Abstract
Trichloroethylene (TCE) is a major occupational hazard and environmental contaminant that can cause multisystem disorders in the form of occupational medicamentosa-like dermatitis. Development of dermatitis involves several proinflammatory cytokines, but their role in TCE-mediated dermatitis has not been examined in a well-defined experimental model. In addition, few animal models of TCE sensitization are available, and the current guinea pig model has apparent limitations. This study aimed to establish a model of TCE-induced skin sensitization in BALB/c mice and to examine the role of several key inflammatory cytokines on TCE sensitization. The sensitization rate of dorsal painted group was 38.3%. Skin edema and erythema occurred in TCE-sensitized groups, as seen in 2,4-dinitrochlorobenzene (DNCB) positive control. Trichloroethylene sensitization-positive (dermatitis [+]) group exhibited increased thickness of epidermis, inflammatory cell infiltration, swelling, and necrosis in dermis and around hair follicle, but ear painted group did not show these histological changes. The concentrations of serum proinflammatory cytokines including tumor necrosis factor (TNF)-α, interferon (IFN)-γ, and interleukin (IL)-2 were significantly increased in 24, 48, and 72 hours dermatitis [+] groups treated with TCE and peaked at 72 hours. Deposition of TNF-α, IFN-γ, and IL-2 into the skin tissue was also revealed by immunohistochemistry. We have established a new animal model of skin sensitization induced by repeated TCE stimulations, and we provide the first evidence that key proinflammatory cytokines including TNF-α, IFN-γ, and IL-2 play an important role in the process of TCE sensitization.
Introduction
Trichloroethylene (TCE) is a volatile organic compound widely used as an industrial solvent for about a century. It is commonly used for cleaning metal parts and cold cleaning of other materials in some of the industrial processes, as it is an effective grease remover. 1 Due to its widespread commercial use and improper waste disposal, TCE has become a major occupational hazard and environmental pollutant found in air emissions, wastewater, and soil. It has been estimated to be present in about one-third of municipal water supplies in the United States. 2 In China, most of TCE is used in the coastal industrialized regions, and the total annual volume used remains high. 3
Exposure to TCE has been reported to result in several adverse health effects in occupationally exposed workers and animal models, including epidermolysis bullosa, exfoliative dermatitis, multiforme erythema, and Steven-Johnson syndrome, with some patients exhibiting damage of liver and kidney. 4 Due to characteristics of the skin lesions similar to drug eruption, these disorders of skin lesions have been classified as “occupational medicamentosa-like dermatitis (OMLDT) induced by TCE” by the Chinese National Diagnostic Criteria. 5 Dermatologists and occupational health physicians classify TCE-induced OMLDT as type IV allergy. Based on epidemiological and clinical features and similarity to drug-induced hypersensitivity syndrome, TCE-induced OMLDT is also known as a delayed-type hypersensitivity (DTH) reaction that depends on the T-cell-mediated immune response. 6,7 The initiation and maintenance of skin inflammation directly involves a number of proinflammatory cytokines such as tumor necrosis factor (TNF) α, interleukin (IL) 2, and interferon (IFN) γ. These are signaling molecules secreted by immune cells and are important in immune cell recruitment and regulation of inflammatory responses, including maintaining homeostasis between cell-mediated and humoral immune responses.
Increasing evidence demonstrates that allergens and irritants can directly stimulate keratinocytes (KCs) to produce several types of cytokines that are responsible for the inflammatory reactions in skin. These cytokines include TNF-α, IFN-γ, IL-1α, IL-3, IL-6, IL-8, granulocyte macrophage colony-stimulating factor (CSF), and macrophage CSF. 8 –10 There is evidence that TCE alters the level of various key cytokines, including IL-2, IFN-γ, and other factors, in the mice with spontaneous autoimmune diseases 1,11 and in occupationally TCE-exposed workers. 12 It has been reported that TNF-α was associated with TCE-induced hypersensitivity dermatitis in humans. 13 Interleukin 2, a cytokine involved in lymphocyte proliferation, 14 activates an inflammatory cytokine cascade in the epidermal immune system. 15 Interferon-γ is a cytokine that is critical for innate and adaptive immunity against viral and intracellular bacterial infections and for antitumor effect. 16 However, the role of these cytokines in contributing to the hypersensitivity has not been demonstrated in a well-defined experimental model of skin sensitization in response to TCE. In recent years, there has been growing research on TCE and the application of animal models of allergic reaction by TCE. The guinea pig sensitization/maximization test has been the best established animal model. 17,18 The model is easy to use and is reliable for identifying a skin sensitizer and type IV immune response. However, the guinea pig model of skin sensitization induced by TCE is greatly limited for pathogenesis investigation due to the lack of guinea pig-specific antibodies and related bioreagents. More importantly, recent advance has revealed immunological-related systemic lesions beyond the skin reaction, including liver and kidney. 3,19 The need to gain molecular insight with physiological relevance necessitates the use of a in vivo animal model with well-characterized genetics and readily available genetically modified phenotypes which the guinea pig models can no longer provide. In this study, we established a model of skin sensitization in BALB/c mice. Using this model, we have also explored the expression of proinflammatory cytokines in order to provide new insight into the immunological mechanisms of TCE sensitization.
Materials and Methods
Animals
One hundred and five healthy BALB/c female mice (aged 6-8 weeks) were provided by Hunan Slac Jingda Laboratory Animal Co, Ltd (Hunan, China). All animals were housed in 6 per cage at 20°C and maintained in a 12 hour/12 hour day/night cycle (day: 07.00-19.00). All mice received tap water and a standard diet ad libitum. After a week of acclimation, animals were randomly divided into untreated control group, solvent (olive oil and acetone) control group, TCE treatment group, and sensitization-positive (DNCB) group. To compare the effect of the back painting and the ear swelling test, we divided TCE-treated mice into 2 subgroups: TCE was applied on the back of the animals (dorsal painted group) or TCE was applied on the right ear (ear painted group). The use of animals and the experimental protocol were approved by the Animal Care and Use Committee of Anhui Medicine University.
