Abstract
Xanthine oxidase activation occurs in sepsis and results in the generation of uric acid (UrAc) and reactive oxygen species (ROS). We aimed to evaluate the effect of xanthine oxidase inhibitors (XOis) in rats stimulated with lipopolysaccharide (LPS). LPS (10 mg/kg) was administered intraperitoneally (i.p.) immediately after allopurinol (Alo, 2 mg/kg) or febuxostat (Feb, 1 mg/kg) every 24 h for 3 days. To increase UrAc levels, oxonic acid (Oxo) was administered by gavage (750 mg/kg per day) for 5 days. Animals were divided into the following 10 groups (n = 6 each): (1) Control, (2) Alo, (3) Feb, (4) LPS, (5) LPSAlo, (6) LPSFeb, (7) Oxo, (8) OxoLPS, (9) OxoLPSAlo, and (10) OxoLPSFeb. Feb with or without Oxo did not aggravate sepsis. LPS administration (with or without Oxo) significantly decreased the creatinine clearance (ClCr) in LPSAlo (60%,
Keywords
Introduction
According to the Third International Consensus 2016, sepsis should be defined as a life-threatening organ dysfunction caused by a dysregulated host response to infection. 1 In addition, septic shock should be defined as a subset of sepsis associated with a greater risk of mortality than sepsis alone, 1 especially when kidney failure is established by acute renal injury and accompanied by multiple organ failure. The Toll-like receptor 4 (TLR4) stimulation caused by lipopolysaccharide (LPS) or TLR 7 and 8 by (+) ssRNA virus induces xanthine oxidase (XO) activation with monosodium urate crystal (i.e. uric acid (UrAc)) and reactive oxygen species (ROS) formation. 2 XO is also activated by urate crystals that stimulate TLR-2 and 4 and hypoxemia due to hemodynamic changes in sepsis, which is commonly observed in ischemia/reperfusion (I/R) models.2,3
Prior administration of allopurinol (Alo) has a well-documented protective effect in the first 24 h in animal models of sepsis,4–11 renal I/R,
3
liver,12,13 intestinal I/R,
14
tourniquets on rat hind-limbs,
15
brain I/R,
16
cocaine-induced diastolic dysfunction,
17
hyperthermia,
18
fructose-fed rats,19,20 streptozotocin-induced diabetes,
21
and
Although Alo has anti-inflammatory and protective effects in several experimental models, including sepsis, there are still controversial results, especially after 24 h. Its effect has not yet been entirely elucidated following its administration immediately prior to the induction of sepsis, and few studies have compared the effects of diverse xanthine oxidase inhibitors (XOis). Therefore, we conducted experiments to investigate these effects.
Material and methods
Lipopolysaccharide
Bacterial LPS from
Xanthine oxidase inhibitors
The following XOi were administered by gavage every 24 h for 3 days: Alo (2 mg/kg, GlaxoSmithKline do Brasil, RJ, Brazil) and febuxostat (Feb, 1 mg/kg, Takeda, IL, USA).
Oxonic acid
Oxonic acid (Oxo, Sigma-Aldrich), which was used to increase UrAc levels, was administered by gavage (750 mg/kg·per day), diluted in 0.25% methylcellulose in saline, once daily for 5 days before administration of LPS.
Animal ethics
The experimental protocol was approved by the Ethics Committee of the Universidade Federal de São Paulo (0220/12). Each experimental group was composed of six animals.
Experimental groups
Male Wistar rats weighing 200–250 g were divided into 10 groups: (1) Control (received water by gavage), (2) Alo, (3) Feb, (4) LPS, (5) LPSAlo, (6) LPSFeb, (7) Oxo, (8) OxoLPS, (9) OxoLPSAlo, and (10) OxoLPSFeb. At 0 and 17 h of the experimental period, blood samples were collected from the orbital sinus, and the animals were maintained in individual metabolic cages for 24 h for urine collection. Animals were euthanized 72 h after the experiment commenced under anesthesia (ketamine/xylazine, 10:1, 0.2 mL/100 g/kg, i.p.). Blood was collected, and the kidneys were perfused with saline solution, removed, and subsequently histopathologically analyzed. 
