Abstract
Recent years have seen substantial shifts in cultural attitudes towards cannabis for medical and recreational use. However, legalizing recreational marijuana may have adverse effects on individual and public health. As the most widely used illicit agent, cannabis is commonly reported to disrupt learning and memory. Unfortunately, the molecular mechanisms underlying behavioral impairment by cannabis abuse remain poorly understood. Tetrahydrocannabinol (THC), a major component in cannabis, causes short-term effects on the visual system, but little is known about persisting visual disturbances. This study was to investigate the effects of systemic administration with THC on retina and explore its underlying mechanisms. BALB/c mice were treated with 1 or 2 mg/kg THC intraperitoneally daily for 2 months, mice treated with vehicle as negative control. The retinal function was tested by electroretinography after THC treatment. Morphology and pathology changes of retina were detected by hematoxylin and eosin staining. Terminal deoxynucleotidyl transferase dUTP nick end labeling assay was used to detect the apoptosis in photoreceptor cells. Enzyme-linked immunosorbent assay was used to show the inflammatory responses and oxidative stress. mRNA and protein changes were measured by real-time polymerase chain reaction and Western blot to explore the underlying mechanisms. Results indicated that 2-month treatment with THC caused retinal damage, evidenced by its functional loss and increased apoptosis in photoreceptor cells through inducing inflammatory responses and oxidative stress. Our study demonstrated that systemic administration with THC caused toxic effects on retinas of BALB/c mice, suggesting the potential mechanisms for the retina damage caused by cannabis abuse.
Introduction
As the world’s most widely cultivated, trafficked, and abused illicit substance, cannabis is used by approximately 2.5% of the world’s population (147 million people). 1,2 In recent years, cannabis use peaks during adolescence, with greater decline in cognitive functions, including learning and memory performance. 3 The initial state of acute intoxication formulates the symptoms such as euphoria and perception alterations. The active ingredient is believed to be tetrahydrocannabinol (THC), which is also responsible for intoxication. Studies showed that inhaled doses of 2–3 mg of THC and ingested doses of 5–20 mg THC can cause impairment of memory, attention, executive functioning, and short-term memory, although with potential use as a novel topical therapy for the treatment of glaucoma after short-term application. 4 Chronic use may lead to long-term effects on cognitive performance. 5,6 Impairment of sensory perception is among the more common effects with hallucinogen persisting perception disorder after marijuana consumption. Although much progress has been made in understanding the neurotoxicity of cannabis abuse, 7 –9 its toxic effects on the visual system after long-term treatment have not been elucidated, specifically on retina. Therefore, it is necessary to investigate its effects and the underlying mechanisms.
Cannabinoids act on two cannabinoid binding (CB) receptors, CB1 and CB2. CB1 is primarily centrally located and mainly involved in central effects, including consequences on learning, memory cognition, emotion, sensory perception, and so on. CB2 is primarily peripherally located and found to affect inflammation and immune system regulation. The wide distribution of CB1 in both the anterior eye and the retina suggests that cannabinoids influence several different physiological functions in the eye. 10 Stimulation of CB1 may result in the stimulation of various neurotransmitters, including L-glutamate, dopamine, and 5-hydroxytryptamine, which contribute to the central and peripheral effects observed in cannabinoid toxicity. 11 Neuroinflammation has been considered as an important cause relating with neurological disorders. 12 THC has been characterized as a full agonist at CB1 receptors on gamma-aminobutyric acid neuron axon terminals in the hippocampus, 13 which may contribute to THC-induced neurotoxicity, because activated neurons produce pro-inflammatory cytokines and reactive oxygen species (ROS) to amplify the neuron damage. 14
The use of hallucinogenic drugs causes prolonged adverse reactions. Among these reactions are hallucinogen persisting perception disorder and persistent psychosis which are usually seen after repeated drug administrations. This study was to investigate whether systemic administration with THC would cause toxic effects on retinas of BALB/c mice, providing the potential mechanisms for the retinal damage caused by cannabis abuse.