Chemicals
Trichloroethylene and Freund complete adjuvant (FCA) were purchased from Sigma (St Louis, Missouri). Olive oil (vehicle for TCE) was provided by Shanghai Chemical Reagent Company. Filter paper and nonirritating tape were purchased from XinHua (Beijing, China). Histostain plus kit and 3, 3′-diaminobenzidine (DAB) substrate kit were purchased from ZSJQ (Beijing, China). Anti-TNF-α, anti-IL-2, and anti-IFN-γ antibodies were obtained from Abcam (Cambridge, Massachusetts). Mouse TNF-α, mouse IFN-γ, and mouse IL-2 enzyme-linked immunosorbent assay (ELISA) kits were purchased from Uscnlife (Wuhan, China).
Establishment of TCE-Induced Skin Sensitization Model in BALB/c Mice
We established a model of TCE sensitization in BALB/c female mice by reference to guinea pig maximization test (GPMT) model and Naoki Inagaki mouse allergic dermatitis model 17 –20 (Figure 1). Female mice appeared to be more sensitive than male mice. 21 Twenty-four hours before the intradermal injection, an area of 2 × 2 cm on the dorsalis of the mice was shaved with an electric clipper. On the first day, BALB/c mice in TCE sensitization group were given intradermal injection of 100 µL mixture of 50% TCE (TCE:olive oil:acetone = 5:2:3) and 50% FCA for the first stimulation. A 100 µL of 50% TCE (the same proportions of TCE, olive oil, and acetone but without FCA) was then painted on the shaved area of the skin for continued sensitization on the 4th, 7th, and 10th day. A week later, 100 µL of 30% TCE (TCE:olive oil:acetone = 3:2:5) was painted twice to challenge the dorsalis skin or the right ear on 17th and 19th day. Each animal in TCE treatment group after being painted with TCE, the shaved area was covered with filter paper and sealed with nonirritating tape for 24 hours. In the solvent control group, mice were treated similarly with the same proportions of olive oil and acetone but without TCE and also covered with filter paper and tape for 24 hours. The positive group followed the same procedure using DNCB instead of TCE: 0.25% DNCB for the first sensitization and challenge and 0.5% DNCB for continued sensitization. The untreated control group received no treatment. Twenty-four hours after the last challenge (study day 20), the cutaneous reactions in the mice dorsalis skin were photographed, and the thickness of the ear was measured by Vernier caliper and scored on a 4-point scale: 0, no reaction; 1, scattered mild redness; 2, moderate and diffuse redness; and 3, intensive erythema and swelling. 19,22 If the cutaneous reaction score of any animal was equal to 1 or greater, it would be judged as a positive reaction and classified as sensitization positive or otherwise as sensitization negative. Frequency of sensitization and percentage of sensitized animals were calculated. Groups of mice were sacrificed on the first day (study day 20), the second day (study day 21), the third day (study day 22), and the seventh day (study day 26) after the last challenge, respectively.
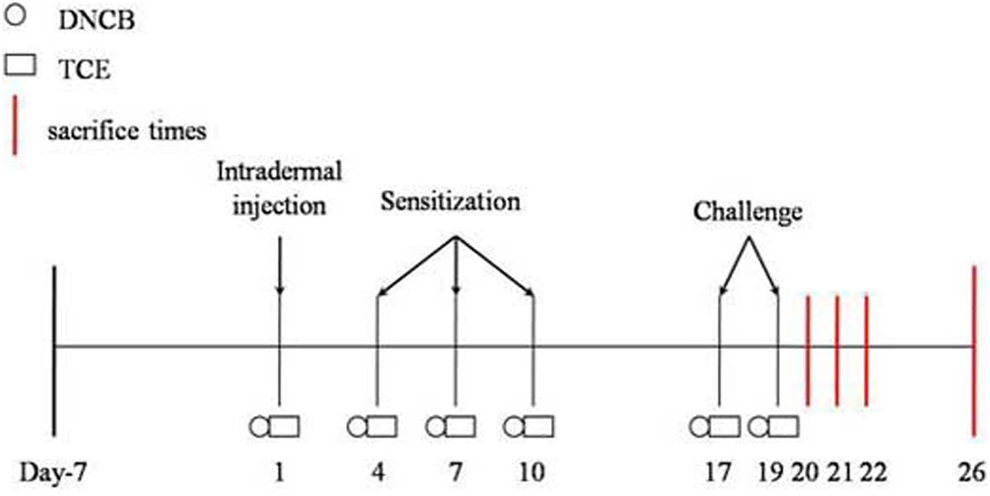
Experimental schedule for the induction of sensitization in BALB/c mouse model by trichloroethylene (TCE) and DNCB. After 7 days of acclimation, DNCB and TCE were applied to the back of animals by intradermal injection. After 3 days, DNCB and TCE were topically painted on the back of mice, repeated for a total of 3 times, with a 3-day interval. One week later, animals were challenged with DNCB and TCE twice, 3 days apart. Animals were sacrificed and tissues were taken at 20th , 21st , 22nd , and 25th days.
Histological Examination
The shaved area of the all mice skin tissues was excised and fixed in 10% neutral-buffered formalin for at least 48 hours and then embedded into paraffin blocks. The tissue was sectioned in 5 mm and stained with hematoxylin and eosin according to the routine protocols for histopathological examination, and the thickness of epidermis was measured under the microscope.
Enzyme-Linked Immunosorbent Assay
One day, 2, 3, and 7 days after the last challenge, blood samples were collected from mice and then centrifuged at 3000 rpm for 15 minutes at 4°C to separate the serum. The concentrations of TNF-α, IFN-γ, and IL-2 in serum were determined by commercially available ELISA kits per manufacturer’s instructions. Briefly, 10 µL serum and 40 µL sample diluent were applied into each testing well. Then, 100 µL of horse raddish peroxidase conjugate reagent was added to each well and incubated for 60 minutes at 37°C. Then after the plate was washed 5 times, 50µL chromogen solution A and 50µL chromogen solution B were added to each well, and the plate was incubated in the dark for 15 minutes at 37°C. As the last step, 50 µL of stop solution was added into each well, and the optical density (OD) was read at 450 nm immediately. The standard curve from the OD values was used to calculate the contents of TNF-α, IFN-γ, and IL-2.
