Abstract
Research in the past has indicated associated long-term and low levels of exposure of bisphenol A (BPA) in early life and neuroendocrine disorders, such as obesity, precocious puberty, diabetes, and hypertension. BPA and its analogs bisphenol B (BPB), bisphenol F (BPF), and bisphenol S (BPS) have been reported to have similar or even more toxic effect as compared to BPA. Exposure of rats to BPA and its analogs BPB, BPF, and BPS resulted in decreased sperm production, testosterone secretion, and histological changes in the reproductive tissues of male rats. In the present study, BPA, BPB, BPF, and BPS were administered in drinking water at concentrations of (5, 25, and 50 μg/L) from pregnancy day (PD) 1 to PD 21. Body weight (BW), hormonal concentrations, antioxidant enzymes, and histological changes were determined in the reproductive tissues. BPA and its analogs prenatal exposure to female rats induced significant statistical difference in the antioxidant enzymes, plasma testosterone, and estrogen concentrations in the male offspring when compared with the control. Histological parameters of both testis and epididymis revealed prominent changes in the reproductive tissues. The present study suggests that BPA and its analogs BPB, BPF, and BPS different concentrations led to marked alterations in the development of the male reproductive system.
Introduction
Bisphenol A (BPA) is among the highly produced chemicals of the world and its production is on 2.7% increase from approximately 5.5 million tons since 2010. 1 It is predicted that this amount is going to increase by an annual rate of 5% by 2019. 2 BPA is used for the manufacturing of epoxy resins and polycarbonate plastics and is found in many consumer products, including thermal papers receipt, toys, medical equipment, food, and beverage containers. 3 BPA used for the manufacturing of different kinds of daily use item has led to widespread contamination in the global environment and it has been detected in various human bodily fluids like urine, blood, and breast milk. 4 –6 Humans are exposed to BPA through diet as it is a common endogenous metabolite formed in situ in addition to external exposure source such as dermal contact and in inhalation of dust. 7 –9 In some in vivo studies, BPA has estrogen-like effects and its continuous exposure to human lead to many health concerns like metabolic disease and reproductive disorders. 2,10 –14 This has prompted the release of strict regulations of European Union on the applications of BPA in many daily use items like infant feeding bottles, food, and beverages packages and sippy cups by government organizations. 15 Consequently, several global manufacturers voluntarily phased out BPA and have started to develop various BPA alternatives molecules all containing the common 2-phenol groups with only the side group differences including bisphenol B (BPB), bisphenol F (BPF), and bisphenol S (BPS). 16,17 BPA analogs BPB, BPF, and BPS have the same common structure of BPA and these analogs are used for the manufacturing of polycarbonate plastics, paper products, and phenolic resins. 18 –20 However, in vitro and in vivo studies have shown that some of these analogs have stronger estrogenic activities as compared to BPA. 21 –24 Due to the increased production of these alternatives in the manufacture of many BPA-free items, human exposure to these chemicals is at rise which is very alarming. 25 BPA analogs like BPF and BPS have been detected in indoor dust, surface water, and human urine in different population across the globe which is comparable to BPA. However, BPA analogs internal and external exposure risks remain poorly understood in the general population. 26 –33
In particular, the potential effects of exposure to BPA on the development of the reproductive system have been noted as an area of special concern by the National Toxicology Program (NTP). 34 –36 The NTP expressed “some concerns” for adverse effects of BPA and its analogs on the brain and reproductive system based upon “limited evidence of adverse effects” of low doses in rodent studies. 37 –39 Relevant to the current study which examined the effects of exposure to BPA and its analogs on the development of reproductive system in male rat, it is noteworthy that none of the exposed studies where rats exposed during perinatal life have been conducted with BPA and its analogs so far. 40 –43 One objective of the current study was to determine if relatively low oral doses of BPA or BPA analogs would alter sexual differentiation, the age at puberty, and the fertility of female rat offspring when administered orally to the dam during gestation period. 44 –46 Pregnant rats were exposed via drinking water from gestation day (GD) 1 until the day of the parturition to expose their offspring during the period of sexual differentiation of the reproductive organs development as well as the neonatal period of sexual differentiation of the brain. Since the organization of some rodent sexually dimorphic behaviors during perinatal life is imprinted by androgens and others by estrogens, exposure to endocrine disrupting chemicals (EDCs) that act via these mechanisms can permanently imprint the nervous system. 47 –51 Research in the past has indicated the association between long-term and low level of exposure of BPA in early life and neuroendocrine disorders, such as obesity, precocious puberty, diabetes, anxiety, and hypertension. 52,53 BPA analogs have been reported to have similar or even more toxic effects as compared to BPA in many animal exposure studies. 54 –57 There are even some studies available which have reported that some of the BPA analogs have similar estrogenic potency as BPA. 58 –61 These findings highlight that BPA analogs marked as safer alternatives to BPA may similarly induce widespread and varied health effects. However, few studies have assessed the endocrine disrupting potentials of these alternatives to BPA in vivo. The current study directly addresses some of the concerns regarding the estrogenic mode of action and toxicity inducing potentials of low dose effects of BPA and its analogs BPB, BPF, and BPS on sexual development of pre-natal male rats.
