Abstract
Hydroethanolic preparations of Acmella oleracea is used in the north of Brazil as a female aphrodisiac. Thus, the objective of this study was to evaluate the action of the hydroethanolic extract of Acmella oleracea (EHFAo) flowers (21.873 and 44.457 mg/kg) and spilanthol (3 mg/kg) administered orally on reproductive performance and effects on the embryonic development of zebrafish F1 generation. It was observed that in the groups in which males and females received EHFAo and spilanthol, the spawning was interrupted, whereas in the groups in which only the females were treated, spawning occurred during the 21 days. Thus, in the histopathological evaluation of the gonads, it was possible to observe that the percentage of mature cells in the spermatozoa and females was significantly reduced. Only the embryo groups in which parental generation was treated with EHFAo showed lethal and teratogenic effects. On the other hand, the parental groups treated with the spilanthol presented only the lethality. Spilanthol and some metabolites showed good oral availability and important toxicological properties. Thus, it is suggested that the treatment of parental generation of zebrafish with EHFAo and spilanthol caused severe changes in the gonads and on fertility. However, on the embryo, the most striking effects in the development were recorded in the groups in which the parental generation was treated with the EHFAo, while the spilanthol influenced the lethality of the embryos.
Introduction
The Acmella oleracea (L) R. K. Jansen, popularly known as jambu, 1 belongs to the family Asteraceae, native to the Eastern Amazon and most cultivated in the Brazilian states of Pará and Amapá, where it integrates local cuisine and is widely used in folk medicine. 2,3
Spilanthol, (2E,6Z,8E)-N-isobutyl-2,6,8-decatrienamide, is the main chemical constituent found in this plant species. It is an alkylamide very well known for its numerous pharmacological properties. 4 –7 Several popular leaves and inflorescence preparations have been used for the treatment of diseases of the mouth and throat, tuberculosis, 6,7,8 used as diuretics, 3,9,10 in the treatment of rheumatism, 11 as anti-inflammatory, analgesic and local anesthetic, 12 –17 in the treatment of influenza and cough, as antibacterial, antifungal, antimalarial, 18 –21 and as an insecticide. 11,18,22,23 Also, the inflorescences, 2 the part of the plant in which the spilanthol is more concentrated, 13,14,15 is commonly used as a local anesthetic in toothaches.
Spilanthol has been suggested in previous studies as a sexual stimulator. 24,25 However, there is still little knowledge about its action on gonads and embryo–fetal development, while there is a growing interest in the pharmacological evaluation of this species and its major component.
The search for new therapies that aid in the understanding of various diseases has grown, and in this sense, the advantages of zebrafish as an experimental model have become more evident, such as small size, extrauterine development, transparent embryos, and the short reproductive cycle. It is an important organism in terms of translation due to the degree of homology with the human genome. 26 –28
Methodologies that assess reproductive toxicity by immersion are already widely used 28,29 and have shown efficiency in the evaluation of the toxicological potential of substances present in water. However, there are no reports in the scientific literature of studies evaluating oral reproductive toxicity.
Bioavailability, metabolism, and biotransformation affect the safety and efficacy of drugs and other xenobiotics for humans. 30 Considering the restrictions for the study of the metabolism of xenobiotics in the human body in experimental in vitro and in vivo tests, a computational approach is currently known as “the method of choice.” 31,32
In this sense, the objective of this study was to evaluate the reproductive performance of zebrafish treated orally with EHFAo and spilanthol and evaluate the effects on the embryonic development of the F1 generation, correlating with the in silico studies of the metabolites of the major compound of EHFAo, spilanthol, and its toxicity in the human organism.
Materials and methods
Chemical substances
The solvents used were of analytical quality: hexane (Hex), ethanol (EtOH), ethyl acetate (EtOAc), dichloromethane (DCM), and methanol (MeOH) obtained from Tedia® Company (Fairfield, Connecticut, USA). To obtain the nuclear magnetic resonance (NMR) spectra, deuterated chloroform (CDCl3;Cambridge Isotope Laboratories, Andover, Massachusetts, USA) was used.
Plant material
The flowers of Acmella oleracea were collected in August of 2017 in the municipality of Santa Izabel, State of Pará, Brazil. The exsiccata was deposited in the Herbarium of the Brazilian Agricultural Research Company, EMBRAPA/CPATU, Eastern Amazonia under registration 197388.
A total of 7.0 kg of Acmella oleracea flowers were collected, then dried in an oven with temperature and controlled humidity (T = 40°C, U = 40%) according to the method described by Salazar et al., 33 and the material was ground in a mill of knives and hammer.
Preparation of the extract
The extract was obtained by maceration with EtOH, in the ratio of 1.0 kg of biomass to 5.0 L of solvent, after which the solution was concentrated in a rotary evaporator Syncore BÜCHI (Flawil, Switzerland) to obtain the extract of flowers (EHFAo).
High-performance thin-layer chromatography
Chromatographic analysis was performed using a high-performance thin-layer chromatography (HPTLC) robotic thin-layer chromatographic system composed of the application modules (Automatic TLC Sample 4 (ATS4)), densitometer (TLC Scanner 4), and the photodocumentary (TLC Visualizer) of the brand CAMAG (Muttenz, Switzerland).
Sample preparation and application
For chromatographic analysis (HPTLC-densitometry), a stock solution of the EEFAo extract at 5000 ppm in MeOH was prepared. Aliquots 5 and 10 μL (25 and 50 μg, respectively) were applied (triplicate) on silica gel chromatographs TLC Aluminum F-254 60 Ă (SILICYCLE, Quebec, Canada).
Chromatographic procedures
The chromate plates were eluted in CAMAG glass vat (Muttenz, Switzerland) with an isocratic system consisting of Hex/EtOAc (7:3) and a chromatographic path of 90 mm. After elution, the plates were photo-documented (TLC Visualizer) under ultraviolet radiation at wavelengths of 254 and 366 nm.
Densitometry
The chromatograms and UV/Visible spectrum were obtained by HPTLC-densitometry using deuterium lamp, Savitsky–Golay 15 filter, wavelength 229 nm, slit 4.00 × 0.30 mm2, with scanner speed of 20 mm/s, resolution of data 100 μm/step.
