Abstract
Safrole, a component of piper betle inflorescence, is a documented rodent hepatocarcinogen and inhibits bactericidal activity and the release of superoxide anion (O2−) by polymorphonuclear leukocytes (PMNs). In the present study, we investigated the effects of safrole on immune responses, including natural killer (NK) cell cytotoxicity, phagocytic activity and population distribution of leukocytes from normal BALB/c mice. The cells population (cell surface markers) and phagocytosis by macrophages and monocytes from the peripheral blood mononuclear cells (PBMCs) were determined, and NK cell cytotoxicity from splenocytes of mice after oral treatment with safrole was performed using flow cytometric assay. Results indicated that safrole did not affect the weights of body, spleen and liver when compared with the normal mice group. Safrole also promoted the levels of CD11b (monocytes) and Mac-3 (macrophages) that might be the reason for promoting the activity of phagocytosis. However, safrole reduced the cell population such as CD3 (T cells) and CD19 (B cells) of safrole-treated normal mice by oral administration. Furthermore, safrole elevated the uptake of Escherichia coli-labelled fluorescein isothiocyanate (FITC) by macrophages from blood and significantly stimulated the NK cell cytotoxicity in normal mice in vivo. In conclusions, alterations of the cell population (the increase in monocytes and macrophages, respectively) in safrole-treated normal BALB/c mice might indirectly influence the immune responses in vivo.
Introduction
The human immune system is divided into innate immunity and adaptive immunity. 1 The innate immune system has been recognized to serve as the first line of host defense against infectious agents by detecting the presence of microbial infection through germ line-encoded pattern recognition receptors. 2 Recent studies have shown that toll-like receptors (TLRs) on CD4+ T cells can directly modulate adaptive immune responses by affecting the survival and proliferation of activated CD4+ T cells. 3 Physical exercise affects both immune function and platelet activity because responses depend on type, intensity and duration of exercise. 4 Past years, many studies have focused on agents from natural plants to promote immune responses. 5–7
The International Agency for Research on Cancer (IARC) has categorized safrole as a group 2B carcinogen (a documented rodent carcinogen). However, there is no adequate study elucidating the relationship between exposure to safrole and human cancers. 8 Wang and Hwang demonstrated that chewing betel quid containing piper betle inflorescence can generate a high concentration of safrole (68 μg/ml) in the saliva. 9 Numerous studies have demonstrated that safrole caused marked [Ca2+]i elevation, decreased cell viability in human osteosacoma cells, 10 and induced apoptosis in human oral cancer HSC-3 cells. 11 Safrole bound to DNA resulted in safrole–DNA adducts formation. 12–14 Safrole induced a significant dose-dependent increase in hepatic lipid hydroperoxides and 8-hydroxy-29-deoxyguanosine levels in Sprague-Dawley rat liver. 15 Therefore, oxidative DNA damage may be involved in the hepatocarcinogenic effect of safrole. Reports have shown that the formation of stable safrole-DNA adducts in chewers’ oral tissues 15 and peripheral blood lymphocytes 16 might have a possible role in oral carcinogenesis.
It was reported that exposure of polymorphonuclear leukocytes (PMNs) to safrole prior to activation reduced the release of superoxide anions from PMNs, 17 and it also reduced the bactericidal activity of PMNs against Actinobacillus actinomycetemcomitans (American Type Culture Collection [ATCC] 33384, serotype c). 18 Currently, there are few reports to show the effects of safrole on the immune response. In the present study, we investigated whether safrole affected the immune modulation and regulated phagocytosis by macrophages and monocytes in normal BALB/c mice in vivo.
Materials and methods
Chemicals and reagents
Safrole, dimethyl sulfoxide (DMSO) and propidium iodide (PI) were purchased from Sigma-Aldrich Corp. (St Louis, MO, USA).
Male BALB/c mice
Male BALB/c mice of 4–6 weeks of age and approximately 20–26 g in weight were purchased from the Laboratory Animal Center, College of Medicine, National Taiwan University (Taipei, Taiwan).
