Abstract
In this study, we aimed to evaluate the effect of microRNA-126 (miR-126) on neuronal apoptosis in cardiopulmonary resuscitation rats and to explore the related molecular mechanism. The expression of miR-126 in brain tissues of rats after cardiopulmonary resuscitation was measured by real-time fluorescence quantitative polymerase chain reaction (RT-qPCR). The basic parameters of cardiopulmonary resuscitation were recorded by miR-126 mimic injection in rats after cardiopulmonary resuscitation. Hematoxylin–eosin staining was used to observe the pathological changes of hippocampus. Immunohistochemistry was used to observe the expression of p38 and caspase-3 protein. Furthermore, the expression of p38 mitogen-activated protein kinase (p38MAPK), Jun N-terminal kinase (JNK), and extracellular signal-regulated kinase 1/2 (ERK1/2) in rat hippocampus was detected by RT-qPCR and Western blot. In order to confirm whether miR-126 takes part in the p38MAPK pathway in the hippocampus of rats after cardiopulmonary resuscitation, the p38MAPK pathway inhibitor (SB203580) and activator (anisomycin) were added. The results showed overexpression of miR-126 could significantly increase the neurological function score and improve the pathological morphology of hippocampus in rats after cardiopulmonary resuscitation. miR-126 overexpression also could reduce the neuronal apoptosis, p38, and caspase-3 expression in the hippocampus. Moreover, the p38MAPK and JNK expression was downregulated and ERK1/2 expression was upregulated after miR-126 mimic injection (p < 0.05). The results of inhibition of p38MAPK pathway were consistent with those of overexpression of miR-126 (p > 0.05). This study indicated miR-126 could significantly reduce neuronal apoptosis of hippocampus in rats after cardiopulmonary resuscitation, which might be involved in the regulation of p38MAPK pathway.
Introduction
With the development of cardiopulmonary resuscitation and organ support technology, more and more patients with cardiac arrest can resume spontaneous circulation and continue to survive, but after the return of spontaneous circulation (ROSC), the surviving patients often have severe neurological dysfunction. Therefore, how to further improve the neurological prognosis of patients after ROSC has become a hot and difficult point in the research field of cardiopulmonary resuscitation. 1 –3
A growing number of studies have found that microRNAs (miRNAs) play an important regulatory role in the pathophysiological cell and molecular signaling pathways of neurological dysfunction. 4,5 A variety of miRNAs have been found to be involved in the process of apoptosis in cerebral ischemia, hypoxia, and reperfusion injury. 6 microRNA-126 (miR-126) is high accumulation in vascular endothelial cells. It can specifically bind to the messenger RNA (mRNA) of the target gene and play a regulatory role after transcription. As a regulatory gene, miR-126 participates in the regulation of growth, proliferation, differentiation, apoptosis, and other biological processes in cells. 7 Researchers showed that miR-126 could promote the proliferation of neural stem cells and inhibit their apoptosis during in vitro neural stem cell culture. 8 In addition, studies also confirmed that elevated levels of miR-126 might play a functional role in dopaminergic (DA) neurons and in Parkinson’s disease (PD) pathogenesis by downregulating insulin-like growth factor-1 (IGF-1)/phosphatidylinositol 3-kinase (PI3K)/protein kinase B (AKT) signaling and that its inhibition could be a mechanism of neuroprotection. 9 These reports indicate that miR-126 plays an important regulatory role in the growth and differentiation of neural stem cells. Whether miR-126 has the effect of alleviating nerve cell damage in rats after cardiopulmonary resuscitation has not been reported.
The main objective of this study was to explore the effect of miR-126 on the apoptosis of neuronal cells of hippocampus in rats after cardiopulmonary resuscitation and its molecular mechanism.
Materials and methods
Research objects
Ninety specific pathogen free (SPF) male Wistar rats, weighing 180–200 g and aged 6–8 weeks, purchased from Jinan Pengyue Experimental Rat Breeding Co., Ltd (Jinan, China) with rat production license number SCXK (Lu) 20140007 were selected in this study. The feeding environment was as follows: 22 ± 1°C, relative humidity 60 ± 10%, 12-h light and 12-h dark cycle, free diet, and drinking water. All animal procedures performed the National Institutes of Health (NIH) guidelines (NIH Pub. No. 85-23, revised 1996) and the work was approved by the Animal Protection and Use Committee of Weihai Municipal Hospital.