Materials and methods
Animal dosage levels and administration of chemicals
Adult female Sprague Dawley rats (Department of Animal Sciences, Quaid-i-Azam University Primate and Rodent Facility, Islamabad, Pakistan) of approximately 80–90 days of age were mated with adult male rats in the present study. Animals were fed with laboratory feed (soy and alfalfa free) and water was available ad libitum. All the experimental protocols were approved by the Ethical Committee of the Department of Animal Sciences, Islamabad, Pakistan. All the females were paired with adult males in separate breeding cages and animals were let to copulate for next few days and mating was confirmed by sperm presence in vaginal smears (day of sperm plug positive = GD1). Males were separated from females on the day when a vaginal plug was more visible and was considered as day 1 of gestation (GD 1), while females with no vaginal plug were excluded from the study. Animals were weighed and housed in separate breeding cages provided with wooden nesting material. All the animals were placed under standard colony conditions (with a 14:10 light:dark cycle at 20–24°C and 40–50% relative humidity). All caging used in the current experiment was made of glass and without evidence of significant wear. Pregnant and lactating dams were also fed standard laboratory feed for (Rat SPR 1002) ad libitum, and weanling and adult rats were fed standard laboratory feed for (Rat SPR 1002) ad libitum and water was provided in polysulfone bottles. Control animals used in this study were also taken from the same supplier and maintained, as above. After isolation from males, pregnant rats were randomly assigned to treatment groups and all the female rats were randomly divided into 13 groups (n = 8 female rats/group). Control group was provided with water containing 0.1–0.5% ethanol while treatment groups were provided with 5, 25, and 50 μg/L of BPA and its analogs BPB, BPF, and BPS (Sigma–Aldrich, St Louis, Missouri, USA) in drinking water. BPA and its analogs BPB, BPF, and BPS were dissolved in ethanol and stock solutions were diluted with water. The final concentration of ethanol in the water was kept below 0.5%. The doses were in accordance with the study of. 37,62,63 During the experiment, the water containing BPA, BPB, BPF, and BPS was removed from the animals on the day the pups were born (PND 1) and were provided with tap water.
Early development study
Ano-genital distance (AGD), nipple retention (NR), and organs weight
On postnatal day 1 (PND 1), pups were weighed and the number of male and female were counted. The pups were checked for abnormalities. AGD was measured in male pups under an ocular stereomicroscope. All the pups were weighed on PND 6, PND 14, and PND 16. On PND 14, pups were examined for the number of NR. On PND 16, two males/per litter were anesthetized with carbon dioxide, euthanized by decapitation and different organs (testes, epididymis, ventral prostate, seminal vesicles, bulbocavernosus muscles, bulbourethral glands, adrenals, thyroid, retroperitoneal fat pad, and liver) were dissected out and weighed.
Late developmental study
Puberty in male pups was checked through the appearance of external signs of puberty according to the methods described elsewhere. 64 All the males were checked daily for preputial separation from day 35 till the pubertal day 1. All the groups were observed and the day puberty took place was noted.
Determination of biochemical assays
To determine the oxidative stress level and antioxidant enzymes, the reproductive tissues were homogenized with automatic homogenizer in phosphate buffer saline and centrifuged at 3,000 × g for 30 min. The supernatants were separated and used for the measurement of hormonal analysis, protein estimation, and antioxidant enzymes assays. To determine catalase (CAT) activity, the method used by Aebi 65 was followed and the change in the absorbance due to H2O2 was measured in the testicular tissues. In this assay, 50 µl homogenate was diluted in 2 ml of phosphate buffer with pH of 7.0. After mixing it thoroughly, the absorbance was read at 240 nm with an interval of 15 and 30 s. Change in the absorbance of 0.01 as unit/min was defined as one unit of CAT. Superoxide dismutase (SOD) activity was estimated by the method developed by Kakkar et al. 66 In this assay, the amount of chromogen formed was measured at 560 nm. The results were expressed in units per milligram of protein.
Peroxidase (POD) activity in homogenate was determined by spectrophotometric method of Carlberg and Mannervik. 67 In this assay, 0.1 ml homogenate was mixed with 0.1 ml of guaiacol, 0.3 ml of H2O2, and 2.5 ml of phosphate buffer and the absorbance was read at 470 nm. Change in the absorbance of 0.01 as unit per minute was defined as one unit of POD. Activity of T-BARS was determined in the homogenate by the method used by Iqbal et al. 68 and the results were expressed as TBARS per minute per milliliter of plasma. In this assay, 0.1 ml of homogenate was mixed with 0.29 ml phosphate buffer, 0.1 ml of trichloroacectic acid, 1 ml of trichlorobarbituric acid followed by heating at 95°C for 20 min and then shifted to ice bath before centrifuging at 2500 r/min for 10 min. The samples were read with the help of spectrophotometer at 535 nm.