Safrole treatment
Fifty BALB/c mice were randomly divided into five groups receiving different treatments. Group I served as a control (10 animals). Group II mice were treated with olive oil (10 animals). Groups III, IV and V animals were treated with safrole in olive oil at 4, 8 and 16 mg/kg, respectively. Safrole was administered by oral gavage to treat groups for 21 days before being weighed. 19,20
Spleen and liver tissue analyses
All animals were weighed and blood withdrawn. Spleen and liver samples were then isolated, photographed and weighed individually. 21
Whole blood samples and immunofluorescence staining for surface markers
At the end of the experiments, 1 ml of blood samples from each animal of each group was collected individually before the mice were killed. Blood was immediately treated with 1× Pharm LyseTM lysing buffer (BD Biosciences, San Jose, CA, USA) for lysing the red blood cells. And then all the samples were centrifuged for 15 min at 1500 rpm at 4°C. The isolated white blood cells were stained by the fluorescein isothiocyanate (FITC)-antimouse CD3, phycoerythrin (PE)-antimouse CD19, PE-antimouse Mac-3 and FITC-antimouse CD11b antibodies (BD Pharmingen Inc, San Diego, CA, USA) before determining the percentage of cell marker by flow cytometry (Becton Dickinson, FACS CaliburTM, Franklin Lakes, NJ, USA), as previously described. 22,23
Quantification of phagocytic activity of macrophages and monocytes
The phagocytosis of macrophages and monocytes was measured using the PHAGOTEST kit (Glycotope Biotechnology GmbH, Heidelberg, Germany), as previously described.20,23 Approximately 1 × 10 5 leukocytes in 100 μl of whole blood or peritoneal cavity from each animal was incubated for 1 h at 37°C with Escherichia coli-labeled FITC (20 μl). The quenching solution (100 μl) was individually added to the reaction. After the completion of phagocytosis by monocytes/macrophages, DNA was stained according to the manufacturer’s protocol. Cells were analyzed by flow cytometry, as previously described. 20,23 Fluorescence data were collected to reach 10,000 cells analyzed using the BD CELLQUEST Pro software.
Quantification of cytotoxicity of NK cell
Approximately 1 × 10 5 splenocytes from the spleens of each animal were placed in 1 ml of RPMI-1640 medium and then were cultured in 24-well culture plates for 24 h. YAC-1 cells (2.5 × 10 7 ) were cultured in 15-ml tubes with serum-free RPMI-1640 medium and PKH-67/Dilunt C buffer (Sigma-Aldrich Corp.) was added to the cells, mixed thoroughly for 2 min at 25°C and then 2 ml of phosphate buffered saline (PBS) was added for 1 min. About 4 ml of RPMI-1640 medium was added to each sample and incubated for 10 min followed by centrifugation at 1200 rpm at 25°C. YAC-1 cells were placed on 96-well plates for 12 h before the addition of the splenocytes to the wells. The determination of cell viability in YAC-1 cells was measured using a PI exclusion method by flow cytometry, and cytotoxicity of natural killer (NK) cell was determined as previously described. 19,20
Statistics analysis
The results were expressed as mean ± SD and the difference between groups was analyzed by one-way analysis of variance (ANOVA) followed by Dunnett’s multiple comparison test. A p value of less than or equal to 0.05 was considered significant.
Results
Safrole affected the weights of body, spleen and liver tissues of BALB/c mice
Animals from each group were individually weighed, and spleen and liver tissues were isolated and were weighed as can be seen in Figure 1. The data from Figure 1A indicated that safrole did not change the body weight of the normal and examined mice. Also, the weights of spleen and liver tissues of safrole-treated mice are not significantly different when compared with untreated control group.