Skin Immunohistochemistry
Skin tissue sections of 5 mm were deparaffined and rehydrated through graded xylene and alcohol series. Endogenous peroxidase was inhibited by 3% H2O2 for 30 minutes. The slides were dipped in 0.01 mol/L sodium citrate buffer for antigen retrieval in a microwave oven. The sections were incubated with polyclonal rabbit anti-TNF-α, anti-IFN-γ, and anti-IL-2 antibodies overnight at 4°C after blocking nonspecific binding with 5% normal goat serum in phosphate-buffered saline for 15 minutes. In the morning of the next day, the sections were processed with Histostain Plus kit and DAB substrate kit for peroxidase reactions. The semiquantitative scoring of immunoreaction depositions took into account not only the area but also the intensity of staining in the tissue. The grading of immunoreaction intensity was estimated with 4 standard grades: 0, no positive reaction; 1, light brown; 2, moderate brown; and 3, dark brown. The grading of immunoreaction deposition in the skin was based on a semiquantitative assessment of deposition area with the 5 standard assigned grades as follows: 0, the area of positive reaction was less than 5%; 1, less than 25%; 2, less than 50%; 3, less than 75%; and 4, more than 75%. The scores of immunoreactions were calculated by the following formula: mean scores = score of positive reaction area + score of reaction intensity. Five tissue sections were selected in each group to assess the intensity of deposition. Slides were coded so that the counter was blind to the identity of the slides. 3 Biopsy scores were estimated by 3 pathologists, respectively.
Statistical Analysis
Throughout the text, data were presented as mean ± standard deviation. All statistical analysis was performed using SPSS 11.0 software package (IBM SPSS Statistics). The comparison of each intervention group with the appropriate control was made with 1-way analysis of variance followed by post hoc least significance test and Dunnett test. P < .05 was considered statistically significant.
Results
Sensitization Rate of BALB/c Mice From the Model
During the experiment, there were no animal deaths. The animals were then divided into dermatitis [+] group and sensitization-negative (dermatitis [–]) group according to the score of cutaneous reactions. The sensitization rates and scores of BALB/c mice are shown in Table 1. The sensitization rate in the dorsal painted group was 38.3% and 100% in the DNCB challenge group. However, no sensitization responses were observed in mice in the ear painted group. The animals showing swelling as well as redness on the skin induced by the TCE challenge or the DNCB challenge were compared with the control group. There was no swelling or redness in the ears to which TCE was applied. These results suggest that TCE painting on the dorsal site was effective, and the ear swelling test was ineffective. We therefore chose the dorsal painted group to perform the subsequent experiments.
Sensitization Rate and Scores of BALB/c Mice in the Model.
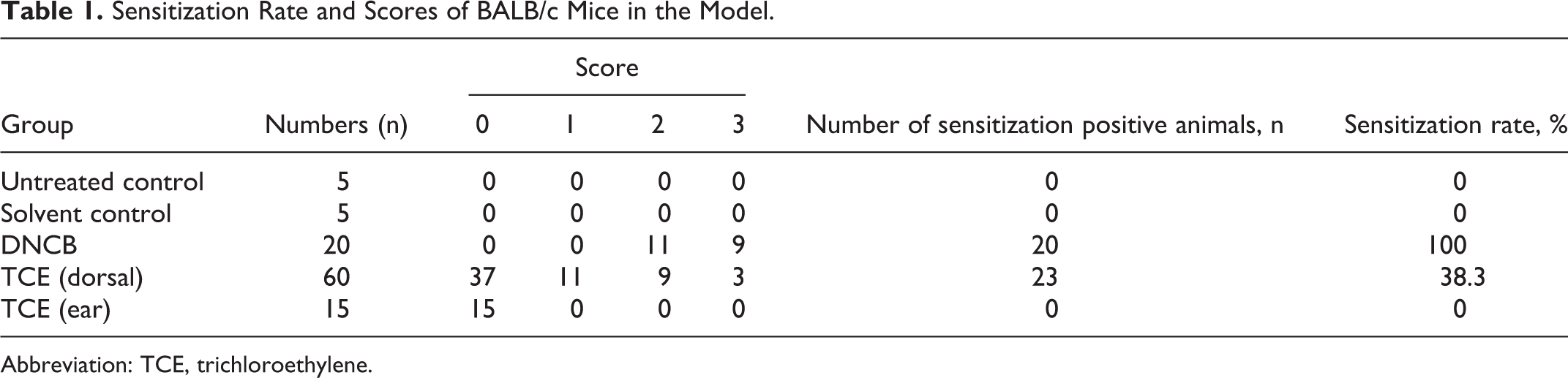
Abbreviation: TCE, trichloroethylene.
Histopathological Results
Histological examination of the skin from untreated control group and solvent control group showed no obvious abnormalities (Figure 2A and B). However, the TCE dermatitis [+] group, especially at 24-hour, 48-hour, and 72-hour, and the positive (DNCB) group showed clearly increasing thickness of epidermis and inflammatory cell infiltration and many neutrophilic granulocytes (Figure 2C and D). Swelling and necrosis were found in dermis and around hair follicle. Some inflammatory infiltrates were observed in the subcutaneous loose connective tissue and adipose tissue, especially capillary vessels (Figure 2A-E). The thickness of epidermis in TCE dermatitis [+] group and the positive control (DNCB) group was significantly greater than that in untreated control group, solvent control group, and TCE dermatitis [–] group (P < .05, Figure 2F), which suggests sensitization-specific histological changes.
