Abstract
The repairing effect and potential mechanism of miR-137 on cerebral ischemic injury in rats was investigated. The volume of cerebral infarction and calculated brain water content was detected by triphenyltetrazolium chloride staining. The expression of inflammatory factors was detected by enzyme-linked immunosorbent assay. The pathological damage of brain tissue was analyzed by hematoxylin and eosin and Nissl staining. The apoptosis in ischemic brain tissue was detected by terminal deoxynucleotidyl transferase-mediated deoxyuridine triphosphate nick-end labeling. The levels of STAT1 and JAK1 proteins were analyzed by Western blot. The expression of miR-137 in primary hippocampal neurons was detected by reverse transcription polymerase chain reaction. miR-137 overexpression significantly improved brain damage in rats. miR-137 overexpression can reduce the expression of TNF-α, IL-1β, and IL-6. miR-137 overexpression can reduce the degree of brain tissue damage and inhibit the expression of JAK1 and STAT1 proteins. miR-137 overexpression can reduce oxygen-glucose deprivation (OGD)/R-induced cell damage, improve cell proliferation, and reduce apoptotic rate. JAK1 and STAT1 protein expression was inhibited in hippocampal neurons after OGD/R treatment after transfection with miR-137 mimic. After the addition of the Filgotinib inhibitor, the levels of JAK1 and STAT1 proteins were significantly reduced. The results suggested that miR-137 overexpression can effectively improve ischemic injury after focal cerebral ischemia and protect against by inhibiting JAK1/STAT1 pathway.
Introduction
Cerebrovascular disease is a common and frequently occurring disease that causes death and disability. 1,2 Cerebral infarction, also known as ischemic stroke, is a general term for brain tissue necrosis caused by transient or permanent occlusion of the cerebral blood vessels and insufficient blood supply to the brain. 3,4 Clinical data suggest that earlier the treatment of patients with ischemic stroke is, the greater the chance of being rescued. 5 Moreover, current opinion supports the necessity for combination thrombolytic therapy with neuroprotective therapy.
Currently, more than 1600 human miRNAs have been identified and proved to be involved in a variety of pathophysiological processes, such as tumor metastasis, cell differentiation, inflammation, and embryonic development. 6 Studies have shown that miRNAs could control diverse aspects of brain disease. 7,8 miR-137, one important type of miRNA, can promote endothelial progenitor cell proliferation and angiogenesis in cerebral ischemic stroke mice by targeting NR4A2 through the Notch pathway. 9
The Janus kinase-signal transducer and transcriptional activator (JAK-STAT) signaling pathway is a newly discovered intracellular signal transduction pathway that acts as an interferon in cells. It has been clarified that it is one of the important pathways for cytokine signal transduction, not only involved in inflammatory response but also in oxidative stress, cell damage, and apoptosis. 10,11 Recently, the JAK-STAT pathway has been reported that this pathway is involved not only in the physiological processes of nerve cells but also in the pathological processes of neurological diseases, especially in the neurological processes, such as cerebral ischemia injury and central nervous system tumors. 12
In ischemic environment, whether miR-137 activates JAK-STAT signaling pathway in brain tissue remains to be further studied. Therefore, in this study, middle cerebral artery occlusion (MCAO) rat model is used to explore the effect of miR-137 on cerebral ischemic injury in rats and the changes of JAK-STAT signaling pathway.
Materials and methods
Cell culture and oxygen-glucose deprivation
Rat hippocampal neurons were isolated from neonatal rats (17–19 days) of Jinan Pengyue Experimental Animal Breeding Co., Ltd (China). Under sterile conditions, the rats were dissected, hippocampus tissue was taken, grounded, and trypsin was added for digestion. Hippocampus cells were suspended in neurobasal medium (Invitrogen, Carlsbad, California, USA) containing 2% B27 and 1% streptomycin/penicillin in a poly-
Cell groups
The experiment was divided into six groups: normoxia group (cultured under normal oxygen conditions, without OGD treatment), blank group (without any transfection after OGD treatment), miR-137 mimic group (transfected with miR-137 mimic after OGD treatment), miR-137 mimic negative control group (mimic-NC, miR-137 mimic negative control after OGD treatment), miR-137 inhibitor group (transfected with miR-137 inhibitor after OGD treatment), and miR-137 inhibitor negative control group (inhibitor-NC, transfection of miR-137 inhibitor negative control after OGD treatment). Cells at the logarithmic phase were inoculated at six-well culture plates, and cells were transfected according to lipofectamin 2000 instructions (Invitrogen, Carlsbad, California, USA) when the cells confluence reached to 30–50%. The final concentration of miR137 mimic or inhibitor was 50 nM.
