Abstract
Chemical modification of cellulose is currently attracting attention as researchers attempt to take advantage of the abundance of hydroxyl groups on its surface to introduce extra biological functionality. However, the possible deleterious effect of exposure to functionalized nanocellulose (CSN) remains a concern. Therefore, this study aims to explore the potential mechanisms of hepatotoxicity of CSN modified with oxalate ester (NCD) in rats. A 7-day repeated oral toxicity study of NCD at the doses of 50 and 100 mg kg−1 body weight was conducted, and plasma and liver tissue samples were assayed using biochemical analysis, liver histopathology, and protein expression. NCD, at both doses, did not significantly (p > 0.05) alter the relative weight of liver, alkaline phosphatase activity, and lipid peroxidation levels of the animals. However, NCD at the dose of 100 mg kg−1 body weight significantly elevated aspartate aminotransferase, alanine aminotransferase, and myeloperoxidase activities. NCD also enhanced the immunohistochemical expression of inducible nitric oxide synthase and Bcl-2-associated X protein in the liver of rats. Histological observations revealed necrosis and severe cellular infiltration at the high-dose treatment. Our study provides an experimental basis for the safe application of NCDs.
Introduction
Cellulose (CSC), abundant in plants, animals, and bacteria, can be nanoscaled to sizes ranging from 1 nm to 100 nm in at least one dimension. As a natural nanoscaled material, nanocellulose (CSN) possesses several characteristics different from traditional materials, including peculiar morphology and geometrical dimensions, crystallinity, high specific surface area, rheological properties, liquid crystalline behavior, alignment and orientation, mechanical reinforcement, barrier properties, surface chemical reactivity, biocompatibility, and biodegradability. 1 Due to these unique physicochemical properties, a diverse range of biomedical applications has been attributed to CSN-containing materials including wound healing, bone and cartilage regeneration, cornea replacement, tissue engineering, cancer targeting, dental applications, antibacterial agents, drug delivery, and biology–device interface. 2,3
CSN, however, is very polar, and its utilization has been restricted to applications involving hydrophilic media. With the presence of a large number of functional groups within their structure, various chemistries have been developed to modify their surfaces to confer specific properties, which can diversify their applications. Specifically, during modification, certain functional groups are introduced to the parent molecule to increase the amount of stable positive or negative charges for better dispersion and the improvement of compatibility when combined with hydrophobic matrices. 4,5 Wei et al. 6 functionalized CSN by transesterification with fatty acid methyl ester of canola oil. In other studies, surface modification of CSN was achieved in aqueous and organic media using octadecyl, monoallyl-poly (ethylene glycol), benzyl, and propargyl triazinyl derivatives. Cationization, carboxylation, and silylation modification chemistries have also been deployed. 7,8 Hydrophobicity of CSN is desirable in applications requiring water repellency, corrosion prevention, and friction reduction. 9 –11 Since oxalate esters are hydrophobic compounds; we hypothesized that the introduction of this functional group at the surface of CSN might increase its hydrophobicity.
Despite remarkable advances in nanomedicine, the toxicity and health risks of exposure to nanomaterials are still attracting considerable and increasing concern globally. It has become certain that the chemical composition, particle size, size distribution, shape, surface charge, and solubility of nanoscaled materials contribute to their toxicity. 12 Several studies highlight the toxicity of inorganic nanoparticles, such as iron, zinc, and silicon oxide nanoparticles, 13 –15 graphene and carbon nanotubes, 16,17 and silver and gold nanoparticles. 18,19 However, toxicological evaluations on CSN and its modified derivatives are still scanty and contradictory. The in vivo toxicity evaluation of CSC nanocrystals, after pharyngeal aspiration, has been reported to elicit dose-dependent oxidative stress, tissue damage, and robust inflammatory responses in the lungs of C57BL/6 mice. 20 Contrariwise, Daphnia magna treated with CSC nanofibrils (0.206–20.6 mg L−1) showed no clinical signs of toxicity (no mortality, no apparent translocation of CSC nanofibrils across the alimentary canal, and no aberrant behavior) after 21 days of treatment. 21 Also, another study showed low toxicity potential of surface-modified CSC-nanocrystals and CSC-nanofibers in embryonic zebra fish. 22 We had also previously reported that oral administration of sulfonated CSN could cause mild toxicity in rat renal tissues. 23
Given the myriad of potential uses of CSN, there is a need to investigate how potential structural and chemical alterations to CSN can impact its toxicity. Furthermore, existing information on the relationship between CSN oxalate ester and liver toxicity is limited, especially in rodent models. The present study evaluated the effects induced on rat liver by short-term repeated oral administration of CSN modified with oxalate ester (NCD) by assessing liver functional indices, markers of oxidative stress, histopathology, and expression studies on key proteins of inflammation (inducible nitric oxide synthase (iNOS)) and apoptosis (Bcl-2-associated X protein (Bax)).