Establishment of rats ROSC model
According to previous studies, 10,11 3% pentobarbital solution (40 mg/kg, Seebio®, Shanghai, China) was administered intraperitoneally to anesthetize rats, and cardiac arrest was induced by ventricular fibrillation using a 12 V/50 Hz alternating current through the esophageal electrode. Cardiopulmonary resuscitation was started with artificial ventilation (FiO2 1.0) after 6 min, and chest compressions were performed with the index finger at a rate of 200 times/min. Then, 20 μg/kg of epinephrine (Seebio®) was administered, and if necessary, transthoracic defibrillation (5 J, DC-defibrillator DEFIPORT SCP912; Hellige, Freiburg, Germany) was performed for 2 min. Sodium bicarbonate was administered until metabolic acidosis was neutralized. The blood pH was measured by gas analyzer BEARS (CIBA Corning Diagnostics Crop., Medfield, Massachusetts, USA). The normal pH is 7.4. 12 The above recovery process was continued until ROSC was restored. If ROSC cannot be reached after the first defibrillation attempt, a single strong shock of 5 J was repeated after each 30 s. If cardiopulmonary resuscitation did not succeed for 5 min, the resuscitation was stopped and the rats were declared dead. After ROSC, the rats returned to the cage and were free to access food and water. The basic parameters (survival rate and vital signs) before and after cardiopulmonary resuscitation were recorded.
Neurological deficit score
According to previous reports, 10,11 the neurological deficit score (NDS) ranged from 80 to 0. A score of 0 indicated brain death, and a score of 80 indicated normal neurological function (Table 1). The neurological outcomes of the successfully resuscitated rats were assessed and the NDS was determined at 24, 48, and 72 h after ROSC. The higher the score was, the better the neurological function. NDS > 60 points can be used as an independent predictor of good neurological outcome in rats. Each group of rats was scored by three professionally trained laboratory workers and averaged. The criteria for cardiac arrest are as follows: arterial pulse fluctuation signals disappear or arterial blood pressure was lower than 10 mmHg. Judgment criteria for spontaneous circulation recovery: supraventricular rhythm (sinus, atrial, and borderline rhythm), accompanied by mean arterial pressure >20 mmHg, lasting more than 5 min. Criteria for successful recovery: restoring autonomous circulation within 10 min was defined as successful recovery.
Neurodeficit scoring for rats (normal = 80; brain dead = 0).a

a Balance beam testing is normal if the rat can cross a 2 cm wide by 1 m long beam suspended 0.5 m above the floor. Abnormal is scored if the rat attempts and does not continue or stays momentarily and falls. Absent is scored when the rat falls off immediately upon placement on the beam. Other behavior reflex subscores evaluated the following: (1) righting reflex (animal placed on its back is able to correct to upright position), (2) turning alley (the animal is made to walk and turn back at the end of a 15 cm by 0.5 m alley), (3) visual placing (the animal is lifted and is able to visually orient itself to objects and depth), and (4) negative geotaxis (animal placed on its back on a plane angled at 45° corrects itself and moves up the incline).
Experimental grouping
Rats were randomly divided into four groups: sham operation (Sham), model (Model), mimic control (NC), and miR-126 mimic groups. Sham group was treated with no cardiac arrest. Model group was treated with cardiopulmonary resuscitation after cardiac arrest. NC group were treated with cardiac arrest after cardiopulmonary resuscitation and 10 μL of normal saline was injected into the lateral ventricle (anteroposterior: −0.72 mm, mediolateral: +1.4 to +2.0 mm, and dorsoventral: −3.8 mm from bregma). 13 miR-126 mimic group was treated with cardiopulmonary resuscitation after cardiac arrest. And 10 μL of miR-126 mimic (10 μmol/mL, HMI0116-5NMOL; Lifes Biological Laboratory Equipment Co., Ltd, Hangzhou, China) was injected into the lateral ventricle. Three percent pentobarbital sodium (40 mg/kg) was anesthetized on the 7th day after ROSC, the rats were killed by cervical dislocation, and the rat brain was removed. Some of the tissue was fixed in 10% formalin for 24 h at room temperature, embedded in paraffin. Some of them were frozen in liquid nitrogen and then transferred to −80°C for storage.