Materials and methods
Mouse treatment
Six-week-old BALB/c mice were purchased from Vital River Ltd (Beijing, China) and housed in humidity and temperature-controlled (23 ± 2°C) facility on a 12-h light/12-h dark cycle. Mice had ad libitum access to food and water in their home cage. All animal studies were performed according to Chinese animal care guidelines under an approved Institutional Animal Care and Use Committee protocol from Harbin Medical University. THC was purchased from National Institutes for Food and Drug Control (Beijing, China). The mixture solution of ethanol, alkamuls-620 (Rhone-Poulenc, Princeton, New Jersey, USA), and saline (prepared according to the ratio: 1/1/18, v/v/v) was used to dissolve THC. BALB/c mice were treated with THC at 1 or 2 mg/kg (n = 10) by intraperitoneal injection daily with mice treated with vehicle (n = 10) as negative control.
Electroretinography
After overnight dark adaptation, mice were anesthetized to perform the electroretinography (ERG) test with UTAS-E3000 Electrophysiology System (LKC Technologies, Gaithersburg, Maryland, USA). 15,16 Mouse pupils were dilated, and the corneal surface was anesthetized with 0.5% proparacaine hydrochloric acid (Alcon, Beijing, China). Reference and ground electrodes were attached to the mouth and placed in the neck-back region subcutaneously. In the light-adapted session, the flash luminance ranged from −0.8 log cd s/m2 to 1.9 log cd s/m2. In the dark-adapted session, the flash luminance ranged from −2.4 log cd s/m2 to 2.1 log cd s/m2 (b wave) and 0 log cd s/m2 to 2.1 log cd s/m2 (a wave). The ERG a-wave is the initial corneal-negative deflection and its amplitude was measured from baseline to the trough of the a-wave. The ERG b-wave is the corneal-positive deflection and its amplitude is measured from the trough of the a-wave to the peak of the b-wave (Figure 1). 17
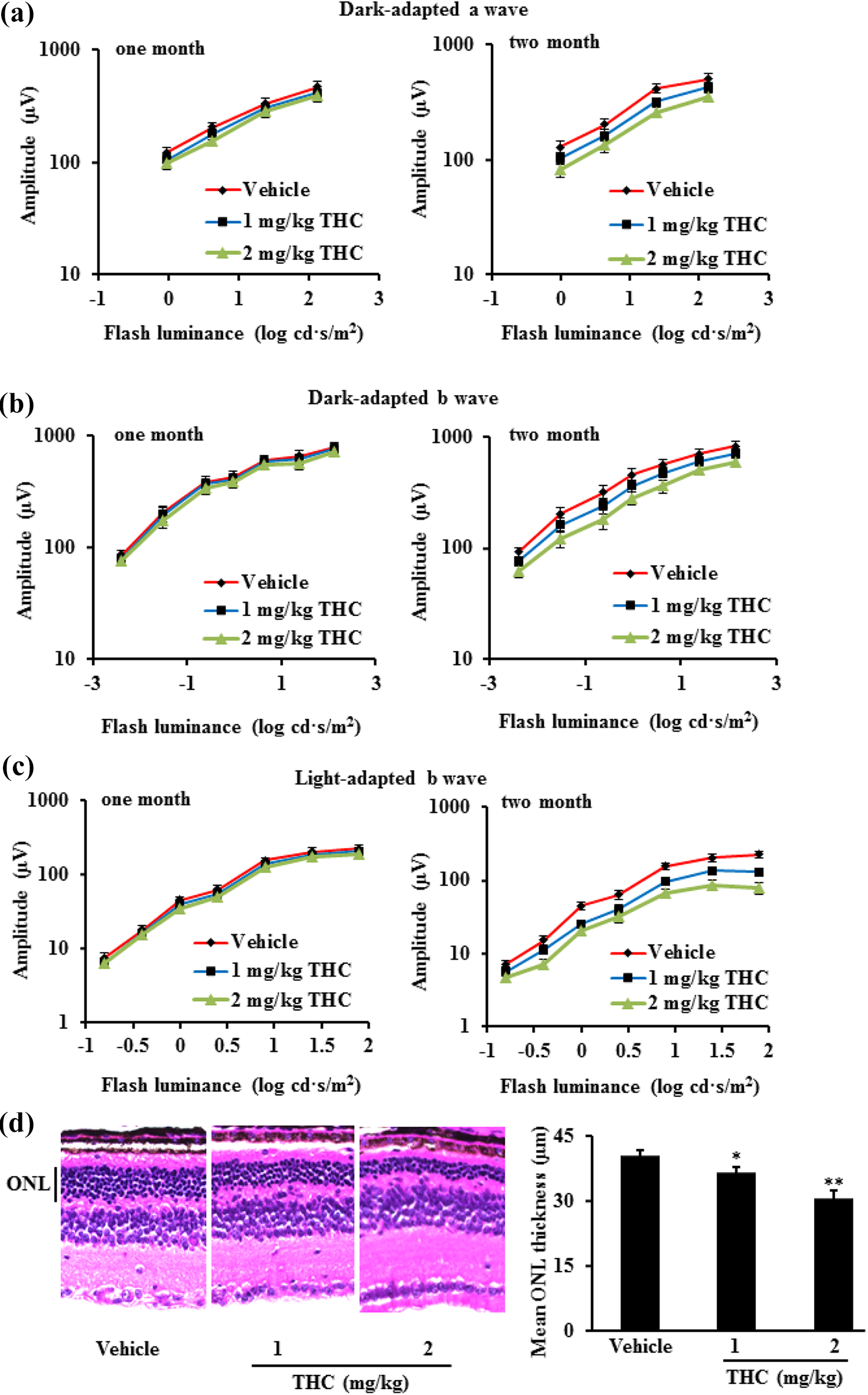
