Abstract
JZL184 is a selective inhibitor of monoacylglycerol lipase (MAGL) that has neuroprotective effect. However, the role of JZL184 in cerebral ischemia/reperfusion (I/R) injury and the exact mechanism have not been fully understood. This study was designed to elucidate the role of JZL184 in cerebral I/R injury induced by oxygen-glucose deprivation/reoxygenation (OGD/R) in hippocampal neurons. Hippocampal neurons were pretreated with various concentrations of JZL184 for 2 h, followed by OGD for 3 h and reoxygen for 24 h. Our results showed that JZL184 improved cell viability in hippocampal neurons in response to OGD/R. JZL184 treatment significantly inhibited the production of reactive oxygen species (ROS) and malondialdehyde (MDA), as well as increased superoxide dismutase (SOD) and glutathione peroxidase (GPx) activities in OGD/R-induced hippocampal neurons. The increased TNF-α, IL-1β, and IL-6 productions in OGD/R-induced hippocampal neurons were decreased after treatment with JZL184. Moreover, the OGD/R-caused intense TUNEL staining in hippocampal neurons was attenuated by JZL184. JZL184 treatment prevented OGD/R-caused increases in bax and cleaved caspase-3 expression and a decrease in bcl-2 expression. Furthermore, JZL184 treatment significantly promoted the activation of Nrf2/ARE signaling pathway in OGD/R-induced hippocampal neurons. Additionally, silencing of Nrf2 reversed the protective effect of JZL184 on hippocampal neurons under OGD/R condition. Taken together, these findings suggested that JZL184 exerted protective effect against OGD/R-induced injury in hippocampal neurons via activating Nrf2/ARE signaling pathway, which provided in vitro experimental support for the therapeutic benefit of JZL184 in cerebral ischemia.
Introduction
Cerebral ischemia is a global health problem that accounts for over 80% of all strokes, which remains one of the leading causes of morbidity and mortality in the world. 1 Therefore, the development of novel therapeutic options is urgently needed to limit injury after cerebral ischemia. Considerable advancements in pathogenesis of cerebral ischemia have been made, which are helpful to explore new therapeutic strategies. 2,3 Among these mechanisms, there is evidence that the accumulation of reactive oxygen species (ROS) and ROS-mediated oxidative stress, inflammation and neuronal apoptosis are considered as important pathogenesis contributing to ischemia/reperfusion (I/R) injury, leading to neurological dysfunction. 4 –7 Hence, the blockage of ROS generation may be therapeutic approach for ischemic stroke.
Monoacylglycerol lipase (MAGL) is a main enzyme responsible for the inactivation of the most abundant brain endocannabinoid, 2-arachidonoylglycerol (2-AG). It has recently been shown that MAGL has become a promising therapeutic target for the treatment of several diseases such as cancers, neuroinflammation, and neurodegenerative disorders, and metabolic disorders. 8 Increasing evidences have demonstrated that blocking MAGL by MAGL inhibitors may produce diverse biochemical and physiological roles in combatting a variety of human diseases through bidirectionally manipulating endocannabinoid, eicosanoid, and other lipid signaling pathways. 9,10
JZL184 is a MAGL selective inhibitor that has been observed to exhibit anti-inflammatory, anti-cancer, anti-hyperalgesic and neuroprotective effects. 11 –14 For example, JZL184 improves behavior and neural properties in a down syndrome model, which is a development disorder with deficient cognition. 15 JZL184 reduces neuroinflammatory response and formation of beta-amyloid (Aβ) in the Alzheimer’s disease mouse model APdE9 and in adult mouse glial cells. 16 JZL184 is neuroprotective in the chronic MPTP-induced Parkinson’s mouse model involving restorative astroglia and microglia activation and the release of neuroprotective and anti-inflammatory molecules. 17 Considering the anti-neuroinflammatory and neuroprotective properties of JZL184, we speculated that JZL184 might have protective effect on cerebral I/R injury.
In the present study, we examined the effect of JZL184 on cerebral I/R injury in vitro using rat primary hippocampal neuronal cells. Furthermore, we investigated whether the protective effect is mediated by Nrf2/ARE signaling pathway. Our results proved that JZL184 exerted protective effect against I/R-induced oxidative stress, inflammation and apoptosis via regulating Nrf2/ARE signaling pathway.
Materials and methods
Preparation of primary hippocampal neuronal cells
Preparation of primary hippocampal neurons was performed as described by Latt et al. 18 Postnatal day 0-2 newborn Sprague-Dawley rats obtained from The First Affiliated Hospital of Xi’an Jiaotong University (Xi’an, China) were used for the isolation of hippocampal neurons. The rats were decapitated, and the cerebral hippocampi were separated from brains after removing meninges. The tissues were dissected in Histidine buffer solution (HBS) and then added to Neurobasal medium with 2% B27 supplement. After trypsinization in 0.25% Trypsin-EDTA at 37°C for 20 min, the fragments were subjected to mechanical dissociation using a Pasteur pipette and mild centrifugation at 2000 rpm for 3 min. Then the cells were maintained in Neurobasal medium with 2% B27 supplement, 0.1% Penicillin-Streptomycin, and 0.25% Glumax and kept in an incubator under 5% CO2 at 37°C. All experimental procedures were performed following the Laboratory Animal Care and Use Guidance Suggestions of The First Affiliated Hospital of Xi’an Jiaotong University.