To further validate the mechanism of action of miR-137, the JAK1 inhibitor Filgotinib (GLPG0634, Selleck.cn, Shanghai, China) had added, and set up five groups: miR-137 mimic group, miR-137 inhibitor group, Filgotinib group, miR-137 mimic + Filgotinib group, and miR-137 inhibitor + Filgotinib group.
Reverse transcription polymerase chain reaction
Total RNA was extracted from cells using Trizol reagent (Invitrogen, Thermo Fisher Scientific, Inc.) according to the relevant kit instructions. The expression level of miR-137 was detected by reverse transcription polymerase chain reaction (RT-PCR). Quantitative RT-PCR was performed using a 7500 Real Time PCR system (Applied Biosystems, Waltham, Massachusetts, USA). The conditions were 94°C for 3 min, 94°C for 15 s, 62°C, and 40 s (40 cycles). The data were processed by the
MTT assay
The hippocampal neurons in logarithmic growth phase were digested, centrifuged (1000 r min−1, 5 min), and the cell concentration was adjusted to 104 mL−1. The cells were inoculated into 96 well plates with 100 μL well−1. After cell adherence, different carriers were added for cell transfection. Six parallel wells were set at each concentration and cultured in a saturated humidity incubator of 37°C and 5% CO2. After 24 h, 20 μL 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT; 5 mg mL−1) was added and cultured in 37°C incubator. After 4 h the supernatant was discarded carefully, and 200 μL DMSO (Sigma–Aldrich, St Louis, Missouri, USA) was added to blow evenly. The optical density (OD) values of each pore were checked at 490 nm by Bio-Rad (Hercules, California, USA). Cell proliferation rate = (ODTest group − ODBlank group)/(ODControl group − ODBlank group) × 100%.
Flow cytometry
After trypsin digestion, the cells were collected. Then, 1 × 106 cells were washed twice with cold 1× phosphate-buffered saline (PBS), centrifuged (1000 r min−1, 5 min), the supernatant was discarded, and the cells were suspended in 200 μL 1× binding buffer. After that, 5 μL Annexin V-FITC and 5 μL PI (SAB, USA) were added. After incubation for 15 min at room temperature, 300 μL of 1× binding buffer was added to flow cytometry (CyFlow Cube, CyFlow Ploidy Analyser, Partec, Germany).
Western blot
Total protein in brain tissue was extracted using a protein extraction kit (Applygen Technologies, Beijing, China). Protein samples were separated by 12% sodium dodecyl sulfate–polyacrylamide gel electrophoresis with a 25 μg loading per lane. Electrophoresis was carried out at a voltage of 80 V, and the protein sample was transferred to a polyvinylidene fluoride membrane (Amersham, Little Chalfont, UK) using an electrotransfer (Bio-Rad, USA), and the 5% skim milk powder solution was blocked for 1 h, and the primary antibody JAK1 (1:2000, ab133666, Abcam, Shanghai, China), STAT1 (1:500, ab31369, Abcam), Bcl-2 (1:1000, ab59348, Abcam), Bax (1:1000, ab53154, Abcam), and β-actin antibody (1:5000, ab6267, Abcam) were added. It was incubated overnight at 4°C. The Tris-buffered saline with Tween 20 (TBST) was rinsed three times, and the secondary anti-goat anti-rabbit immunoglobulin G–horseradish peroxidase (HRP; 1:5000, ab97080, Abcam) dilution was added for 90 min, washed by the TBST for three times, and the enhanced chemiluminescence was developed and photographed. Western blot data were quantified using Image-Pro Plus 6.0 software, and protein expression was normalized using β-actin as a loading control.