THC treatment caused retina dysfunction and damage in BALB/c mice. BALB/c mice were treated with THC at 1 or 2 mg/kg (n = 10) by intraperitoneal injection daily, mice treated with vehicle (n = 10) as negative control. ERG response was obtained from BALB/c mice treated with THC (n = 10) or vehicle (n = 10) after 1 and 2 months, shown by dark-adapted ERG a wave (a), dark-adapted ERG b wave (b), and light-adapted ERG b wave (c). Representative images (×20) of HE staining from BALB/c mice treated with THC or vehicle after 2 months were presented. THC treatment significantly decreased the ONL thickness (d). Data were expressed as mean ± SD. *p < 0.05, **p < 0.01 versus vehicle group. THC: tetrahydrocannabinol; ERG: electroretinography; HE: hematoxylin and eosin; ONL: outer nuclear layer.
Terminal deoxynucleotidyl transferase dUTP nick end labeling assay
After euthanization with carbon dioxide, mouse eyes were fixed with 4% paraformaldehyde for 4 h and then dehydrated in graded sucrose solution. After being embedded in optimal cutting temperature (OCT) compound, 10-µm-thick frozen sections were cut sagittally passing through the optic disc with freezing microtome (Sakura, the Netherlands). Frozen sections were permeabilized with 0.1% Triton X-100/0.1% sodium citrate solution for 2 min on ice and then incubated with the terminal deoxynucleotidyl transferase dUTP nick end labeling reaction mixture (Sigma-Aldrich, St Louis, Missouri, USA) for 60 min at 37°C. After rinsing, sections were sealed with VECTASHIELD mounting medium containing 4′,6-diamidino-2-phenylindole and visualized under the fluorescence microscope.
Hematoxylin and eosin staining
Ten-µm-thick frozen sections were stained with hematoxylin and eosin 18 and visualized under an Olympus BX60 microscope (Olympus, Shinjuku, Japan). The thickness of the outer nuclear layer (ONL) was measured at 200 µm from the edge of the optic disc using ImageJ 1.48v software (National Institutes of Health, Bethesda, Maryland, USA).