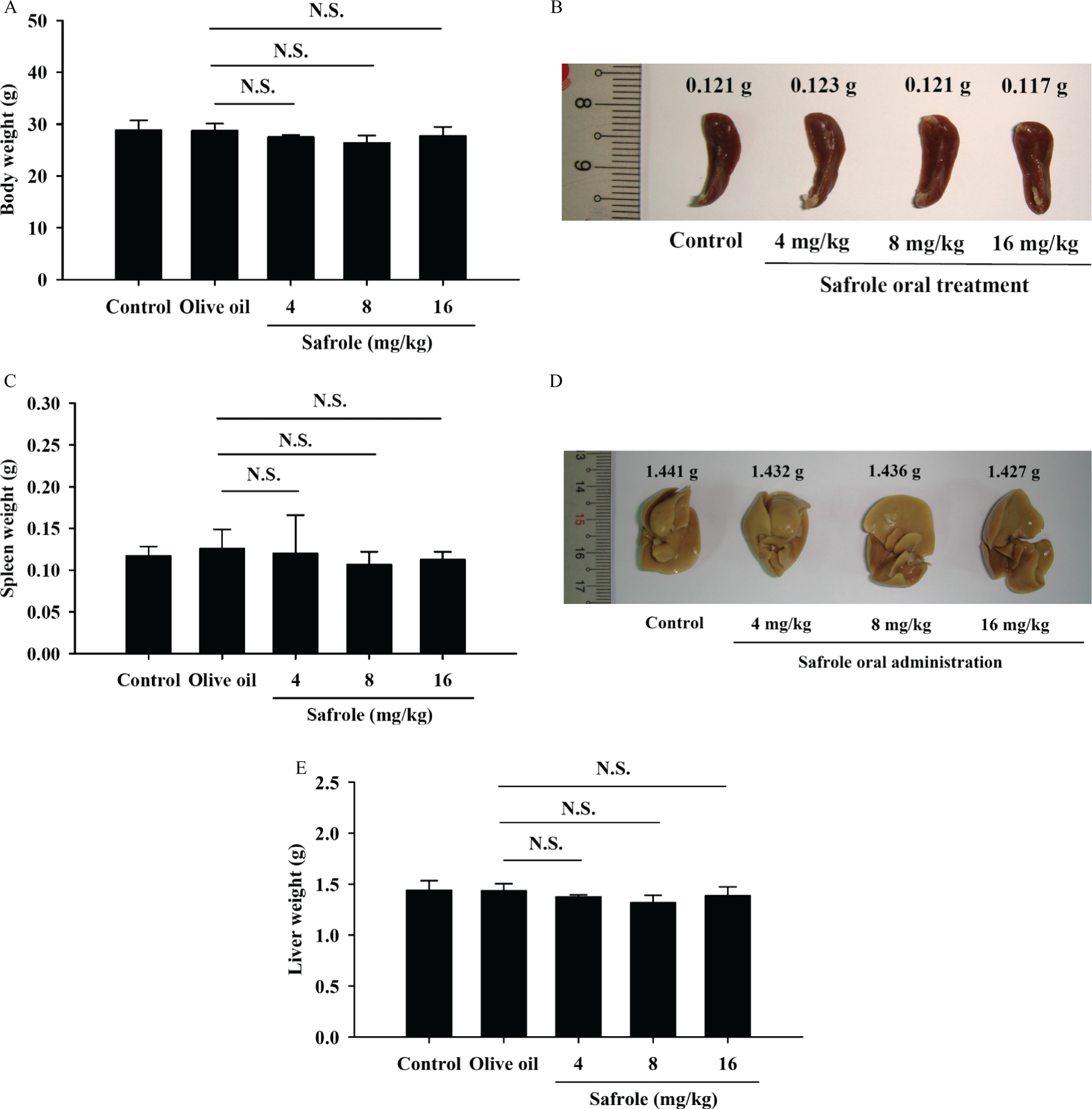
Safrole affected the body, spleen and liver weights of BALB/c mice. The mice were orally treated with or without safrole (4, 8 and 16 mg/kg) for 21 days. Respective profile of body weight was shown (A). The spleen and liver tissues were collected, photographed (B and D) and weighed (C and E) as described in the section on Materials and methods. Each point is mean ± SD (n = 10). NS = not significant when compared with the olive oil-treated mice.
Safrole affected whole blood cell surface markers of BALB/c mice
In order to examine whether safrole altered the levels of cell surface marker, leukocytes from safrole-treated or untreated mice were collected. The levels of CD11b, Mac-3, CD3 and CD19 were determined by flow cytometry. The results of cell markers of white blood cells from each treatment group are shown in Figure 2. The data indicated that safrole increased the levels of CD11b (Figure 2A; 8 mg/kg/day: increased 21.6%; 16 mg/kg/day: increased 26.0%) and Mac-3 (Figure 2B; 8 mg/kg/day: increased 3.7%; 16 mg/kg/day: increased 5.3%), but it decreased the levels of CD3 (Figure 2C; 4 mg/kg/day: decreased 13.9%; 8 mg/kg/day: decreased 16.9%; 16 mg/kg/day: decreased 18.2%) and CD19 (Figure 2D; 4 mg/kg/day: decreased 8.6%; 8 mg/kg/day: decreased 9.0%;16 mg/kg/day: decreased 13.5%) when compared to the only olive oil-treated group.

Safrole treatment altered the levels of cell markers in white blood cells of BALB/c mice. The animals were orally treated with or without safrole for 21 days. Blood was collected from each animal and was analyzed for cell markers (A: CD11b; B: Mac-3; C: CD3 and D: CD19) by flow cytometry as described in the section on Materials and methods. Data are expressed mean ± SD of three experiments. Each point is mean ± SD (n = 10). *p < 0.05 was considered significant when compared with the olive oil-treated mice.