Real-time fluorescence quantitative polymerase chain reaction
Tissue total RNA was extracted using a Trizol kit (Takara, Japan). It was transcribed into the complementary DNA, according to the instructions of the Reverse Transcription Kit (Applied Biosystems, Waltham, Massachusetts, USA). Real-time fluorescence quantitative polymerase chain reaction (RT-qPCR) detection was performed using a Mastercycler® nexus X2 (Eppendorf, Hamburg, Germany). Conditions were as follows: 95°C for 30 s, 95°C for 30 s, and 60°C for 20 s (45 cycles). The data were processed by 2−ΔΔCt method, and the relative expression levels of miR-126 and p38 mitogen-activated protein kinase (p38MAPK), Jun N-terminal kinase (JNK), and extracellular signal-regulated kinase (ERK) was calculated by using U6 and glyceraldehyde-3-phosphate dehydrogenase (GAPDH) mRNA as internal parameters, respectively. The primers (Shanghai Shenggong Bioengineering Technology Service Co., Ltd, Shanghai, China) were as follows: miR-126, forward: 5′-CATTATTACAGGGCAGCGGTCGC-3′; reverse: 5′-ATTATTACGCGGCAGGTGC-CGT-3′. p38MAPK, forward: 5′-CCCGAACGATAC-CAGAACC-3′; reverse: 5′-GCGTGAATGATG-GACTGAAA-3′. JNK, forward: 5′-AGGAATAGTGTGTG-CAGCTTATG-3′; reverse: 5′-CTTCTAGGGATTTCTGTGGT-GTG-3′. ERK, forward: 5′-AGTGCTTCCTCTACTGTGATGC-3′; reverse: 5′-TCTGCCCTCTAAAACCAAGG-3′. GAPDH, forward:5′-TGACCTCAACTACATGGTCTACA-3′; reverse: 5′-CTTCCCATTCTCGGCCTTG-3′.
Hematoxylin–eosin staining
The embedded hippocampus brain tissue was selected and sectioned. The thickness of the section was 5 μm. The sections were conventionally dewaxed with xylene and hydrated by gradient ethanol. The section was stained by hematoxylin (Solarbio, Beijing, China) for 5 min and then rinsed with tap water. The section was differentiated in the ethanolic hydrochloric acid for 30 s. And then, the section was placed in the eosin dye solution (Solarbio) for 2 min. After dehydration, transparency, and mounting, histopathological changes were observed under a 400× optical microscope (Olympus Model BX50; Olympus, Japan).
Terminal deoxynucleotidyl transferase-mediated deoxyuridine triphosphate nick-end labeling
The embedded rat brain tissue was selected (4 μm) (biological tissue paraffin embedding machine, paraffin slicer/Leica Company, Germany). The sections were dewaxed and dehydrated, respectively, by conventional xylene and gradient ethanol and then detected by the terminal deoxynucleotidyl transferase-mediated deoxyuridine triphosphate nick-end labeling method using the apoptosis detection kit (batch number: ZK-8005; Beijing Zhongshan Jinqiao Biotechnology Co., Ltd, Beijing, China). Under the 400× optical microscope (BX50/Olympus), five fields of view were randomly selected to observe the apoptosis of tissue. The apoptotic cells were brown or brown-yellow. Apoptotic index (AI) was calculated to reflect the degree of apoptosis. AI = (number of apoptosis cells/total number of cells) × 100%.