SOD and catalase activities, MDA and GSH content measurement
Retinas were harvested after the ERG recording. The retinas were grinded with ReadyPrep Mini Grinders (1632146, Bio-Rad, Hercules, California, USA) in lysis buffer (ReadyPrep Protein Extraction Kit, 1632086, Bio-Rad). Cell lysate was used to determine the superoxide dismutase (SOD) and catalase activities and malondialdehyde (MDA) as well as glutathione (GSH) levels by enzyme-linked immunosorbent assay (ELISA) kits (Jiancheng Biological Engineering, Nanjing, China) according to the manufacturers’ instructions.
Cytochrome c assay
The concentration of cytochrome c in retinas was determined by the mouse cytochrome c immunoassay kit (R&D Systems, Minneapolis, Minnesota, USA). The optical density was measured on an ELISA plate reader with a wavelength of 490 nm.
Caspase activity measurement
The caspase 3 (Ac-DEVD-Amc, 390/475 nm) and caspase 9 (Ac-LEDH-Afc, 400/505 nm) activities in retinas were determined using the fluorescent assay kit (R&D systems) by a microplate reader, respectively, according to the manufacturer’s instruction.
Cytokine assays
Cytokine levels of interleukin (IL)-1β, IL-6, and tumor necrosis factor α (TNF-α) were measured by ELISA kits (Jiancheng Biological Engineering, Nanjing, China) according to the manufacturer’s instruction.
Nitric oxide measurement
Nitric oxide (NO) in cell lysate of retinas was measured by Griess assay (Sigma-Aldrich). 19
Real-time polymerase chain reaction
RNA was extracted from retinas after the ERG test with TRIzol using manufacturer’s protocol. RNA concentration was quantified by a spectrophotometer, and mRNA was reverse transcribed into cDNA with SuperScript master mix (Bio-Rad). Quantitative polymerase chain reaction was conducted using SYBR Green Supermix (Bio-Rad) with comparative C t value method to quantify the expression in different samples. The mRNA levels were normalized to that of a housekeeping gene β-actin. The gene-specific primer sequences are the following. For SOD, forward: ccgaggagaagtaccacgag, reverse: gaaccttggactcccacaga, and GenBank reference: Z18857.1; for Catalase, forward: ttgacagagagcggattcct, reverse: agctgagcctgactctccag, and GenBank reference: AK075853.1; for glucose-regulated protein 78 kDa (GRP78), forward: cgtatgtggccttcactcct, reverse: tttcttctggggcaaatgtc, and GenBank reference: D78645.1; for activating transcription factor 6 (ATF6), forward: acatgctcatgtggtttcca, reverse: agggctgaaccacaaatcac, and NCBI reference: NM_001081304.1; for inositol-requiring enzyme 1α (IRE1α), forward: gaatctggttttgcctgcat, reverse: tccacagcattgctaacgag, and GenBank reference: AB031332.1; for nitric oxide synthase (NOS), forward: ctcactgggacagcacagaa, reverse: gcttgtctctgggtcctctg, and GenBank reference: U43428.1; for IL-1β, forward: tgaaatgccaccttttgaca, reverse: tgtcctcatcctggaaggtc, and NCBI reference: NM_008361.4; for IL-6, forward: gagcccaccaagaacgatag, reverse: tccacgatttcccagagaac, and NCBI reference: NM_031168.2; for TNF-α, forward: gattatggctcagggtccaa, reverse: ctccctttgcagaactcagg, and NCBI reference: NM_013693.3; and for β-actin, forward: tgttaccaactgggacgaca, reverse: ggggtgttgaaggtctcaaa, and NCBI reference: NM_007393.5.
