Abstract
Bleomycin (BL) is a powerful chemotherapy drug that has devastating effects on spermatogenic function and may make cancer survivors at risk of infertility. Protective effects of thymoquinone (TQ), a phytochemical compound with antioxidant and anticancer influences, were investigated on sperm parameters, testicular structures, and sexual hormones in BL-treated mice. Forty-eight adult male Balb/c mice were randomly divided into six groups. Control group received normal saline; BL group received 10 mg/kg BL; TQ7.5 group received 7.5 mg/kg TQ; TQ15 group received 15 mg/kg TQ; BL+TQ7.5 group received 10 mg/kg BL and 7.5 mg/kg TQ; BL + TQ15 group received 10 mg/kg BL and 15 mg/kg TQ. BL was intraperitoneally used every day through 35 days, and TQ was intraperitoneally injected 3 days before administration of BL and continued twice per week for 35 days. Results showed that BL significantly decreased count, viability, morphology, maturity, and progressive movement of sperm, testosterone, seminiferous tubule diameters, the ratio of testis weight to body weight, number of spermatogonia, spermatocytes, spermatids, and Sertoli cells per tubule, and expression of
Introduction
Nowadays, the survival rate of patients has improved due to advances in early cancer detection and treatments; consequently, it is necessary to promote quality of life in these patients after treatment. 1 Since many cancer treatment procedures have a risk of developing long-term side effects on reproductive potential, childbearing and parenthood are major concerns in cancer survivors. Therefore, establishing a multidisciplinary treatment program toward fertility preservation in these patients is of remarkable importance.
Chemotherapeutic agents and standardized regimes of drugs are cytotoxic and can destroy cancer cells in a variety of ways. These drugs, by inhibition of mitosis, prevent the growth and division of any cells with the rapid division leading to apoptosis. 2 Furthermore, the induction of apoptosis via reactive oxygen species (ROS) is potentially an alternative mechanism in many chemotherapeutic agents. 3
Bleomycin (BL), a water-soluble glycopeptide antibiotic derived from
Besides the beneficial effects of BL in the treatment of cancers, it has various devastating side effects such as pneumonitis and gonadal damages dependent on the dose and the duration of its usage.6–8 Therefore, chemotherapy can cause cytotoxicity for both cancer cells and normal cells. Germinal epithelium of male gonads is a target of chemotherapeutic drugs that can be affected by treatment procedures. 2 Thus, there is an intense debate on the concurrent use of antioxidants during conventional cancer treatments to prevent ROS-induced damage to tissues. 9
Nowadays, the use of herbal medicine as a complementary and alternative medicine for the prevention and treatment of diseases is strongly suggested.10,11 The beneficial effects of traditional medicine in promoting reproductive function and alleviating some male and female reproductive systems disorders have been shown in many studies.12–16
Therefore, based on all this evidence, in the current study, the possible protective effect of TQ in two different dosages on BL-induced toxicity on male reproductive parameters was investigated.
Material and methods
Animals and experimental procedures
Forty-eight healthy Balb/c mice (8 week old), weighing 30 ± 2 g, were obtained from the animal house of Shiraz University of Medical Sciences and held in standard cages, 12-h light, and 12-h dark, with ad libitum access to food and water. All parts of the procedure related to animals were confirmed by the ethical committee of Shiraz University of Medical Sciences (Ethical code: IR.SUMS.REC.1397.124).
After 2 weeks, animals were randomly divided into six groups of eight and treated with BL sulfate (Nippon Kayaku, Tokyo, Japan; CAS No. 9041-93-4) and TQ (Sigma-Aldrich, USA; CAS No. 490-91-5) as follows:
Control group received normal saline; BL group received BL (10 mg/kg); TQ7.5 group received TQ (7.5 mg/kg); TQ15 group received TQ (15 mg/kg); BL+TQ7.5 group received BL (10 mg/kg) + TQ (7.5 mg/kg); BL + TQ15 group received BL (10 mg/kg) + TQ (15 mg/kg). All reagents were intraperitoneally administered. Since one cycle of spermatogenesis in mice takes 34.5 days, we defined the duration of the experiment to be 35 days to be sure that the treatment has an effect at least on an entire spermatogenesis cycle. BL was injected every day through 35 days, and TQ was started 3 days before administration of BL and continued twice per week for 35 days.