Animals
A total of 120 SPF male SD rats, weighing 260–300 g, were purchased from Jinan Pengyue Experimental Animal Breeding Co., Ltd, China, under license number SCXK (Lu) 20140007. Rats were adaptively reared for 5 days. All animal experiments were conducted in accordance with the guidelines of the institutional ethics committee and approved by the Affiliated Huaian No. 1 People’s Hospital of Nanjing Medical University. The experiment was conducted in accordance with the National Laboratory for Laboratory Animal Care Guidelines.
Model and groups
The MCAO model was prepared by the modified Longa method. 13 The rats were weighed and intraperitoneally injected with 3% pentobarbital (45 mg/kg) for anesthesia. The left internal carotid artery was clamped by nylon thread suture. The median incision of the neck was 3–4 cm, and then the subcutaneous tissue was separated to expose the left common carotid artery. The proximal and distal end of the left common carotid artery was ligated. The oblique artery was cut with the ophthalmic scissors on the left common carotid artery. The bolt was inserted until resistance occurred. The bolt entered nearly 18–20 mm. The subcutaneous tissue and skin were sutured, and the MCAO model was established. Focal cerebral ischemia was MCAO for 2 h, followed by reperfusion 24 h (MCAO/R).
Experimental groups were divided into sham group, model group, miR-137 agomir group (agomir), agomir negative control group (agomir-NC), miR-137 antagomir group (antagomir), and antagomir negative control group (antagomir-NC). miR-137 agomir, agomir-NC, miR-137 antagomir, and antagomir-NC were obtained from Genepharma (Shanghai, China), and the injection was made with a electronic design limited (CED) microinjection apparatus (Harvard) at 0.5 µL/min (12 mg/kg). 14 The administration was performed at 30 min after ischemia. In the sham group, rats were anesthetized and exposed to the left common carotid artery, but bolt not inserted. The room temperature was controlled at 23–25°C, and the temperature of rectum was maintained at (37 ± 0.5°C) with electric blanket. Routine feeding was performed before and after surgery.
Neurological deficit score
A blind assessment was performed by two observers, and neurological deficits in MCAO rats were scored using a five-level classification by Bederson et al, as presented in Table 1.
Bederson five-point scale.

Triphenyltetrazolium chloride
After the anesthetized rats were intraperitoneally injected with 3% sodium pentobarbital (45 mg/kg), brain tissue was taken and frozen in a refrigerator at −20°C for 30 min. Coronal sections (5–6 pieces, 2 mm) were sliced and placed in a six-well plate. Then, 2% triphenyltetrazolium chloride (G3005, Solarbio® Life Sciences, Beijing, China) was added, and a circular cover slip was covered for uniform staining. After staining, it was taken out in 4% paraformaldehyde for 24 h, and then photographed by a digital camera. The infarct volume was analyzed by computer image analysis software Image J 1.43 (National Institutes of Health, Bethesda, Maryland, USA).
Brain tissue water content
On the first day after MCAO surgery, the ipsilateral and contralateral hemisphere cerebellum were taken. The brainstem was removed, and the wet weight (WW) was immediately weighed, then dried in a gravity oven at 100°C for 24 h to obtain a dry weight (DW). Water content = (WW − DW)/WW × 100%.
Enzyme-linked immunosorbent assay
After 24 h of ischemia–reperfusion, the rats were anesthetized with 3% sodium pentobarbital (45 mg/kg) and the brain was decapitated. The whole brain was weighed, rinsed with ice-cold saline, and placed in a homogenized tube. Then, 10% tissue homogenate was prepared by adding an appropriate amount of physiological saline at 4°C. The sample was centrifuged at 3000 r/min for 15 min and the supernatant was taken. The expression of inflammatory factors IL-1β (ab100768, Abcam), IL-6 (ab100772, Abcam), and TNF-α (ab100785, Abcam) was detected by enzyme-linked immunosorbent assay. It was operated in strict accordance with the corresponding kit instructions during the experiment.
Hematoxylin and eosin and Nissl staining
After the anesthetized rats were intraperitoneally injected with 3% sodium pentobarbital (45 mg/kg) and perfused with 0.9% sodium chloride, the brain tissue was fixed with 4% formaldehyde for 48 h and immersed in 20% sucrose solution to observe the sucrose solution. When the brain tissue sinked to the bottom, it can be taken out and then embedded and frozen. For Nissl staining, the frozen sections were placed in an incubator at 37°C overnight, rehydrated, subjected to Nissl staining, dehydrated with gradient ethanol, and sealed with a transparent, neutral gum. Hematoxylin and eosin (HE) staining was done according to the literature method. 15 The brain tissue sections were observed under an optical microscope (Olympus, Tokyo, Japan).