Western blot
Protein was extracted from retinas after the ERG test with radioimmunoprecipitation assay (RIPA) buffer (Sigma-Aldrich), supplemented with protease inhibitors (Roche, Basel, Switzerland) and phosphatase inhibitors (Thermo Scientific, Waltham, Massachusetts, USA). Protein was quantified with the bicinchoninic acid assay kit (Thermo Scientific, Waltham, MA, USA). Equal amounts of protein (40-μg per lane) were mixed with 4× loading buffer and subjected to sodium dodecyl sulfate polyacrylamide gel electrophoresis with 4–12% gel. Protein was transferred to polyvinylidene fluoride membranes, blocked for 1 h at room temperature in 1% bovine serum albumin, and then incubated with primary antibodies (anti-cleaved caspase 3, rabbit pAb, ab49822, 1:1000; anti-cleaved caspase 9, rabbit mAb, #9509, 1:1500; anti-Bcl-2, rabbit pAb, ab196495, 1:1000; anti-Bax, rabbit mAb, #14796, 1:2000; anti-nicotinamide adenine dinucleotide phosphate (NADPH) oxidase 4, rabbit mAb, ab195524, 1:1000; anti-GRP78, rabbit pAb, ab21685, 1:1000; anti-ATF6, rabbit pAb, ab37149, 1:1000; anti-p-IRE1α, rabbit pAb, ab48187, 1:1000; anti-p-P38, rabbit pAb, ab47363, 1:1000; anti-NF-κB (nuclear factor kappa-light-chain-enhancer of activated B cells) rabbit mAb, #8242, 1:1000; and anti-β-actin, rabbit pAb, ab16039,1:3000) overnight at 4°C. Bound antibodies were washed and incubated with the secondary antibody (goat anti-rabbit IgG H&L HRP, ab6721, 1:3000) for 1 h. Membranes were washed and exposed to PierceTM ECL substrates (Thermo Scientific), followed by the X-ray film development.
Statistical analysis
Data were expressed as mean ± SD and analyzed with SPSS 19.0 software (SPSS Inc., Chicago, Illinois, USA). Two-way repeated analysis of variance (ANOVA) was used to analyze the ERG data. All other comparisons were made by one-way ANOVA followed by Least Significant Difference (LSD) post hoc analysis. A value of p < 0.05 was considered as significant difference.
Results
Systematic treatment with THC caused retinal dysfunction and damage in BALB/c mice
ERG can be used to test the function of outer retina or monitor disease progression, because it reflects the mass response of photoreceptor cells. Our results showed that the amplitude of ERG significantly decreased after 2-month treatment with THC, indicating THC treatment caused retinal dysfunction, which was also evidenced by the decreased thickness of ONL (Figure 1).
THC treatment caused photoreceptor cell apoptosis
THC treatment significantly increased apoptotic cells in the ONL, together with increase of caspase activities and cytochrome c release (p < 0.01). Proapoptotic protein Bax and caspase expression increased but antiapoptotic protein Bcl-2 decreased after THC treatment (Figure 2).

THC treatment caused photoreceptor cell apoptosis. BALB/c mice were treated with THC at 1 or 2 mg/kg (n = 10) by intraperitoneal injection daily, mice treated with vehicle (n = 10) as negative control. Mice were euthanized after 2 months and retinas were harvested for the following assays. The red spots indicated the TUNEL-positive cells (×20) (a). THC treatment significantly increased DNA fragmentation (a), cytochrome c release (b), and caspase activities (c). THC treatment decreased antiapoptotic protein but increased proapoptotic protein expression (d). Data were expressed as mean ± SD. *p < 0.05, **p < 0.01 versus vehicle group. THC: tetrahydrocannabinol; TUNEL: Terminal deoxynucleotidyl transferase dUTP Nick End Labeling; ONL: outer nuclear layer; INL: inner nuclear layer; GLC: ganglion cells.
THC treatment increased oxidative stress in retinas
Compared to vehicle control, oxidative stress significantly increased after 2-month treatment with THC, evidenced by increased MDA level, decreased GSH level, SOD, and catalase activity, together with the decrease of mRNA expression of SOD and catalase, and the increase of NADPH oxidase 4 protein expression, suggesting THC caused retina damage through increasing oxidative stress (Figure 3).