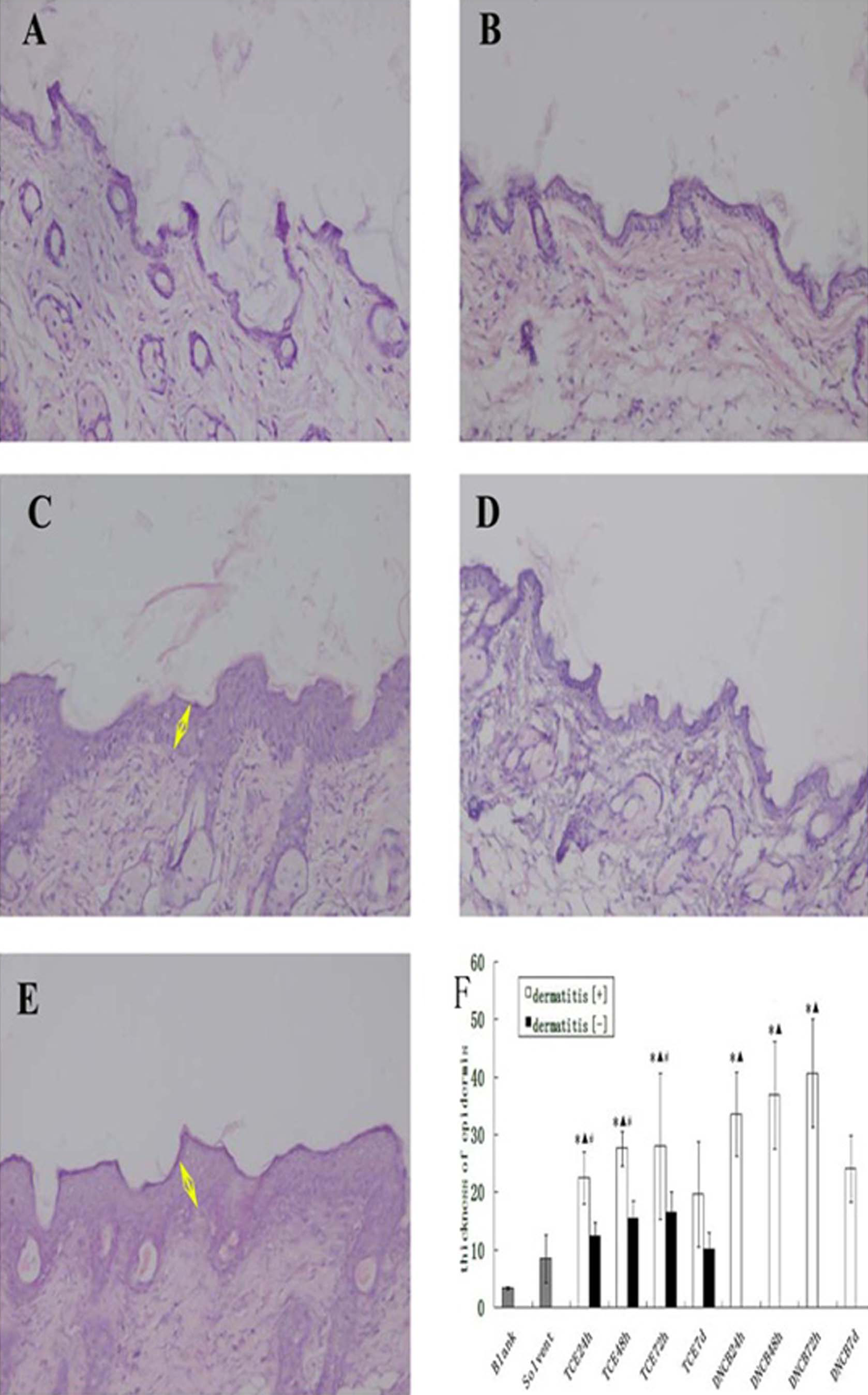
Photomicrographs of skin histopathological sections from BALB/c mice of the model in different groups, stained with hematoxylin–eosin and quantification of epidermis thickness in different groups of the model under light microscopy. (A) Untreated control group and (B) solvent control group: no abnormality; (C) trichloroethylene (TCE) sensitization-positive (dermatitis [+]) group and (E) positive (DNCB) group: increased thickness of epidermis, inflammatory cell infiltration, swelling, and necrosis in dermis and around hair follicle; (D) TCE sensitization-negative (dermatitis [–]) group: a small degree of inflammatory cell infiltration, swelling, and necrosis. (F) Significantly increased thickness of epidermis in TCE dermatitis [+] 1-day, 2-day, and 3-day groups and positive (DNCB) group. Data are shown as mean ± SD. *P< .05, compared with untreated control group. ▴ P< .05 compared with the solvent control group. # P< .05, compared with the corresponding sensitization-negative group. SD indicates standard deviation; yellow arrow, epidermis thickness (magnification ×200).
Serum TNF-α, IFN-γ, and IL-2 Concentrations
Compared to the untreated control group and solvent control group, the concentrations of TNF-α, IFN-γ, and IL-2 were significantly higher in 24-hour, 48-hour, and 72-hour dermatitis [+] groups and positive control (DNCB) group and peaked at 72 hours. The levels of TNF-α, IFN-γ, and IL-2 in the 72-hour dermatitis [+] group were slightly higher than those from the untreated control group, solvent control group, and 72-hour dermatitis [–] group. There was also statistically significant difference between TCE-sensitized positive groups and TCE-sensitized negative groups (Figure 3), suggesting that elevations in these cytokines are sensitization specific but not a simple tissue lesion due to a cytotoxic effect of TCE.