Oxygen-glucose deprivation/reperfusion (OGD/R) model and treatment
OGD/R is an in vitro model that mimics in vivo I/R injury and initiates a series of devastating cascades that lead to the overproduction of ROS, mitochondrial dysfunction, oxidative stress and finally cell death through lipid degeneration, protein oxidation and DNA damage. An in vitro I/R injury model was established using the OGD/R model. In brief, primary hippocampal neurons were maintained in glucose-free medium and incubated in a hypoxic chamber (Billups-Rothenberg, MC-101, San Diego, CA, USA) containing 1% O2, 5% CO2, and 94% N2 for 3 h at 37°C. Then the medium was replaced by the normal complete culture medium and cultured in the normoxic incubator containing 5% CO2 and 95% air for 24 h at 37°C for reperfusion, to induce OGD/R injury. JZL184 was added in to the medium 2 h before OGD until the end of reoxygenation.
Lactate dehydrogenase (LDH) release assay
To assess the cytotoxicity effect of JZL184 on primary hippocampal neurons, the neurons were treated with 0, 5, 10, 20, or 40 μM of JZL184 for 24 h. After appropriate treatments, the culture supernatant was collected for the measurement of LDH using LDH Cytotoxicity Assay Kit (Biovision Research Products, Mountain View, CA, USA) according to the instructions. The absorbance at 450 nm wavelength was measured using the microplate reader (Tecan, Männedorf, Switzerland). The percentage of LDH release (%) was calculated relative to the untreated group.
Cell viability assay
Hippocampal neurons in logarithmic growth stage were collected and the cell density was adjusted to 2 × 104 cells/ml. The cells (100 µl cell suspension per well) were inoculated in 96-well plates and subjected to indicated treatments. Hippocampal neurons in control group were cultured in normoxic condition. Hippocampal neurons in OGD/R group were subjected to 3 h of OGD and 24 h of reperfusion. Hippocampal neurons in JZL184 treatment groups were pretreated with 5, 10, or 20 μM of JZL184 for 2 h, followed by OGD for 3 h and reoxygen for 24 h. For detecting the cell viability, 10 µl MTT solution (Beyotime Biotechnology, Shanghai, China) was added to each well and then incubated in the incubator for 1 h. At the termination of culturing, the medium in each well was discarded and 100 µl DMSO was added. Then the 96-well plate was read using the microplate reader (Tecan) to measure the absorbance at 490 nm wavelength.
Assessment of ROS level
Total intracellular ROS in hippocampal neurons was determined by staining cells with the stable nonpolar dye dichlorofluorescin diacetate (DCFH-DA). Cells with appropriate treatments were incubated with 10 μM DCFH-DA for 20 min at 37°C and gently mixed every 5 min. Then the neurons were analyzed at 485 and 500 nm excitation and emission wavelengths.
Analysis of biochemical parameters
The oxidative indicators including malondialdehyde (MDA), superoxide dismutase (SOD) and glutathione peroxidase (GPx) were detected by corresponding specific commercial kits (Beyotime) according to the manufacturer’s instruction. The secretion levels of TNF-α, IL-1β, and IL-6 in the cell culture medium were measured by ELISA using commercial kits (R&D Systems, Minneapolis, MN, USA).
Terminal deoxynucleotidyl transferase dUTP nick end labeling (TUNEL) assay
Apoptosis of hippocampal neurons was evaluated by TUNEL staining using a TUNEL detection kit (Millipore, Billerica, MA, USA). Briefly, cells seeded on the coverslips and incubated for 24 h for the attachment. After the staining of TUNEL, cells were washed with PBS and subjected to DAPI nucleus staining. Fluorescent intensity was detected by fluorescence microscopy (Olympus IX71, Olympus, Tokyo, Japan) and the quantitative analysis was performed using image analysis software.
Quantitative real time PCR
Total RNA extracted from hippocampal neurons by TRIzol reagent (Invitrogen, Carlsbad, CA, USA) was used for the reverse transcription according to the manufacturer’s protocol. The RNA was reverted to cDNA using the PrimescriptTM RT reagent kit (TaKaRa, Ohtsu, Japan), followed by the qPCR reaction using a SYBR Green PCR Master Mix (TaKaRa). The relative gene expression levels of TNF-α, IL-1β, IL-6 were calculated by 2−ΔΔct method. The β-actin was used as an internal reference. The PCR primers used in the study were: TNF-α forward primer: 5’-AGC ACA CAA GTG GCA CAA CG-3’, reverse primer: 5’-CAT GCG GTC GTA GTC CAT AAT-3’; IL-1β forward primer: 5’-GCT GCT TCC AAA CCT TTG AC-3’, reverse primer: 5’-AGC TTC TCC ACA GCC ACA AT-3’; IL-6 forward primer: 5’-TGC CTT CTT GGG ACT GAT GT-3’, reverse primer: 5’-ATA CTG GTC TGT TGT GGG TGG T-3’; β-actin forward primer: 5’-GAG ACC TTC AAC ACC CCA GC -3’, reverse primer: 5’-ATG TCA CGC ACG ATT TCC C-3’.