Safrole affected the phagocytosis by macrophages and monocytes from peripheral blood mononuclear cell (PBMC) and peritoneal cavity on BALB/c mice
To investigate the safrole-affected phagocytosis in mice, the macrophages from PBMC and peritoneal cavity of safrole-treated or untreated mice were isolated and phagocytic activity was examined. The results shown in Figure 3A and B demonstrated that safrole (8 and 16 mg/kg/day) promoted the activity of phagocytosis (8 mg/kg/day:18.4%; 16 mg/kg/day: 9.4%) from PBMC, but all doses of safrole treatment did not affect the activity of phagocytosis from peritoneal cavity of mice.

Safrole treatment stimulated the phagocytic activity of PBMC and peritoneal cavity in BALB/c mice. Macrophages were isolated from peripheral blood mononuclear cell (PBMC) (A) or peritoneal cavity (B) of each mice after oral administration of 0, 4, 8 and 16 mg/kg/day of safrole for 21 days. The percentages of phagocytosis with phagocyte green fluorescent particles (FITC-Escherichia coli) at safrole oral treatment were determined by flow cytometric analysis as described in the section of Materials and methods. Each point is mean ± SD (n = 10). *p < 0.05 was considered significant and NS = not significant when compared with the olive oil-treated mice.
Safrole affected the NK cell cytotoxicity of splenocytes in BALB/c mice
Splenocytes from each treatment group with or without safrole were isolated and cytotoxicity of NK cell was determined by flow cytometry. The results are shown in Figure 4 and indicated that the YAC-1 target cells were killed by NK cells of the mice treated with safrole at 4, 8 or 16 mg/kg/day at target cells ratio of 100:1 and 50:1. This dose was effective at both target ratios and increased the activity of NK cells (4 mg/kg/day: 21.3%, 6 mg/kg/day: 18.2%, 16 mg/kg/day: 17.6% at target cells ratio of 100:1; 4 mg/kg/day: 20.4%, 8 mg/kg/day: 15.5%, 16 mg/kg/day: 15.9% at target cells ratio of 50:1).
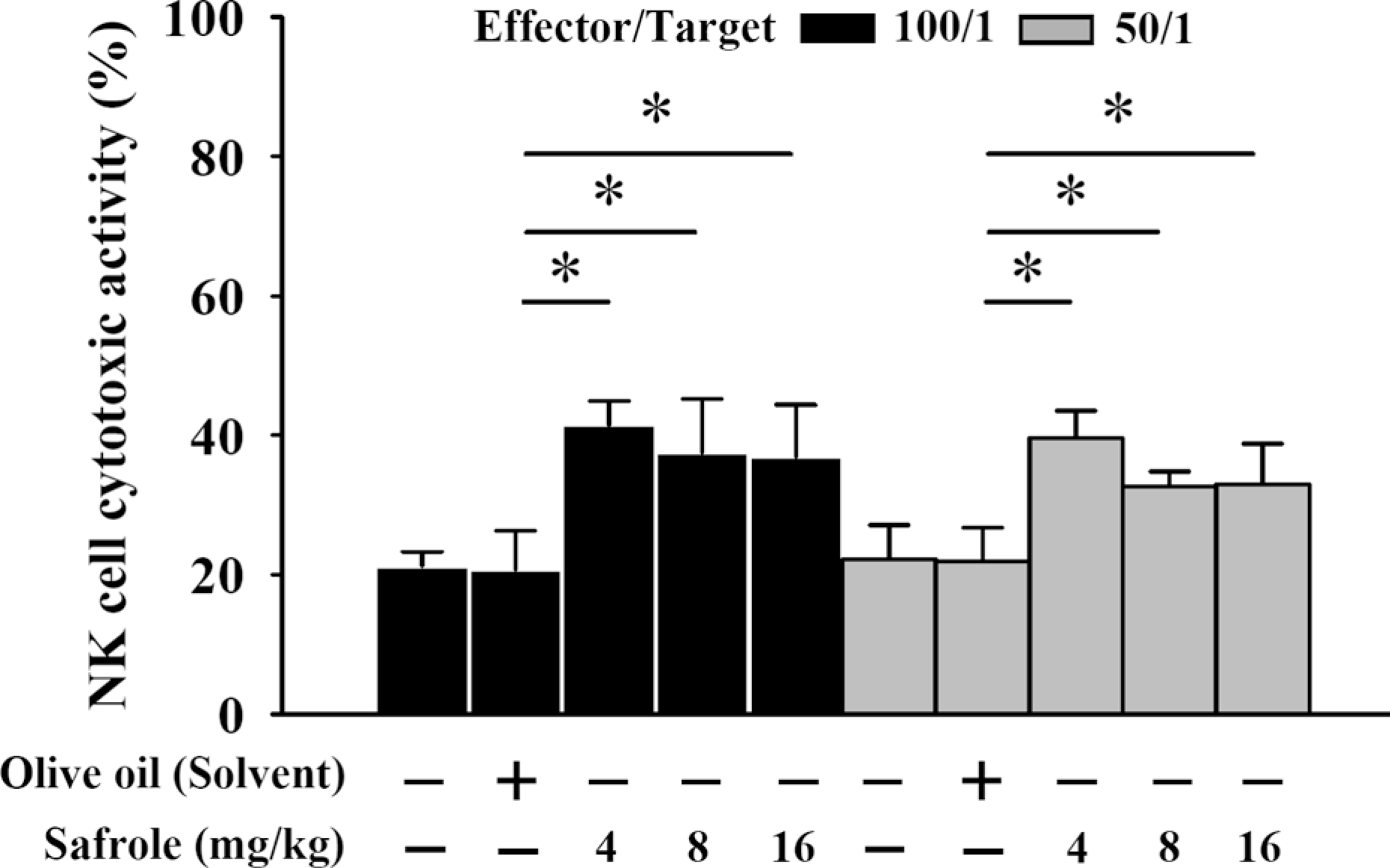
Safrole treatment promoted the cytotoxicity of natural killer (NK) cells in BALB/c mice. The YAC-1 target cells were killed by NK cells of the mice treated with 0, 4, 8 and 16 mg/kg/day of safrole by oral administration in target cells ratio of 100:1 and 50:1. Each point is mean ± SD (n = 10).*p < 0.05 was considered significant when compared with the olive oil-treated mice.
Discussion