Hormonal assay
After 35 days, each mouse was anesthetized using chloroform (KimiaExir Co, Tehran, Iran) and blood samples were obtained from dorsal aorta puncture and centrifuged at 2000 r/min at 25°C for 20 min. Then, the clear serums were isolated and stored at −20°C until used. The testosterone (DRG Company, Germany; SKU: EIA-1559), LH (Padtan Elm Company, Tehran, Iran; Cat. No.:1112015), and FSH (Padtan Elm Company, Tehran, Iran; Cat. No.:1112014) concentrations of each sample were measured by their specific kits and determined by the radioimmunoassay solid-phase method.
Sperm analysis
After collecting the blood sample, the end of the right epididymis tail (1 cm) was cut and put in 500 μl of pre-incubated Ham’s F10 and incubated at 37°C for 30 min. After this time, the motility, count, morphology, vitality, and maturation status of sperm were evaluated in all groups.
Sperm motility
We put one drop (10 μl) of sperm suspension on the slide and examined the movement of 200 sperms using a light microscope (Nikon ECLIPSE E200, Japan) at 400 × magnification. Based on World Health Organization (WHO) guidelines, sperm movement is graded in four categories: (a) fast progressive movement; (b) slow progressive movement; (c) non-progressive movement; and (d) immotile, and the percentage of each movement was recorded. 28
Sperm count
A 1:5 dilution of semen suspension with distilled water was made, and 10 μl of the diluted sample was placed on the hemocytometer counting chamber. The total concentration of spermatozoa (C) is the total counted sperms (
Sperm morphology
To evaluate morphology, sperm were stained by a diff-quick staining kit (Avicenna, Tehran, Iran; Cat. No.: DQ082101). In each sample, 200 sperms were counted randomly and the percentage of sperms with normal morphology was determined.
Sperm vitality