Isolation and structural identification
Fractionation and isolation
A 10 g aliquot of the EHFAo was dissolved in 1 L of 8:2 MeOH-H2O and partitioned with Hex and DCM, resulting in the FrHEX and FrDCM fractions, and then analyzed by TLC (Hex/EtOAc 7:3). The FrDCM fraction was purified by column chromatography (isocratic Hex/EtOAc 7:3), resulting in the isolation of spilanthol (330 mg) (S1).
Compound characterization
The isolated compound (S1) was identified by applying proton nuclear magnetic resonance (1H NMR) and Carbon-13 nuclear magnetic resonance (13C NMR) spectroscopy. NMR spectra were obtained on a Bruker, Ascend™ (Rheinstetten, Germany) spectrometer, operating at 400 MHz for 1H and 75 MHz for 13C. The TopSpin 3.6.0, software, was used for control and data processing and displacements were recorded in dimensionless values δ (ppm), with the solvent reference being the internal reference.
Animals
The project was approved by the Animal Use Ethics Committee of the Federal University of Amapá—CEUA-UNIFAP, receiving protocol number 002/2018.
Animals of both sexes of Danio rerio species (wild AB strain, 6 months old, 3.5–4.0 cm long and weighing on average 650 mg, out of a total of 84 fish) were obtained from Acqua New Aquarium and Fish Ltd (Igarassu-PE, Brazil). The animals were quarantined and later acclimated for a week before the experiments at the Zebrafish Platforms of the Laboratory of Research on Drugs of the Federal University of Amapá.
These animals have been kept in a maintenance system with running water, at a temperature of 25–29°C, pH 8.4–8.6; toughness: 140–145 mg/L calcium carbonate; O2 dissolved 90%, with an average density of one fish/L, and photoperiod of 12 h light/12 h dark in an aquarium with 25 L of water. The concentrations (in mg L−1) of the predominant ions in the water of the maintenance system were
Experimental design
After the acclimation period, the animals were randomly divided into seven groups with 12 animals in each of them, in the ratio of two males to one female. One week before the start of the experiment with EHFAo and spilanthol, the fertility test was performed with maintenance water only, during which time the eggs were collected, quantified, and evaluated at up to 96 hpf.
After the fertility test, oral administration for 21 days of EHFAo at doses of 21.873, 44.457 mg/kg12 and spilanthol at a dose of 3 mg/kg were performed in a semi-static test with 70% daily water renewal, according to the methods described by Carvalho et al., 35 Borges et al., 36 Sampaio et al., 37 and Melo et al. 38 The doses were established from a preliminary acute toxicity test (48 h).
Eggs, mortality, and teratogenicity were evaluated at up to 96 hpf. The following groups were established: A—Control group (Saline solution); B—21.873 mg/kg of EHFAo diluted in 1 ml of saline solution (treated males and females); C—21.873 mg/kg of EHFAo diluted in 1 ml of saline solution (treated females only); D—44.457 mg/kg of EHFAo diluted in 1 ml of saline solution (treated males and females); E—44.457 mg/kg EHFAo diluted in 1 ml saline solution (treated females only); F—3 mg/kg of spilanthol diluted in 1 ml of saline solution (males and females treated); and G—3 mg/kg of spilanthol diluted in 1 ml of saline solution (females only).
Collection and maintenance of eggs
After mating and spawning, fertilization occurred within 30 min after the lights went on. The eggs were collected, quantified, and washed with water, then stored in inert Petri dishes containing water from the maintenance system with 70% daily renewal and incubated in an oven (SOLAB SL-102/630) at 28°C ± 2 per 96 hpf. 39
Histopathological analysis of gonads
After 21 days of treatment with EHFAo and spilanthol, the animals were euthanized according to the methodology described by Castro 40 : fixed in bouin solution, decalcified in 7% ethylenediaminetetraacetic acid solution, processed, sectioned in the thickness of 6 μm, and stained with hematoxylin and eosin. 41 The sections obtained were examined under light microscopy (Olympus BX41), scanning by electron microscopy and photographed using a digital camera (ScopeImage 9.0 USB, Bioimager Inc., USA).
The ovaries of the four females and the testes of the eight males of each treatment were analyzed individually, in a total of 10 sections per animal and classified according to OECD. 42
The ovarian follicles were classified into five stages: oogonia/oocytes (Oo), perinucleolar oocytes (PO), cortical alveolar oocytes (CAO), early vitellogenic oocytes (EVO), and late mature oocytes. The testis, in four stages: spermatogonia (Sg), spermatocytes (Sc), spermatids (St), and spermatozoa (Sz), according to Liang et al. 29 and Blüthgen et al. 43
The gonad data were evaluated by calculating the number of each stage (follicular and testis) and expressed as the percentage of pathological changes.
Assessment of the lethal and teratogenic effect
Embryos were classified according to the severity of morphological defects and signs of toxicity according to Table 1, based on daily observations as 24, 48, 72, and 96 hpf evaluations.
Lethal and teratogenic effects observed on zebrafish embryos throughout development.

+: hour at which the change was observed in the embryos; hpf: hours postfertilization.
aCoagulated eggs are milky white and appear dark under an optical microscope.
bThere is no record of heartbeat for at least 1 min.
Embryo photomicrographs live in lateral orientation (N = 10 embryos/treatment) were analyzed using Scion Image software (Scion Corporation, Maryland, USA) to determine body length (mm).
Statistical analysis
For analysis of the egg production data and histopathology of the gonads, one-way analysis of variance (ANOVA) followed by the Tukey test was used for comparison with the control and the doses between them. The results with p <0.05 were considered statistically significant.