Western blotting
Protein samples were prepared using RIPA buffer according to the manufacturer’s protocol. Totally, 50 µg proteins from each sample were loaded on the 10% dodecyl sulfate sodium salt (SDS)-polyacrylamide gel electrophoresis (PAGE). After being transferred onto PVDF membranes, the membranes were blocked with 5% nonfat milk solution. The proteins on the membranes were incubated with respective primary antibodies including anti-bax (1:1500; ab232479), anti-bcl-2 (1:2500; ab182858), anti-cleaved caspase-3 (1:2000; ab214430), anti-nuclear Nrf2 (1:1000; ab31163), anti-lamin B1 (1:1000; ab133741), anti-HO-1 (1:1500; ab68477), anti-NQO-1 (1:1000; ab34173), and anti-β-actin (1:800; ab8227; Abcam, Cambridge, UK) overnight at 4°C. Thereafter, the membranes were washed with PBST buffer and then incubated with respective secondary antibody (1:1000; ab97051; Abcam, Cambridge, UK) for 1 h at room temperature. The protein bands were visualized by enhanced chemiluminescence kit (Amersham Biosciences, Piscataway, NJ, USA) and quantified with Image J software (NIH, Bethesda, MD, USA).
Cell transfection
Commercial si-Nrf2 and scrambled nontarget negative control siRNA (si-control) were obtained from Invitrogen. Hippocampal neurons (5 × 105 cells/well) were transiently transfected with si-Nrf2/si-control using Lipofectamine 2000 (Invitrogen) according to the manufacturer’s protocol.
Statistical analysis
All data were presented as mean ± standard error of the mean (SEM). One-way analysis of variance (ANOVA) was carried out to analyze the differences among groups using GraphPad Prism 5.0 (GraphPad Software, San Diego, CA, USA). P-values less than 0.05 were regarded as statistically significant.
Results
JZL184 improved cell viability in OGD/R-induced hippocampal neurons
LDH is a cytosolic enzyme that can be released into the extracellular space when the plasma membrane is damaged in injured cells. Therefore, the increased LDH content is usually used for an indicator of cell damage. LDH release assay proved that the hippocampal neurons did not show any changes in LDH content after treatment with 5, 10, or 20 μM of JZL184. However, the LDH content was slightly increased when treated with 40 μM of JZL184 (Figure 1(a)). Hence, the concentrations of 5, 10, and 20 μM were used for the following experiments. Furthermore, we found that OGD/R treatment resulted in a significant reduction in cell viability of hippocampal neurons as compared to the control group. However, treatment with JZL184 exhibited improvement in cell viability of hippocampal neurons exposed to OGD/R (Figure 1(b)).

Improvement of cell viability in OGD/R-induced hippocampal neurons. (a) Cytotoxicity effect of JZL184 on hippocampal neurons was evaluated by LDH release assay after incubating with 5, 10, 20 or 40 μM of JZL184. *p < 0.05 versus hippocampal neurons without JZL184 treatment. (b) Cell viability was determined by MTT assay after pretreatment with 5, 10, or 20 μM of JZL184 and the following OGD/R treatment. *p < 0.05 versus hippocampal neurons without any treatment. # p < 0.05 versus hippocampal neurons with OGD/R treatment.
JZL184 ameliorates OGD/R-induced oxidative stress in hippocampal neurons
It has been confirmed that increased ROS production and ROS-mediated oxidative stress play crucial roles in the I/R injury. Therefore, we evaluated the effect of JZL184 on OGD/R-induced oxidative stress through detection of parameters associated oxidative stress including ROS, MDA, SOD and GPx. Hippocampal neurons with OGD/R treatment showed significant increases in ROS and MDA production as compared to normal control. While JZL184 treatment suppressed the production of ROS and MDA with the increasing of the concentration (Figure 2(a) and (b)). Besides, OGD/R induction induced significant decreases in the activities of SOD and GPx in hippocampal neurons, whereas the changes were prevented by JZL184 treatment (Figure 2(c) and (d)).

JZL184 exhibited anti-oxidative activity in OGD/R-induced hippocampal neurons. Hippocampal neurons were pretreated with 5, 10, or 20 μM of JZL184, followed by OGD/R treatment. (a and b) Quantitative levels of ROS and MDA in hippocampal neurons. (c and d) Activities of SOD and GPx in hippocampal neurons. *p < 0.05 versus hippocampal neurons without any treatment. # p < 0.05 versus hippocampal neurons with OGD/R treatment.