Biochemical analysis
The blood creatinine (Cr) and UrAc levels were assayed spectrophotometrically according to standard procedures using commercially available diagnostics kits (Labtest Diagnostica, MG, Brazil).
Lipid peroxidation
Lipid peroxidation was measured using the thiobarbituric acid reactive substances (TBARS) assay. The reactive substances combine with thiobarbituric acid to form a red compound. Malondialdehyde (MDA) was used to construct a standard curve, and the results are expressed as MDA (mM)/mg protein. Urine samples were added to a solution of 0.375% thiobarbituric acid, 15% trichloroacetic acid, and 0.25 N HCl (Sigma-Aldrich). The samples were continually agitated while being heated to 95°C for 20 min, and then they were allowed to cool to 22°C. The absorbance was spectrophotometrically determined at 535 nm. The results were expressed as 10−7 M/mg creatinine.
Cytokine analysis
Immunoassays, based on Luminex xMAP (multi-analyte profiling) technology, were performed to simultaneously detect and quantify multiple cytokines (interleukin (IL)-1α, IL-4, IL-1β, IL-2, IL-6, IL-10, and tumor necrosis factor (TNF)-α) using the Cat#RECTYMAG65K-08 granulocyte-macrophage colony-stimulating factor (GM-CSF) kit. The assays were performed at the beginning (time 0), and at 17 and 72 h in all groups, but for Oxo-treated animals, the assays were also performed 5 days before.
Histopathology
Hematoxylin and eosin (H&E) staining of the paraffin-embedded kidney tissue slices was performed.
Immunohistochemistry
Paraffin-embedded tissues were cut into 4-μm thick sections using a rotary microtome (Leica Microsystems, Herlev, Denmark). The kidney slices were deparaffinized and rehydrated, and then the sections were boiled in a target retrieval solution (1 mmol/L Tris, pH 9.0, with 0.5 mM ethylene glycol tetraacetic acid (EGTA)) for 10 min for antigen retrieval. Nonspecific binding was prevented by incubating the sections in phosphate-buffered saline (PBS) containing 1% bovine serum albumin (BSA), 0.05% saponin, and 0.2% gelatin. Endogenous peroxidase activity was blocked with 5% hydrogen peroxidase (H2O2) in absolute methanol for 10 min at 22°C.
The sections were incubated with primary antibodies against proliferating cell nuclear antigen (PCNA, 1:500) or cleaved caspase-3 (1:100), overnight at 4°C. PCNA is a nuclear protein linked to DNA replication, also known as cyclin, which plays a role in initiating cell proliferation by enhancing the DNA polymerase enzyme. After the sections were washed, they were incubated with horseradish peroxidase–labeled polymer conjugated to a secondary antibody (Dako, Denmark) for 1 h at room temperature. The sites of antibody-antigen reactions were visualized using 0.5% 3,3′-diaminobenzidine tetrachloride (Dako) dissolved in distilled water with 0.1% H2O2. A total of 10 sections per animal along the kidney cortex were analyzed, and labeled cells (staining light to dark brown) were counted.
Statistical analysis
The results are expressed as the mean ± standard error (SE). The data were analyzed using survival Kaplan–Meier curves and a one-way analysis of variance (ANOVA) followed by Tukey tests using GraphPad Prism 5.
Results
Coadministration of Alo but not Feb aggravated septic shock by increasing mortality
LPS administration significantly reduced the mean arterial blood pressure (BP) of rats, measured using a tail-cuff method, in all groups. The LPS group at 72 h showed a mean arterial BP of 95.5 ± 13.2 (

Blood pressure (BP) (mmHg) and survival curves. (a) BP in the LPS, LPSAlo, and LPSFeb groups and (b) BP in the Oxo, OxoLPS, OxoLPSAlo, and OxoLPSFeb groups compared to the control group showed significantly lower values (*
Oxo pre-treatment increase BP, blood UrAc levels and decreased mortality
After 5 days from Oxo administration, a significant increase in BP occurred (from 124.7 ± 4.3 to 149 ± 20.9 mmHg,

Biochemistry. UrAc: (a) LPS, LPSAlo, and LPSFeb groups and (b) OxoLPS, OxoLPSAlo, and OxoLPSFeb groups. UrAc values of treated groups were significantly higher than those of control groups, *
LPS increases ROS especially with coadministration of Oxo
In addition, we observed high levels of ROS (TBARS: nMol/mg urinary Cr,
Inflammatory cytokine
In all the groups who received LPS, the levels of TNF-α increased about three times in relation to control groups in 17 h and returned to basal levels in all groups, except in the LPSAlo and OxoLPSAlo groups where this late decline was not observed (Figure 3(a) and (b); Table 1).