The assay of reactive oxygen species (ROS) was done according to the method of Hayashi et al. 69 and for the presentation of mean values the assay was repeated multiple times. In this assay, 5 ml of H2O2 standards and homogenate was mixed with 140 ml of sodium acetate buffer with pH 4.8 in 96 wells plate and incubated at 37°C for 5 min. After the incubation, 100 ml of N, N-diethyl-pera-phenylenediamine (DEPPD) and ferrous sulfate were mixed and were added in each well with a ratio of 1:25 and incubated at 37°C for 1 min. With an interval of 15 s for 3 min, the absorbance was read at 505 nm at micro plate reader. Total protein content was determined by (AMEDA Laboratory diagnostic kit, Krenngasse, Graz, Austria.) for total protein in tissue. The results of protein were measured by plotting absorbance of the standard against samples. These values were expressed as milligrams per gram of tissue.
Dissections and histopathology
Male rats (n = 8 male pups/group) were weighed at PND 80, anesthetized with carbon dioxide, euthanized by decapitation, trunk blood was collected for hormonal analysis while different organs were collected, weighed, and processed for histopathology.
Histopathological examination
Testicular tissues (testes and epididymis) were fixed in 10% buffered formalin for 48 h. Dehydrated with different grades of alcohol and cleared with help of xylene, the paraffin sections (5 µm) were cut and stained with hematoxylin and eosin stains to assess standard histology and morphometry. Testicular sections (5 µm thick) 10–20 per group were digitalized under Leica Microscope (New York Microscopes, Lauman Ln, Ste A, Hicksville, New York, USA) equipped with a digital camera (Canon, Japan).
For the morphometry, the images were taken at 20× and 40× and the results were done with Image J software (version ImageJ2, Wayne Rasband). Area of different sections was calculated by the method described elsewhere of. 70 From 20× images, 30 pictures per animal were selected and known area of different area of intestinal space, epididymis tubules, and seminiferous tubules were measured by the software. The number of different cell types (spermatids, spermatogonia, and spermatocytes) and the area were calculated and comparison of different groups with control was done.
Daily sperm production
Prior to the homogenization, frozen at −80°C, testicular tissues were thawed at room temperature, tunica albuginea was removed, and the parenchyma was weighed and homogenized in 5 ml of solution, containing 0.9% NaCl and 0.5% Triton X-100 for the 30 s using a rotor stator homogenizer (IKA-Werke, Staufen, Germany). The homogenate was diluted fivefold, a volume of 20 ml homogenate was transferred to a Neubauer chamber, and 19th stage spermatids were counted under a light microscope at 40× magnification. A total of three readings were taken for calculation of the average number of spermatids in each sample. These values were used to obtain the number of spermatids per testis and were divided by 6.3 (number of days the spermatids remain in seminiferous epithelium) to determine DSP.
Number of sperm in different parts of epididymis and sperm transient time
Immediately after dissection, the cauda epididymis was cut slightly with a scissor in 0.5 ml pre-warmed (37°C) phosphate-buffered saline (PBS, pH 7.3) containing a drop of nigrosin stain. An aliquot of 50 μl was taken and placed on a pre-cleaned and warmed (37°C) glass slide and was observed under a light microscope at 40×. A total of 100 sperm/sample were analyzed for motility by a technician blinded to the treatment groups. Each sample was analyzed three times and the average value was used as the total sperm motility. For viability, a drop of eosin and nigrosin was added to the sperm sample. A volume of 10 μl was placed on a pre-warmed and cleaned glass slide and observed under a microscope at 100×. Ten fields were analyzed by a person blinded to the treatment groups. A total of 100 sperm/field were checked for eosin staining and the number of live and dead sperm was estimated. Each sample was repeated three times and the average number was reported and expressed as the percentage of live sperm.
Hormonal analysis
For the measurement of different plasma hormone (testosterone, estradiol, luteinizing hormone (LH) and follicle stimulating hormone (FSH)) concentrations in the tissues, quantitatively EIA kits (Amgenix Inc., California, USA) was used and the assay was performed by the instructions provided with the kits. All the above assays were repeated in triplicates with both inter and intra assay variations for more precise results.
Statistical analysis
Values were expressed as mean ± SEM. One-way analysis of variance followed by Dunnet’s multiple comparison tests was applied to compare different groups with control using Graph Pad Prism 5 software (Version 5, GraphPad Software, Inc.). Values were considered significant at p < 0.05.
Results
Effects of BPA and its analogs BPB, BPF, and BPS exposure on dams, litter size, and offspring BWs
Exposure to different concentrations of BPA and its analogs BPB, BPF, and BPS during the gestational period from GD1 to GD20 showed no external toxic effects on mothers in the current study. There was no significant difference observed in the BW gain during the pregnancy, litter size, and sex ratio (Table 1). Similarly, BPA and its analogs BPB, BPF, and BPS different concentrations also did not show significant difference in the male pups birth weights and litter size as compared to the control (Table 1).