In order to distinguish the live sperm from the dead, we put one drop of sperm suspension (10 μl) on the slide and followed the protocol of eosin-nigrosin staining. Nigrosin increases the contrast between the background and sperm heads, making sperm easier to visualize. Eosin stains only the dead sperm, turning them a dark pink, whereas live sperm appears white. In each sample, 200 sperms were counted in random fields and the results were converted to a percentage.
Sperm nuclear maturity
Sperm maturation requires chromatin condensation, which is accomplished by replacing histone with protamine. Thus, the mature sperm have protamine in their nucleus instead of histone. Acidic aniline blue binds to histones and therefore discriminates mature sperms from immature. Sperm smears were prepared and fixed by 3% glutaraldehyde in 0.2 Sperm nuclear maturity analysis by acidic aniline blue staining. The sperm were classified into three categories: not stained (mature; dashed arrow), light blue (intermediate; solid arrows), and dark blue (immature; head arrow).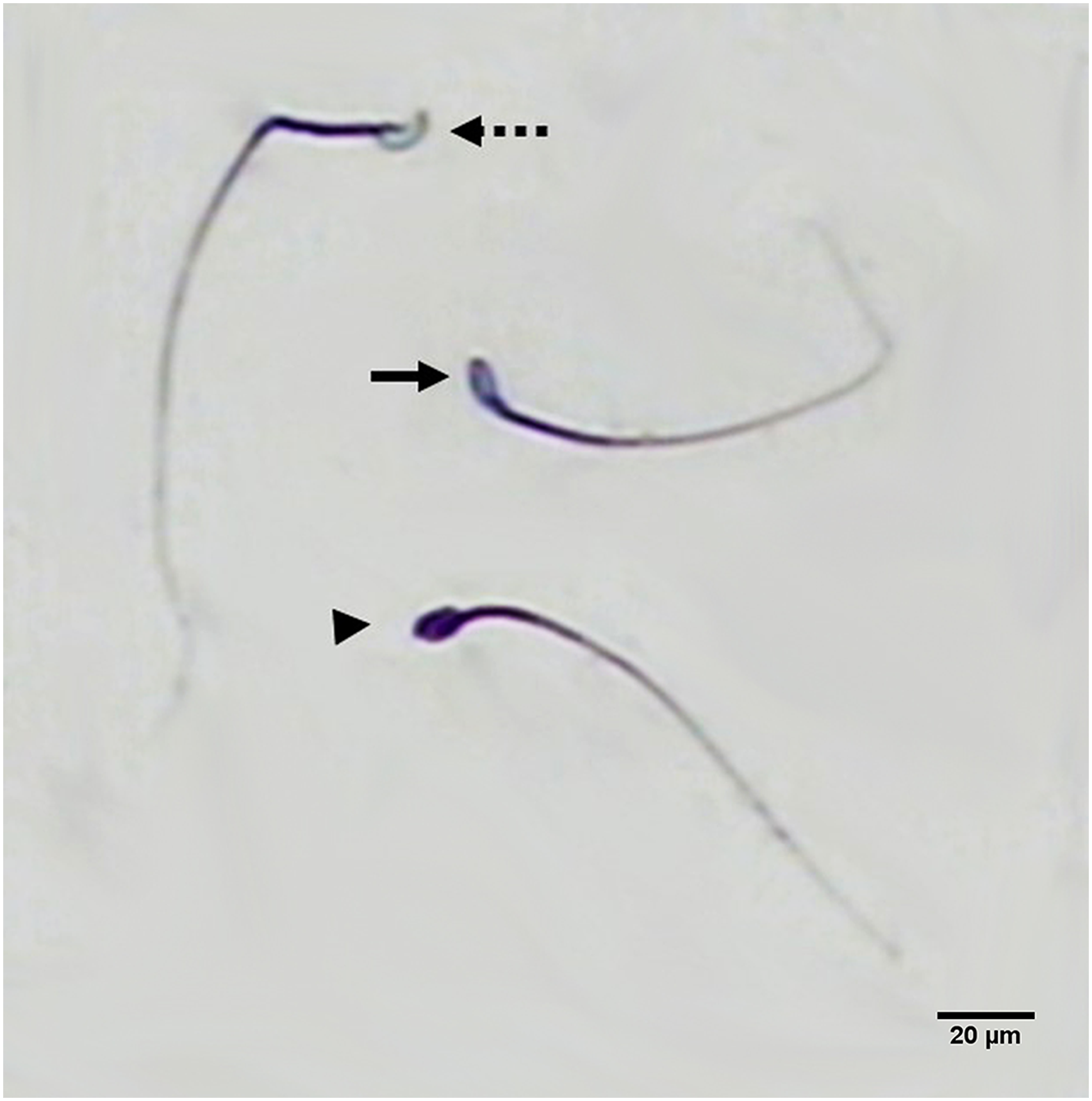
Histopathological evaluation
After dissecting the testis and removing all the fatty tissue around it, the left testis was fixed in Bouin’s solution. Then, the routine paraffin embedding was done and 5 μm sections were cut using a rotary microtome. After that, hematoxylin–eosin staining was performed and sections were examined by a light microscope (Nikon ECLIPSE E200, Japan). The diameters of seminiferous tubules, the thickness of tunica albuginea, germinal epithelium thickness, and the number of spermatogonia, spermatocytes, spermatids, and Sertoli cells per tubule were evaluated.
Lipid peroxidation assessment
After blood sampling, the right testis of each mouse was removed and weighed. Then, 30 mg of tissue was separated from the whole right testis and stored at −20°C until used. The lipid peroxidation status of each sample was determined by a colorimetric and fluorometric assay kit (Navand salamat, Iran; REF: NS-15023) according to the manufacturer’s instruction.
Evaluation of Bax and Bcl2l1 genes expression
Details of primers used for quantitative real-time RT-PCR.