Cytokine analysis (pg/mL): TNF-α: (a) LPS, LPSAlo, and LPSFeb groups and (b) Oxo, OxoLPS, OxoLPSAlo, and OxoLPSFeb groups. All groups treated with LPS alone or with drugs showed significantly higher values than the control group ***
Cytokine analysis.

GM-CSF: granulocyte-macrophage colony-stimulating factor. TNF-α (pg/mL): in relation to controls: significantly higher values ***
The levels of IL-6 in relation with controls were elevated during 17 h, by 19 times the superior values in the LPS group; by 26 times in the LPSAlo group; by 22 times in the LPSFeb group; by 25 times in the OxoLPS group; by 30 times in the OxoLPSAlo group; and by 25 times in the OxoLPSFeb group. After 72 h, lower IL-6 values were present in about 50% of cases in all groups, except in LPSAlo and OxoLPSAlo groups where these values remained higher, both about 15 times in relation to the control group (Figure 3(c) and (d)).
After 17 and 72 h, all animals treated with LPS alone or with LPS with drugs showed significantly higher IL-1α values than those of the control group, (
Compared to the control group after 17 h, all groups showed significantly higher IL-10 levels, (
All groups that received LPS alone or with drugs showed significantly higher IL-2 and GM-CSF levels (
Histopathological changes, proliferation (PCNA), and apoptosis rates
Minimal alterations were observed in the histopathological analysis, with rare areas that indicated circulatory disruption where vasodilatation and hemorrhage occurred. However, no significant difference (