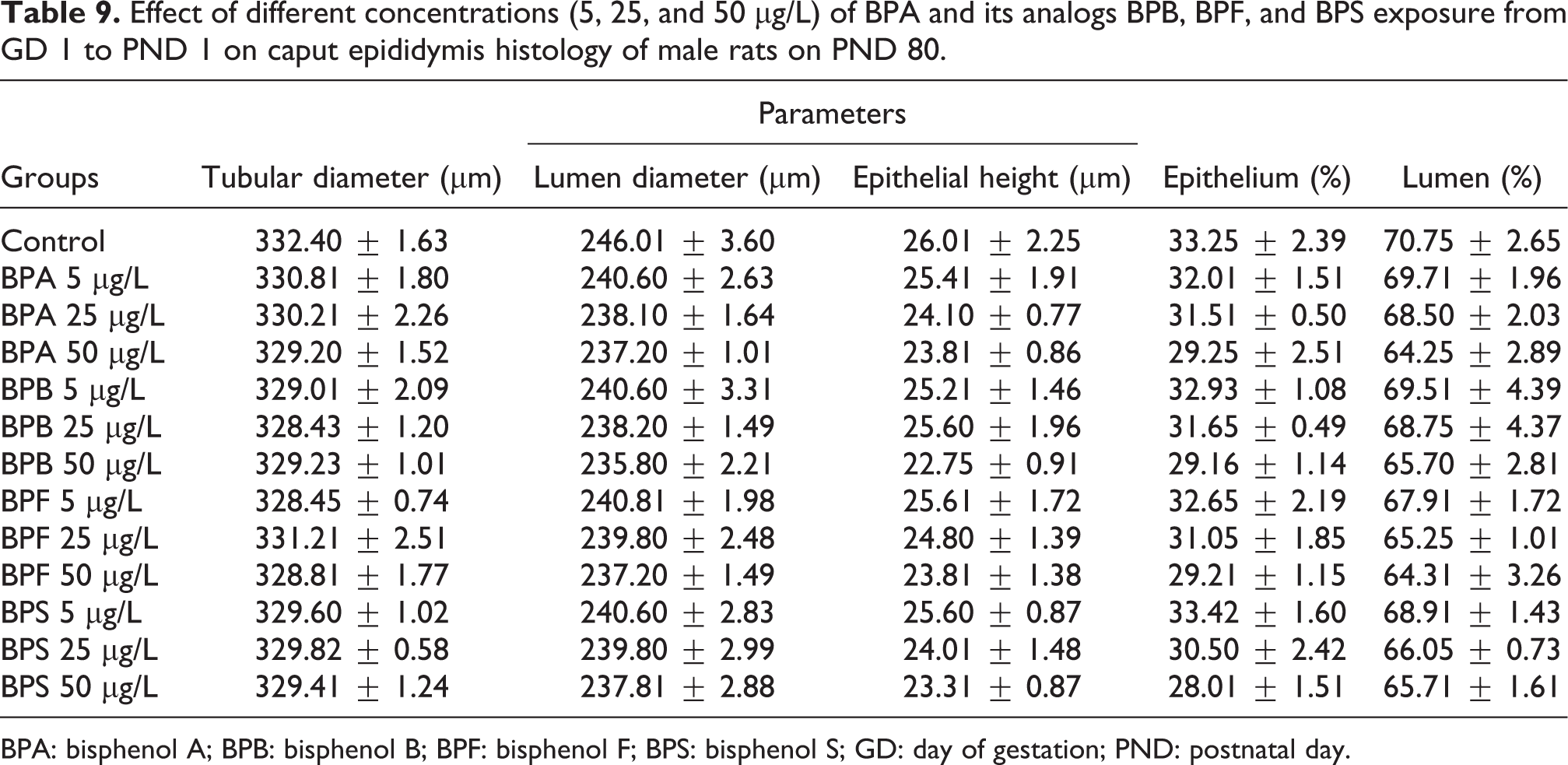
Effect of different concentrations (0, 5, 25, and 50 μg/L) of BPA and its analogs BPB, BPF, and BPS exposure from GD 1 to PND 1 on offspring.