Materials and methods
Preparation and characterization of NCD
Cotton seed (CS) was obtained from a local market in Ibadan, Oyo state, Nigeria. The seeds were air-dried, ground into powder, and used for the isolation of CSC as previously described by Neto et al. 24 Briefly, alkali solution (2 wt% sodium hydroxide (NaOH)) was added to a 5-L beaker containing CS (300 g) and heated at 80°C for 5 h with continuous stirring using a Fisatom mechanical stirrer. After cooling and filtration, the residue was bleached in equal volumes of acetate buffer (27 g NaOH and 75 mL glacial acetic acid, diluted to 1 L of distilled water) and aqueous sodium chlorite (1.7 wt% NaClO2 in deionized water). The mixture was stirred at 80°C for another 5 h. The resulting fibers were washed repeatedly in deionized water until the pH of the fiber became neutral. The bleaching step was repeated twice until the fiber became completely white and dried in an air-circulating oven at 50°C for 24 h giving rise to CSC of an estimated yield of about 87%. CSC was then converted to CSN by acid hydrolysis with 65% sulfuric acid solution (v/v) at 50°C as described by Adewuyi and Pereira. 25 Powdery CSN was obtained by freeze-drying. Subsequently, NCD was synthesized by weighing CSN (20 g) into a round-bottomed flask containing toluene (80 mL). Then, 5 mL of triethylamine and then 10 mL of oxalyl chloride were added, after a cooling process. The mixture was refluxed for 3 h, cooled in ice, and the product formed was washed severally with deionized water. The resulting NCD gave about 82% yield after drying at room temperature. The reaction can be described as shown in Figure 1.

Synthesis of NCD. NCD: nanocellulose modified with oxalate ester.
The functional groups in NCD were determined using Fourier transform infrared (FTIR; Perkin Elmer, spectrum RXI 83303), while the X-ray diffraction pattern was obtained with X-ray diffractometer (XRD-7000X-Ray diffractometer, Shimadzu, Japan ) using filtered copper K α radiation operated at 40 kV and 40 mA. The XRD pattern was recorded from 10°C to 80°C of 2θ s−1 with a scanning speed of 2.0000° of 2θ min−1. The surface morphology of NCD was studied with scanning electron microscopy (SEM; FEI quanta 200 EDAX EDS, USA). Particle size distribution and zeta potential of NCD were determined at 25°C using a zeta potential analyzer (DT1200, Dispersion Technology, New York, USA).