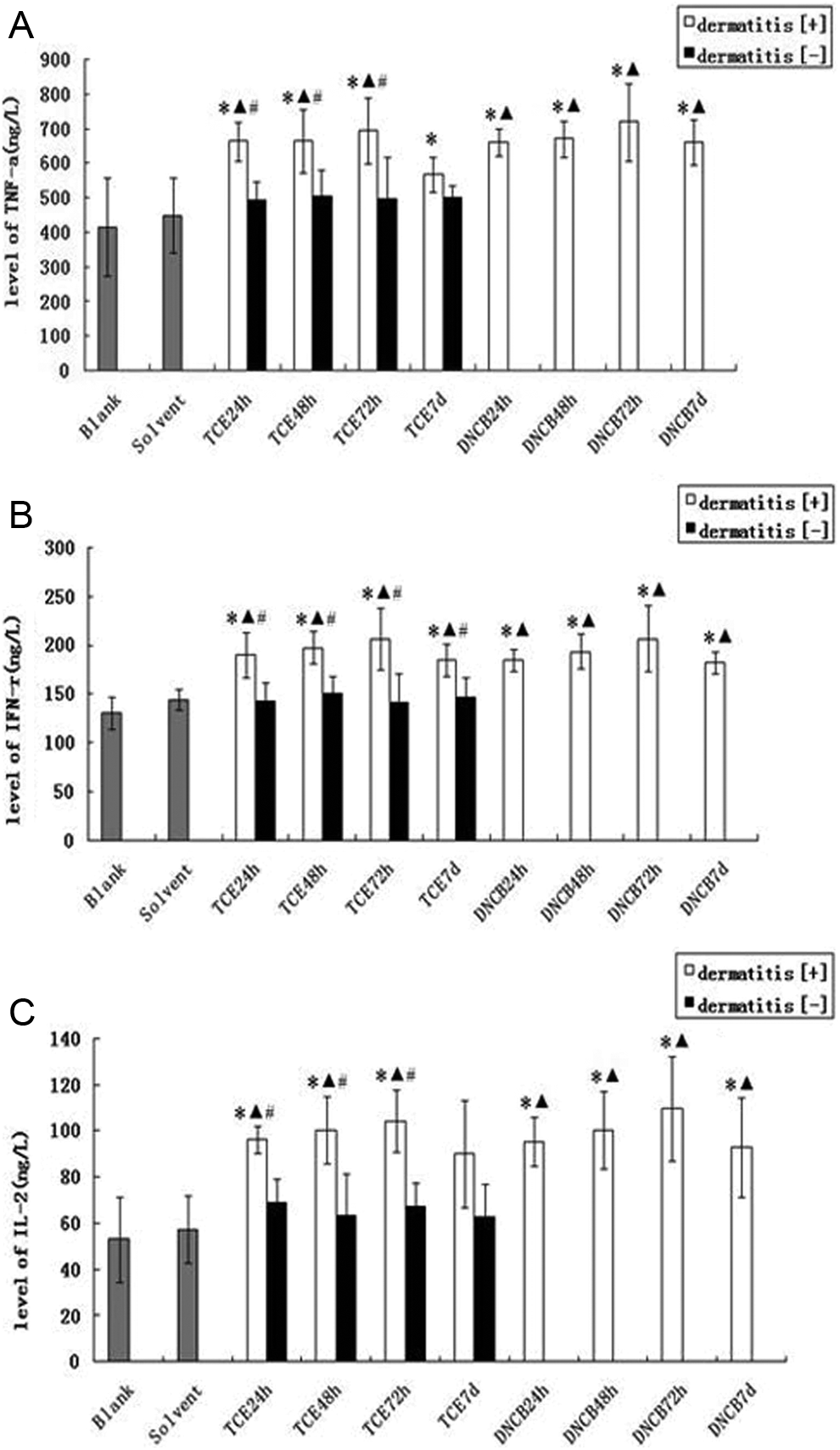
Serum levels of TNF-α, IFN-γ, and IL-2. A, B, and C represent concentrations of TNF-α, IFN-γ, and IL-2 in serum, respectively. Data are shown as mean ± SD. *P < .05, compared with untreated control group. ▴ P < .05 compared with the solvent control group. # P < .05, compared with the corresponding sensitization-negative group. SD indicates standard deviation; TNF, tumor necrosis factor; IL, interleukin; IFN, interferon.
Deposition of TNF-α, IFN-γ, and IL-2
The local expression of TNF-α, IFN-γ, and IL-2 in the skin was also examined. The mean scores of the deposition of TNF-α, IFN-γ, and IL-2 in skin tissue from different experimental groups are summarized in Table 2. Representative micrographs are shown in Figures 4 to 6. There were no immunoreactant depositions in untreated control group and solvent control group. As shown in Table 2, TNF-α, IFN-γ, and IL-2 depositions were increased significantly (P < .05) in 24-hour, 48-hour, and 72-hour dermatitis [+] groups and peaked in the 72-hour dermatitis [+] group (mean score: 3.91 ± 1.18, 3.83 ± 0.88, and 4.46 ± 0.66, respectively). These changes were absent in the dermatitis [–] groups but manifest more pronounced in the positive control (DNCB) group. These data further suggest that classic skin sensitizer DNCB produced consistent deposition of these proinflammatory cytokines into the skin tissue in this BALB/c mice model. This effect was also observed following TCE sensitization, suggesting that these cytokines were involved in TCE-induced sensitization. The lack of effect in TCE dermatitis [–] mice indicates again that this effect was via sensitization, not a direct cytotoxic effect of TCE.
Score of TNF-α, IFN-γ, and IL-2 on Skin Tissue Paraffin Sections From BALB/c Mice of the Model (TCE Dorsal Painted Group).