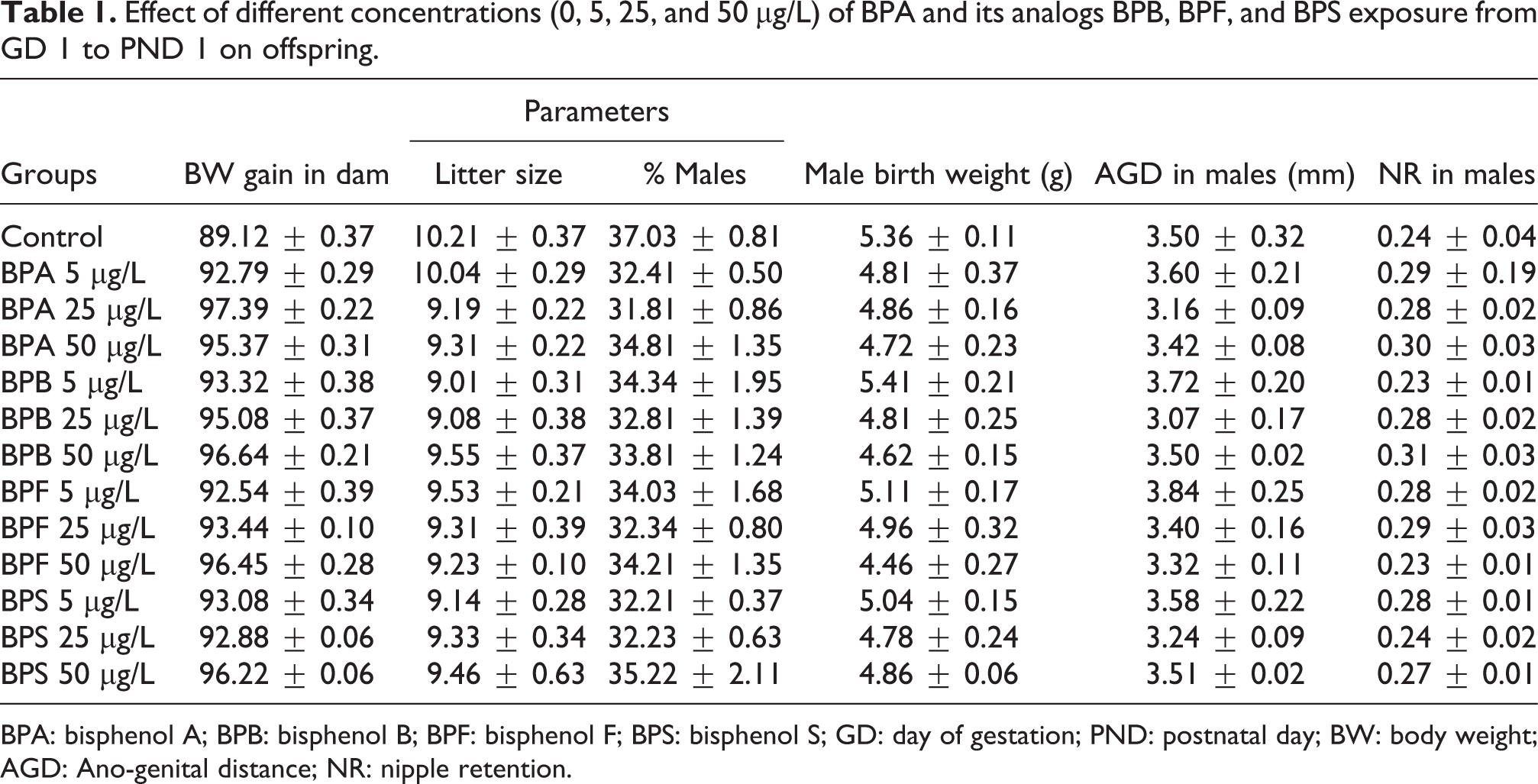
BPA: bisphenol A; BPB: bisphenol B; BPF: bisphenol F; BPS: bisphenol S; GD: day of gestation; PND: postnatal day; BW: body weight; AGD: Ano-genital distance; NR: nipple retention.
Effects of BPA and its analogs BPB, BPF, and BPS exposure on AGD and NR
AGD and NR in the different treatment groups and control are presented in Table 1. There was no significant difference observed in the AGD in the male offspring after prenatal exposure to different concentrations of BPA and its analogs BPB, BPF, and BPS. Similarly, there was also no significant difference observed in the number of the nipple in the prenatal stage of groups exposed to different concentrations of BPA and its analogs BPB, BPF, and BPS as compared to the control. The average number of nipples in control (0.24 ± 0.04) was not statistically different as compared to the BPA, BPB, BPF, and BPS exposed groups (Table 1).
Effects of BPA and its analogs BPB, BPF, and BPS exposure on the BWs and different organs weight on PND 16
BWs in male pups were determined on PND 16 and different organs were weighed after dissection represented in Tables 2 and 3. Prenatal exposure to different concentrations of BPA, BPB, BPF, and BPS did not affect the weight of reproductive organs like testis, epididymis, seminal vesicles, prostate, bulbourethral gland, and bulbocavernosus muscles in comparison to the control. Similarly, nonreproductive organs like adrenals, liver, and retroperitoneal fat pad were also unaffected in the groups exposed to different concentrations of BPA, BPB, BPF, and BPS (Tables 2 and 3).
Effect of different concentrations (5, 25, and 50 μg/L) of BPA and its analogs BPB, BPF, and BPS exposure from GD 1 to PND 1 on organ and body weights of offspring on PND 16.

BPA: bisphenol A; BPB: bisphenol B; BPF: bisphenol F; BPS: bisphenol S; GD: day of gestation; PND: postnatal day.
Effect of different concentrations (5, 25, and 50 μg/L) of BPA and its analogs BPB, BPF, and BPS exposure from GD 1 to PND 1 on organ and body weights of offspring on PND 16.

BPA: bisphenol A; BPB: bisphenol B; BPF: bisphenol F; BPS: bisphenol S; GD: day of gestation; PND: postnatal day.