Statistical analysis
All the data obtained from experiences were carried out using IBM® SPSS® Statistics 24.0 for Windows, and they were analyzed by one-way analysis of variance (ANOVA) followed by Tukey test for post hoc comparisons. Results were expressed as mean ± standard error (SE).
Results
Hormonal assay
Evaluation of LH, FSH, and testosterone levels in the serum of all groups showed no significant differences between the control and TQ7.5 and TQ15 groups ( The concentration of LH, FSH, and testosterone in serum, and MDA concentration in the testis tissue. Data are presented as means ± SE. *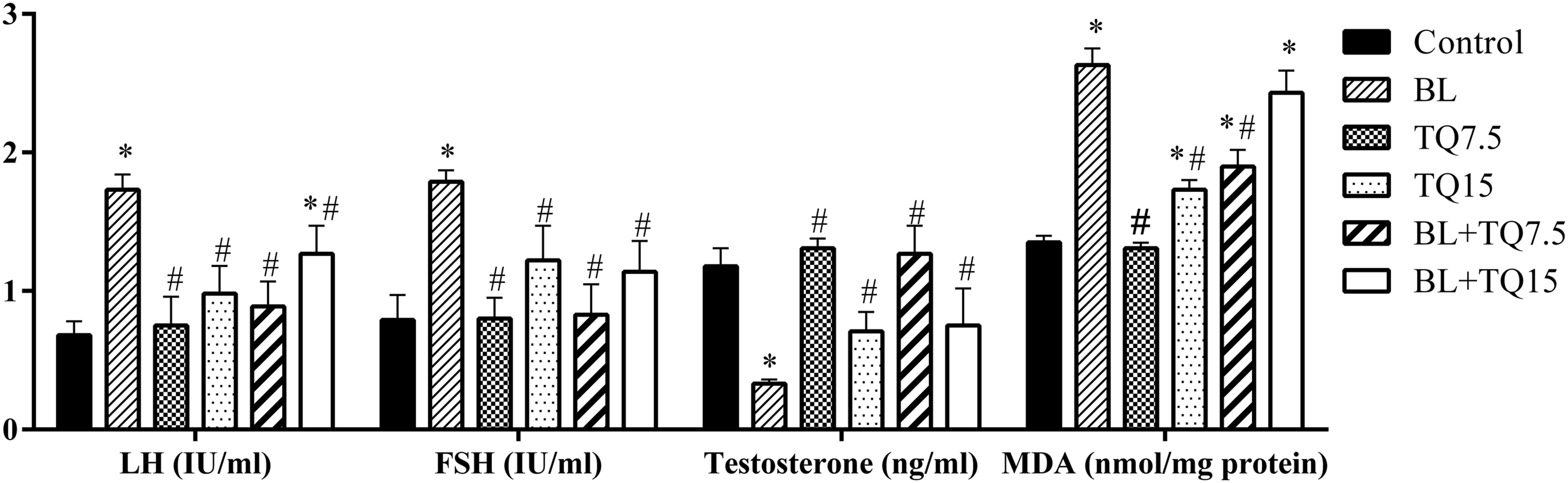
Lipid peroxidation
The formation of malondialdehyde (MDA), an indication of membrane lipid peroxidation, was significantly increased in the BL and TQ15 groups compared to the control group (
Sperm count
The number of sperm in the BL and TQ7.5 was decreased and increased, respectively, compared to the control group ( The sperm count, viability, and morphology. Data are presented as means ± SE. *
Sperm viability
There was a significant decrease in sperm viability in the BL, TQ15, BL + TQ7.5, and BL + TQ15 groups compared to the control group (
Sperm morphology
The percentage of sperm with normal morphology was decreased in the BL, BL + TQ7.5, and BL + TQ15 groups compared to the control group (
Sperm maturation status
Analysis of the maturation status of sperm (Figure 1) revealed that the percentage of mature sperm was significantly decreased in the BL and TQ15, BL + TQ7.5, and BL + TQ15 compared to the control group ( The status of sperm maturity. Data are presented as means ± SE. *
Sperm movement
The number of sperm with fast and slow progressive movement was significantly lower ( The sperm movement is graded in four categories: (a) Fast progressive movement; (b) slow progressive movement; (c) non-progressive movement; and (d) immotile. Data are presented as means ± SE. *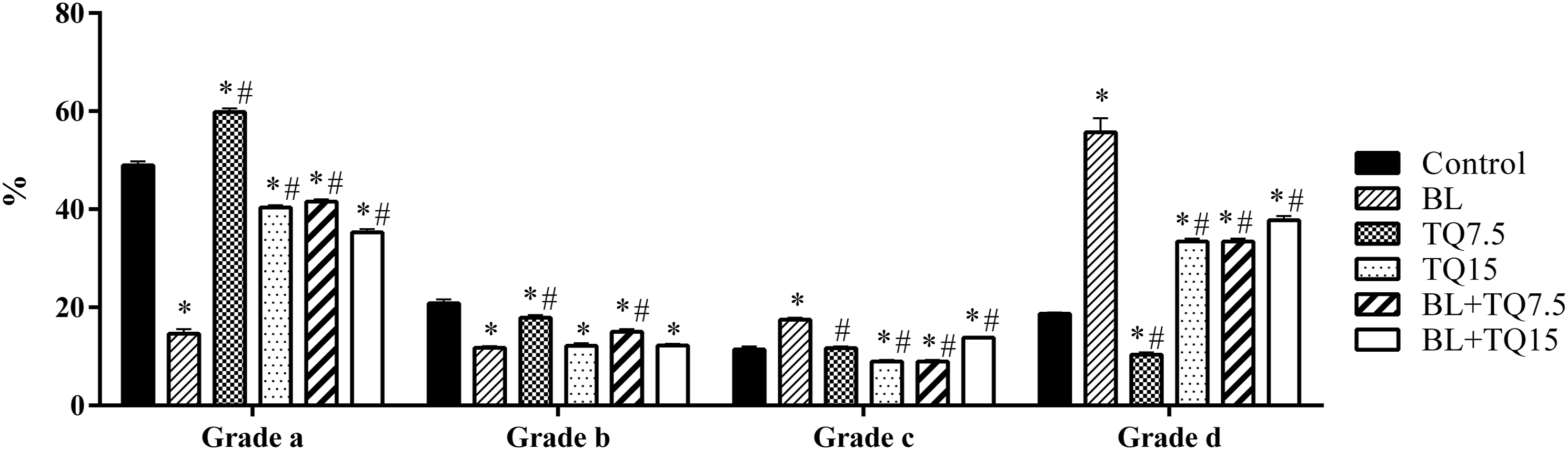
Histopathological evaluation