TUNEL assay
The cellular apoptosis in brain tissue was analyzed using terminal deoxynucleotidyl transferase-mediated deoxyuridine triphosphate nick-end labeling (TUNEL) Apoptosis Detection Kit (C1098, Beyotime). The paraffin section was dewaxed and hydrated, and 20 μg/mL of DNase-free proteinase K was added dropwise, and the mixture was allowed to act at 37°C for 20 min, and washed three times with PBS. It was incubated with 3% hydrogen peroxide solution for 20 min and washed three times with PBS. Then, 50 μL of TUNEL working solution was added to each well and incubated at 37°C for 60 min in the dark. Then the sample was washed with PBS, 0.3 mL of labeled reaction stop solution was added, incubated for 10 min at room temperature, and washed three times with PBS. Then, 50 μL of streptavidin-HRP working solution was added and incubated for 15 min at room temperature. Then, 0.5 mL of DAB color solution was added dropwise and incubated for 30 min. After hematoxylin counterstaining, ethanol gradient dehydration, xylene transparent, and neutral resin sealing, it was observed under an optical microscope.
Data analysis
Data were analyzed by SPSS19.0 statistical software. The results were expressed as mean ± standard deviation (mean ± SD). Data analysis among multiple groups was analyzed by one-way analysis of variance. The least significant difference test was used in subsequent analysis. The p < 0.05 meant the difference was statistically significant.
Result
miR-137 expression reduced in hippocampal neurons after OGD treatment
As can be seen from Figure 1(a), after OGD treatment, the miR-137 content in hippocampal neurons was decreased in a time-dependent manner. In Figure 1(b), after transfection of miR-137 mimic, the expression of miR-137 was increased in cells, and the expression of miR-137 was decreased in cells after transfection with miR-13 inhibitor.

RT-qPCR detected the expression of miR-137 in hippocampal neurons after OGD/R treatment. (a) Expression levels of miR-137 after OGD treatment for different times. *p < 0.05: compared with normoxia; @ p < 0.05: compared with 12 h; $ p < 0.05: compared with 24 h. (b) RT-qPCR analysis of miR-137 expression after hippocampal neuronal cells treated with OGD for 24 h and transfected with miR-137 mimics or miR-137 inhibitor. *p < 0.05: compared with the normoxia group; # p < 0.05: compared with blank group. OGD: oxygen-glucose deprivation; RT-qPCR: reverse transcription quantitative polymerase chain reaction.
Overexpression of miR-137 reduced OGD/R-induced cell damage
In Figure 2, compared with the normoxia group, the apoptotic rate was significantly increased in the blank group, and the proliferation rate was significantly decreased in the blank group (p < 0.05). The apoptotic rate was significantly decreased in the brain tissue of the miR-137 overexpression group compared with the blank group (p < 0.05). The proliferation rate was significantly increased in the brain tissue of the miR-137 overexpression group compared with the blank group. Compared with the blank group, the apoptotic rate was significantly increased in the brain tissue of the miR-137 inhibitor group, and the proliferation rate was significantly decreased in the brain tissue of the miR-137 inhibitor group (p < 0.05). These results indicated that overexpression of miR-137 increased cell proliferation and decreased apoptotic rate, thereby reducing OGD/R-induced cell damage.

Effect of miR-137 on OGD-induced cell damage. (a) and (b) Flow cytometry to detect apoptosis of hippocampal neurons after hippocampal neuronal cells transfected with miR-137 mimic or miR-137 inhibitor after OGD/R treatment. (c) MTT assay for hippocampal neuronal cell proliferation after hippocampal neuronal cells transfected with miR-137 mimic or miR-137 inhibitor after OGD/R treatment. *p < 0.05: compared with the normoxia group; # p < 0.05: compared with the blank group. OGD: oxygen-glucose deprivation.