Animal rearing and treatment
Albino rats were purchased from the primate colony of the Department of Veterinary Pathology, University of Ibadan (Ibadan, South-west Nigeria). Animals were housed in plastic cages in our animal facility with controlled light and dark cycle of 12 h each at 25 ± 2°C. For the present study, male rats were used in experiments at 8–12 weeks of age. The rats were maintained on standard laboratory rat chow and water ad libitum. All animals received humane care following institutional guidelines for experimental research as established by the Redeemer’s University Research Ethics Committee. The study approval was coded RUN/BCH/12/4594. A 7-day repeated oral toxicity study of NCD was conducted in male Wistar rats. A total of three groups (six rats per group) were compared: (1) control (normal saline), (2) NCD 1 (50 mg kg−1 body weight), and (3) NCD 2 (100 mg kg−1 body weight). Selection of doses for this study was based on 0.025 and 0.05 of the median lethal dose previously established for orally administered CSC nanocrystals. 26 General conditions including weight loss, reduced activity, breathing difficulties, and diarrhea were monitored daily for 7 days, after which blood was collected by cardiac puncture into ethylenediaminetetraacetic acid sample bottles and processed for sero-clinical analyses including aspartate aminotransferase (AST), alanine aminotransferase (ALT), alkaline phosphatase (ALP), and albumin (ALB). The liver was excised, and a portion was fixed in 4% buffered formalin. The remaining tissue was rinsed with 1.15% potassium chloride solution (4°C), blotted, and stored in the freezer for further processing. Later, the remaining liver tissues were homogenized in four volumes of 0.1 M phosphate buffer (pH 7.4). The homogenates were centrifuged at 12,000 × g for 15 min to obtain post-mitochondrial supernatant fraction which was kept at −20°C until analysis.
Liver function biomarkers and growth performance indices
Plasma samples from rats were used for the determination of AST, ALT, ALP, and ALB using commercial kits (Randox Laboratories Limited, UK). Mortality and changes in weight were observed in rats. After the animals were euthanized, the liver was harvested and weighed. The relative liver weight was calculated using the formula (relative liver weight = total liver wet weight/body weight × 100%).
Assessment of oxidative stress biomarkers and protein expression studies
Total protein concentration was determined according to the method described by Gornal et al. 27 Superoxide dismutase (SOD) activity was assayed according to the method described by Misra and Fridovich. 28 Catalase (CAT) activity was assayed using hydrogen peroxide as a substrate according to the method described by Luck. 29 Glutathione peroxidase (GPx) activity was assayed according to the method described by Rotruck et al. 30 Reduced glutathione (GSH) was determined according to the method described by Jollow et al. 31 Hydrogen peroxide level (H2O2) was assayed according to the method described by Wolff. 32 Malondialdehyde (MDA) level, an index of lipid peroxidation, was assayed according to the method described by Varshney and Kale. 33 In the liver, activity of myeloperoxidase (MPO) served as an index of neutrophil accumulation and stimulation and was determined as previously described. 34 Macrophage activation, captured by nitric oxide (NO) production, was measured as nitrite (NO2) using Griess reaction as described by Bryan and Grisham. 35
Immunohistochemical staining of iNOS and Bax protein expressions on liver tissue was performed using a streptavidin–horse radish peroxidase detection system with diaminobenzidine (DAB) as the chromogen. The images were captured using a digital camera (Leica Biosystems, UK) attached to the microscope. The resulting images were processed and analyzed with open source ImageJ software (v1.51a NIH). Three micrographs from each group were subjected to ImageJ analysis. These were deconvolved to filter microscope-associated noise, and color threshold was adjusted to the region of positive DAB stain. The analysis entailed determining the mean intensity of immunoreactivity after removing the background intensity relative to the total intensity of the image. This was expressed as a percentage of high positive DAB intensity.
Histology
A portion of the excised hepatic tissues from each group (n = 3) was collected and preserved in 10% neutral-buffered formalin for histopathological studies. These tissues were processed and embedded in paraffin wax after which thin sections of 5–6 µm thickness were cut and stained with hematoxylin and eosin. The thin sections of liver were made into permanent slides and examined photomicroscopically
Statistical analysis
The results are expressed as mean ± standard deviation. Statistical significance was determined by one-way analysis of variance and complemented with Duncan’s multiple comparisons between control and treated animals in all groups using GraphPad Prism statistical software. The values of p < 0.05 were considered to indicate statistical significance.