Effects of BPA and its analogs BPB, BPF, and BPS different concentrations exposure on the puberty onset and organ weights on PND 80
All the male pups exposed to different concentrations of BPA and its analogs BPB, BPF, and BPS were analyzed daily for preputial skin after postnatal day 35. The day 1 of puberty was considered as the preputial skin separated in the pups. External signs of puberty analysis showed that BPA and its analogs BPB, BPF, and BPS exposure did not have any effect on this parameter in comparison to the control group. In the BW of male rats, significant increase was observed only in the highest exposure groups of bisphenols as BPA 50 μg/L (p < 0.05), BPB 50 μg/L (p < 0.05), PBF 50 μg/L, and BPS 50 μg/L (p < 0.05) when compared to the control. All the male rats were dissected on PND 80 and different reproductive parameters were observed as presented in Table 4. There was also increase observed in fat pad, liver, kidney, and adrenals weight but the increase was not statistically significant in comparison to the control. Similarly, a nonsignificant reduction was also observed in the prostate of the exposed groups of BPA and its analogs BPB, BPF, and BPS as compared to the control. On the other hand, significant decrease was observed in the seminal vesicle of higher exposure groups as BPA 50 μg/L (p < 0.05), BPB 50 μg/L (p < 0.05), PBF 50 μg/L, and BPS 50 μg/L (p < 0.05) as compared to the control (Table 5). However, there was no significant difference observed in the other treated groups as compared to the control.
Effect of different concentrations (5, 25, and 50 μg/L) of BPA and its analogs BPB, BPF, and BPS exposure from GD 1 to PND 1 on organ and body weights of male offspring on PND 80.a

BPA: bisphenol A; BPB: bisphenol B; BPF: bisphenol F; BPS: bisphenol S; GD: day of gestation; PND: postnatal day.
aValues are presented as mean ± SEM.
bSignificance at p < 0.05 versus control.
Effect of different concentrations (0, 5, 25, and 50 μg/L) of BPA and its analogs BPB, BPF, and BPS exposure from GD 1 to PND 1 on organ and body weights of male offspring on PND 80.a

BPA: bisphenol A; BPB: bisphenol B; BPF: bisphenol F; BPS: bisphenol S; GD: day of gestation; PND: postnatal day.
aValues are presented as mean ± SEM.
bSignificance at p < 0.05 versus control.
Effects of BPA and its analogs BPB, BPF, and BPS on the antioxidant enzymes, lipid peroxidation (LPO), and ROS in the adult male rats after prenatal exposure
Antioxidant enzymes, ROS, and LPO levels in the adult offspring rats testicular tissues after maternal exposure to various concentrations of BPA, BPB, BPF, and BPS are presented in Table 6. The activities of all antioxidant enzymes (CAT, SOD, and POD) were decreased relative to controls for all the high dose groups, whereas ROS and LPO concentrations were significantly increased in the testes of the same groups.
Effects of different concentrations (5, 25, and 50 μg/L) of BPA and its analogs BPB, BPF, and BPS from GD 1 to PND 1 on antioxidants, LPO and ROS in male offspring on PND 80.a
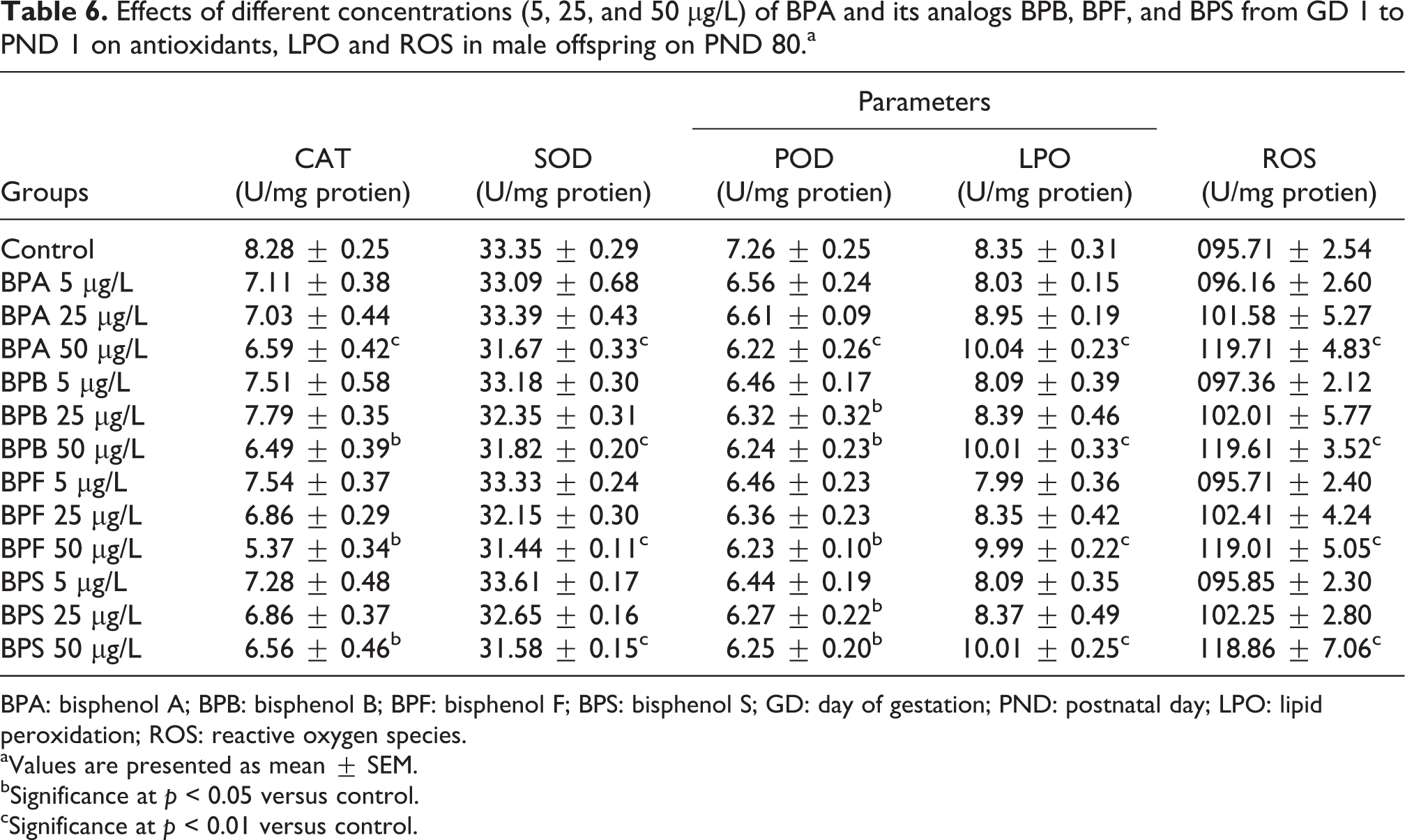