miR-137 mimic inhibited the expression of JAK1 and STAT1 in hippocampal neurons after OGD/R treatment
We have studied the effects of miR-137 mimic on the expression of JAK1 and STAT1 after OGD/R treatment (Figure 3). After transfection of miR-137 mimic, the expression of phosphorylated JAK1, and phosphorylated STAT1 protein in hippocampal neurons was inhibited after OGD/R treatment. The expression levels of JAK1 and STAT1 proteins were significantly reduced after the addition of the Filgotinib inhibitor (p < 0.05). After both of them acted on hippocampal neurons, the expression of JAK1 and STAT1 protein was significantly lower than that of miR-137 mimic and Filgotinib (p < 0.05). In addition, miR-137 overexpression inhibited the activation of JAK1/STAT1 pathway, upregulated the content of Bcl-2, downregulated the expression of Bax, thereby reducing the volume of cerebral infarction and protecting against cerebral ischemia–reperfusion injury.

Western blot analysis of the mechanism of miR-137 on hippocampal neuronal injury. (a) and (b) Western blot analysis of JAK1, STAT1, Bcl-2, and Bax protein expression, in hippocampal neurons transfected with miR-137 mimic or miR-137 inhibitor after OGD/R treatment. *p < 0.05: compared with the normoxia group; # p < 0.05: compared with the blank group. (c) and (d) Western blot analysis of the effect of JAK1 inhibitor Filgotinib on the expression of JAK1, STAT1, Bcl-2, and Bax protein after hippocampal neuronal cells transfected with miR-137 mimic or miR-137 inhibitor after OGD/R treatment. ^p < 0.05: compared with miR-137 mimic; & p < 0.05: compared with miR-137 inhibitor; △ p < 0.05: compared with the Filgotinib group. OGD: oxygen-glucose deprivation.
Overexpression of miR-137 significantly ameliorated brain damage
It can be seen from Figure 4 that the MCAO rats had a significantly larger cerebral infarct size and a higher neurological function score. Compared with sham group, the cerebral infarct size was significantly greater and the neurological function score was significantly higher in the model group (p < 0.05). However, compared with the model group, the cerebral infarct size and neurological function score of the miR-137 overexpression group were significantly lower. Then compared with the model group, the cerebral infarct size and neurological function score of the antagomir group were significantly higher (p < 0.05). These results above indicated that miR-137 overexpression reduced the infarct size of MCAO rats, decreased the neurological function score, and reduced the water content of brain tissue.

miR-137 overexpression significantly improved brain damage. (a) Triphenyltetrazolium chloride staining and cerebral infarct size, (b) neurological function score, and (c) brain water content. *p < 0.05: compared with the sham group; # p < 0.05: compared with the model group.
Overexpression of miR-137 can decrease the expression of inflammatory factors TNF-α, IL-1β, and IL-6 in brain tissue
In Figure 5, compared with the sham group, the levels of inflammatory factors TNF-α, IL-1β, and IL-6 in the brain tissue of the model group were significantly increased (p < 0.05). Compared with the model group, the levels of inflammatory factors TNF-α, IL-1β, and IL-6 were significantly lower in the brain tissue of the miR-137 overexpression group. And then, compared with the model group, the levels of inflammatory factors TNF-α, IL-1β, and IL-6 were significantly higher in the brain tissue of the antagomir group (p < 0.05). These results indicated that miR-137 overexpression can reduce the expression of inflammatory factors TNF-α, IL-1β, and IL-6 in brain tissue.

Enzyme-linked immunosorbent assay was used to detect the expression of inflammatory factors TNF-α, IL-1β, and IL-6 in brain tissue. *p < 0.05: compared with the sham group; # p < 0.05: compared with the model group.
Overexpression of miR-137 reduced the degree of brain damage
After 2 h of ischemia and reperfusion for 24 h, HE (Figure 6(a)) and Nissl (Figure 6(b)) staining were used to detect the degree of brain tissue damage. In Figure 6(a), the hippocampus and parietal cortex were observed under light microscope. The sham operation group had regular cell arrangement, abundant cytoplasm, round nucleus, and clear nucleoli. Pathological changes were obvious in the model group, with a large number of neurons necrosis and degeneration, disordered arrangement, neuron cytoplasm concentration, nucleus pyknosis, and unclear nucleolus. Compared with the model group, neuronal loss and injury in the miR-137 overexpression group were lighter, and some neurons showed degeneration and necrosis, neuronal loss and injury in the antagomir group were more serious compared with the model group. In Figure 6(b), rat neurons in the sham operation group are rich in Nissl bodies, with normal structure, clear and complete, and abundant cytoplasm. In the model group, normal neuronal structure was destroyed during ischemia–reperfusion, and the number of Nissl bodies was reduced. The number of Nissl bodies in the miR-137 overexpression group increased compared with the model group. Compared with the model group, the number of Nissl bodies in the antagomir group was significantly lower. These results indicated that miR-137 overexpression had protective effect on ischemia-reperfusion injury.