Abbreviations: SD, standard deviation; TCE, trichloroethylene; TNF, tumor necrosis factor; IL, interleukin; IFN, interferon; dermatitis[+], sensitization-positive; dermatitis[−], sensitization-negative.
a P < .05, compared with untreated control group.
b P < .05 compared with the solvent control group.
c P < .05, compared with the corresponding sensitization-negative group.
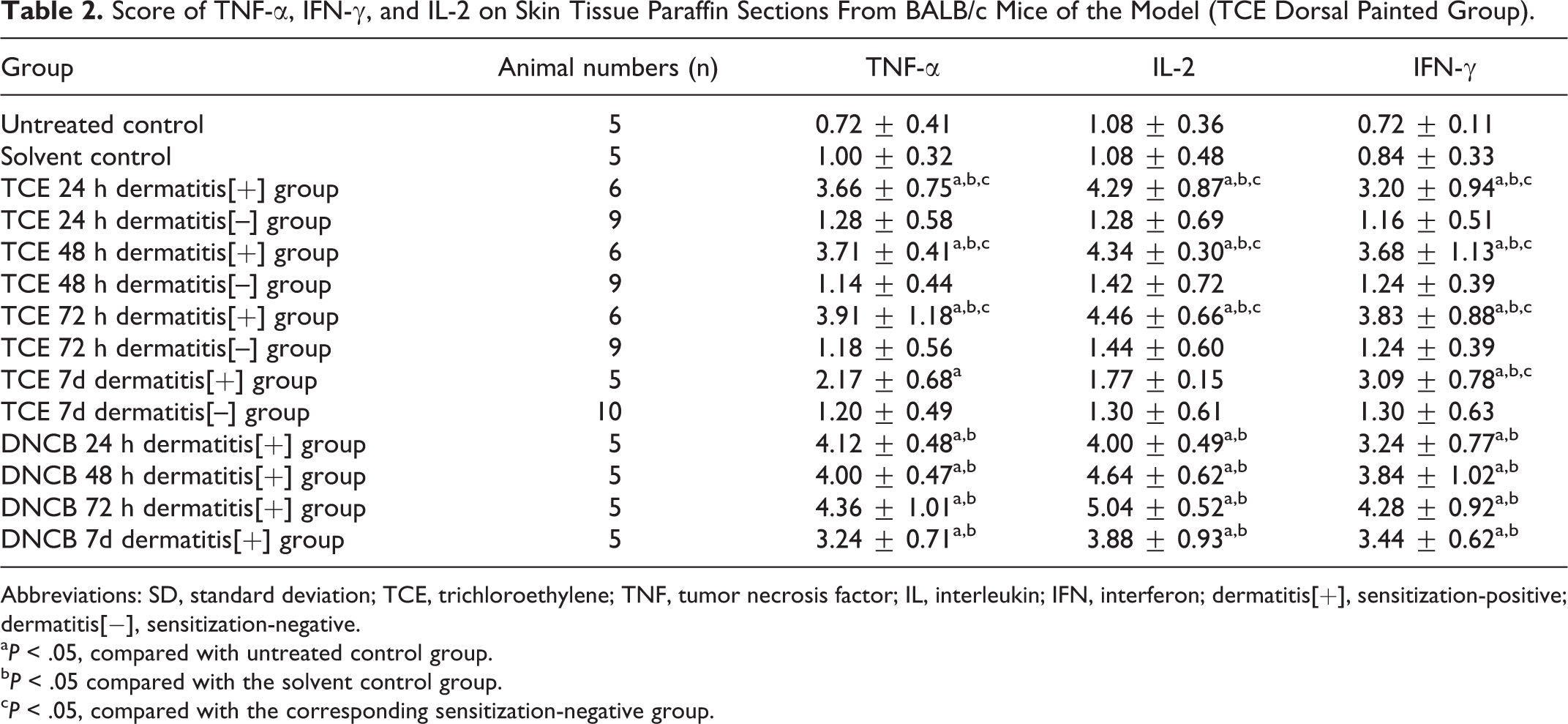
Deposition of tumor necrosis factor (TNF) α in skin by immunohistochemistry detected with DAB kit. (A) Untreated control group and (B) solvent control group: no deposition of TNF-α; (C) 24-hour trichloroethylene (TCE) sensitization-positive group and (D) 24-hour DNCB sensitization-positive group, apparent TNF-α deposition; (E) 48-hour TCE sensitization-positive group and (F) 48-hour DNCB sensitization-positive group: more TNF-α deposition; (G) 72-hour TCE sensitization-positive group and (H) 72-hour DNCB sensitization-positive group: strong deposition of TNF-α compared with untreated control and solvent control group; (I) 7-day TCE sensitization-positive group; and (J) 7-day DNCB sensitization-positive group: reduced deposition of TNF-α (magnification ×200).
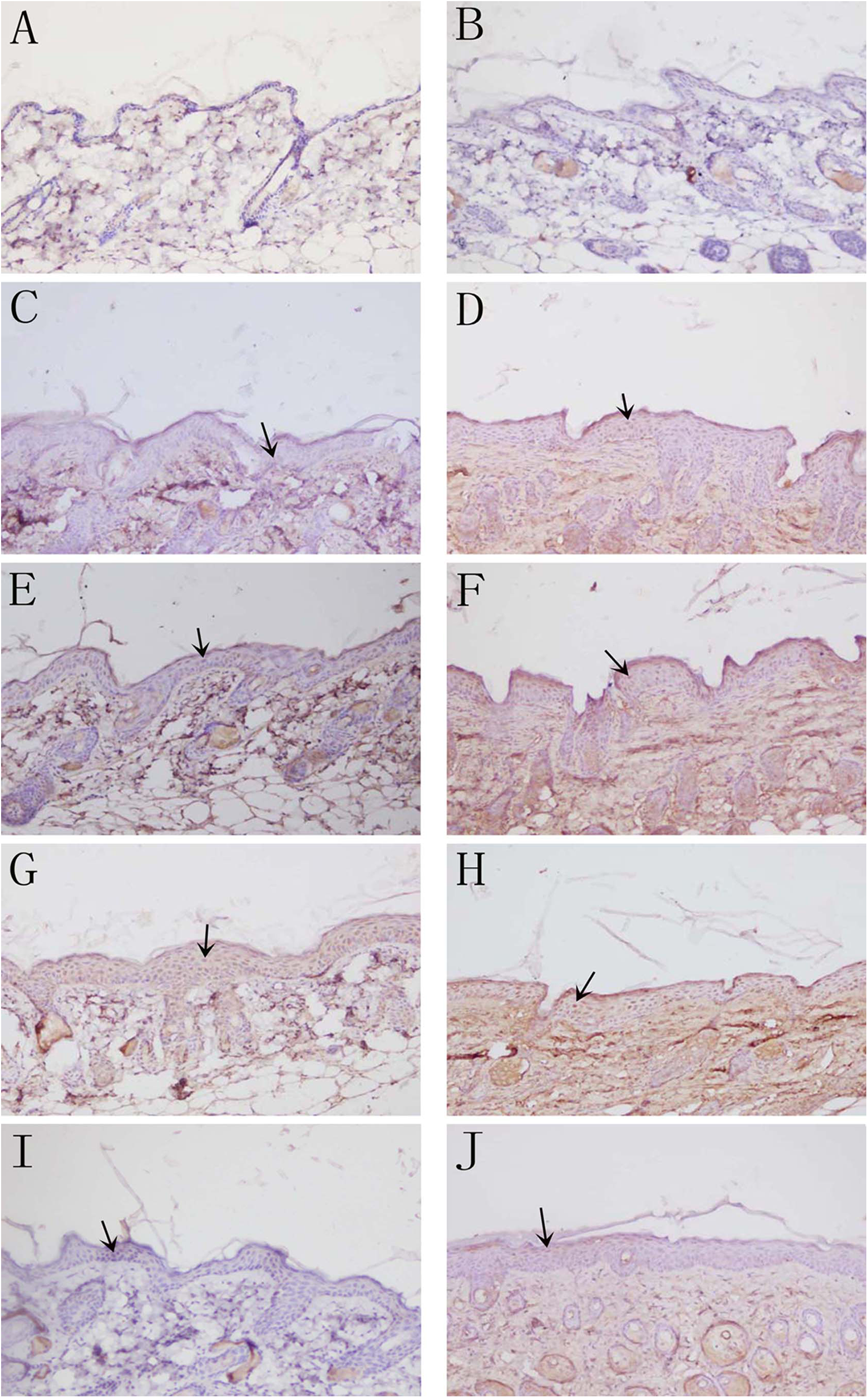
Deposition of interferon (IFN)-γ in skin by immunohistochemistry detected with DAB kit. (A) Untreated control group and (B) solvent control group: no deposition of IFN-γ; (C) 24-hour trichloroethylene (TCE) sensitization-positive group and (D) 24-hour DNCB sensitization-positive group, apparent IFN-γ deposition; (E) 48-hour TCE sensitization-positive group and (F) 48-hour DNCB sensitization-positive group: more IFN-γ deposition; (G) 72-hour TCE sensitization-positive group and (H) 72-hour DNCB sensitization-positive group: strong deposition of IFN-γ compared with untreated control and solvent control group; (I) 7-day TCE sensitization-positive group; and (J) 7-day DNCB sensitization-positive group: less deposition of IFN-γ; (magnification ×200).