BPA: bisphenol A; BPB: bisphenol B; BPF: bisphenol F; BPS: bisphenol S; GD: day of gestation; PND: postnatal day; LPO: lipid peroxidation; ROS: reactive oxygen species.
aValues are presented as mean ± SEM.
bSignificance at p < 0.05 versus control.
cSignificance at p < 0.01 versus control.
CAT activity was expressed as units per milligram of tissue and in BPA, BPB, BPF, and BPS 50 μg/L significant (p < 0.05) reduction was observed in the exposed group as compared to the control. On the other hand, BPA, BPB, BPF, and BPS 5 and 25 μg/L groups did not show any significant reduction in the cat activity when compared to the control (Table 6).
The activity of SOD was expressed as milliunits per milligram of protein and is presented in Table 6. Significant reduction was observed in BPA, BPB, BPF, and BPS 50 μg/L (p < 0.01) groups when compared to the control. However, BPA, BPB, BPF, and BPS (5 and 25 μg/L) groups did not show any significant difference when compared to the control presented in Table 6.
POD activity expressed as units per milligram of protein in the testis after prenatal exposure reduced significantly (p < 0.05) in BPA 50 μg/L group as compared to the control. Similarly, BPB 25 and 50 μg/L also showed significant reduction (p < 0.05) in the activity of POD as compared to the control. POD activity was reduced significantly (p < 0.05) in BPF 25 and 50 μg/L treated group when compared to the control. Similarly, BPS treatment caused significant reduction (p < 0.05) at the dose level of 25 and 50 μg/L when compared to the control. However, there was no significant reduction observed in other treated groups of BPA, BPB, BPF, and BPS when compared to the control (Table 6).
LPO activity in the different treatment groups and control after prenatal exposure is presented in Table 6. All the high doses groups exposed to prenatal exposure of BPA, BPB, BPF, and BPS (50 μg/L) showed significant increase (p < 0.01) in the LPO activity as compared to the control. However, there was no significant difference observed in 5 and 25 μg/L groups of BPA, BPB, BPF, and BPS as compared to the control.
ROS activity in the testicular tissues of adult male rats after prenatal exposure to different concentrations of BPA, BPB, BPF, and BPS is presented in Table 6. Significant increase was observed in BPA, BPB, BPF, and BPS 50 μg/L (p < 0.01) group when compared to the control. On the other hand, all the other doses (5 and 25 μg/L) of BPA, BPB, BPF, and BPS did not show significant reduction in the ROS activity as compared to the control (Table 6).
Effects of BPA and its analogs BPB, BPF, and BPS different concentration exposure on DSP and sperm parameters of the epididymis
Results of DSP in the control and exposed groups of BPA and its analogs BPB, BPF, and BPS are presented in Table 7. Significant reduction was observed in the DSP of higher exposure groups of BPA, BPB, BPF, and BPS 50 μg/L (p < 0.05) when compared to the control. However, there was no significant difference observed in 5 and 25 μg/L exposed groups of BPA, BPB, BPF, and BPS when compared to the control.
