kg bw during 35 days (group 3) displayed a disorganized germinative epithelium with sloughing of immature germ cells into the tubular lumen (Figure 1c). Severe disorganization of testicular seminiferous epithelium was also noted in animals treated with 7.5 mg NaAsO2/kg bw followed by exposure to distilled water during 70 days (group 4; Figure 1d) with disruption of seminiferous epithelium, sloughing of immature cells and the presence of intraepithelial vacuoles (Figure 1d).
Epididymis histopathology
Histopathological assessment of mice epididymis revealed a normal cellular structure for all animal groups (Figure 2a–d).
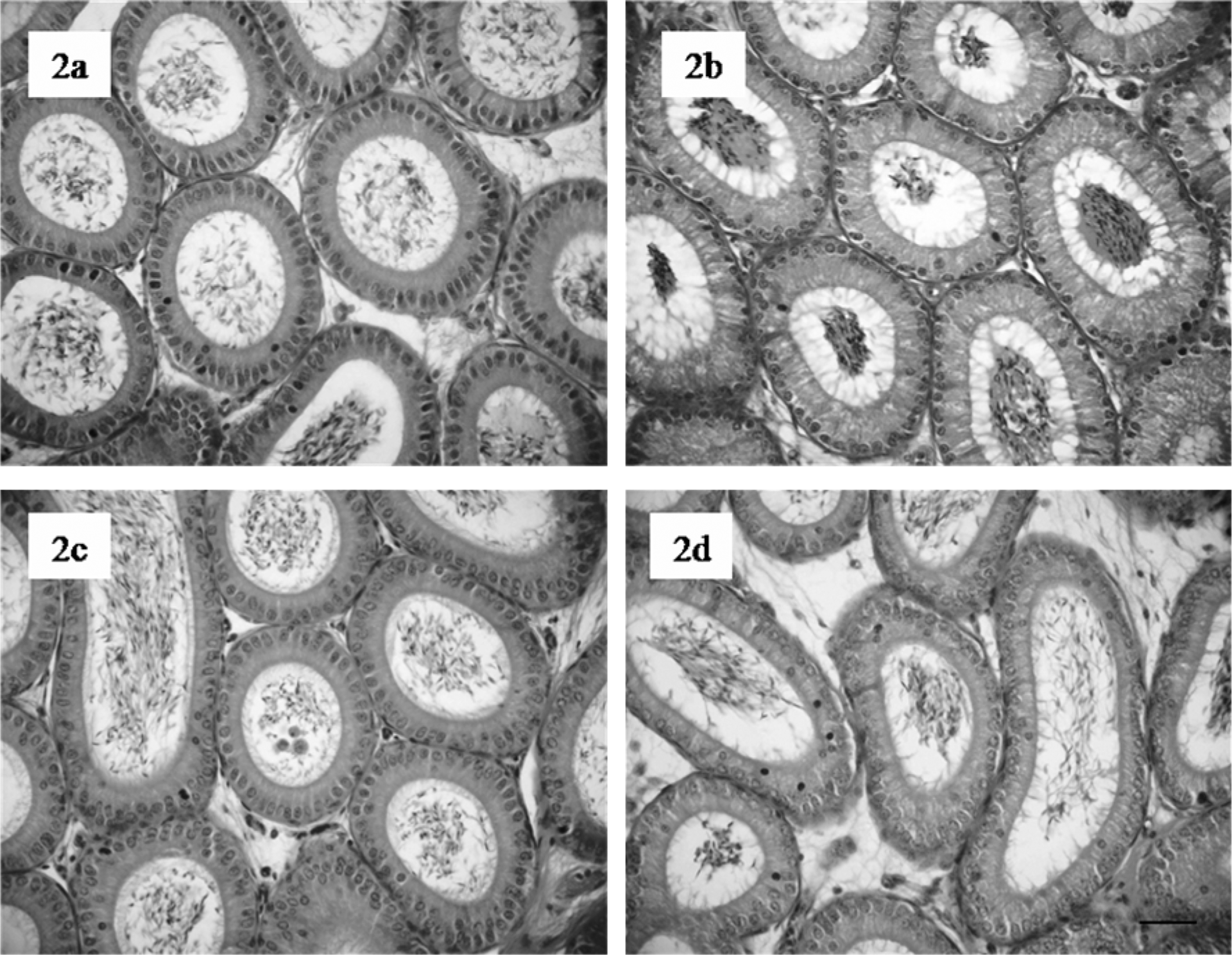
Histological sections of mice epididymis head following 7.5 mg sodium arsenite (NaAsO2)/kg bw treatment (groups 3 and 4). Representative section from control epididymis (a) and (b) shows normal histological organization. No significant alterations are noted in mice epididymis 35 days (c) and 70 days after treatment (d). Haematoxylin and eosin (HE), scale bar = 100 μm.