Results
Preparation and characterization of NCD
The FTIR of NCD is presented in Figure 2. The spectra revealed a peak at 3440 cm−1 which may be attributed to the OH functional group. The peak at 2901 cm−1 suggests the C–H stretching of CH2, while the sharp peak at 1741 cm−1 assigned to the C=O stretching of acetyl group and uranic ester groups suggests the formation of the oxalate ester in NCD. The XRD pattern of NCD is shown in Figure 2. The diffraction planes showed three 2θ peaks at 14.6°, 18°, and 22°. The SEM profile of CSN and NCD is presented in Figure 3. The surface of CSN is homogeneous and showed an average particle size of around 100 nm. The zeta potential ranging from −50 mV to −10 mV increased with increasing pH (Figure 4). Agglomeration of particles was also observed in NCD.

(a) FTIR and (b) XRD profile of NCD. FTIR: Fourier transform infrared; XRD: X-ray diffraction; NCD: nanocellulose modified with oxalate ester.

Scanning electron microscopy analysis of (a) CSN and (b) NCD. CSN: nanocellulose; NCD: nanocellulose modified with oxalate ester.

(a) Particle size distribution and (b) zeta potential of NCD. NCD: nanocellulose modified with oxalate ester.
Plasma markers of hepatotoxicity, total protein, liver coefficient, and growth performance indices
The body weights and liver coefficients of rats after oral administration of NCD at 50 and 100 mg kg−1 doses were not significantly different (p > 0.05, Table 1). However, there was a dose-dependent increase in ALT and AST activities in the plasma of rats exposed to NCD (p < 0.05; Table 1). Plasma ALP activity was not significantly different across all treatment groups. In animals exposed to NCD, ALB levels were also significantly increased at the doses of 50 and 100 mg kg−1, when compared with the control group, while the hepatic total protein levels were significantly elevated in rats administered with NCD at 100 mg kg−1 body weight (Table 1).
ALT, AST, ALP, ALB, and liver total protein level: body weight and relative liver weight (g/100 g b.w.) in control and NCD (NCD 1: 50 mg kg− 1 b.w. and NCD 2: 100 mg kg− 1 b.w)-treated rats.a