JZL184 inhibited OGD/R-induced inflammatory response in hippocampal neurons
In addition to oxidative stress, ROS-mediated production of inflammatory cytokines is another major mechanism responsible for I/R injury. We next validated the effect of JZL184 on the production of inflammatory cytokines including TNF-α, IL-1β, and IL-6. We assessed the increase in the mRNA levels of TNF-α, IL-1β, and IL-6 in OGD/R-induced hippocampal neurons. However, the increased TNF-α, IL-1β, and IL-6 mRNA levels were decreased after treatment with JZL184 (Figure 3(a)–(c)). Additionally, the levels of TNF-α, IL-1β, and IL-6 in culture supernatant were also increased in OGD/R-induced hippocampal neurons, which were decreased in JZL184-treated hippocampal neurons, as evident in Figure 3(d)–(f).

JZL184 exhibited anti-inflammatory activity in OGD/R-induced hippocampal neurons. Following indicated treatments, the levels of inflammatory cytokines including TNF-α, IL-1β, and IL-6 were determined. (a–c) The mRNA levels of TNF-α, IL-1β, and IL-6 were determined using qRT-PCR. (d–f) The levels of TNF-α, IL-1β, and IL-6 in culture supernatant were determined using ELISA. *p < 0.05 versus hippocampal neurons without any treatment. # p < 0.05 versus hippocampal neurons with OGD/R treatment.
JZL184 inhibited OGD/R-induced cell apoptosis in hippocampal neurons
Importantly, ROS can regulate the apoptosis-associated pathway to induce cell apoptosis. We first evaluated cell apoptosis by TUNEL staining, showing that hippocampal neurons under OGD/R condition had intense TUNEL staining when compared with control hippocampal neurons. However, the TUNEL staining in JZL184-treated hippocampal neurons were slighter than OGD/R-induced cells (Figure 4(a)). Moreover, western blot analysis proved that the increased bax and cleaved caspase-3 expressions and decreased bcl-2 expression in OGD/R-induced hippocampal neurons were attenuated by JZL184 treatment (Figure 4(b) and (c)).
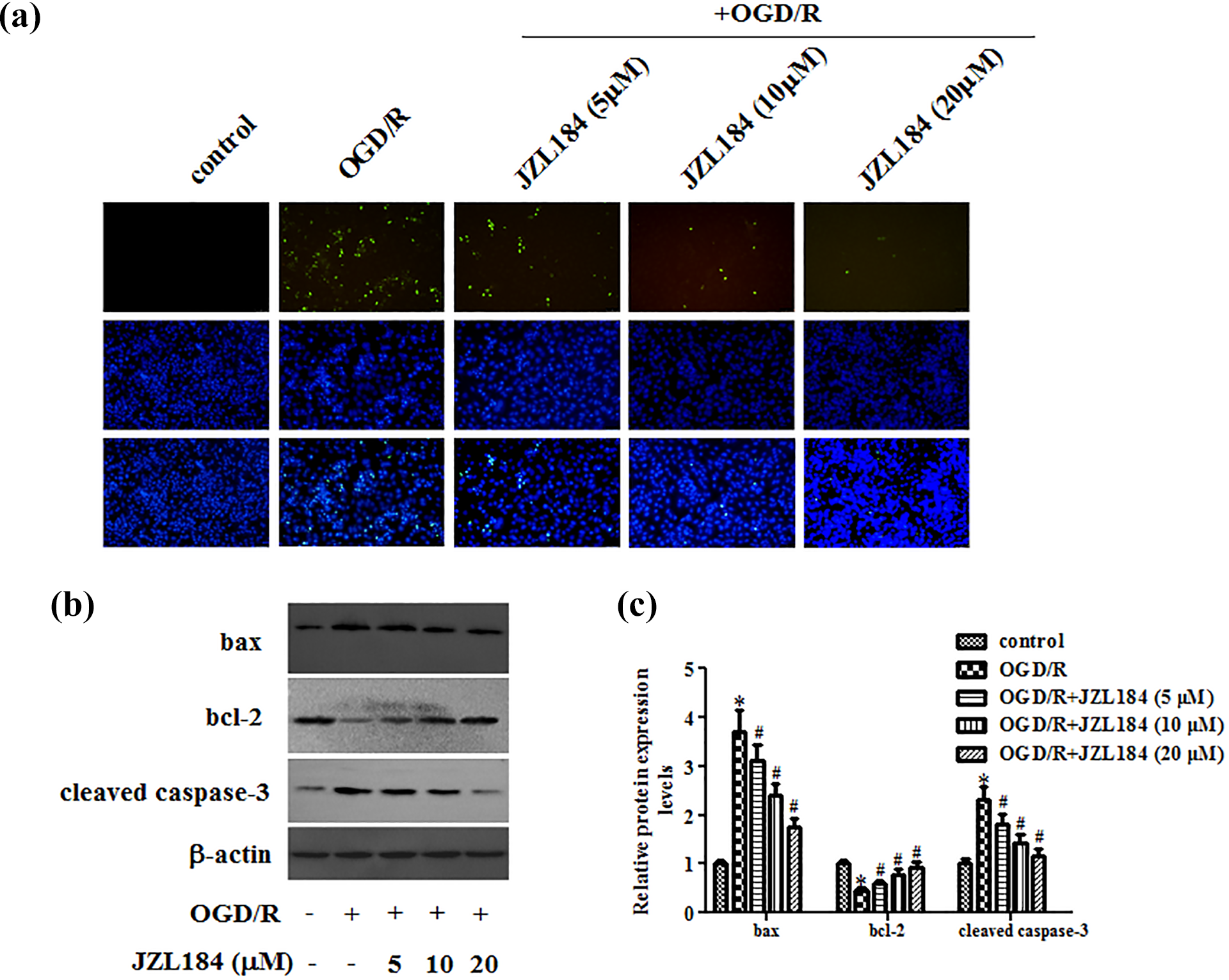
JZL184 exerted anti-apoptotic activity in OGD/R-induced hippocampal neurons. (a) Cell apoptosis of hippocampal neurons was evaluated by TUNEL staining. (b) Western blot analysis was used to detect the expression levels of bax, bcl-2 and cleaved caspase-3. (c) Quantification analysis of bax, bcl-2 and cleaved caspase-3. *p < 0.05 versus hippocampal neurons without any treatment. # p < 0.05 versus hippocampal neurons with OGD/R treatment.