Functional parameters of sperm cells
Motility
The evaluation of sperm cells motility in animals exposed to 7.5 mg NaAsO2/kg bw during 35 days (group 3) revealed significant decrease (
Effects of NaAsO2 on sperm functional parameters, 35 days and 70 days after start of the exposure to 7.5 mg NaAsO2/kg bw

bw: body weight, NaAsO2: sodium arsenite.
a
b
c
Morphology
Evaluation of sperm cells morphology revealed a significant reduction (

Morphological classification of epididymal sperm cells from mice. (a) Normal sperm cell, (b) abnormal mid-piece (→), (c) coiled tail (→), (d) abnormal head (→), (e) abnormal hook (→), and (f) multiple abnormalities (head [] and mid-piece [→]). Haematoxylin and eosin (HE), scale bar = 10 μm.
Density, viability and acrosome integrity
No significant differences were revealed in sperm cells density for all NaAsO2 treatments respective to the control animals (Table 2).
Exposure to 7.5 mg NaAsO2/kg bw during 35 days (group 3) resulted in a significant (
Treatment with 7.5 mg NaAsO2/kg bw followed by exposure to distilled water during 70 days (group 4) affected acrosome integrity of sperm cells, with a significant decrease (
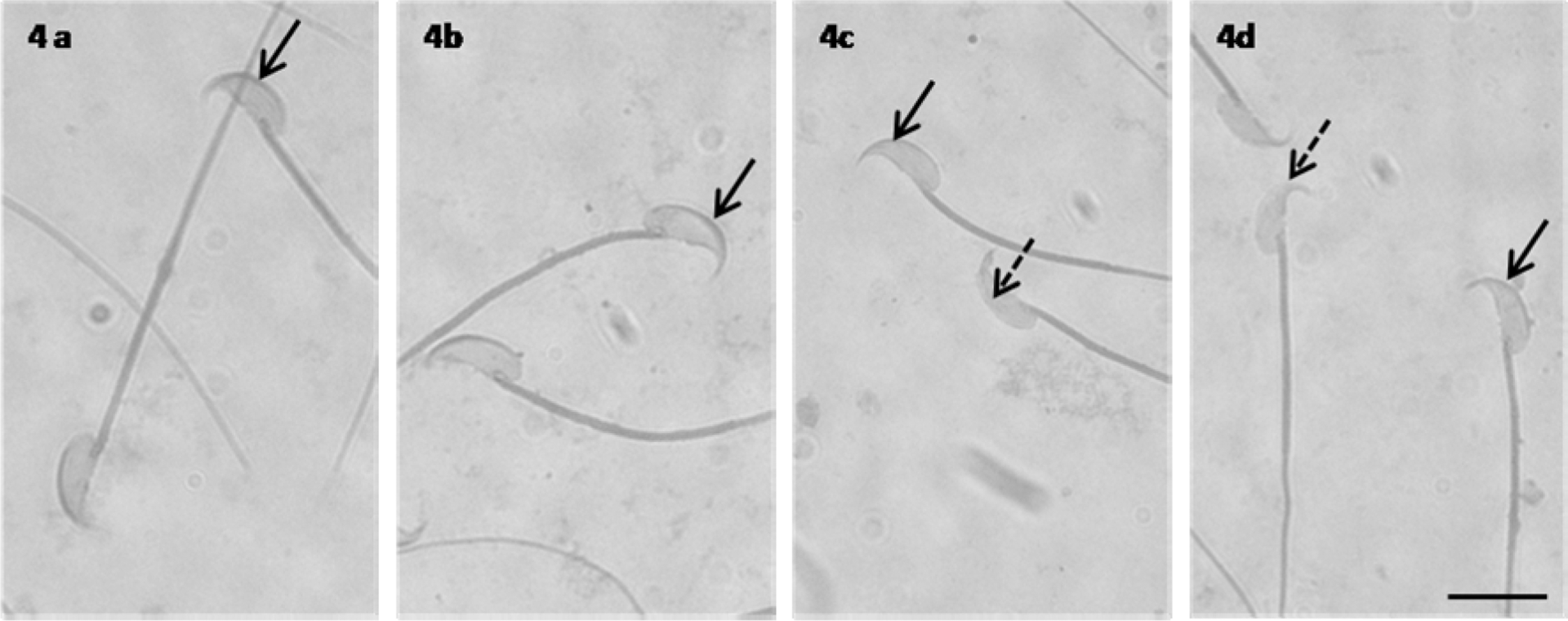
Mice sperm stained with Coomassie blue. (A and B) Control sperm (groups 1 and 2) showing intact acrosome intensely stained (arrow). (C and D) Sperm from 7.5 mg sodium arsenite (NaAsO2)/kg bw treatment (groups 3 and 4) showing reacted acrosome (dashed arrow), scale bar = 10 μm.