In silico oral bioavailability study
The oral bioavailability of spilanthol and the molecules M8, M18, M26, M28, M33, and M36 were analyzed using the Molinspiration server, seen at http://www.molinspiration.com, based on the rule of the five of Lipinski et al. 44 The rule states that most of the “drug-like” compounds have a molecular weight (PM ≤ 500 Da), number of hydrogen bond acceptors (HBA ≤ 10), number of hydrogen bond donors (HBD ≤ 5), and partition coefficient octanol/water (log P ≤ 5). Compounds that do not fall into more than one of these rules may have problems with bioavailability. Molinspiration calculates important molecular properties such as molecular weight (MW), log P, polar surface area, number of hydrogen bond donors and acceptors. To identify the undesirable toxic properties of spilanthol and the six metabolites, the toxicity prediction server (Protox), seen at website http://tox.charite.de/tox/, was used in this study. The prediction was based on group similarity for the reference compounds with the in vitro and in vivo validated compounds present in this database. Toxic properties, such as toxicity class, generation of toxic fragments, median lethal dose (LD50) values in mg/kg, toxicity targets, relevant drug properties, log S (solubility), MW, and overall drug score were calculated. 45,46 This approach was based on studies by Roy et al. 32
Determination of toxicological properties
Spilanthol and its metabolites were calculated using the PreADMET online server available at https://preadmet.bmdrc.kr/. This server calculates toxicological properties such as mutagenicity and carcinogenicity.
Results
Chromatographic study
The chromatographic analyses by HPTLC-densitometry allowed characterizing the EEFAo extract as to its complexity, presence, and content of spilanthol. The plates after elution were photo-documented under ultraviolet radiation (254 and 366 nm) and indicated a low complexity—viewing of few bands (Figure 1). From the densitometric analysis it was possible to obtain the chromatogram at the wavelength (λ) 229 nm and the UV/Visible spectrum of the substances present in the extract, thus it was possible to identify spilanthol (Rf 0.41) from its UV/Visible spectrum and its abundance (Figure 2).

Chromate plate of the ethanolic extract of Acmella oleracea flowers (EHFAo), visualized under ultraviolet radiation 254 and 366 nm. Visualization under irradiation: (a) 254 nm; (b) 366 nm; (E1) EHFAo aliquot of 25 μg/spot; (E2) EHFAo aliquot of 50 μg/spot. EHFAo: hydroethanolic extract of Acmella oleracea.

The chromatogram in λ = 229 nm of the ethanolic extract of the flowers of A. oleracea (EHFAo) and UV/visible spectrum of the band Rf 0.41 (Spilanthol). (a) Chromatogram under irradiation 229 nm; (b) UV/visible spectrum of the Rf band 0.41. EHFAo: hydroethanolic extract of Acmella oleracea. UV: ultraviolet.
Structural elucidation: Compound 1
Compound S1 (yellow oil; Figure 3) showed solubility in chloroform and its structure was determined by 1H and 13C NMR (CDCl3, 400 and 75 MHz), with the values: [(H-2: [δH 5.79 d (J = 14.9)/C-1 δC 124.1]; H-3: [δH 6.83 dt (J = 14.9;7.1)/C-3 δC 143.6]; H-4: [δH 2.27–2.35 m/C-4 δC 32.1]; H-5: [δH 2.27–2.35 m/C-5 δC 26.3]; H-6: [δH 5.27 dt (J = 10.9; 7.1)/C-6 δC 127.6]; H-7: [δH 5.96 dd (J = 11.1; 15)/C-7 δC 129.4]; H-8:[δH 6.28; dd (J = 11.1; 15)/C-8 δC 126.7]; H-9: [δH 5.71 dq (J = 14.9; 7)/C-9 δC 130.0]; H-10: [δH 1.79 d (J = 7)/C-10 δC 18.3]; H-N: [δH 5.48 s/C-10 δC 166.0]; H-1′: [δH 3.15 dd (J = 7; 6) / C-1′ δC 46.9]; H-2′:[δH 1.77 m/C-2′ δC 28.5]; H-3′/H-4′:[δH 1.77 m/C-3′/4′ δC 20.1], and by comparison with the literature data. 47
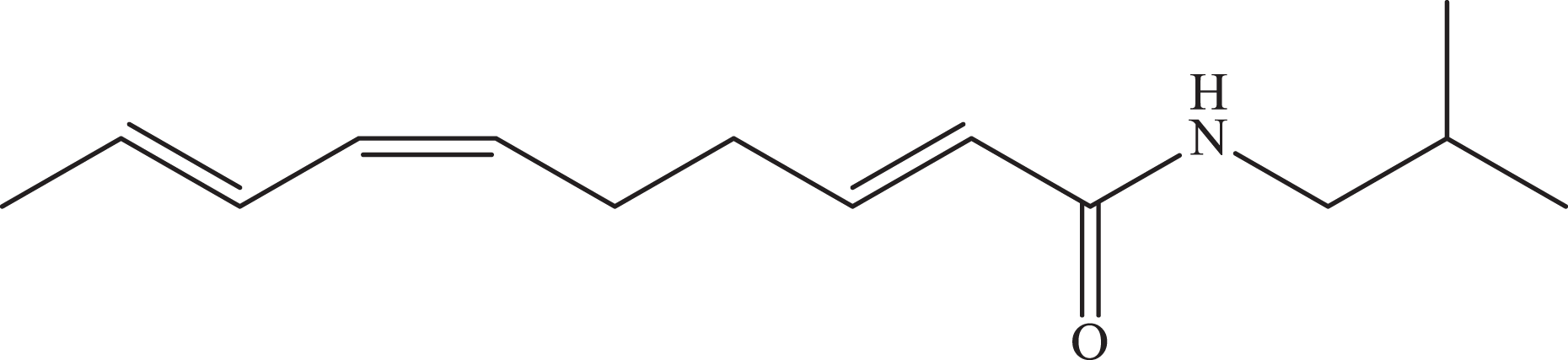
Molecular structure of spilanthol.
Fertility
In the pretreatment period, spawning occurred at regular intervals for all groups with 100% fertilization and hatching (Figure 4).

Total cumulative eggs per group during the 7-day pretreatment period and control treatment (saline solution) and doses of 22.873 and 44.457 mg/kg EHFAo and 3 mg/kg of spilanthol for 21 days. The pre-exposure period is indicated for 1–7 days, followed by the indicated exposure period of 8–28 days. EHFAo: hydroethanolic extract of Acmella oleracea.