JZL184 induced Nrf2/ARE pathway activation in hippocampal neurons under OGD/R condition
Nrf2/ARE has been observed to exert crucial role in response to I/R injury. To elucidate the potential mechanism underlying the protective effect of JZL184, we determined the effect of JZL184 on Nrf2/ARE pathway in hippocampal neurons under OGD/R condition. As indicated in Figure 5(a)–(c), as compared with the control group, OGD/R treatment increased the protein expression levels of nuclear Nrf2, HO-1 and NQO-1 in hippocampal neurons. However, these effects were enhanced by JZL184.
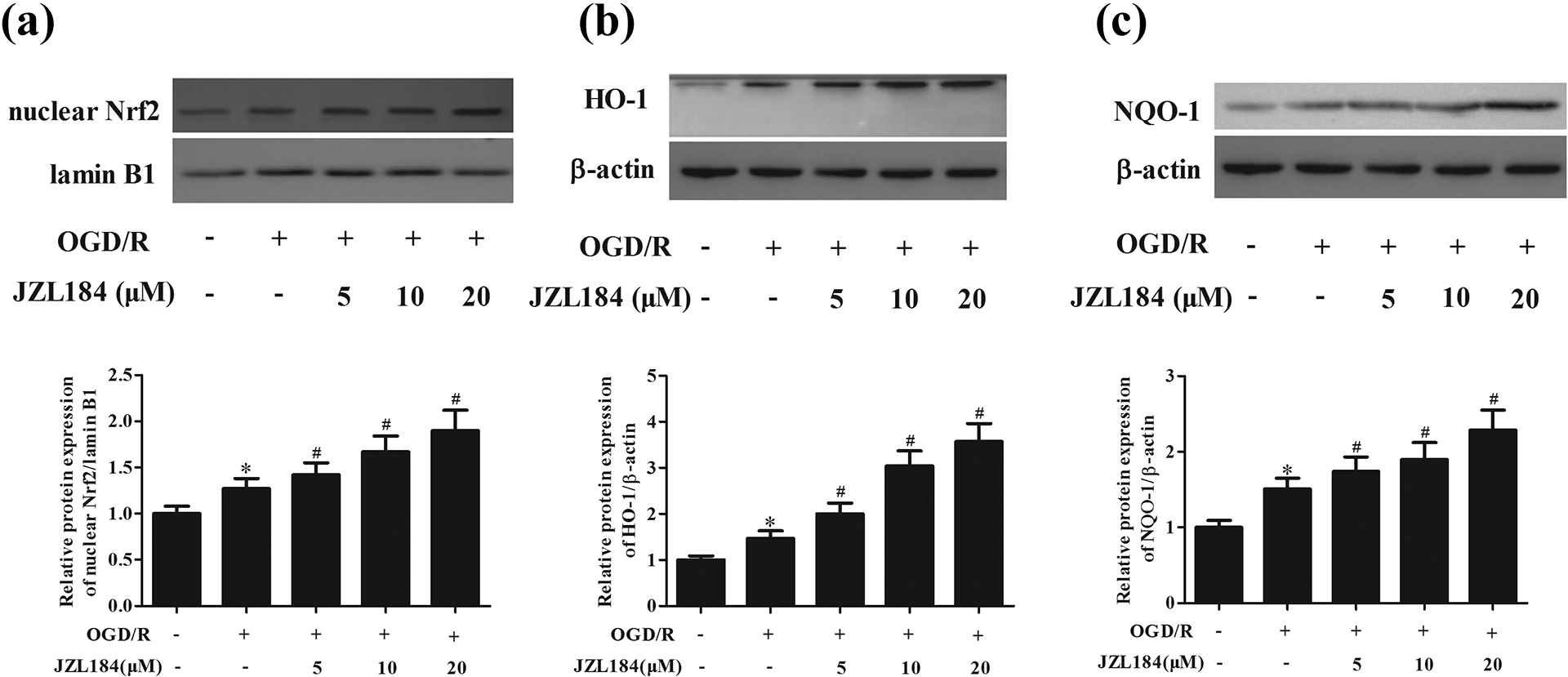
Enhanced activation of Nrf2/ARE pathway caused by JZL184 in hippocampal neurons under OGD/R condition. (a) Western blot analysis was performed to detect the expression levels of nuclear Nrf2. (b) Western blot analysis was performed to detect the expression levels of HO-1. (c) Western blot analysis was performed to detect the expression levels of NQO-1. *p < 0.05 versus hippocampal neurons without any treatment. # p < 0.05 versus hippocampal neurons with OGD/R treatment.