Enzymatic assays
The assessment of the toxic effects on several enzymatic key systems was performed through the quantification of the GSTs activity. However, no changes were observed after the exposure of test organisms to arsenic. Animals from groups 3 and 4 were not statistically different from control animals (Figure 5
). On the contrary, animals from group 3 had significantly lower selenium-dependent GPx when compared to control (group 1; d.f. = 5,

Glutathione-S-transferase (GST) activity in testicular homogenate following 7.5 mg sodium arsenite (NaAsO2)/kg bw treatment. No differences were observed after 35 days (group 3) and 70 days (group 4) of treatment.

Se-dependent glutathione peroxidase (Se-GPx) activity in testicular homogenate following 7.5 mg sodium arsenite (NaAsO2)/kg bw treatment. A significant decrease in Se-GPx activity was observed after 35 days (group 3) of treatment *
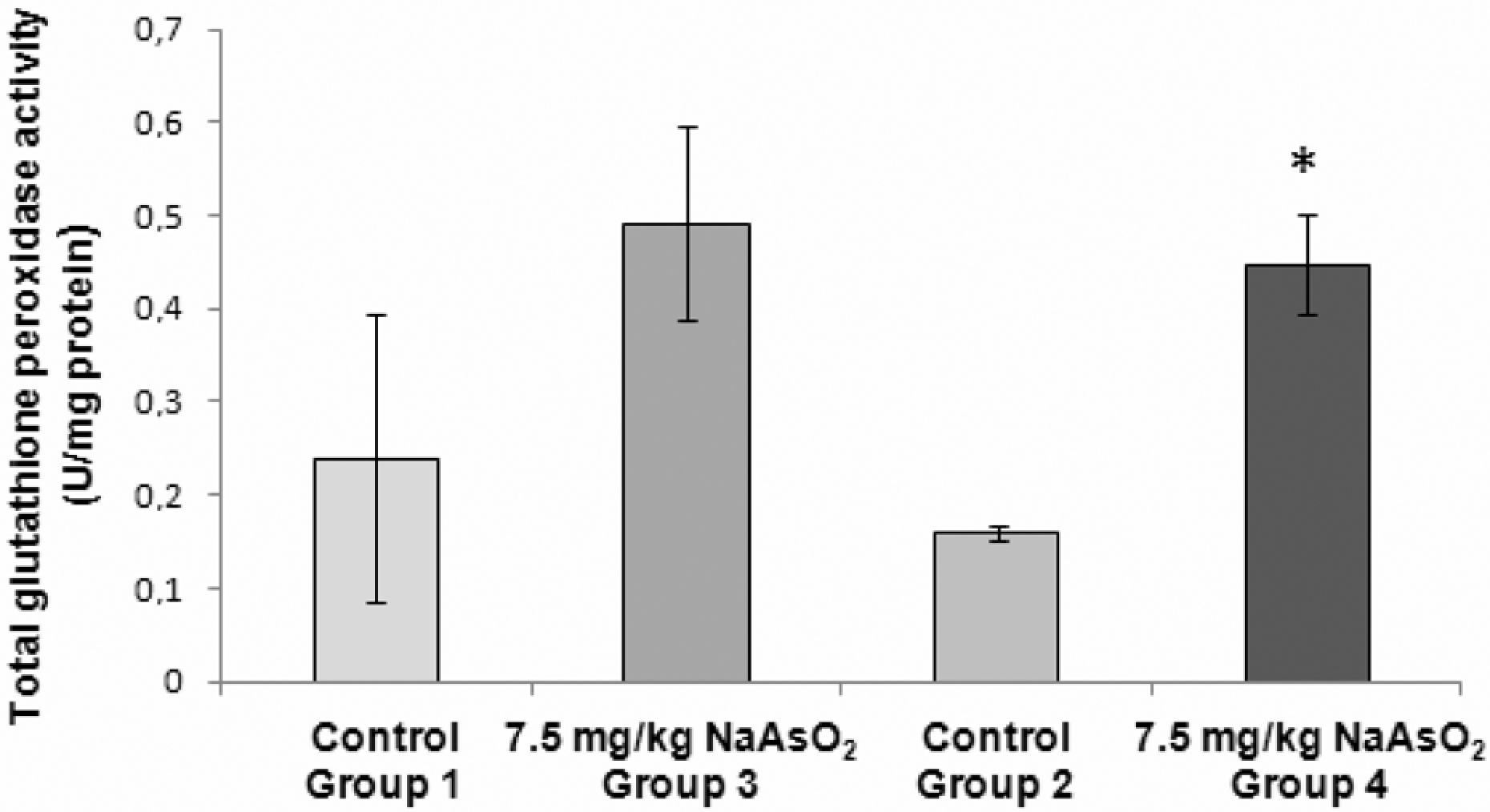
Total glutathione peroxidase (GPx) activity in testicular homogenate following 7.5 mg sodium arsenite (NaAsO2)/kg bw treatment. A significant rise in GPx activity was observed after 70 days (group 4) of treatment *
Discussion
The present study confirms the cytotoxic effects of arsenic compounds on spermatogenesis. Previous studies suggested that arsenic had a suppressive effect on reproductive functions by affecting the testicular weight, the seminiferous tubules diameter and the sperm quality. 16,17 The results described in this manuscript are in agreement with this author since animals treated with NaAsO2 during one spermatogenic cycle (group 3) revealed a significant decrease in testis/bw ratio (%) and seminiferous tubules diameter. According to Russell et al., 34 there is a strong correlation between testis histopathology and seminiferous tubules weight decrease accompanied by the decrease of spermatogenic activity in testis. Previous studies related the effect of NaAsO2 in mice with a reduction of sexual hormones, namely testosterone, luteinizing hormone (LH) and follicle-stimulating hormone (FSH), that consequently promotes spermatogenesis suppression with loss of testis weight and inhibition of testicular enzymes. FSH inhibits the normal degeneration of germ cells, and the decrease of this hormone can cause the shrinkage of seminiferous tubules. 35
In the present study, NaAsO2 exposure during one spermatogenic cycle affected the seminiferous epithelium with sloughing of immature germ cells into the tubular lumen. The occurrence of immature germ cells in tubular lumen is possibly related with disruption of germinal epithelium carried out by intercellular bond cleavages and decrease of fluid secretion by Sertoli cells. 34 Moreover, reduction of testosterone might lead to the degeneration of germinal epithelium necessary for the maintenance of seminiferous tubules morphology, since testosterone is crucial for the continuous production of different populations of germ cells in seminiferous tubules. Sloughing of germ cells from seminiferous epithelium might also occur with the reduction of testosterone levels. 36