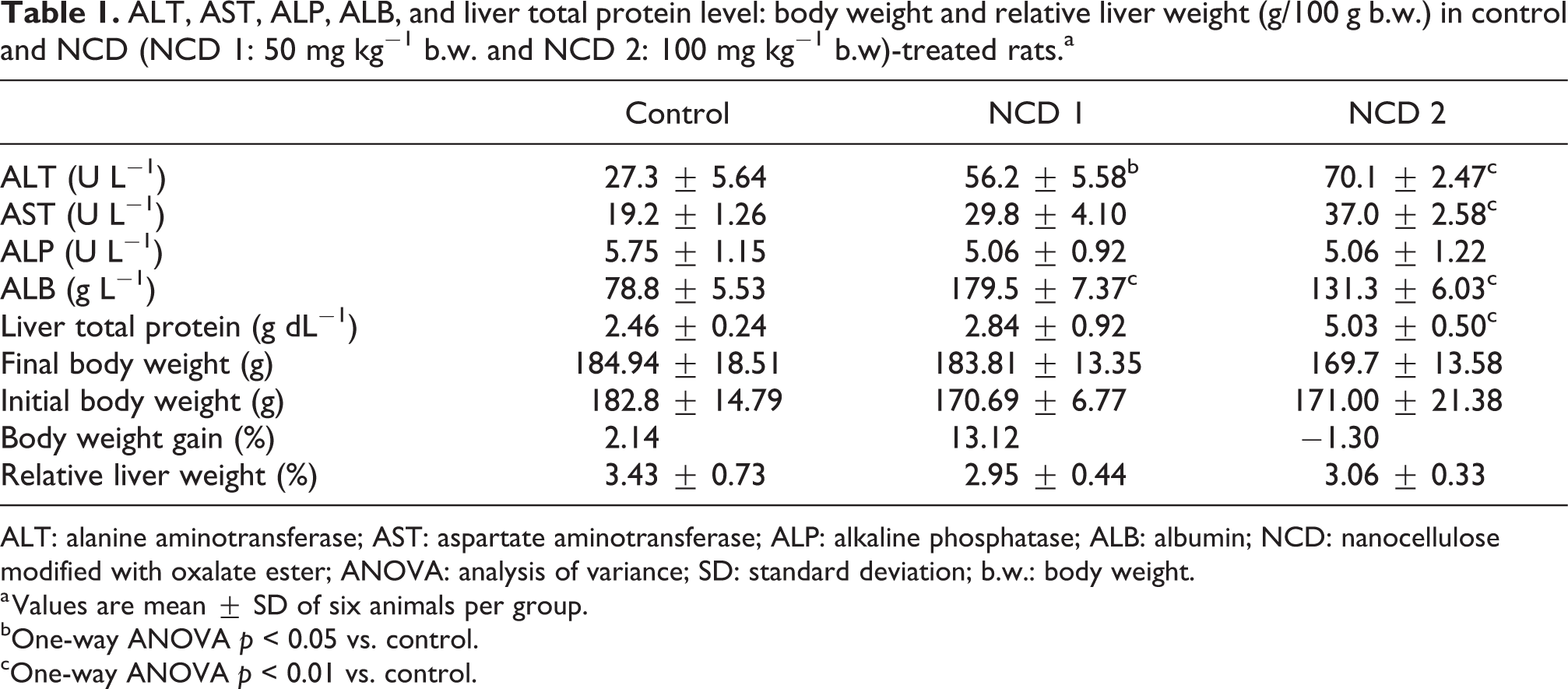
ALT: alanine aminotransferase; AST: aspartate aminotransferase; ALP: alkaline phosphatase; ALB: albumin; NCD: nanocellulose modified with oxalate ester; ANOVA: analysis of variance; SD: standard deviation; b.w.: body weight.
a Values are mean ± SD of six animals per group.
bOne-way ANOVA p < 0.05 vs. control.
cOne-way ANOVA p < 0.01 vs. control.
Assessment of oxidative stress biomarkers and protein expression studies
To investigate the effects of NCD on redox status and pro-inflammation, CAT, GPx, SOD, GSH, H2O2, MDA, NO, and MPO concentrations were measured in the liver homogenates after treatment of rats to NCD at the doses of 50 and 100 mg kg, as depicted in Figure 5. NCD elicited a dose-dependent and significant decrease in the CAT when compared to the control. The percentage decrease in CAT activity was 46% and 70% at 50 and 100 mg kg−1 doses, respectively, when compared with the control. On the other hand, a significant decrease (14%) in GPx activity was observed at the 100 mg kg−1 dose. Conversely, NCD treatment significantly elevated MPO activity when compared with the control. However, SOD, GSH, H2O2, NO, and MDA levels were not different between the NCD-treated and control rats. The effects of NCD on inflammation and apoptosis were evaluated by analyzing the immunohistochemical expression of iNOS and Bax in the liver of exposed animals, as shown in Figure 6. There were significant increases in the intensities of iNOS and Bax staining in the liver of the treated rats when compared with the control. However, these changes were not dose dependent.

Activities of CAT, GPx, and SOD, and levels of GSH, H2O2, MDA, NO and in the liver of rats treated with NCD. NCD 1: 50 mg kg−1 body weight and NCD 2: 100 mg kg−1 body weight. Values are means ± SD of six animals per group. *p < 0.05 and **p < 0.01 vs. control. CAT: catalase; GPx: glutathione peroxidase; SOD: superoxide dismutase; GSH: reduced glutathione; H2O2: hydrogen peroxide level; MDA: malondialdehyde; NO: nitric oxide; NCD: nanocellulose modified with oxalate ester; SD: standard deviation.