The diameters of seminiferous tubules were significantly decreased in the BL group compared to the control group ( The seminiferous tubule diameters, tunica albuginea, and germinal epithelium thickness, and ratio of testis weight to body weight in all groups. Data are presented as means ± SE. *
Administration of BL caused a significant decrease in the number of spermatogonia, spermatocytes, spermatids, and Sertoli cells per tubule compared to the control group ( The number of spermatogonia, spermatocytes, spermatids, and Sertoli cells per tubule. Data are presented as means ± SE. *
The seminiferous tubules of testis were histologically normal in the control group and mice received 7.5 and 15 mg/kg TQ. These tubules consisted of spermatogonia, primary spermatocytes, secondary spermatocytes, spermatids, and spermatozoids (Figure 8(a),(c),(d)). In mice receiving BL, the normal structure of seminiferous tubules was significantly disturbed and severe necrosis was seen in almost all seminiferous tubules. In some parts, the identification of seminiferous tubules was not easy. The cells involved in the process of spermatogenesis were very small and not clear. The connective tissue between seminiferous tubules was noticeably weak. It was hard to detect between the lumen of seminiferous tubules and the interstitial tissue (Figure 8(b)). Treatment of mice receiving BL with a lower dose of TQ (7.5 mg/kg) improved testicular structure and prevented testicular destruction because the normal structure of the testicles in these mice was seen as a control group (Figure 8(e)). However, the beneficial effects of a high dose of TQ (15 mg/kg) on repairing damaged testicular structure were less (Figure 8(f)). The histological structure of seminiferous tubules in (a) control, (b) bleomycin, (c) 7.5 mg/kg thymoquinone, (d) 15 mg/kg thymoquinone, (e) bleomycin +7.5 mg/kg thymoquinone and (f) bleomycin + 15 mg/kg thymoquinone by hematoxylin and eosin staining. Condensation of germ cell nuclei (white arrow), and Sertoli cell vacuolization (head arrow) were observed in bleomycin group. SG: spermatogonia; SP: spermatocyte; ST: spermatid; S: Sertoli cell; GE: germinal epithelium; *: spermatozoa.