Immunohistochemical detection
After routine sectioning of rat brain tissue, the baked pieces were dewaxed with xylene and hydrated by a gradient ethanol solution. Inactivated by 3% H2O2 methanol solution for 20 min, heat-fixed for 10 min in citrate buffer (pH 6.0), and blocked with 5% bovine serum albumin (Solarbio) for 20 min. Rabbit anti-mouse p38 (1:200, ab31828; Abcam, UK) and caspase-3 (1:500, ab31828; Abcam) polyclonal antibodies were added dropwise and reacted overnight at 4°C. After rewarming, horseradish peroxidase-labeled goat anti-rabbit IgG (1:1000, #7074; Cell Signaling Technology, Danvers, Massachusetts, USA) was used for secondary antibody incubation. 3,3'-Diaminobenzidine tetrahydrochloride (DAB) (Solarbio) was used for color development, and hematoxylin was used for counterstaining. And then it was dehydrated, transparent, and sealed. The results were observed at 400 times magnification (Olympus). The positive cell scoring criteria were as follows: less than 5% was 0 point, 6–25% was 1 point, 26–50% was 2 points, 51–75% was 3 points, and more than 75% was 4 points. The grading and scoring criteria of staining intensity were as follows: no color development was 0 point, light yellow was 1 point, yellow or dark yellow was 2 points, and brown or tan was 3 points, and the product of the two scores was used as the final score. 14
Western blot
The tissue was lysed, centrifuged (2000 r/min, 20 min), and the supernatant was taken. The protein concentration was measured by using a bicinchoninic acid (BCA) kit (Beijing Solebao Bio). Four microliters of the protein sample were added to the 5× sample buffer, applied to 10% sodium dodecyl sulfate-polyacrylamide gel electrophhoresis, and electrophoresed at a voltage of 80 V, and the proteins were separated by molecular weight. Protein samples were transferred to polyvinylidene fluoride membranes (Merck, Darmstadt, Germany) under semidrying conditions of 20 V and 30 min with an electrotransfer (Bio-Rad, Hercules, California, USA). Then, they were washed and blocked with 5% skim milk powder for 2 h. Each protein primary antibody (p-p38MAPK, 1:500, orb14942; Biorbyt, Cambridge, UK; p38MAPK, orb14630; p-ERK1/2, 1:500, orb229648; ERK1/2, 1:500, orb106403; p-JNK, 1:500, orb184400; JNK, 1:500, orb77280; and β-actin, 1:2000, orb178392) was added and incubated overnight at 4°C. After washing, they were incubated with goat anti-rabbit IgG-HRP (1:1000, #7074; Cell Signaling Technology) for 30 min. The results were observed and recorded using a Roche Elecsys-2010 chemiluminometer (Switzerland). The protein expression level was normalized according to β-actin, and gray scale scanning and quantification were performed by Image J software (V1.8.0.112, NIH).
In order to further determine whether miR-126 reduced neuronal apoptosis in cardiopulmonary resuscitation rats through the p38MAPK pathway, the p38MAPK pathway inhibitor and activator were added. Groups were as follows: Model, miR-126 mimic, p38MAPK pathway inhibitor (SB203580) and miR-126 overexpression + p38MAPK pathway activator (miR-126 + anisomycin (ANS)). In the p38MAPK pathway inhibitor group, 10 μL of 10 μmol p38MAPK pathway inhibitor SB203580 was injected with cardiopulmonary resuscitation. In miR-126 + ANS group, 10 μL of 10 μmol p38MAPK pathway activator ANS and miR-126 mimic mixture were injected simultaneously with cardiopulmonary resuscitation.
Statistical methods
The data were analyzed with SPSS19.0 statistical software. The results were expressed as mean ± standard deviation. The data analysis between multiple groups was based on one-way variance analysis, and the Fisher’s least significant difference test was used for subsequent analysis. Difference was statistically significant at p < 0.05.
Results
miR-126 overexpression ameliorated survival rate and NDS in cardiopulmonary resuscitation rats
The expression of miR-126 in rat brain tissue after cardiopulmonary resuscitation was detected by RT-qPCR (Figure 1(a)). Compared with the Sham group, the expression of miR-126 was significantly decreased in the Model group (p < 0.05). After miR-126 mimic injection, the level of miR-126 in rat brain tissue was significantly higher than that in the Model group (p < 0.05). miR-126 mimic treatment also significantly increased the survival rate in cardiopulmonary resuscitation rats (Figure 1(b), p < 0.05). The survival rate was 59.1% in the miR-126 mimic group, but only 34.8% and 27.3% survival rate occurred in the Model and NC groups. The NDS scores of rats in each group were shown in Figure 1(c). Compared with the Sham group, the NDS scores of rats in other groups were significantly reduced (p < 0.05). Compared with the Model group, the NDS scores of the miR-126 mimic group were significantly increased (p < 0.05).