THC treatment increased oxidative stress in retinas of BALB/c mice. BALB/c mice were treated with THC at 1 or 2 mg/kg (n = 10) by intraperitoneal injection daily, mice treated with vehicle (n = 10) as negative control. Mice were euthanized after 2 months. Retinas were harvested to measure the oxidative stress: MDA content, GSH level, and SOD activity (a); catalase activity (b); mRNA expression of SOD and Catalase (c) and oxidative stress-related protein expression (d). Data were expressed as mean ± SD. *p < 0.05, **p < 0.01 versus vehicle group. THC: tetrahydrocannabinol; MDA: malondialdehyde; GSH: glutathione; SOD: superoxide dismutase.
THC treatment increased endoplasmic reticulum stress in retinas
As a central regulator for endoplasmic reticulum (ER) stress due to its role as a major ER chaperone and its ability to control the activation of transmembrane ER stress sensors IRE1 and ATF6, a large number of studies established that induction of GRP78 is a marker for ER stress. 20 IRE1α and ATF6 are signaling proteins in ER stress. IRE1α stimulates activation of the apoptotic-signaling kinase-1, which activates downstream of stress kinases to promote apoptosis. As a transcriptional factor, ATF6 translocates to the Golgi compartment where it is cleaved upon ER stress. 21 ER stress markers were detected after THC treatment. Results showed that THC increased the mRNA and protein expression of GRP78, ATF6, and IRE1α (Figure 4).

THC treatment increased ER stress in retinas of BALB/c mice. BALB/c mice were treated with THC at 1 or 2 mg/kg (n = 10) by intraperitoneal injection daily, mice treated with vehicle (n = 10) as negative control. Mice were euthanized after 2 months and retinas were harvested for the following assays. ER stress was evaluated by examining ER stress-related proteins (GRP78, ATF6, and p-IRE1α) and their mRNA expression through Western blot (a) and real-time PCR (b). Data were expressed as mean ± SD. *p < 0.05, **p < 0.01 versus vehicle group. THC: tetrahydrocannabinol; ER: endoplasmic reticulum; GRP78: glucose-regulated protein 78 kDa; ATF6: activating transcription factor 6; IRE1α: inositol-requiring enzyme 1α; PCR: polymerase chain reaction.
THC treatment induced inflammatory responses in retinas
Production of NO and IL-1β, IL-6, and TNF-α in retinas of BALB/c mice significantly increased after 2-month treatment with THC. mRNA expression of NOS, IL-1β, IL-6, and TNF-α and protein expression of p-P38 and NF-κB in the BALB/c mouse retinas also increased after THC treatment (p < 0.01) (Figure 5).

THC treatment induced inflammatory responses in retinas of BALB/c mice. BALB/c mice were treated with THC at 1 or 2 mg/kg (n = 10) by intraperitoneal injection daily, mice treated with vehicle (n = 10) as negative control. Mice were euthanized after 2 months and retinas were harvested for the following assays. THC treatment promoted inflammatory responses, evidenced by the increase of pro-inflammatory factors: NO (a), IL-1β (b), IL-6 (c), and TNF-α (d). THC treatment increased mRNA expression of pro-inflammatory factors (e) and the protein expression of p-P38 and NF-κB (f). Data were expressed as mean ± SD. *p < 0.05, **p < 0.01 versus vehicle group. THC: tetrahydrocannabinol; NO: nitric oxide.
Discussion
Marijuana for recreational use has many negative health effects. The drug is addictive, with mounting evidence for the existence of a withdrawal syndrome. Furthermore, it has been shown to have adverse effects on mental health, intelligence (including irreversible declines in cognition), and the respiratory system. Medical marijuana should also be subject to the same rigorous approval process as other medications prescribed by physicians. 22 Impairments of learning and memory by cannabis abuse have been reported and much progress has been made in understanding its neurological consequence. 23,24 However, few studies have investigated the effects of cannabis abuse on retinas. 25 There is limited knowledge about the chronic toxicity induced by cannabis abuse and no effective treatment is available for this injury. After administration with THC at the same dosage (5 mg/kg), absorption after intraperitoneal (ip) route was rapid and plasma disappearance was slow, which produced approximately four to six times higher plasma concentration than that after per os or subcutaneous routes. Moreover, the brain disappearance of THC was slower after repeated administration than that after single treatment. 26 Evaluation of abuse liability of substances via ip administration is comparable to vapor inhalation. 27 This study was to investigate the effects of systemic administration with THC, the major component in cannabis, on mouse retinas and its underlying mechanisms. For the first time, we explored the effects of dosing and administration duration (up to 2 months) that reflect cannabis abuse situations, complementing studies into the mechanisms underlying retina damage caused by cannabis abuse.