Bcl2l1 and Bax genes expression
The expression of The expression of 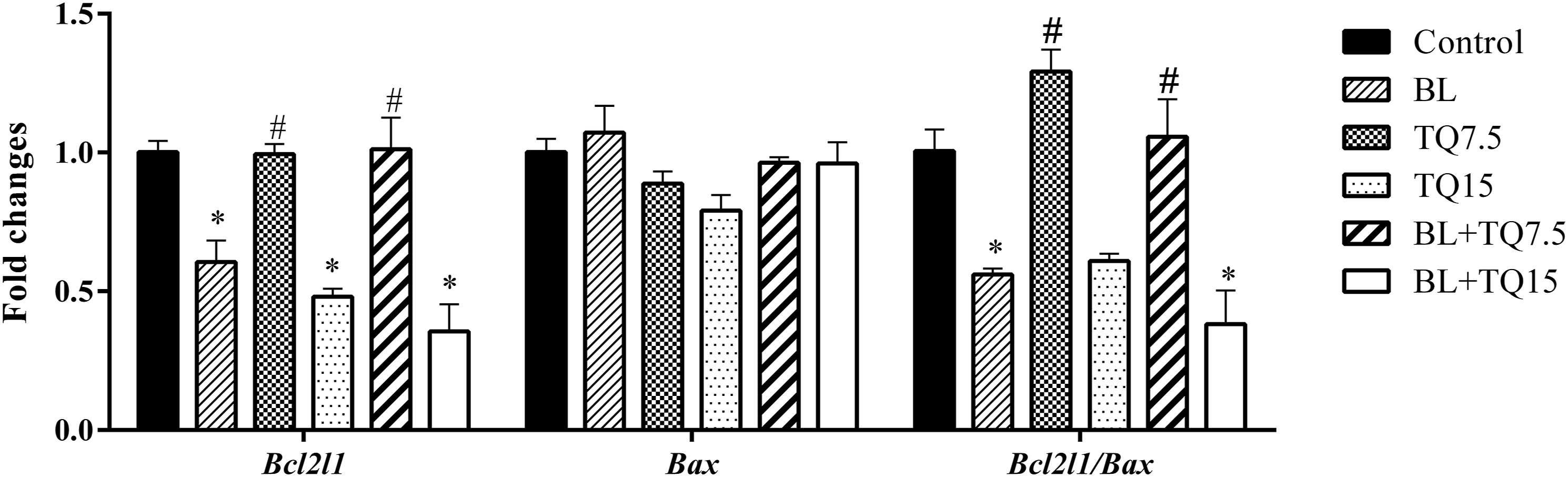
Discussion
In the current study, influence of TQ as the main constituent of
At the level of 7.5 mg/kg, TQ induced some fertility parameters including the number of spermatogonia, spermatid and Sertoli cells, sperm count, mature sperm, and fast progressive motility when compared to the control group. Mohammad et al. suggested that
According to our results, concentration of 15 mg/kg of TQ increased MDA level and had remarkable detrimental issue on sperm motility, viability and maturation and number of spermatocytes and spermatids per seminiferous tubule; however, the total number of produced sperms was not altered by 15 mg/kg of TQ. Both 7.5 and 15 mg/kg TQ had no effect on seminiferous tubules, tunica albuginea and germinal epithelium thickness and testis weight. Rosli et al. (2019) demonstrated that oral administration of 5 mg/kg TQ to rats for 30 days significantly decreased the viable sperms, while the number of sperm and level of testosterone hormone was not affected by TQ, 35 which was similar to our results at level of 15 mg/kg TQ. It seems that the beneficial or detrimental effects of TQ on male reproductive system depend on the dose applied, and also the duration of administration. In our study, the lower dose of TQ had positive effects, but at a higher dose showed some adverse effects on some male fertility parameters in mice.