Si-Nrf2 reversed the effects of JZL184 on cell viability and ROS level in hippocampal neurons under OGD/R condition
To confirm that Nrf2/ARE pathway contributes to the regulation of the JZL184-mediated neuroprotective effect, we examined the effect of Nrf2 knockdown on the JZL184-mediated effect in OGD/R-exposed hippocampal neurons. As illustrated in Figure 6(a), transfection of si-Nrf2 significantly decreased the nuclear expression of Nrf2 in hippocampal neurons treated with JZL184 following OGD/R exposure. Moreover, the increased cell viability and reduced ROS generation caused by JZL184 were reversed by si-Nrf2 (Figure 6(b) and (c)).
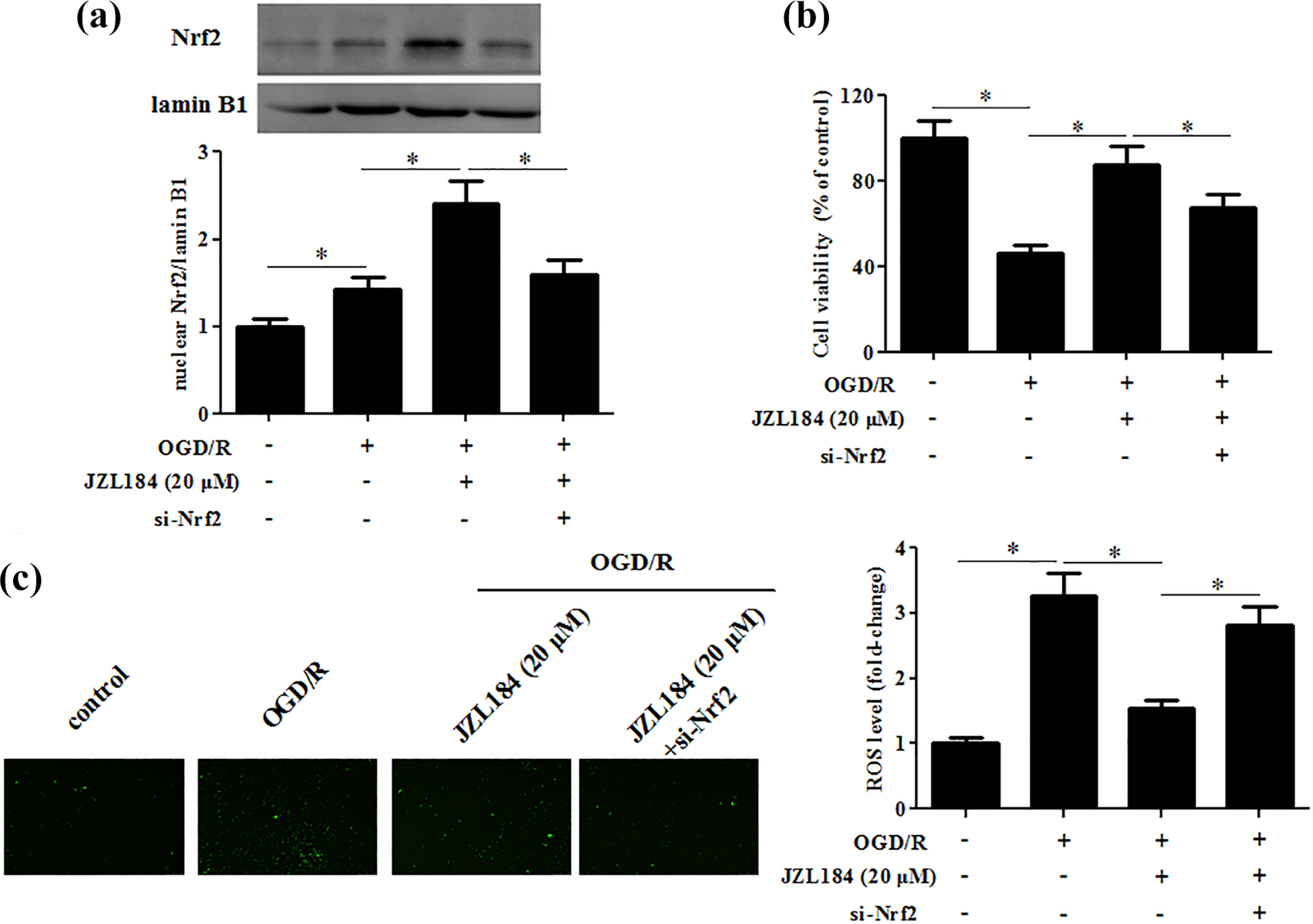
Si-Nrf2 reversed the effects of JZL184 on cell viability and ROS level in hippocampal neurons under OGD/R condition. Hippocampal neurons were transfected with si-Nrf2 in the presence of JZL184 (20 μM), followed by an OGD/R stimulation. (a) Nrf2 nuclear protein expression was measured by western blot. (b) Cell viability was determined by MTT assay. (c) Quantitative level of ROS in hippocampal neurons. *p < 0.05.