Several reports have demonstrated that safrole, a carcinogen, is able to bind to DNA, and it also caused cytotoxic effects in human cancer cells. 12,13 However, there is no report to show safrole affected the immune responses in normal mice. Therefore, our study is the first one to show that safrole could affect the immune response in normal BALB/c mice and promoted the levels of CD11b (monocytes) and Mac-3 (macrophages; Figure 2A and B). It is well documented that healthy food might promote immune responses that will increase the levels of macrophages and promote the phagocytosis of macrophages. 24 Although the previous studies indicated that safrole exhibited carcinogenicity and typically was found to cause DNA adduct formation, 12,16 our results provided critical events addressing that lower concentration (<16 mg/kg) of safrole might not affect the mice in toxicity study and there is no alteration in the survival rate (100%; data not shown).
It was reported that the inflammatory infiltrate present in periodontal lesions also includes PMNs and other leukocytes. 25 Other reports have demonstrated that safrole, a component of piper betle inflorescence, inhibited the bactericidal activity of PMNs. 18 Furthermore, in vitro studies demonstrated that safrole interfered with these cells’ ability to kill bacteria 18 and to carry out phagocytosis. Based on these short-term treatment observations, the authors have suggested that safrole might affect the normal activation activity of PMNs. 26 In the present studies, normal mice after oral treatment with safrole was examined for phagocytosis by macrophages and from PBMC and peritoneal cavity, and the results indicated that safrole promoted the phagocytosis from PBMC (Figure 3A), but it did not induce significant effects on phagocytosis from peritoneal cavity (Figure 3B). However, the interesting point is that safrole promoted the cytotoxic activities of NK cells from splenocytes of normal mice after oral treatment of safrole as seen in Figure 4. Furthermore, all the three safrole-treated doses did not show significant effects on the weights of body, spleen and liver (Figure 1). Overall, the oral treatment of safrole led to increase in the activities of macrophages and NK cells of normal mice (Figures 3A and 4). And these findings are matched to the increased levels of cell population of CD11b and Mac-3 (Figure 2A and B).
In summary, our results showed that oral administration of safrole could promote the activities of macrophages and NK cells of normal BALB/c mice. In the promotion of macrophage phagocytosis, it was also mated to increase the cell marker population such as CD11b and Mac-3. Additionally, NK cell cytotoxicity was suppressed remarkably in mice treated with safrole. However, safrole suppressed the levels of cell marker population of CD3 (T cells) and CD19 (B cells). The results led us to pursue further research on in vivo studies by safrole in the future.
This work was supported by a grant CMU98-asia-10 from China Medical University, Taichung, Taiwan.