Immunoreactivity of a to c (iNOS) and e to g (Bax) in the liver of rats treated with NCD for seven consecutive days. Brown color indicates specific immunostaining, while the light blue color depicts hematoxylin staining. iNOS and Bax expression was quantified with ImageJ. *Statistical significance at p < 0.05 compared with control. Error bars show the standard deviation of the specific intensities of three micrographs per group (magnification ×400). NCD 1: 50 mg kg−1 body weight; NCD 2: 100 mg kg−1 body weight. iNOS: inducible nitric oxide synthase; Bax: Bcl-2 associated X protein; NCD: nanocellulose modified with oxalate ester.
Histological observations
The histopathological changes caused by administration of NCD in the liver were observed after 7 days of treatment (Figure 7). In the control group, the architectural layout of hepatocytes appeared normal. All the nuclei were round, and no breakage of organelles was observed. On the other hand, treatment with NCD 1 was characterized by dilated sinusoidal spaces, while NCD 2 presented with vacuolated hepatocytes and cellular infiltration.

(a to c) Representative photomicrographs of liver slides from nanocellulose oxalate ester-treated rats. NCD 1 received 50 mg kg−1 body weight and NCD 2 received 100 mg kg−1 body weight (magnification ×100) H&E. Normal architectural layout of hepatocyte with no visible lesion (control), dilated sinusoidal spaces (arrowed; NCD 1), vacuolated hepatocytes (short arrow), and cellular infiltration (arrowhead; NCD 2). NCD: nanocellulose modified with oxalate ester; H&E: hematoxylin and eosin.
Discussion
Nanoparticles are poorly absorbed from the gastrointestinal tract (GIT). The acidic milieu of the GIT and its high content of degradative enzymes contribute to poor absorption properties of nanosized materials as well as other orally administered xenobiotics. Orally administered nanoparticles absorbed from the intestine could be transported either through mesenteric vessels to the liver or through the lymphatic system en route the systemic circulation. 36 Fortunately, there has been a report of the retention of nanoparticulates in the liver after ingestion. Schleh et al. 37 showed that absorption of nanoparticles across intestinal membranes and subsequent accumulations in secondary organs were dependent on the particle size and surface charges. The authors concluded that smaller size and a negative charge improved absorption and accumulation of nanoparticles. Corroborating this study was another research that showed the preferential accumulation of charged nanoparticles, over neutral and zwitterions nanoparticles, in the hepatic tissue. 38 In our present study, we report a monomodal particle size of 0.0168 µm and zeta potential range of −50 to −10 mV for NCD. These are favorable physicochemical properties that boost absorption and target organ accumulation. This study, therefore, evaluated the hepatotoxicity and oxidative stress potential of graded doses of CSN modified with oxalate ester. ALT and AST, classified as aminotransferases because of their roles in transamination reactions during amino acid metabolism, are clinical markers of liver function. The elevation in the plasma levels of these enzymes, as observed in this present study, is an indication of the onset of hepatic injury. 39 Albumin is a blood plasma protein synthesized in the liver. It has been reported that reprioritization of acute-phase protein response involves the decrease of acute-phase proteins, including albumin and transferrin. This adaptive response paves the way for the increased synthesis of the positive acute-phase proteins involved in the host defense mechanism. 40,41 However, reports on albumin synthesis sequel to inflammation are conflicting. While some studies showed a decrease in albumin levels, 42,43 albumin synthesis was found to be increased in others. 44,45 The increase in albumin as observed in the present study could be explained in line with the suggestion of Mansoor et al. 46 that there may be a biphasic pattern to albumin synthesis in response to extrinsic insult. It is also possible that the elevated total hepatic protein levels, observed in the current study, contributed to the elevated albumin synthesis at the site of albumin messenger ribonucleic acid transcription. 47 Interestingly, the elevation in albumin levels was more pronounced in the smaller dose than in the larger. The surface properties of NCD could have contributed to this effect. Higher doses of nanoparticles have been reported to favor agglomeration. 