(a) HE staining. (b) Nissl staining. Magnification 400×.
Overexpression of miR-137 reduced apoptosis in brain tissue
As shown in Figure 7, in the model group, the nucleus was brownish yellow or brown under high magnification, and the cells not stained by cytoplasm were TUNEL-positive cells. A small number of apoptotic cells were observed in the sham operation group rats. Compared with the model group, the number of TUNEL-positive cells was significantly decreased in the miR-137 overexpression group. The number of TUNEL-positive cells was significantly higher in the antagomir group. These results indicated that the overexpression of microRNA-137 had protective effect on ischemia–reperfusion injury.
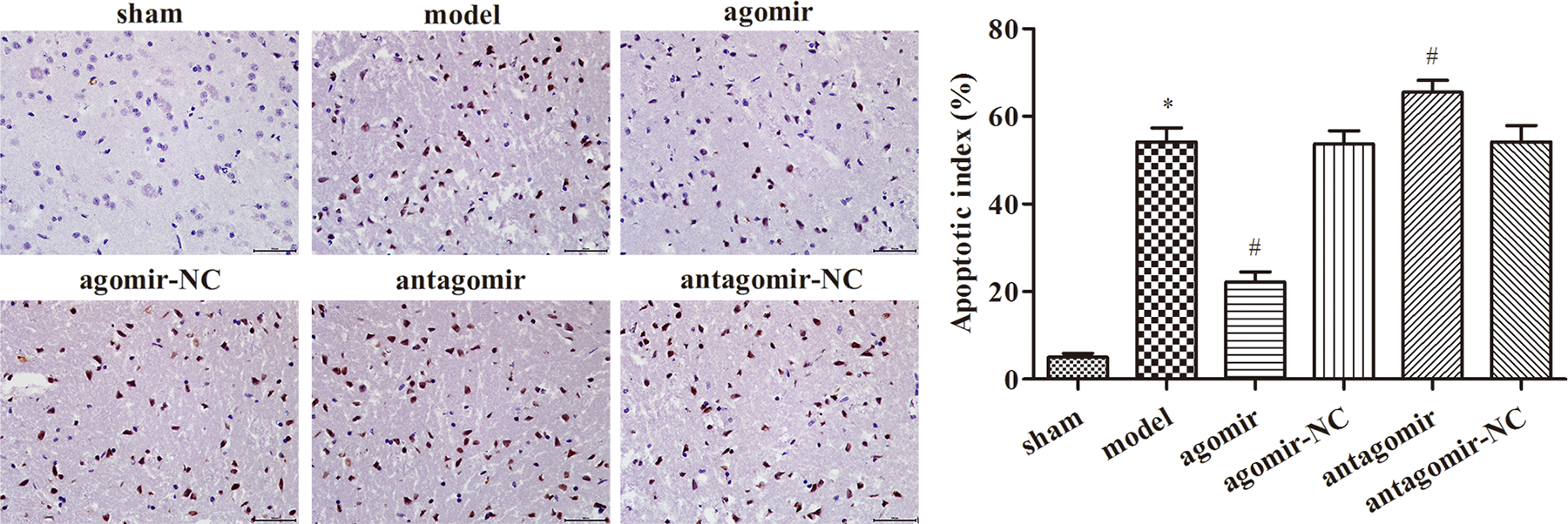
Apoptosis in brain tissue of rats in each group detected by TUNEL. Magnification 400×. *p < 0.05: compared with the sham group; # p < 0.05: compared with the model group. TUNEL: terminal deoxynucleotidyl transferase-mediated deoxyuridine triphosphate nick-end labeling.
Overexpression of miR-137 inhibited the expression of JAK1 and STAT1 proteins in brain tissue
As shown in Figure 8, the expression levels of JAK1 and phosphorylated STAT1 proteins were significantly increased in the model group compared with the sham group, while the overexpression of miR-137 significantly inhibited the expression of JAK1 and STAT1 proteins in brain tissue (p < 0.05).