miR-126 overexpression ameliorated survival rate and NDS in cardiopulmonary resuscitation rats. (a) The expression of miR-126 in the hippocampus of cardiopulmonary resuscitation rats was analyzed by RT-qPCR. (b) Effect of miR-126 expression on survival rate in cardiopulmonary resuscitation rats. (c) miR-126 overexpression improved the NDS score in cardiopulmonary resuscitation rats. Note: *p < 0.05, **p < 0.01, compared with Sham group; # p < 0.05, compared with Model group. miR-126: microRNA-126; NDS: neurological deficit score; RT-qPCR: real-time fluorescence quantitative polymerase chain reaction.
miR-126 overexpression improved histomorphology and neuronal apoptosis in rat hippocampus after cardiopulmonary resuscitation
Hematoxylin–eosin staining was performed on the hippocampus of each group of rats, and the results were shown in Figure 2(a). The neurons in the hippocampal CA1 area of the Sham group were closely arranged and the structure was clear, while the neurons in the Model group and NC group were edema, blurred, or invisible. In miR-126 mimic group, the arrangement of neurons in rats was still regular, and some intact cell structures were visible, and the degree of neuronal damage was reduced. The neuronal apoptosis in rat brain tissue was shown in Figure 2(b). Compared with the Sham group, the neuronal apoptosis rate of each experimental group was significantly increased (p < 0.05). Compared with the Model group, the apoptotic rate of neurons decreased significantly (p < 0.05) in the miR-126 mimic group.
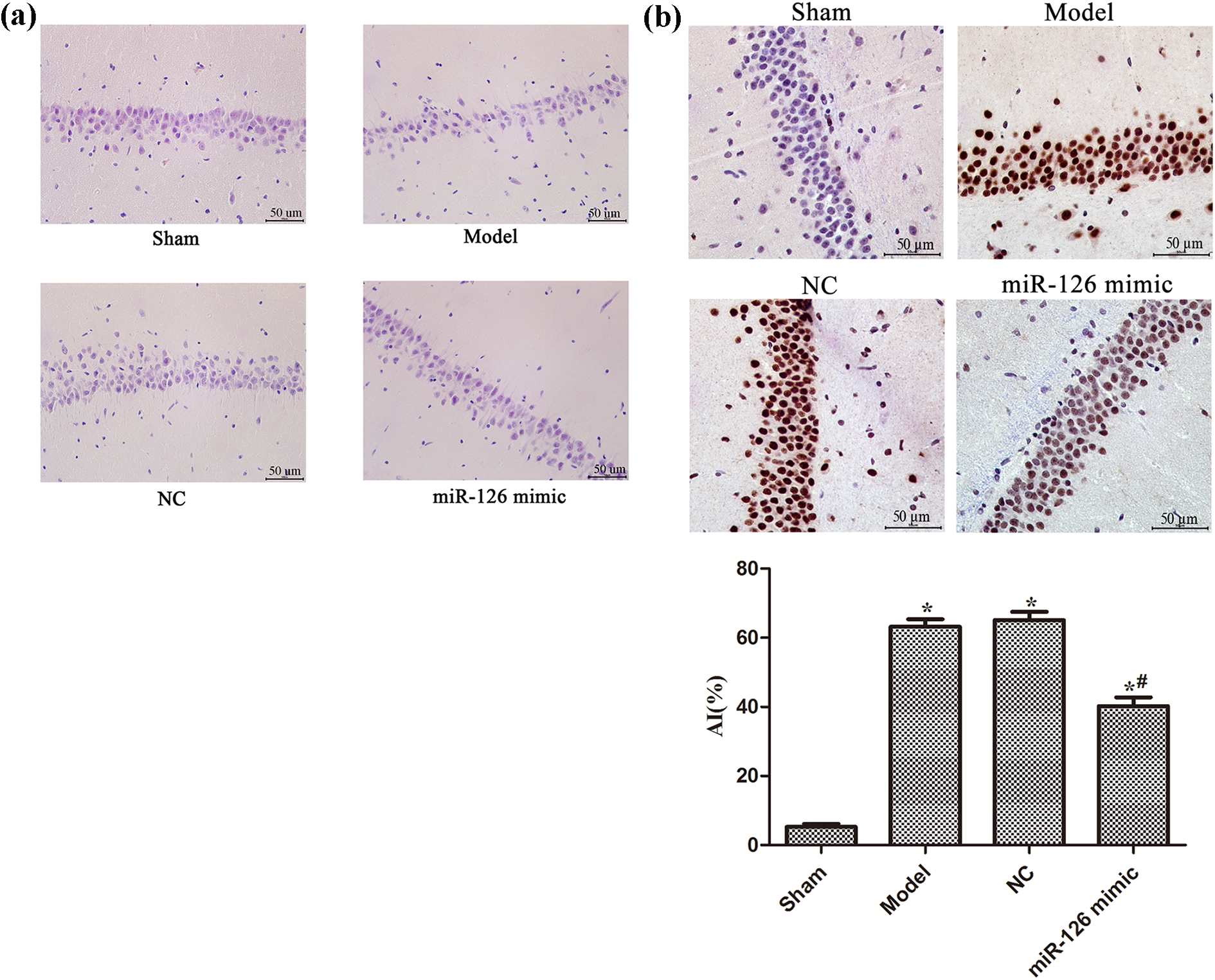
miR-126 overexpression improved histomorphology and neuronal apoptosis in rat hippocampus after cardiopulmonary resuscitation. (a) The histomorphology of the rat hippocampus was analyzed by HE staining. (b) The neuronal apoptosis in the rat hippocampus was analyzed by TUNEL. Note: *p < 0.05, compared with Sham group; # p < 0.05, compared with Model group. miR-126: microRNA-126; HE: hematoxylin–eosin; TUNEL: terminal deoxynucleotidyl transferase-mediated deoxyuridine triphosphate nick-end labeling.