Results described in this study point to the absence of spermatogenesis regeneration in animals subjected to NaAsO2 during one spermatogenic cycle followed by the administration of distilled water in the subsequent spermatogenic cycle (group 4). In fact, remarkable alterations on seminiferous epithelium were also observed, with disruption of seminiferous tubules, sloughing of immature cells and the presence of intraepithelial vacuoles. Nevertheless, studies report the possibility of partial spermatogenesis recovery after arsenic exposure; this recovery is only described in the particular case where spermatocytes have been the first cells affected by NaAsO2, accompanied by the survival of Sertoli cells. 34
Functional parameters of spermatozoa were also studied in the present work. A trend of lower spermatozoa count concerning each treatment (groups 3 and 4) was observed, which is in agreement with other reports where a reduction of spermatozoa density as a consequence of arsenic insult was demonstrated. 16,17 Furthermore, motility results obtained after the current experiment showed a significant decrease in the percentage of sperm cells with rapid progressive motility for animals exposed to NaAsO2 during 35 days (group 3). This finding is possibly associated with one of the toxicity mechanisms elicited by arsenic, since this metalloid reacts with thiol groups in vivo. The chromatin and the flagellum of mammal’s spermatozoa being enriched in thiol protamines and sulphydril groups, respectively, are prone as toxicological targets for arsenic. 16,17 Consequently, the decrease of motility and vitality of spermatozoa might be due to high arsenic concentration accumulated in epididymis ducts lumen, 37 where sperm cells proceed into maturation process and acquire motility. 16,17
With respect to the estimation of sperm cells morphology, the present work revealed increasing percentages of abnormal spermatozoa for all group of animals exposed to NaAsO2 (groups 3 and 4). These results suggested that following NaAsO2 exposure, a reduction of FSH hormone released by Sertoli cells may occur, which might affect normal spermatogenesis. 38 Concerning the assessment of acrosome integrity, mice treated with NaAsO2 during 35 days followed by other 35 days with distilled water (group 4) revealed a major decrease in the percentage of spermatozoa enclosed by an intact acrosome. This fact might be related to the adverse effects of arsenic observed in Sertoli cells and germ cells, which have consequences in morphogenesis of acrosome. 38 Also, NaAsO2 might alter the association of a range of proteins to spermatozoa membranes, which is crucial for sperm integrity and function. 39
Oxidative stress was also assayed in this work. Studies corroborate that arsenic is a compound that exerts oxidative stress, a scenario well documented in plants.