Western blot analysis of JAK1 and STAT1 protein expression in brain tissue.
Discussion
In clinic, middle cerebral artery embolism is the main cause of stroke. Therefore, we use special nylon thread to embolize the middle cerebral artery of rats to imitate stroke, so we can establish a more similar clinical stroke model, namely MCAO. The model has little trauma, no need of craniotomy, strong repeatability and stability, which is close to clinical pathophysiological changes. It can achieve local reperfusion after removal of thread embolus. 16
Some studies have indicated that miRNAs play an important role in the growth, development, and physiological functions. 17 Recently, microRNAs were found that they were involved in the pathogenesis and development of ischemic stroke, including atherosclerosis, brain edema, ischemia–reperfusion injury, and angiogenesis after cerebral infarction. 18,19 It is known that there are many kinds of specific expression of microRNAs in brain tissue. In the rat model of MCAO, the level of microRNA-124 increased after 6 h of ischemia and lasted until 48 h. 20 As one of the types of miRNAs, miR-137 is involved in a variety of tumorigenesis and progression processes and is closely related to tumor cell proliferation, apoptosis, migration, invasion, and tumor angiogenesis. 21 The study found that miR-137 is lowly expressed in the serum of patients with acute ischemic stroke and can accelerate brain tissue damage by promoting calmodulin expression. 22 This study indicated that miR-137 overexpression significantly improved brain damage in rats and reduced cerebral infarct size in MCAO rats, as demonstrated by HE staining and Nissl staining. Our result showed that miR-137 overexpression reduced the infarct size of MCAO rats, decreased the neurological function score, and reduced the water content of brain tissue. miR-137 overexpression can decrease the number of apoptotic cells significantly with TUNEL.
In addition, a large number of cytokines, including TNF-α, IL-1β, IL-6, and so on, can be produced after cerebral ischemia–reperfusion injury. TNF-α, IL-1β, and IL-6 are all important inflammatory factors, which can produce many biological effects and activate inflammatory cells, such as leukocytes and monocytes. Stimulating the release of other inflammatory factors can overamplify the inflammatory response due to its biological amplification effect. 23 In our study, overexpression of miR-137 can reduce the expression of inflammatory factors TNF-α, IL-1β, and IL-6 in brain tissue, reduce inflammatory factor levels, reduce inflammatory cell invasion, and control inflammatory response, which may be miR-137 overexpression has another important aspect of brain protection.
After stroke, brain tissue produces excessive oxygen free radicals and releases a large number of inflammatory cytokines. Numerous studies have shown that these cytokines and oxidative stress processes utilize the JAK/STAI signal transduction pathway to induce cell differentiation, proliferation, or apoptosis. 24 Researchers found that AG490 (JAK kinase inhibitor) is able to intervene and regulate JAK-STAT signaling, thereby inhibiting GFAP (astrocyte-specific protein) expression and regulating the expression of activated astrocytes in ischemic injury. At present, the mechanism of this effect is not clear, probably through the induction of metallothionein in astrocytes. 25 In addition, another studies have found that JAK-STAT signaling pathway activated in central nervous system disorders not only participates in a variety of neuroinflammatory pathological processes but also regulates this inflammatory pathway, regulating JAK-STAT signaling pathways can alleviate neuronal damage caused by inflammation. 26 Our study found that overexpression of miR-137 inhibited the expression of JAK1 and STAT1 proteins in brain tissue. After transfection with miR-137 mimic, the expression of JAK1 and STAT1 protein was significantly lower than that of miR-137 mimic and Filgotinib on hippocampal neurons. In addition, miR-137 overexpression inhibited the activation of JAK1/STAT1 pathway, upregulated the content of Bcl-2, downregulated the expression of Bax, thereby reducing the volume of cerebral infarction and protecting against cerebral ischemia–reperfusion injury.
Conclusion
This study found that miR-137 overexpression can effectively improve ischemic injury after focal cerebral ischemia. It can reduce the infarct size of MCAO rats and the expression of inflammatory factors in brain tissue. Its mechanism may be through inhibiting JAK1/STAT1 signaling pathway to play a protective role.