During the treatment period, it was observed that the spawning average of the control group (36.14 ± 4.79; Figure 5) as the cumulative total was higher in comparison to the other treatment groups, indicating that the EHFAo extract (22.873 and 44.457 mg/kg) and spilanthol (3 mg/kg) caused a reduction in egg production during the 21 days of treatment.

Average production of zebrafish eggs collected daily during the 21 days of treatment with EHFAo and spilanthol. Egg production was significantly reduced in the groups treated with the highest dose of EHFAo and spilanthol. The total number of eggs of each concentration was compared to each other and to the control group using GraphPad Prism 5.0 software and the statistically significant results are represented by “a” (p > 0.05). EHFAo: hydroethanolic extract of Acmella oleracea.
At the doses of 22.873 and 44.457 mg/kg of EHFAo, it was observed that the groups in which only the females were treated spawned throughout the 21 days of the experiment. However, the group treated with doses of 44.457 mg/kg presented a statistically significant reduction when compared to the control group. On the other hand, groups in which females and males were treated with the same doses did not spawn during the 21 days of treatment.
In the case of spilanthol with the dose of 3 mg/kg, and according to Figure 2, it is possible to observe that in the group in which only the females received the treatment, the spawning occurred during the 21 days of treatment, and in the group in which males and females were treated, the spawning was interrupted from the 13th day of treatment (Figure 4). It was observed that spawning in these groups was lower than the treatment groups with EHFAo in the doses of 22.873 and 44.457 mg/kg, and the reduction in egg production was statistically significant when compared to the control (Figure 5).
Histopathology of the gonads
All stages of ovarian and testis development (Figures 6 and 8) of adult zebrafish were observed in control (saline solution) and at different treatment doses of EHFAo (22.873 and 44.457 mg/kg) and spilanthol (3 mg/kg). This treatment resulted in visible histopathological changes in female ovaries and male testicles (Figures 7 to 9).

Histological section of ovaries of adult zebrafish females after 21 days of the control treatment (saline solution) (a), 22.873 mg/kg EHFAo (b), 44.457 mg/kg EHFAo (c), and 3 mg/kg spilanthol (d). The ovaries were categorized in the following stages: Oo, PO, CAO, EVO, and LMO (H&E). EHFAo: hydroethanolic extract of Acmella oleracea; Oo: oogony/oocyte; PO: perinuclear oocytes; CAO: cortical alveolar oocytes; EVO: early vitellogenic oocytes; LMO: late mature oocytes; H&E: hematoxylin and eosin.

Effects of EHFAo and spilanthol on gonadal development of zebrafish. Percentages of each stage in the ovaries of zebrafish females (Oo, PO, CAO, EVO, and late mature ovarian oocytes). Values represent the average ± SEM of four individually analyzed animals. Significance between control and treatment groups is indicated by a, b, c, and d with p < 0.05. EHFAo: hydroethanolic extract of Acmella oleracea; Oo: oogony/oocyte; PO: perinuclear oocytes; CAO: cortical alveolar oocytes; EVO: early vitellogenic oocytes; SEM: standard error of mean.

Testicular histological section of adult zebrafish males after 21 days of treatment at the control group (saline solution) (a), 22.873 mg/kg EHFAo (b), 44.457 mg/kg EHFAo (c) and 3 mg/kg spilanthol (d). The testes were categorized into the following stages: Sg, Sc, St, and Sz (H&E). EHFAo: hydroethanolic extract of Acmella oleracea; Sg: spermatogonia; Sc: spermatocytes; St: spermatids; Sz: spermatozoids; H&E: hematoxylin and eosin.

Effect of EHFAo and spilanthol on gonadal development of zebrafish. Percentages of each stage in the testicles of zebrafish (Sg, Sc, St, Sz). Values represent the average ± SEM of eight animals individually analyzed. Significance between control and treatment groups is indicated by a, b, c, and d with p < 0.05. EHFAo: hydroethanolic extract of Acmella oleracea; Sg: spermatogonia; Sc: spermatocytes; St: spermatids; Sz: spermatozoids; H&E: hematoxylin and eosin.
In females, Figure 7 shows that the percentage of oocyte/oocyte (Oo) and PO were significantly increased in the groups treated with EHFAo and spilanthol (Figure 3(b) to (d)). However, for the percentage of CAO, only the groups treated with the doses of 22.873 and 44.457 mg/kg of EHFAo were statistically reduced.
In contrast, the percentage of immature (EVO) and mature ovarian (EVO) oocytes were strongly decreased after treatment with the doses of 22.873 and 44.457 mg/kg of EHFAo and 3 mg/kg of spilanthol (Figures 6(b) to (d) and 7).
In males, there was no significant difference in the percentage of Sg after treatment with EHFAo and spilanthol (Figures 8(a) to (d) and 9), whereas a significant increase in the percentage of Sc was observed in the group treated with a dose of 22.873 mg/kg EHFAo (Figure 9). However, the percentage of St was significantly increased only in the group treated with the doses of 44.457 mg/kg of the EHFAo (Figure 9). The percentage of Sz was significantly reduced in all treatment groups, being more evident in the group treated with the 44.457 mg/kg dose of EHFAo.
Lethal and teratogenic effect
The coagulation and absence of heartbeat were evidencing embryonic death, recorded at up to 96 hpf, were the lethal effects observed in the embryos where the parental generation was treated with the extract with the doses of 22.873 and 44.457 mg/kg of the EHFAo (Table 2).
Overview of the teratogenic and lethal effects of EHFAo and spilanthol in zebrafish embryos at 96 hpf.