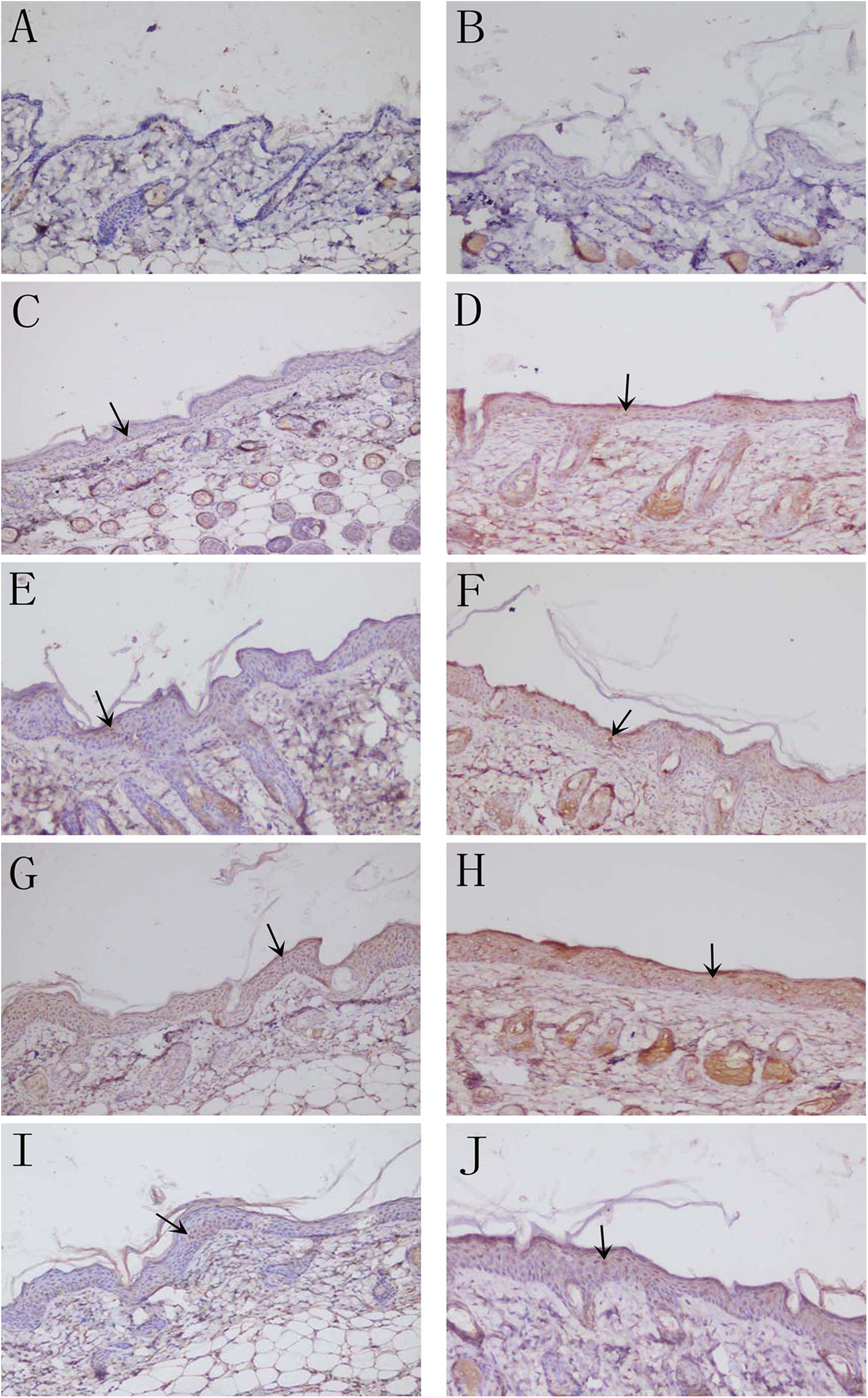
Deposition of interleukin (IL) 2 in skin by immunohistochemistry detected with DAB kit. (A) Untreated control group and (B) solvent control group: no deposition of IL-2; (C) 24-hour trichloroethylene (TCE) sensitization-positive group and (D) 24-hour DNCB sensitization-positive group, apparent IL-2 deposition; (E) 48-hour TCE sensitization-positive group and (F) 48-hour DNCB sensitization-positive group: more IL-2 deposition; (G) 72-hour TCE sensitization-positive group and (H) 72-hour DNCB sensitization-positive group: strong deposition of IL-2 compared with untreated control and solvent control group; (I) 7-day TCE sensitization-positive group; and (J) 7-day DNCB sensitization-positive group: decreased deposition of IL-2; (magnification ×200).
Discussion
We have successfully established a mouse model of skin sensitization by repeated skin challenges with TCE as evidenced by gross pathological evaluation and histological examinations, validated by both positive control and negative control. To our knowledge, this is a practical model of skin sensitization by TCE in mice. Mice have well-characterized genetic background, and genetically-modified models are readily available, and many molecular research tools have been developed for this species. Establishment of a mouse model of TCE sensitization thus overcomes several limitations of guinea pig sensitization model and permits exploration of molecular mechanisms underlying TCE-mediated immunotoxicity.
Trichloroethylene is a widely used solvent in several industrial processes, and the associated health hazard is an important public health issue. Data on TCE over the years show that the routes for TCE exposure are mainly through the respiratory tract and skin, and the OMLDT is one of major health hazards. Most researchers in occupational medicine categorize the pathogenesis of OMLDT as type IV allergy, also known as a DTH reaction and allergic contact dermatitis (ACD) that depends on the T-cell-mediated immune response. With the increasing number of OMLDT, TCE has attracted increasing attention and hence stimulated many studies on TCE-induced OMLDT including both human studies and animal models. In many animal models, most allergens and irritants were repeatedly injected intraperitoneally or painted the ear or toe, which are different from routes of entry for TCE in humans. Therefore, we used one intradermal injection at the dorsum of the mice and intermittently painted the skin by 2 different concentrations of TCE which was closer to the human route of TCE absorption. Furthermore, we compared the difference between the painted ears and the painted dorsalis. The results showed that the sensitization rate of BALB/c mice was 38.3% in the dorsal painted group, but there were no sensitized mice in the ear painted group. These observations suggest that the use of dorsalis skin is more effective than the use of the ear as a site for sensitization.