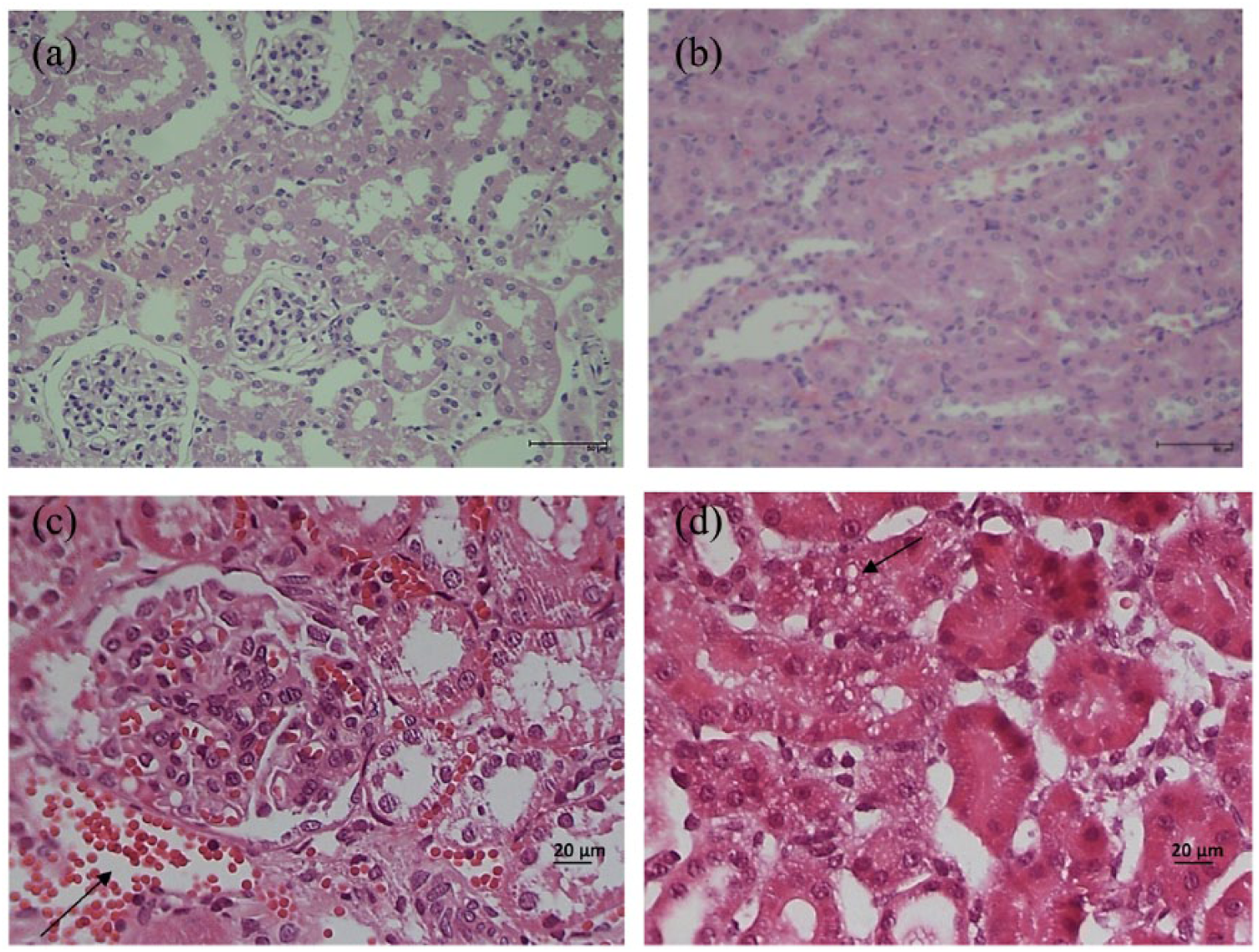
Histopathological sections of kidney were obtained at 72 h (n = 3 rats/group). (a) LPS, normal and (b) LPS, normal, 100× scale bar = 50 µm. (c) LPS, sector with vasodilatation and hemorrhaging (arrow) and (d) LPS, vacuolar degeneration of proximal renal tubules (arrow). 400× scale bar = 20 µm.
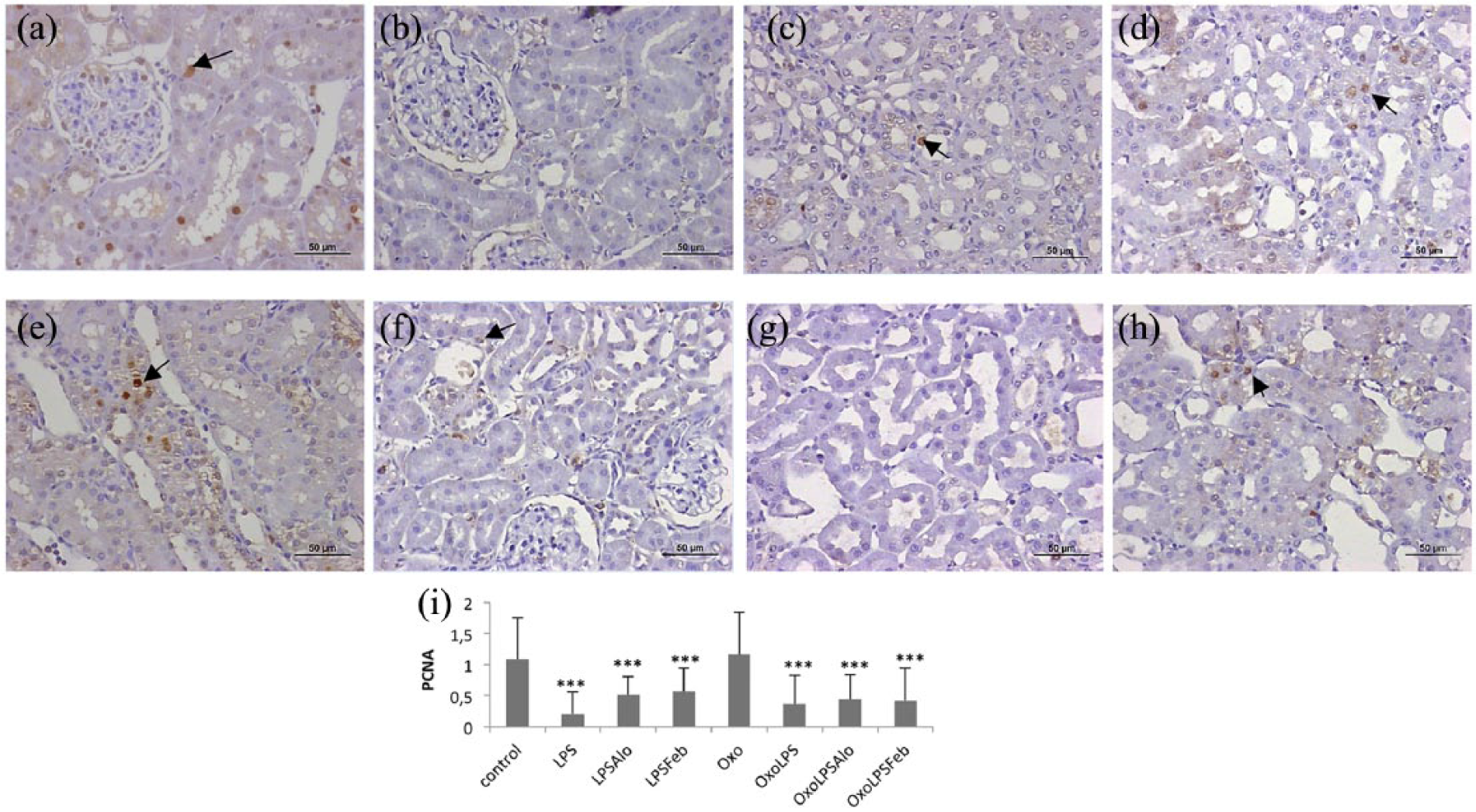
Immunohistochemical staining of PCNA (1:500 µm2): (a) Control; (b) LPS; (c) LPSAlo; (d) LPSFeb; (e) OxoLPS; (f) OxoLPS; (g) OxoLPSAlo; and (h) OxoLPSFeb. All groups (i) treated with LPS alone or with drugs demonstrated a significantly lower proliferation rate (***

Staining for cleaved caspase-3 (1:100): apoptotic cell number/field. (a) Control; (b) LPS; (c) LPSAlo; (d) LPSFeb; (e) Oxo; (f) OxoLPS; (g) OxoLPSAlo; and (h) OxoLPSFeb. All groups (i) the number of apoptotic cells (arrow) per field was significantly higher in groups treated with LPS alone or with drugs compared to the control group (
Discussion
Sepsis has a high mortality rate, mainly due to septic shock, aggravated by renal insufficiency and multiple organ failure. 1 In this study, we would like to be clear that LPS induces endotoxemia and systemic inflammatory response syndrome (SIRS) but not sepsis as defined in the third consensus definition. LPS-treated groups had lower BP, impaired renal function, and higher ROS levels compared to the control group. Furthermore, immunohistochemistry