miR-126 overexpression suppressed p38 and caspase-3 expression in the hippocampus of rat brain after cardiopulmonary resuscitation
Immunohistochemistry was used to detect the expression of p38 and caspase-3 protein in the hippocampus of each group of rats. It was found (Figure 3) that the positive expression intensity of p38 and caspase-3 in the brain tissue of rats in each experimental group increased significantly (p < 0.05), compared with the Sham group. The expression intensity of p38 and caspase-3 in the miR-126 mimic group was significantly decreased compared with the Model group (p < 0.05).

miR-126 overexpression suppressed p38 and caspase-3 expression in the hippocampus of the rat brain after cardiopulmonary resuscitation. The expression of p38 and caspase-3 protein in the hippocampus was analyzed by immunohistochemical detection. Note: *p < 0.05, compared with Sham group; # p < 0.05, compared with Model group. miR-126: microRNA-126.
miR-126 overexpression inhibited the phosphorylation of p38MAPK pathway in rat brain after cardiopulmonary resuscitation
As shown in Figure 4, compared with the Sham group, the expression of phosphorylation of p38MAPK, JNK, and ERK1/2 in the brain tissue of the cardiopulmonary resuscitation group increased significantly (p < 0.05). In the miR-126 mimic group, the expression of phosphorylation of p38MAPK, JNK, and ERK1/2 was significantly lower than those in the Model group (p < 0.05).

miR-126 overexpression inhibited the phosphorylation of the p38MAPK pathway in rat hippocampus after cardiopulmonary resuscitation. (a) The levels of p-p38MAPK, p-JNK, and p-ERK1/2 mRNA in the hippocampus were measured by RT-qPCR and (b) the phosphorylation of p38MAPK, JNK, and ERK1/2 expression in the hippocampus was measured by Western blot. Note: *p < 0.05, compared with Sham group; # p < 0.05, compared with Model group. miR-126: microRNA-126; p38MAPK: p38 mitogen-activated protein kinase; JNK: Jun N-terminal kinase; ERK1/2: extracellular signal-regulated kinase 1/2; RT-qPCR: real-time fluorescence quantitative polymerase chain reaction.
miR-126 reduced neuronal apoptosis in cardiopulmonary resuscitation rats by regulating the p38MAPK pathway
In order to further determine whether miR-126 reduces neuronal apoptosis in cardiopulmonary resuscitation rats by regulating the p38MAPK pathway, p38MAPK pathway inhibitor and activator were added to observe neuronal apoptosis in rat brain tissue. The results of adding p38MAPK pathway inhibitor were similar to those of miR-126 mimic, which could significantly improve the neurological function score and the pathological morphology of rat hippocampus (Figure 5(a) and (b)). Meanwhile, the p38MAPK pathway inhibitor could reduce the neuronal apoptosis (Figure 5(c)) and the expression of p38 and caspase-3 protein in brain tissue (Figure 6). At the same time, it could downregulate the phosphorylation of p38MAPK, JNK, and ERK levels (p > 0.05, Figure 7). Overexpression of miR-126 and activation of the p38MAPK pathway at the same time significantly reversed the effect of overexpression of miR-126.