We have achieved a sensitization rate of 38.3% in the dorsal painted group. Although the rate was not as high as 60% sensitization rate of GPMT in guinea pigs in our previous studies, 3,23 the nature of sensitization was consistent with GPMT. Our study also showed that simply painting the TCE on the ear did not cause swelling and/or redness. Furthermore, histological changes in the skin by painting TCE on the dorsal area (Figure 2) were similar to those for the GPMT. 23 The thickness of epidermis in TCE dermatitis [+] group was significantly increased, as seen in positive (DNCB) mice, further indicating the success of the sensitization model (Figure 3).
The process of allergic response is regulated by a complex network of numerous cytokines of which the proinflammatory cytokines TNF-α, IL-2, and IFN-γ and their effects in immunological inflammatory response have attracted particular attention. Tumor necrosis factor α is a key cytokine with a variety of biological activity in vivo, and is an important mediator of immunological and inflammatory reactions. 24 Specifically, it may play a crucial role during contact allergic reactions 25 DTH and the susceptibility of ACD. 26 Tumor necrosis factor α amplifies inflammation by stimulating resident skin cells to synthesize chemokines that attract inflammatory cells and membrane molecules important for the retention and activation of T cells. The expression levels of proinflammatory cytokines TNF-α and IFN-γ are positively correlated. 27 Interferon-γ is the molecular basis of DTH and plays an important role in the pathogenesis of ACD by its effects on antigen presentation, enhancing the cytotoxic effect of T cells and inducing the production of inflammatory cytokines. 28 Iavicoli et al 12 reported that TCE-exposed workers had higher IL-2 and IFN-γ levels compared to TCE nonexposed workers in the printing area of the same factory. Interleukin 2 is a broad-spectrum cytokine and has an important regulatory role in humoral immunity and cell immunity. Interleukin 2 promotes T cell-mediated immune responses and induces the activity of regulatory T cells. 29 In addition, IL-2 also plays a major role in maturation/development of lymphocytes and monocytes and stimulates natural killer cells to secrete IFN-γ, 30 which in turn promotes macrophage activation and T-cell differentiation. 31 Of importance for understanding TCE sensitization, we have obtained the first experimental evidence that cytokines TNF-α, IFN-γ, and IL-2 are increased following TCE sensitization. The concentrations of TNF-α, IFN-γ, and IL-2 in TCE dermatitis [+] group were significantly higher than those from the untreated control group and solvent control group, as seen in positive (DNCB) control group. Consistently, TNF-α, IFN-γ, and IL-2 were also deposited into the skin in TCE dermatitis [+] mice, indicating the involvement of skin KCs in releasing these cytokines. The upregulation of the key proinflammatory cytokines is sensitization specific, as this was not seen in TCE-treated but dermatitis [−] mice. The consistently increased expression of these cytokines in both serum and skin tissue supports the notion that TEC-induced sensitization and cytokines are involved in local immune responses. Thus, these cytokines are key mediators in TCE-induced immune response.
These data are also consistent with our previous observations on GPMT, in which the expression of TNF-α in serum 32,33 and skin 34 was increased in TCE dermatitis [+] groups. We interpret the elevations in these proinflammatory cytokines as follows: repeated skin applications of TCE lead to skin sensitization and immune response, which cause stimulation of body immune systems and activation of macrophages, monocytes, and KCs; this is followed by the release of inflammatory mediators (TNF-α) and further systemic release of other cytokines.
The release of proinflammatory cytokines can amplify immune response by stimulating macrophages, monocytes, and other inflammatory cells which in turn release more cytokines and lead to further injury to the tissue. 35 These may explain increased levels of serum cytokines in patients with ACD. Evaluation of specific actions of TNF-α, IFN-γ, and IL-2 in mediating inflammatory reaction and tissue damage is of high importance, given the increased secretion of these cytokines in TCE-induced skin sensitization. This will provide further insight into the molecular basis of chemical-induced DTH. Our data suggest that TNF-α, IFN-γ, and IL-2 are importantly involved in generating the TEC-induced DMLT, and their mechanisms are worthy of further investigations.
In summary, our study has established a practical animal model for the study of TCE sensitization and pathogenesis. The BALB/c mouse model overcomes the drawbacks of other existing animal models of TCE-induced sensitization and more realistically simulates human TCE exposure. We have also demonstrated the role of key proinflammatory cytokines, such as TNF-α, IFN-γ, and IL-2, in TCE-sensitized mouse model. Specific mechanisms of TCE-mediated immunity and the actions of cytokines in vivo deserve further study.
Footnotes
Acknowledgments
We thank Clinical Laboratory and Central Laboratory of The First Affiliated Hospital of Anhui Medical University for valuable help with part of our experiment.
Author Contribution
Hui Wang contributed to conception and design, contributed to acquisition, analysis, and interpretation, drafted manuscript, and critically revised manuscript; Jia-xiang Zhang contributed to conception and design, contributed to acquisition, analysis, and interpretation, drafted manuscript, and critically revised manuscript; Shu-long Li contributed to conception, contributed to interpretation, drafted manuscript, and critically revised manuscript; Feng Wang contributed to design, contributed to interpretation, drafted manuscript, and critically revised manuscript; Wan-sheng Zha contributed to conception, contributed to interpretation, drafted manuscript, and critically revised manuscript; Tong Shen contributed to conception and design, contributed to acquisition, analysis, and interpretation, drafted manuscript, and critically revised manuscript; Changhao Wu contributed to conception, contributed to acquisition and analysis, drafted manuscript, and critically revised manuscript; Qi-xing Zhu, contributed to conception and design, contributed to acquisition, analysis, and interpretation, drafted manuscript, and critically revised manuscript. All authors gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Authors’ Note
Hui Wang and Jia-xiang Zhang are cofirst authors. These authors contributed equally to this article
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was supported by grants from National Natural Science Foundation of China (81371730), Specialized Research Fund for the Doctoral Program of Higher Education of China (20133420110001), and Scientific research fund project of Anhui Medical University (2015xkj005).