for PCNA revealed less cell proliferation and that for cleaved caspase-3 indicated considerable apoptosis that accompanied the permanently elevated IL-10 levels related to septic shock.4,34–36 ROS are involved in the caspase-3 apoptosis cascade 37 in renal tubular cells in response to systemic sepsis stress, which is a mechanism of acute kidney injury (AKI), 35 where increased expression of renal IL-10 has been observed. 37
Alo is readily absorbed from the gastrointestinal tract. In our experiment, LPS administration immediately after daily treatment with 2 mg/kg Alo paradoxically worsened the sepsis compared to Alo pre-treatment at doses between 10 and 100 mg/kg body weight from 4 days to 20 min before LPS treatment.5,38–40 This contrasts with the effects observed in the first 24 h, when Alo protected against experimental sepsis.5,38–40 It was previously shown that the protective effects of Alo may disappear after 24 h, with mortality rates reaching 100%. 22 Matuschak et al. 41 showed the potent suppressive effect of combined hypoxic stress/reperfusion (H/R) and EC stimulation on the activation and nuclear translocation of nuclear factor (NF)-Kb, in contrast with the robust hypoxic stress-induced transactivation of NF-kB without sepsis. However, administration of the anti-inflammatory compound Alo in H/R + EC increased the NF-kB transactivation, which could, 41 in part, explain the paradoxical effect observed in the LPSAlo and OxoLPSAlo groups.