Apoptosis was considered as the main pathway of cell death in retinal degeneration. 28,29 Cytochrome c release from the mitochondria to nucleus would activate caspase 9 to facilitate the formation of apoptosome complex. 30 As an apoptotic executor, caspase 3 activates DNA fragmentation factors, which in turn activate endonucleases to cleave nuclear DNA, ultimately resulting in cell death. 31 Our data showed that apoptotic cells significantly increased after THC treatment and this is the first report that apoptosis was induced in retinas after 2-month of daily THC systematic administration. Furthermore, THC treatment increased cytochrome c release and caspase 3/caspase 9 activities. Antiapoptotic protein expression decreased but proapoptotic protein expression increased.
Oxidative stress plays a pivotal role in the mode of action of “ecstasy” through affecting various biological macromolecules and cellular function. 32,33 A high content of polyunsaturated fatty acids are found in retinas, which is particularly susceptible to ROS. 34
MDA is well-known as a widely used marker for oxidative damage and resultant thiobarbituric acid reactive substances, which are proportional to lipid peroxidation and oxidative stress. 35 Importantly, intracellular antioxidants limiting oxidative damage caused by ROS, SOD, and GSH constitute a first-line defense system in cells. The depletion of GSH and the decreased activities of SOD compromise cellular defense pathway. In this study, THC treatment resulted in the depletion of GSH and decrease of SOD activity, together with markedly increase of MDA and protein expression of NADPH oxidase 4, a major enzyme responsible for the production of superoxide by transferring electrons across the membrane from NAD(P)H to molecular oxygen, 36 in retinas of mice, suggesting THC systematic treatment caused oxidative stress.
Protein misfolding in the ER causes the pathogenesis of many diseases, and activation of the unfolded protein response leads to oxidative stress and resultantly apoptosis. 37 ER stress has been established as a pathogenic factor contributing to photoreceptor cell death. 38 Chronic ER stress promotes inflammatory responses through activating NF-κB and mitogen-activated protein kinase (MAPK) pathway to induce autophagy and apoptosis. 39 –41 In this study, THC treatment upregulated the expression of GRP78, ATF6, and IRE1α, indicating THC at least partially caused retinal damage through inducing ER stress.
Inflammatory response has been considered as a possible factor in the pathogenesis of retinal degeneration. During chronic inflammation, intracellular responsive cascades such as NF-κB and MAPKs become activated, increasing expression of pro-inflammatory products, including IL-1β, IL-6, TNF-α, and COX-2, as well as genes responsible for regulating cell survival and growth. 42,43 Overproduction of pro-inflammatory factors stimulates the production of oxidants with subsequent peroxidative damage to biological macromolecules, causing intracellular toxic events. 44 Studies have demonstrated that the levels of pro-inflammatory cytokines and chemokines were substantially increased in the retinas of retina-degenerated mice and these events preceded the photoreceptor cell loss. 45,46 NF-κB has been considered as a prototypical pro-inflammatory signaling pathway. 47 Additionally, the p38 MAPK pathway is associated with several inflammatory diseases such as Alzheimer’s disease, rheumatoid arthritis, and inflammatory bowel disease. 48 Our results indicated that THC treatment increased the gene expression of NOS, IL-1β, IL-6, and TNF-α and, consequently, increased their levels in the retinas, through activating MAPK and NF-κB pathways.
In conclusion, our data demonstrated that systemic administration with THC caused retina damage in BALB/c mice through promoting inflammatory response and increasing oxidative stress, providing scientific rationale for the retina damage caused by the cannabis abuse and potential for harm to many more users.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Health and Family Planning Commission of Heilongjiang province (Grant No. 2016-014).