miR-126 reduced neuronal apoptosis in cardiopulmonary resuscitation rats by regulating the p38MAPK pathway. (a) Rat neurological function score, (b) the histomorphology of rat hippocampus was analyzed by HE staining, and (c) the neuronal apoptosis in rat hippocampus was analyzed by TUNEL. Note: *p < 0.05, compared with Model group; # p < 0.05, compared with miR-126 mimic group. miR-126: microRNA-126; p38MAPK: p38 mitogen-activated protein kinase; HE: hematoxylin–eosin; TUNEL: terminal deoxynucleotidyl transferase-mediated deoxyuridine triphosphate nick-end labeling.

miR-126 reduced p38 and caspase-3 expression in the hippocampus of the rat brain after cardiopulmonary resuscitation by regulating the p38MAPK pathway. The expression of p38 and caspase-3 protein in the hippocampus was analyzed by immunohistochemical detection. Note: *p < 0.05, compared with Model group; # p < 0.05, compared with miR-126 mimic group. miR-126: microRNA-126; p38MAPK: p38 mitogen-activated protein kinase.

miR-126 overexpression inhibited the phosphorylation of the p38MAPK pathway in rat hippocampus after cardiopulmonary resuscitation. The phosphorylation of p38MAPK, JNK, and ERK1/2 expression in the hippocampus was measured by RT-qPCR (a) and Western blot (b). Note: *p < 0.05, compared with Model group; # p < 0.05, compared with miR-126 mimic group. miR-126: microRNA-126; p38MAPK: p38 mitogen-activated protein kinase; JNK: Jun N-terminal kinase; ERK1/2: extracellular signal-regulated kinase 1/2; RT-qPCR: real-time fluorescence quantitative polymerase chain reaction.
Discussion
This study found that the expression of miR-126 in the brain tissue of rats after cardiopulmonary resuscitation was significantly decreased, and overexpression of miR-126 significantly increased the survival rate of rats, improved the neurological function of cardiopulmonary resuscitation rats. Previous studies have reported that miR-126 promoted homing of endothelial progenitor cells within arterial thrombus in patients with cerebral infarction. 15 miR-126 levels were markedly downregulated in patients with ischemic stroke until 24 weeks. 16 Our results were consistent with the previous reports, which suggested that miR-126 might play an important role in the protection of the brain tissue of rats after cardiopulmonary resuscitation.
In order to study the mechanism of miR-126 in brain tissue, we found that an increase of miR-126 could alleviate the neuronal damage, reduce the apoptosis of neuronal cells, and decrease the caspase-3 and p38 expression in the hippocampal region after cardiopulmonary resuscitation for rats. In neurons, caspase-3 plays a key role in the phase of apoptosis and is related to the neuronal death in several acute and chronic neurodegenerative disorders. 17 p38 plays an important role in death cascades. Inhibition of the p38 pathway was effective against apoptosis induced by a wide variety of oxidative stressors. 18
The p38MAPK signaling pathway is known to have important biological functions in neuronal cells. 19,20 Studies have shown that overexpression of miR-126 can promote the differentiation of mesenchymal stem cells into endothelial cells by regulating the p38MAPK signaling pathway. 21 In endothelial progenitor cells, ginkgolide B promoted cell growth through overexpression of miR-126 and activation of the p38 MAPK signaling pathway. 22 Furthermore, reports showed that miR-126 could promote the proliferation of neural stem cells and inhibit their apoptosis during in vitro neural stem cell culture. 8 In addition, miR-126 may play a functional role in DA neurons and in PD pathogenesis by downregulating IGF-1/PI3K/AKT signaling and that its inhibition could be a mechanism of neuroprotection. 9 In this study, miR-126 significantly decreased the phosphorylation of p38MAPK pathway. But overexpression of miR-126 and activation of the p38MAPK pathway significantly reversed the protective effect of overexpressing miR-126 on rat neurons after cardiopulmonary resuscitation. These results indicated that miR-126 protected neural cells in rats after cardiopulmonary resuscitation through the p38MAPK signaling pathway.
Conclusion
miR-126 could improve the neurological function and decrease neuronal apoptosis of the hippocampus in rats after cardiopulmonary resuscitation, which might be due to the regulation of the p38MAPK pathway.