In cell cultures exposed to LPS, Alo blocked caspase-3 activity and ROS formation in the first 24v h, but this effect disappeared within 48 h, inexplicably. 10 No deaths were observed in the OxoLPS and OxoLPSFeb groups, perhaps because the elevated UrAc level functioned as an antioxidant. UrAc is a potent extracellular antioxidant that evolutionarily confers longevity, 42 maintains BP under conditions with low amounts of dietary sodium, and has a protective effect in multiple sclerosis 43 and severe sepsis. 44 However, it is also an intracellular pro-oxidant, which perhaps explains the increased rates of lipid peroxidation, the levels of proinflammatory ILs and selectively decreases the human blood monocyte production of the natural IL-1 receptor antagonist (IL-1Ra) and changes the production to highly inflammatory IL-1β. 45 In human vaccines, aluminum hydroxide adjuvant boosts adaptive immunity by inducing UrAc and activating inflammatory dendritic cells. 46 Oxo administration for 5 days significantly increased UrAc and BP levels, which was expected. UrAc causes endothelial dysfunction and indirectly decreases levels of nitric oxide, which regulates vascular endothelial cell tone. 47 Furthermore, it activates the renin-angiotensin system and the proliferation of smooth muscle cells by stimulation of inflammatory responses. 47 In the kidney, it triggers afferent arteriopathy, induction of the epithelial-mesenchymal transition, and interstitial tubular fibrosis. 47 Hyperuricemia may be an early marker of sepsis severity. 47
Alo is the drug of choice for reducing UrAc levels, although it is contraindicated in the initial phase of gout treatment. Recent meta-analyses have shown that continuous use of Alo in patients with hyperuricemia preserves renal function, and the subsequent slower induction of proteinuria protects against arterial hypertension and slows the progression to chronic kidney disease and mortality. 48 Alo thus has a beneficial effect on ischemia, inflammation, and chronic heart failure. 48 However, some adverse effects such as hepatitis, nephropathies, and hypersensitivity reactions limit the clinical use of Alo. 49 Although occurring at a frequency of <0.1% in humans, hypersensitivity to Alo increases the risk of death by 27%, 49 with the main risk factors as follows: male sex with arterial hypertension and/or renal insufficiency and overdosage, 49 recent initiation of therapy, use of concomitant diuretics or nonhormonal anti-inflammatory drugs, and the presence of HLA-B*5801. 49
In the LPSAlo group, the shock was possibly triggered by the hemodynamic changes of sepsis evolving into AKI that was aggravated by Alo or the animals developed interstitial nephritis induced by Alo. 49 However, no substrate such as eosinophilia was found in histopathology. There may be another unknown mechanism underlying the paradoxical beneficial therapeutic effect of Alo and negative effect of renal function, as the use of Feb did not worsen the sepsis. Feb does not have structural similarity to purines such as Alo, which may interfere with other stages of purine metabolism. 50
Feb is an XOi with a favorable toxicological profile and high bioavailability, and it reduces UrAc levels in a more sustained and potent manner than Alo with adverse effects in less than 2% of patients; such effects include diarrhea and increased hepatic transaminases, without serious cutaneous reactions.51,52 Feb is primarily metabolized by hepatic oxidation and glucuronidation and eliminated by both the liver and kidney. In contrast to the renal elimination of the active metabolite of Alo (oxypurinol), renal elimination plays a minor role in the elimination of Feb, facilitating safer prescription including for the treatment of tumor lysis syndrome (TLS). Although TLS is classically treated with Alo, studies on Feb in clinical trials have shown a more promising profile.