Si-Nrf2 reversed the effects of JZL184 on inflammatory cytokines production and apoptosis in hippocampal neurons under OGD/R condition
Furthermore, we observed that Nrf2 silencing markedly reversed the inhibitory effects of JZL184 on the production of TNF-α, IL-1β and IL-6 in OGD/R-exposed hippocampal neurons (Figure 7(a)–(c)). In addition, the suppressive effect of JZL184 on OGD/R-induced cell apoptosis was also markedly abolished by Nrf2 silencing (Figure 7(d)).
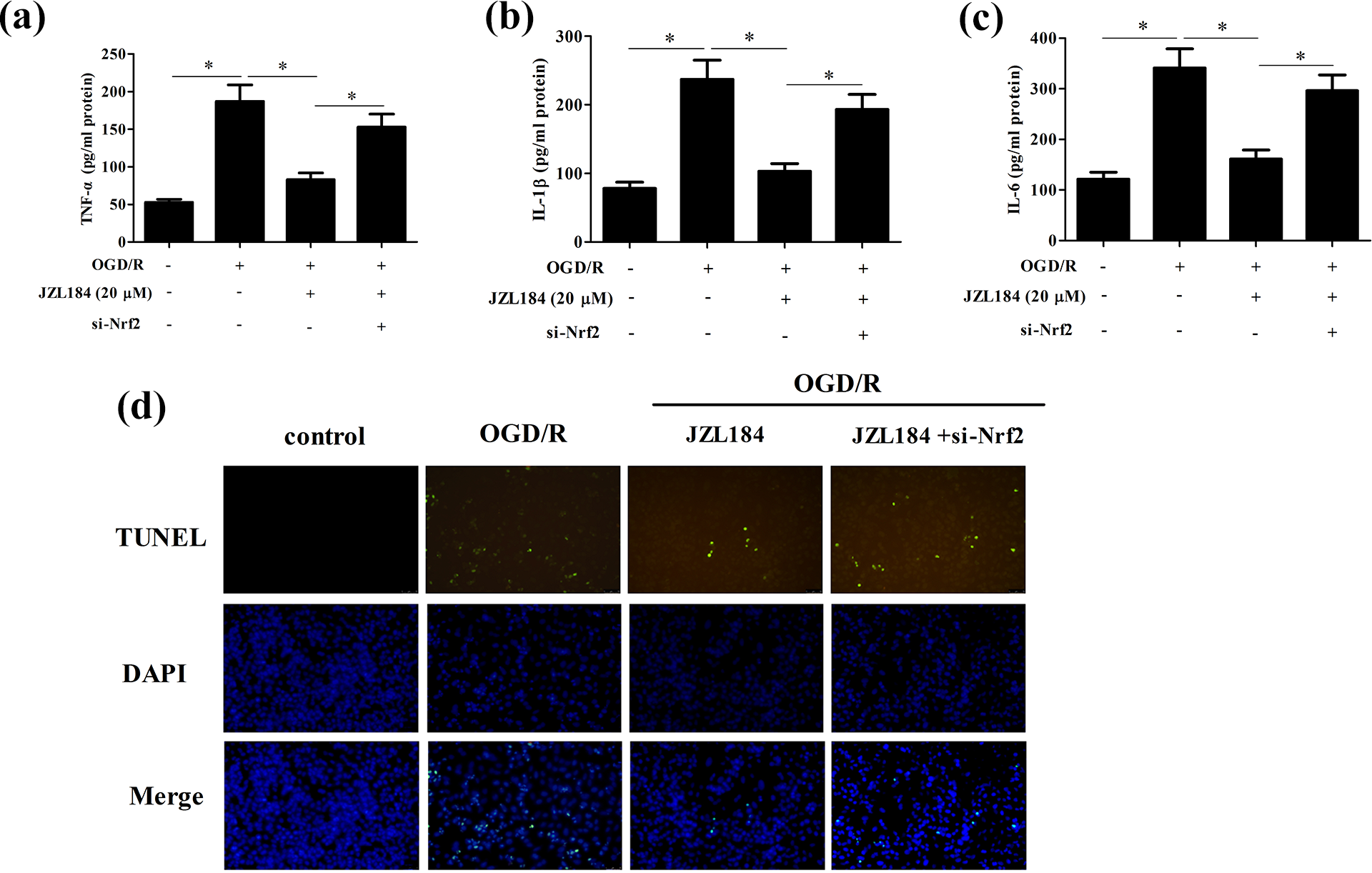
Si-Nrf2 reversed the effects of JZL184 on inflammatory cytokines production and apoptosis in hippocampal neurons under OGD/R condition. Hippocampal neurons were transfected with si-Nrf2 in the presence of JZL184 (20 μM), followed by an OGD/R stimulation. (a–c) The levels of TNF-α, IL-1β, and IL-6 in culture supernatant were determined using ELISA. (d) Cell apoptosis was evaluated by TUNEL staining. *p < 0.05.