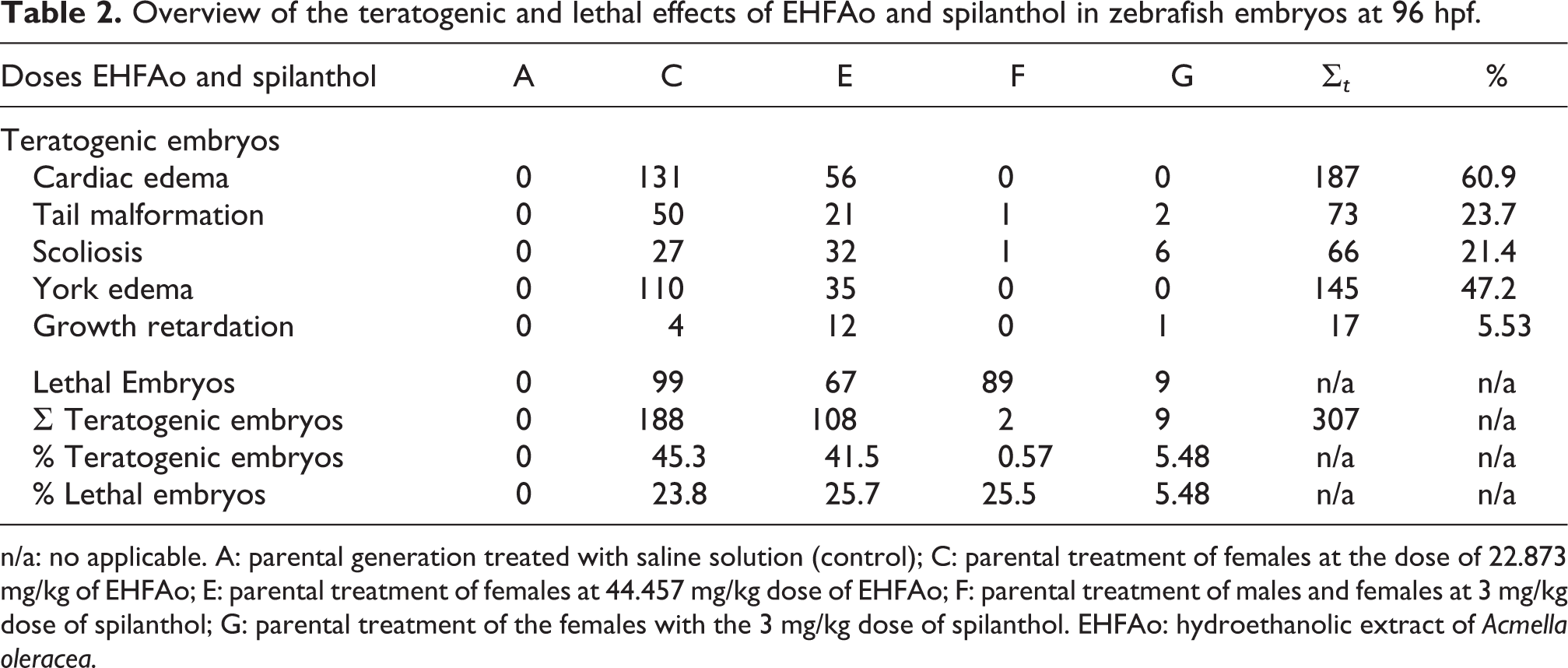
n/a: no applicable. A: parental generation treated with saline solution (control); C: parental treatment of females at the dose of 22.873 mg/kg of EHFAo; E: parental treatment of females at 44.457 mg/kg dose of EHFAo; F: parental treatment of males and females at 3 mg/kg dose of spilanthol; G: parental treatment of the females with the 3 mg/kg dose of spilanthol. EHFAo: hydroethanolic extract of Acmella oleracea.
During the remaining hpf of evaluation, no coagulation was recorded in any of the groups. However, embryonic mortality was recorded at up to 96 hpf, mainly due to the occurrence of several changes in a single embryo.
The number of embryos that presented at least one malformation was registered more frequently between 48 and 72 hpf, since in that period all larvae had already hatched (Figure 10), thus allowing better visualization of the structures; in 96 hpf, there was a decrease in the registry of malformations in all groups.

Representative micrographs of embryos and zebrafish larvae, in which parental generation was treated for 21 days with EHFAo (C and E groups) and spilanthol (F and G groups). A (control group). (a) Normal larvae with 96 hpf. (b)(c) parental treatment of females at the dose of 22,873 mg/kg of EHFAo. (d)(e) parental treatment of females at dose of 44.457 mg/kg of EHFAo. (f) parental treatment of males and females at the dose of 3 mg/kg of spilanthol. (g)(h)(i) parental treatment of the females at the dose of 3 mg/kg dose of spilanthol. EHFAo: hydroethanolic extract of Acmella oleracea; Ce: Cardiac edema; Tm: malformation on the tail; Sc: scoliosis; Ye: edema in the yolk; Dn: no development at 24 hpf; hpf: hours postfertilization.
The most severe and most evident malformations recorded were edema in the yolk (Figure 10(c), (f), and (g)) and cardiac edema (Figure 10(c), (f), and (h)). The total number of cardiac edema recorded was 131 and 56, and edema in the yolk was 110 and 35 for groups C and E of EHFAo, respectively. The larvae that presented these changes did not survive after 96 hpf. In the groups in which parental generation was exposed to 3 mg/kg doses of spilanthol, these changes were not recorded.
The tail deformity (Figure 10(e) to (i)) and scoliosis (Figure 10(b), (d), and (f)) were widely observed changes for both groups in which parental generation was treated with EHFAo and spilanthol in all doses tested. It was observed that these alterations resulted in a reduction of the movement capacity of the larvae after hatching. The least frequent change was the growth retardation in which 4, 12, and 1 occurrence was recorded for groups C, E, and G, respectively (Table 2).
Oral bioavailability
The pharmacological properties of spilanthol and its metabolites, M8, M18, M26, M28, M33, and M35, are shown in Table 3. They were evaluated according to Lipinski’s rule of five, curiously all metabolites and spilanthol that were selected presented weight (MW) in the range of 221.34–526.65 Da. In Table 3, only M28 presented a value higher than 500 Da, which will make it difficult to transport, to diffuse, and to absorb.
ADME properties of spilanthol and metabolites.