BL significantly reduced the number of spermatogenic and Sertoli cells in each tubule and the diameter of the seminiferous tubules, but the thickness of the germinal epithelium was not affected by BL. This means that space between each cell in the germinal epithelium was increased and lumen space was decreased by BL treatment. Regarding all parameters evaluated in this study, it is obvious that BL had strong detrimental effects on male reproductive structure and function which is in line with other studies.36–39 In the male reproductive system, secreted gonadotropin releasing hormone from the hypothalamus has stimulating effect on the anterior pituitary to release LH and FSH hormones which are necessary for sperm production. The Leydig cells in the interstitial tissue of seminiferous tubules have LH receptors. After activation of these receptors by LH, Leydig cells begin to secrete the testosterone necessary for sperm production. Then, sperm becomes motile in epididymis. Finally, blood testosterone level exerts a negative feedback which leads to decrease in the secretion of GnRH from the hypothalamus and results in a depletion in LH and FSH production. 40
The size and weight of testes are intensely associated with the number of Sertoli cells and sperm production, and testis size is reflective of the number of germinal cells in the testis. In our study, besides the remarked decrease in testis weight, histological evaluation demonstrated that BL caused adverse effects on testis structure, including the attenuation of diameters of seminiferous tubules, increase of the thickness of tunica albuginea, and severe degradation of seminiferous tubule cells. Destruction of testes tissue resulted in a depletion in the level of testosterone production and notable decline in number of cells involved in spermatogenesis and also Sertoli cells, impaired sperm production, and elevated LH and FSH levels due to impaired androgen-mediated negative feedback regulation of gonadotropins. Therefore, the elevation of LH, FSH, and attenuation of testosterone hormones in BL-treated rats are consistent with the results of degradation of testis tissue, decrease of seminiferous tubules, and disturbance of spermatogenic cell lineage. All of the mentioned factors significantly resulted in a dramatic reduction in both quantity and quality of sperm such as viability, maturation, morphology, and motility. Oxidative stress and apoptosis are key events involved in causing DNA damage in the germ line.41,42 It is demonstrated that increased ROS along with decreased antioxidant defense result in redox imbalance, reduced sperm motility, and sperm DNA damage. 43
According to our result, it seems that BL had oxidative stress effects through disruption of cell membrane lipids of all testicular cells and resulted in reduced sperm production, sperm motility, and triggered apoptosis.
BL disturbed all aspects of the spermatogenesis process involving sperm production, movement, maturation, sperm vitality, and morphology by induction of apoptosis cascade. Administration of 7.5 mg/kg TQ to BL-treated mice showed an inhibition of cell death by antioxidant mechanism, which plays a pivotal role in the amelioration of the adverse effects of BL on male reproductive system by inhibition of apoptosis, elevation of
Conclusion
BL has toxic effects on all fertility parameters in male mice and TQ as an antioxidant agent has strong protective effects against BL-induced reproductive toxicity. It should be considered that the constructive effects of TQ are in a dose-dependent manner because the beneficial effects of 7.5 mg/kg TQ in alleviating the adverse effects of BL on fertility parameters were more remarkable compared to 15 mg/kg. Most of evaluated parameters in this study returned to the level of the control group by administration of 7.5 mg/kg TQ to BL-treated mice.
Footnotes
Acknowledgments
The authors would like to thank the Research Vice Chancellor of Shiraz University of Medical Sciences, Shiraz, Iran for financially supporting the research (Grant number 13398).
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Shiraz University of Medical Sciences grant number 13398.