52
In this experiment, baseline TNF-α levels were approximately 1 pg/mL and increased significantly (
The LPSAlo and OxoLPSAlo groups still showed significantly increased TNF-α levels after 72 h, which is related to the severity of the condition, shock, and higher death rate.54,55 Similarly, compared to the control levels at 17 and 72 h, all groups treated with LPS with or without XOi or Oxo showed elevation of IL-6, especially the LPSAlo and OxoLPSAlo groups at 72 h. This is related to greater severity of the condition and a worse prognosis, as IL-6 is considered a marker for the early diagnosis, follow-up, and prognosis of sepsis. 55
Significantly higher IL-1β values were observed in all animal groups treated with LPS with or without XOi than in the control group; however, after 72 h, the IL-1β levels in the LPSFeb group were significantly lower than those in all other groups. 56 In this study, IL-10 values gradually increased with significant differences in all groups at 17 and 72 h, which could be interpreted as a good anti-inflammatory response.53,55 This is because IL-10 is the main cytokine dampening the innate immune response. TNF-α and other cytokines stimulate IL-10 synthesis, which by feedback blocks the synthesis of TNF-α, normalizing serum levels of cytokines.39,55 The peak of IL-10 levels is related to a higher risk of shock, and when IL-10 levels remain high, as in the groups that received Alo, it is a factor for the development of multiple organ failure,55,57 septic shock, and death. 57
Here, administration of Feb immediately prior to LPS, with or without previous administration of Oxo, did not aggravate sepsis, mortality rates, or levels of TBARS, TNF-α, IL-6, IL-1α, IL- 2, IL-10, and GM-CSF, with similar values compared to the group that received LPS alone (with or without prior administration of Oxo).
Administration of LPS plus Alo (with or without Oxo) significantly decreased the ClCr values, with a significant increase in mortality and also increased levels of TBARS, TNF-α, IL-6, IL-1α, IL -1β, IL-2, IL-10, and GM-CSF in the first 17 h compared to the levels in groups treated with LPS and LPS plus Feb with or without Oxo. The TNF-α and IL-6 levels remained significantly higher after 72 h in the animals that received LPS plus Alo (with or without Oxo), which was correlated with a higher risk of death.
In this study, LPS administration immediately after Alo in experimental LPS-induced sepsis aggravated septic shock with increased mortality and impaired renal function, with higher values of ROS and proinflammatory ILs. Clinically, the known risk factors for Alo adverse reactions include recent initiation of therapy and renal failure. In contrast, when we administered Feb, no damage to the sepsis-regulating system was observed, probably because it did not interfere with other metabolic events, which probably occurs with Alo. The prescription of Alo requires physician expertise for use in patients with sepsis who at any time might progress to shock and renal failure. In contrast, Feb, a XOi with hepatic metabolism and renal and hepatic excretion, did not aggravate or improve septic shock with renal failure. Further studies are needed to elucidate the role of XOs in septic shock with renal failure.
Supplemental Material
supplements_tables – Supplemental material for Xanthine oxidase inhibitors and sepsis
Supplemental material, supplements_tables for Xanthine oxidase inhibitors and sepsis by Maria Fátima de Paula Ramos, Alceni do Carmo Morais Monteiro de Barros, Clara Versolato Razvickas, Fernanda T Borges and Nestor Schor in International Journal of Immunopathology and Pharmacology
Footnotes
Acknowledgements
Department of Morphology and Genetics, UNIFESP/EPM, São Paulo, Brazil
Declaration of conflicting interests
We have no direct or indirect commercial financial incentive associated with publishing this article. In addition, the authors have no conflicts of interest, and the source of extra-institutional funding is indicated in the manuscript. All authors participated in the design, interpretation of the study, analysis of the data, and review of the manuscript.
Funding
This work was supported by grants from Conselho Nacional de Desenvolvimento Científico Tecnológico (CNPq), Financiadora de Estudos e Projetos (FINEP), Fundação Oswaldo Ramos (FOR), Fundação de Amparo à Pesquisa do Estado de São Paulo (FAPESP) and Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