hpf: hours postfertilization; MW: molecular weight; HBA: hydrogen bond acceptor; HBD: hydrogen bond donor; log P: Logarithm of the partition between of n-octanol and water phases; MPSA: molecular polar surface area; MV: molecular volume.
The number of hydrogen bonding receptors (O and N atoms) and the number of hydrogen bonding donors (NH and OH) for metabolites and spilanthol were found within the Lipinski limit, and the others range from 2 to 3 and 0, this is, less than 10 and 5, respectively, seen at Table 3.
The value of log P varied from 3.55 to −2.54 and the Lipinski rule limit was acceptable for all metabolites, and spilanthol showed the highest value of the metabolites studied.
The molecular polar surface area (MPSA) or topological polar surface area (TPSA) is a very useful parameter for the prediction of drug transport properties.
The values for this property are shown in Table 3 and variation from 29.10 to 213.22 Å2. Molecular volume (MV) determines the transport characteristics of molecules, such as intestinal absorption or blood–brain barrier penetration. In this study, the values varied from 243.19 to 490.48 Å2, seen in Table 3.
Good bioavailability is more likely for compounds with rotational bonds ≤10 and MolPSA of ≤140 Å, and only spilanthol and its metabolites M8, M18, M26, and M35 have 8–9 rotating and flexible bonds, while M28 and M35 have 22 and 10 rotating links, respectively.
Table 4 clearly shows that the LD50 prediction value was 4.378 mg/kg for spilanthol and the other metabolites varied between 225 and 5000 mg/kg. Depending on the toxicity, the classes are defined according to the globally harmonized Classification of Chemicals Labeling System (GHS). Only M26 metabolite was classified as toxic by ingestion, spilanthol and other metabolites were classified as possibly harmful by ingestion.
Prediction of oral toxicity of the compounds.

LD50: median lethal dose.
aValues in mg/kg body weight.
bClass I: fatal if swallowed (LD50 ≤ 5); Class II: fatal if swallowed (5 < LD50 ≤ 50); Class III: toxic if swallowed (50 < LD50 ≤ 300); Class IV: harmful if swallowed (300 < LD50 ≤ 2000); Class V: may be harmful if swallowed (2000 < LD50 ≤ 5000); Class VI: non-toxic (LD50 > 5000).
Pharmacokinetic properties and toxicological properties
Computed metabolism (Table 5) has shown that spilanthol, M8, M26, and M33 are cytochrome P450 2D6 inhibitors. However, only the M28 metabolite presented inhibitory properties against cytochrome P450 3A4 and M18 substrate for cytochrome P450 3A4 and inhibitor for P-glycoprotein.
Metabolic characteristics of spilanthol and metabolites.

Non: no occurrence.
In the Ames method for the evaluation of mutagenicity for spilanthol and other metabolites, it was observed that only the M28 metabolite was predicted as non-mutagenic, spilanthol and other selected metabolites (M8, M18, M26, M33, and M35) present positive predictions, meaning that they were predicted to be mutagenic.
In the prediction of carcinogenicity in rats, the spilanthol and the metabolites M8, M26, M28, and M33 obtained a positive prediction, and there is no evidence of carcinogenic activity. The M18 and M35 metabolites analyzed were negatively predicted, which means that there is carcinogenic activity evidence in rats. In the prediction of carcinogenicity in mice, all selected metabolites showed a positive prediction demonstrating that these metabolites do not present carcinogenic effects (Table 6).
Prediction of toxicological properties of metabolites.

Discussion
As spilanthol is an amphiphilic compound, 47 it can be extracted from plants using solvents that vary in polarity such as Hex 18 and MeOH:H2O (4:1, v/v). 48 Studies report that HPLC quantification with UV detection allows the identification of the content of spilanthol in different extracts, 24,49,50,51 as it was observed in this study. Spilanthol showed solubility in chloroform and it had its structure determined by NMR, according to the data compared in the literature. 52
Spilanthol (Figure 3) is an N-alkylamide with varied bioactivity, 52,53 including analgesic activity, 6,52,54 –61 antinociceptive, 62 antioxidant, 58 anti-inflammatory, 56,63 antimutagenic, 64 anti-wrinkle, 65 antifungal, 52 bacteriostatic, 66 insecticides, 11,57 antimalarial, 57 anti-larvicidal activities against Aedes aegypti and Helicoverpa zea neonates, 18 and anti-molluscicidal activities. 67 There are also reports of its activities as an anticonvulsant, antioxidant, aphrodisiac, pancreatic lipase inhibitor, antimicrobial agent, antinociceptive agent, diuretic, vasorelaxant, human immunodeficiency virus, toothache relief, and as an anti-inflammatory. 52
Several authors affirm that EHFAo and spilanthol present characteristics that facilitate their absorption by the intestine. 53 It is known that oral treatment has the intestine as its first contact organ, which guarantees the absorption of treatment doses.
The results of this present study indicated that treatments with oral EHFAo and spilanthol caused reproductive dysfunction in adult zebrafish. Despite stimulating mating at all doses tested, treatment for 21 days affected gonadal development impairing reproductive success in males. This condition was not observed in rats since the treatment with Spilanthes acmella extract improved sexual performance in males, which was indicated by penile erection and frequency of assembly, intrusion, and ejaculation. 24
After 21 days of treatment with EHFAo and spilanthol, egg production was reduced in all treatment groups, and in the groups where males and females were treated with the doses of 22.873 and 44.457 mg/kg of the extract, there was no production of eggs during the 21 days in this study. This result suggests that both EHFAo and spilanthol at the tested doses showed a progestin profile with a significant effect on the reduction of egg production such as progesterone, 68,69,70 levonorgestrel, 71,72 norethindrone, 73 gestodene, 74 drospirenone, 69 desogestrel, 71 megestrol acetate, 74 and dydrogesterone. 75
Several studies have reported that even at low concentrations several substances can inhibit reproduction in different fish species such as norethindrone and gestodene that inhibited egg production in Japanese minak and medaka, 71,73 drospirenone and desogestrel, 71,72 and progesterone in zebrafish. 71
The effects of EHFAo and spilanthol doses on egg production and fertilization seem to be related to their action on gonadal steroids. It is already well established that the endocrine system mediates the control of reproductive activities, and each hormone plays a specific role in the different stages of the reproductive process. 75 Sexual steroids are necessary for spermatogenesis and spermiogenesis, sexual differentiation and sexual maturation, 76 being also significantly involved in all phases of behavior associated with reproduction. 77 In this study, it was observed that there was a significant decrease in egg production when compared to the control group.