40
–42
Furthermore, other studies show the involvement of this compound in animal toxicity, with the occurrence of damage to intracellular components of such proteins,
36,43
contributing to apoptosis in testicular cells.
44
Animals are prone to toxicity of arsenic compounds with the involvement of an oxidative mechanism, as suggested by others,
45
when assessed the effects of this metalloid on rodents. The involvement of oxidative stress responses in rodents was also shown by others authors,
46
whose work revealed the complex mechanisms underlying arsenic intoxication. Besides oxidative stress, arsenic poisoning significantly altered the biochemical homeostasis of the rat brain, with sphingolipidosis, depleted protein content and impairment of the antioxidant content. Fish species usually face a similar outcome, as described by others,
47
which referred the possible involvement of peroxisomes in the oxidation-based response after chemical insult by arsenic in the fish
Our work showed that the activity of the total GPx content, which may be related to an oxidative alteration, was significantly increased after exposure to NaAsO2 during one spermatogenic cycle, followed by distilled water for 35 days. This means that the occurrence of the oxidative insult is more likely after a significant period of time, since it was only evident when an interval of recovery was allowed. A nonsignificant increase in total GPx was also observed for animals sacrificed immediately after the exposure to arsenite; albeit nonstatistically significant, this finding may be indicative that the occurrence of an oxidative damage is probable. An additional finding sustains this point of view. Selenium-dependent GPx activity was significantly decreased in organisms sacrificed after an exposure to arsenite during 35 days. These findings are of particular significance, because they establish a linkage between the administration of arsenic in a plausible route for most humans, via drinking water, and the occurrence of chronic long-lasting effects, that are even more physiologically significant following a recovery period.
The metabolism of arsenic was also evaluated in the present work. GSTs are enzymes that, in humans, are involved in the detoxification of arsenic compounds. 51 The genes responsible for the expression of glutathione-S-transferase omega 1 (GSTO1), and omega 2 (GSTO2) are involved in the metabolism of arsenic compounds through the final reduction of intermediate metabolites produced after sequential reduction and oxidative methylation of the ingested arsenic compounds. 52 The importance of enzymatic conjugation of arsenic compounds with GSH is not exclusive to humans. The over expression of a form of GST (GST II) was suggested to be the main biochemical mechanism underlying the acquired resistance of several rat cell lines to the chronic exposure to low levels of arsenic compounds, as demonstrated by others. 53 However, our results did not show any sensitive modification in these isoenzyme activities, suggesting that the presence of arsenic in drinking water that was consumed by test organisms was not conjugated with glutathione, with consequent increase in the enzyme’s activity.
The present study is indicative of the deleterious effects of NaAsO2 on male reproductive physiology. However, for a better evaluation of long-term effects on the reproductive system, longer periods of exposure and lower doses may be assayed. Additionally, the inclusion of a broader battery of oxidative stress biomarkers must be considered. Oxidative alterations were unequivocally observed, but the extension of damage, as well as occurrence of recovery and remission of damage, is an important issue to be assessed.
Footnotes
The present work was funded by CICECO and by the Project “BiOtoMetal” (PTDC/AMB/70431/2006) from Fundação para a Ciência e a Tecnologia (Portuguese Government).