48 The larger nano-aggregates of NCD 2, which suppressed intra-surface hyperactivity, could be responsible for the reduced level of albumin when compared with NCD 1. This observation also confirmed the unreliability of nano-sized materials in the determination of the no-observed-adverse-level effects. 48 Oxidative stress occurs when there is a distortion of the antioxidant–oxidant balance in the tissue. It is often accompanied by depletion of GSH and an increase in lipid peroxidation. In this present study, biomarkers (SOD, GSH, H2O2, NO, and MDA) were not modulated by the administration of NCD. In our previous study, we had reported that administration of sulfonated CSN to rats at the doses of 50, 75, and 100 mg kg−1 did not alter GST, MDA, H2O2, and NO levels. 23 Interestingly, NCD treatment decreased CAT and GPx and elevated MPO activities. The triad of SOD, CAT, and GPx are known as the first-line antioxidants. SOD dismutates superoxide radical into H2O2 and molecular oxygen, while CAT and GPx convert H2O2 into H2O. When neutrophils are recruited to the site of inflammation, their intrinsic enzymes, MPO catalyzes the formation of hypochlorous (HOCl) acid by acting on H2O2 and Cl− ions. The observed decrease in CAT and GPx activities could suggest that they were ineffective in removing H2O2 at the site of inflammation. A similar trend was observed with an earlier report showing the inactivation of CAT and GPx by HOCl at inflammatory sites. 49 CAT and MPO are important enzymes in the complex mechanism of maintaining a low level of reactive oxygen species (ROS) in the body, and their interrelationship has been of interest. 50 –52 In this study, MPO activity increased in liver at the high-dose NCD exposure, thereby disrupting the ROS balance. The increase in MPO activity and the subsequent formation of HOCl could be the source of oxidative stress. The observed increase in MPO also correlated with histopathological aberrations in the liver of the animals treated with NCD.
Inflammation and apoptosis, in addition to oxidative stress, are key mechanisms of hepatic injury. As in this present study, the role of hepatic iNOS has been investigated in rodent models. 53 We observed the presence of iNOS immunoreactivity in the hepatocytes of control and treated groups. This finding is consistent with that of a previous study, which showed constitutive iNOS expression in normal hepatocytes, 54 although the intensity of staining, and hence protein expression, was more pronounced in the group administered with NCD 1(50 mg kg−1 body weight) than in the NCD 2 (100 mg kg−1 body weight) group. The increase in the expression of iNOS could contribute to liver injury through inflammation and oxidative stress in NCD-treated rats.
Furthermore, NCD elicited an increased expression of the proapoptotic protein, Bax. This observed elevation in expression could be as a result of the regulatory action of Bax genes which culminated in increased Bax protein levels and subsequent translocation into the mitochondria, thereby disintegrating the membrane and causing cell death. 55 It is plausible that cellular oxidative stress could have triggered this apoptotic process. As was the case with iNOS expression, the upregulation of Bax was not dose dependent. The nonlinearity of the response could be due to the unique properties of NCD. Nanoscaled materials, at a given low dose, can exert either benign or toxic effects depending on their size and surface reactivity. 56 Furthermore, the pathological observations of parenchymal disorganization and severe cellular infiltration observed in the liver of rats treated with NCD, at the various concentrations, are consistent with rat liver histoarchitecture in a recent study. 57
Conclusion
In summary, the introduction of oxalate ester functional group at the surface of CSN might increase its applications. However, short-term exposure of rats to the chemically modified CSN reveals its potential hepatotoxicity via elevation of the aminotransferases, depletion of CAT activity and exacerbation of myeloperoxidase activity, culminating in the induction of iNOS and Bax proteins. These data provide useful information on the mechanisms of toxicity of chemically modified nanoscaled CSC.
Footnotes
Acknowledgements
Authors are grateful to TWAS-CNPq for the provision of postdoctoral fellowship to AA. Authors also acknowledge the support received from Prof. Pereira Vargas Fabiano and the Department of Chemistry, Universidade Federal de Minas Gerais, Minas Gerais, Brazil.
Author contributions
CAO and AA were involved in conceptualization, manuscript preparation, and editing. OA was involved in laboratory experiments and project supervision, while IE was involved in data curation and formal analysis.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