According to Kime, 78 gonadal steroid levels may affect the quantity and quality of both ova and Sz during gametogenesis. Thus, treatment with EHFAo concentrations seems to inhibit the biological pathways involved in the production of sex hormones, either through modifications of steroidogenic enzymes or through indirect feedback effects, 79 leading to decreased production of mature eggs and Sz.
These results are supported by the evaluation of the histopathological changes in the gonads after the 21 days of treatment with the doses of EHFAo and spilanthol. This study revealed a significant decrease in the percentage of immature (EVO) and mature ovarian (EVO) oocytes at all treatment doses, indicating that the reduction in oocyte maturation partially contributes to spawning reduction, suggesting that EHFAo and spilanthol caused reproductive toxicity in zebrafish. Previous studies have shown that some substances, mainly with characteristics of pesticides such as glyphosate, methyl parathion, triadimenol, and bifenthrin, also had adverse effects on fish reproduction. 80 –83
The results obtained by Anholeto et al. 84 demonstrate that the extract of Acmella oleracea affects the germ cells of the ticks (of both males and females), compromising their reproduction.
Studies have shown that follicle stimulating hormone (FSH) and luteinizing hormone (LH) have a significant impact on the regulation of steroidogenesis and gametogenesis by binding to FSH receptor (FSHR) and LH receptor (LHR). 85 According to Clelland and Peng, 86 in females, the growth phase (vitellogenesis) is under control mainly of FSH, whereas LH is involved in the final stages of oocyte maturation.
According to some authors, chemical agents can act by multiple mechanisms, as well as blocking or disrupting agents of hormonal action, and may have not only cumulative but potentiated action in the development of organisms. 87 –89 Some studies suggest that time of exposure is more important than the dose of exposure to these chemicals. It is still not possible to determine the likely mechanism of action of these agents. Its action can occur by mimicking the hormone itself, by blocking the hormonal action, since there is the possibility that these agents are occupying the receptors that would be destined specifically to him, thus preventing his action from being exercised; causing damage to the metabolism of the hormones, that is, in their synthesis or in their destruction and physiological or natural elimination; or even affecting the central nervous system (CNS), which is the main control of hormone production. 90
In rats, Rocha et al. 91 observed that the hydroethanolic extract of the flowers of Acmella oleracea (EHAo) did not cause maternal toxicity. Only the frequency of Proestro (P) and Estros (E) phases were significantly altered in the estrous cycle, which was significantly increased with the doses of 88.91 and 444.57 mg/kg of EHAo to the control. On the other hand, the metestrus (M) and diestrus (D) phases showed a significant reduction in their frequency in the EHAo-treated groups.
In the males treated in this study with the doses of 22.873 and 44.457 mg/kg of the EHFAo and 3 mg/kg of the spilanthol, Figure 6 shows that there was a significant reduction in Sz percentage when compared to the control group. This reduction can also be related to the action of FSH since, according to Schulz et al., 92 it plays a fundamental role in the final stages of sperm maturation.
In a recent study, Souza et al. 93 evaluated the reproductive toxicity of EHFAo in zebrafish by immersion. The male and female animals remained in direct contact with the concentrations of the extract for 21 days and observed that the fertility was not altered when compared to the control group.
According to Forsatkar et al., 28,94 substances of natural and synthetic origin have the potential to affect the life cycle and development of organisms, which may interrupt and interfere with the normal functioning of physiological and behavioral responses. This study analyzed the influence of the treatment of the parental generation with the doses of EHFAo and spilanthol in the development of F1 generation.
In addition to the doses of EHFAo and spilanthol inhibit reproduction, parental treatment over the 21 days negatively affected F1 generation, leading to developmental alterations such as cardiac and yolk edema, growth retardation, tail malformations, and scoliosis (Table 2). These changes have already been reported by several authors, both for direct and indirect action of natural and synthetic substances. 95 –97
It was observed that parental treatment with the lowest dose of EHFAo (22.873 mg/kg) induced a higher proportion of teratogenic effects in the F1 generation. In the groups in which male and female parental received treatment with spilanthol, the lethal effect was more evident, thus suggesting that there is a transference of toxicity from parent to progeny. It is noteworthy that the embryos of the F1 generation in which there was no parental treatment (control) did not present lethality nor any changes in development. Several studies have already reported the transfer of toxicity from the parental generation to F1 generation of various substances such as glyphosate, 93 bisphenol S, 98 fusaric acid, 99 selenomethionine, 100 nanoparticles of ZnO, 101 and azoxystrobin. 102
Based on the results obtained in this study and previous studies, there are two possible mechanisms that enhance the toxicity of parental treatment to various substances in fish progeny, which may include characteristics such as (1) the treatment of the parents can alter the quality of the eggs or Sz, which would affect the development of the progeny, 103 and (2) the bioaccumulation of chemicals or their metabolites in parental fish can induce the transfer and deposition of chemicals from eggs to the progeny. 104 It is not possible to describe the precise mechanism that EHFAo and spilanthol cause toxicity in the F1 generation, since it is not possible to measure the maternal transfer of the compounds present in the extract or its metabolites and the spilanthol, from the adult to the embryo.