Discussion
This study, for the first time, demonstrated that JZL184 protected hippocampal neurons from OGD/R-induced oxidative stress, inflammation and apoptosis. Our results provided in vitro evidence for the protective effect of JZL184 against cerebral I/R injury. In consistent with our study, several previous studies have identified the protective effect of JZL184 on cerebral ischemia in vivo. A recent study has reported that JZL184 administration reduces brain edema, infarct volume, brain levels of TNF-α, MMP9, and also improves behavioral tests in the mice middle cerebral artery occlusion (MCAO) model, which indicate that JZL184 may be a novel therapeutic approach for the treatment of stroke. 19 Rahmani et al. 20 reported that JZL184 reduces brain infarction, edema and inflammatory response in mouse permanent cerebral ischemia (PPMCAO) model. JZL184 reduces infarct volume and improves functional outcome in experimental ischemic stroke model. 21 Collectively, it now seems clear that JZL184 can be considered as a good candidate for inhibition of stroke injury.
Oxidative stress is closely associated with elevated intracellular levels of ROS that can results in damage to lipids, proteins and DNA, thus leading to cell apoptosis. 22 ROS is excessively produced in the I/R injury. In the current study, we found that JZL184 attenuated the oxidative stress in hippocampal neurons in response to OGD/R, suggesting that the protective effect of JZL184 on I/R injury is closely related to its anti-oxidative property.
Neuroinflammation plays a central role in the pathogenesis of ischemic brain injury. 23 Previous studies have shown that OGD/R can promote the levels of pro-inflammatory cytokines in neurons. 24,25 In line with these results, in the present study, we found that OGD/R treatment induced the levels of TNF-α, IL-1β and IL-6 in hippocampal neurons; however, these effects were attenuated by JZL184 pretreatment. These data suggest that JZL184 protects against OGD-induced injury in hippocampal neurons partly through inhibiting inflammatory response.
Accumulating evidence has demonstrated that apoptosis also plays a critical role in the process of neuronal death after cerebral ischemia. 26 –28 Bcl-2 is an anti-apoptotic factor and important for cell survival while bax promotes apoptosis. Caspase-3 acts a key factor in apoptosis, eventually triggers apoptosis under cerebral ischemia. 29 Herein, we observed that OGD/R greatly increased bax expression and reduced the expressions of bcl-2 and cleaved caspase-3. However, these effects were reversed by JZL184, implying that JZL184 protects against OGD-induced injury in hippocampal neurons partly via inhibiting cell apoptosis.
Nrf2 is a transcription factor that induces the expression of a great number of cytoprotective and detoxificant genes via binding to the ARE. 30,31 There are many evidences that Nrf2/ARE pathway is an intrinsic mechanism of defense against oxidative stress and becomes an emerging target against oxidative stress and neuroinflammation in neurogenic diseases. 32 The transcription factor Yin Yang 1 (YY1) potentiates anti-oxidative response in neuronal I/R injury via enhancement of Nrf2-mediated ARE transcription. 33 Levocarnitine attenuates oxidative damage and apoptosis in cerebral I/R rats via activating Nrf2/ARE signaling pathway. 34 An important antioxidant protein, DJ-1, plays a neuroprotection in astrocytes subjected to cerebral I/R injury by the Nrf2/ARE pathway. 35 Our results showed that JZL184 enhanced the activation of Nrf2/ARE pathway. Additionally, knockdown of Nrf2 reversed the protective effect of JZL184 on hippocampal neurons under OGD/R condition. These findings suggested that JZL184 exerted its functions via activating Nrf2/ARE signaling pathway.
Taken together, our results from the present study suggested that JZL184, an MAGL inhibitor, was effective in attenuating cerebral I/R injury through its anti-oxidative, anti-inflammatory and anti-apoptotic effects, which are associated with the activation of Nrf2/ARE signaling pathway. These findings provided in vitro experimental support for the therapeutic benefit of JZL184 in ischemic stroke.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Institution foundation of the First Affiliated Hospital of Xi’an Jiaotong University (2019ZYTS-10) and the Natural Science Foundation of Shaanxi Province (No. 2018JM7021) in China.