In the process of discovery and development of new drugs, the success rate of new candidates selected for clinical development is approximately 20% 105 with most of the difficulties attributed to nonviable pharmacokinetic properties, such as low absorption rate, high hepatic extraction (HD), and hepatic bleaching (HCL)—which cause low bioavailability—and metabolic studies in vitro, providing guidance for subsequent clinical studies. 106
MW is an important aspect in the action of the therapeutic drug; if it increases beyond a certain limit, the volume of the compounds also increases correspondingly, which affects the action of the drug. Drug molecules having M W < 500 are easily transported, diffused, and absorbed compared to heavy molecules. Even some molecules of drugs with a molecular weight higher than that established by the Lipinski five rule (<500 Da), low-molecular-weight compounds (below 800 Da) with high liposolubility show the highest penetration; 28 in this study only M28 presented value greater than 500 Da, which will hinder transport, diffusion, and absorption. 28
The number of hydrogen bonding receptors (O and N atoms) and the number of hydrogen bonding donors (NH and OH) for metabolites and spilanthol were found within the Lipinski limit. 28
The log P (octanol/water partition coefficient) was calculated by the methodology developed by Molinspiration as a sum of contributions based on fragments and correlation factors. 28 The log P coefficient is used as a measure of molecular hydrophobicity affecting drug absorption, bioavailability, hydrophobic drug–receptor interactions, metabolism of molecules, as well as their toxicity. The highest degree of lipophilicity has been found for all compounds which are indicative of good liposolubility and assist in the design of drugs that can interact with membranes. 28 In this study, spilanthol presented log P with a higher value among all metabolites studied.
The MPSA or TPSA is a very useful parameter for predicting drug transport properties. The polar surface area is defined as a sum of polar atomic surfaces (oxygen, nitrogen, and bound hydrogen) in a molecule. It has been discovered that phytochemicals or drugs can be targeted to the CNS with a PSA of less than 60–70 Å2. Spilanthol presented PSA of 29.10 Å2, indicating its action on the CNS. MPSA or TPSA values are two important properties for the prediction of bioavailability of drug molecules. 107
MV determines transport characteristics of molecules, such as intestinal absorption or penetration of the blood–brain barrier. 108 The volume is therefore often used in QSAR studies to model molecular properties and biological activity within the range. 102,109 As the number of rotating bonds increases, the molecule becomes more flexible and more adaptable for efficient interaction with a particular binding pocket. 28 The values are shown in Table 3.
As the number of rotating bonds increases, the molecule becomes more flexible and more adaptable for an interaction efficiency with a particular bond. 28 The M28 metabolite presented the highest number of rotational connections equal to 22.
Pharmacokinetic interactions often occur as a result of a change in drug metabolism. CYP3A4 is an important human liver isoenzyme and is known to metabolize a wide variety of endogenous xenobiotics and biochemicals. 110
Li et al. 111 list a total of 32 chemicals belonging to different structural classes that were evaluated and considered substrates for CYP3A4. Metabolic pathways for these substrates include N-oxidation, C-oxidation, N-dealkylation, O-dealkylation, nitro-reduction, dehydration, and C-hydroxylation. While the main experimental system used to elucidate the role of CYP3A4 in the metabolic transformation of these substrates is the human liver microsome system, cultured human hepatocytes and genetically modified leavened/cultured cells to express CYP3A4 are also employed by the different investigators. In this study, interactions with the CYP3A4 substrate involved the inhibition of P-glycoprotein (P-gP) by the M28 metabolite.
P-gP is a multidrug resistance (MDR1) gene product that acts as an ATP-dependent efflux pump that transports drugs and xenobiotics to the outside of liver, kidney, brain and gastrointestinal tract cells, 112 –114 tumor cells, and hematotecidual barrier cells. It is involved in the process of absorption, distribution, metabolism, and excretion of different drugs, such as chemotherapeutic agents, 115 cardiac glycosides, 116 HIV-1 protease inhibitor, 117 and cyclosporin. 118
The differentiated expression of P-gP indicates that it plays a role in protecting the body from xenobiotic agents by excreting these components in the bile, urine, and intestinal lumen and also by preventing its accumulation in the brain. 119 Only the M28 metabolite showed inhibition of P-gP in this study.
The Ames test is a simple method for testing the mutagenicity of a compound, suggested by Ames et al., 120 where several strains of Salmonella typhimurium bacteria are used with mutations in genes involved in histidine synthesis. The variable being tested is the ability of the mutagen to cause growth reversal in a histidine-free medium. 121 In this method, the spilanthol and the metabolites M8, M18, M26, M33, and M35 presented positive predictions, which means that they were predicted as mutagenic compounds (Table 6).
Carcinogenicity is the ability of a substance to induce changes that lead to cancer. Carcinogenicity tests require an extended period (>2 years), and the main methodologies use “in vivo” assays, using rats or mice, exposing them to a chemical compound, where the observed variable is the existence of cancer.
In this study, the PreADMET server was used to predict the results (Table 3) which are constructed from data from the National Toxicology Program and the US/FDA, which are the results of in vivo tests for carcinogenicity in rats and mice for 2 years. 122 Spilanthol and the metabolites M8, M26, M28, and M33 obtained a positive prediction, which means that there is no evidence of carcinogenic activity.
Conclusion
This is the first study to evaluate the reproductive toxicity and development of the hydroethanolic extract of Acmella oleracea (EHFAo) and its major compound, spilanthol in oral zebrafish (gavage). In a parental generation, this treatment resulted in many tissue changes in the gonads, thus interfering with reproduction; this has important implications for studies of unintentional pharmaceutical treatments. However, the exact mechanism from which EHFAo or spilanthol may induce toxicity at these doses in embryos (F1 generation) could not be fully elucidated since it is not possible to measure the transfer of EHFAo or spilanthol molecules from parent fish to embryos.
Considering the prediction errors, physicochemical properties, pharmacokinetic and toxicological parameters results, it was observed that all selected compounds are lipophilic and obeyed Lipinski’s rule of five. The pharmacokinetic and toxicological parameters results are generally within the limits of clinical relevance reported in the literature for most CYP P450 and P-gP for eight of the major metabolites generated.
Footnotes
Acknowledgement
The authors thanks Dr Clarissa Silva Lima for assistance in obtaining spilanthol.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was financially supported by the Coordination for the Improvement of Higher Education Personnel (CAPES – Procad Amazônia, Proc. 88887.200554/2018-00) and National Council for Scientific and Technological Development (CNPq; Proc. 402332/2013-0).