Effect of different concentrations (5, 25, and 50 μg/L) of BPA and its analogs BPB, BPF, and BPS exposure from GD 1 to PD 1 on DSP and sperm in different parts of epididymis of male rats on postnatal day (PND 80).a
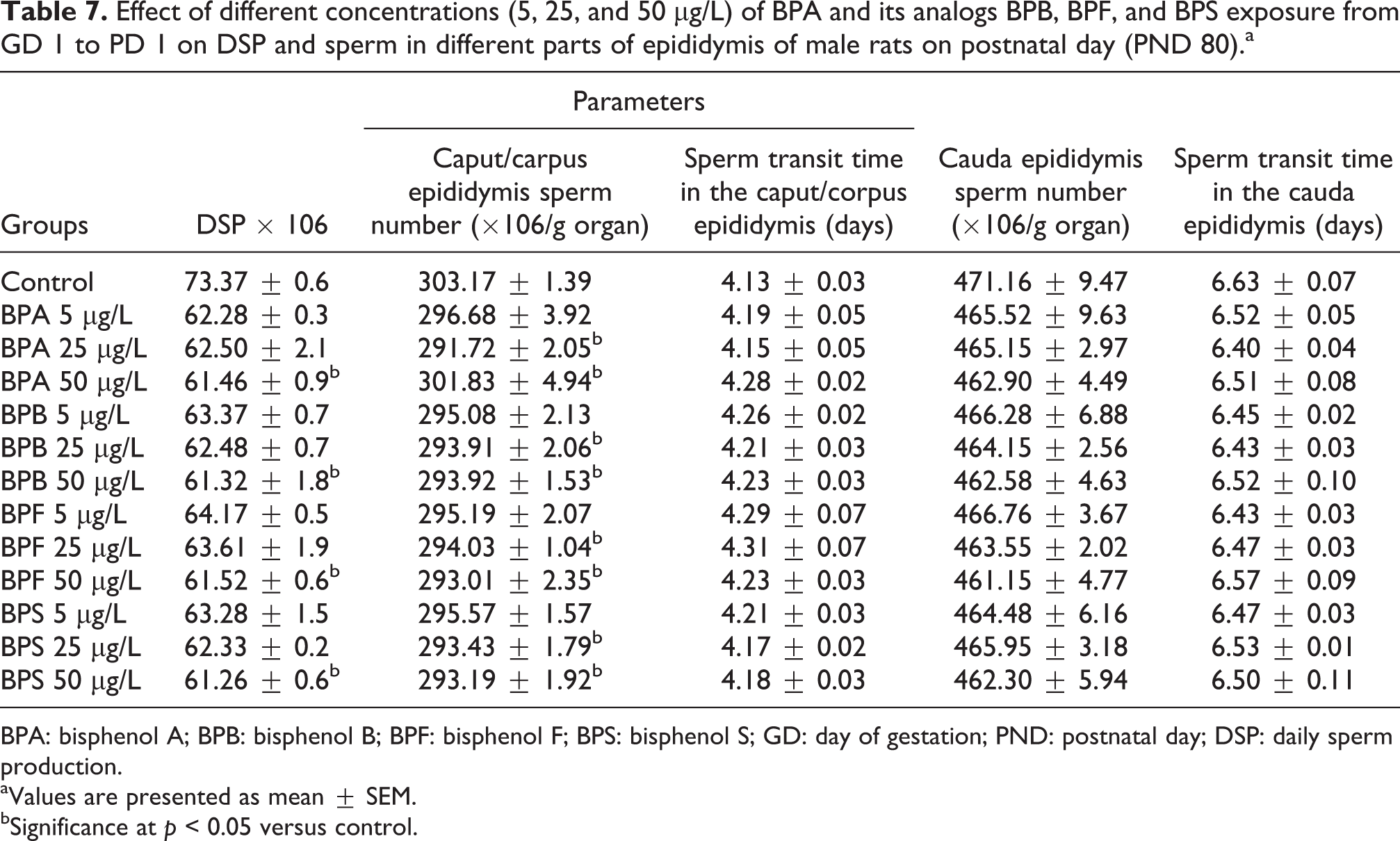
BPA: bisphenol A; BPB: bisphenol B; BPF: bisphenol F; BPS: bisphenol S; GD: day of gestation; PND: postnatal day; DSP: daily sperm production.
aValues are presented as mean ± SEM.
bSignificance at p < 0.05 versus control.
Sperm number in the caput/corpus region of epididymis in the exposed groups was comparable to the control as significant reduction (p < 0.05) was observed in BPA, BPB, BPF, and BPS 25 and 50 μg/L. However, low doses groups 5 μg/L of BPA and its analogs BPB, BPF, and BPS did not reduce the number of sperm in both caput and corpus region as compared to the control. Moreover, sperm transit time in the epididymis and sperm number in the epididymis was not statistically different in the exposed groups as compared to the control group. This suggests that maternal exposure to different concentrations of BPA and its analogs BPB, BPF and BPS do not affect sperm number and sperm transit time in the epididymis of the rats measured on PD 80 (Table 7).
Effects of BPA and its analogs BPB, BPF, and BPS different concentration exposure on histology of testis
Histopathological results in different treatment groups of BPA, BPB, BPF, BPS, and control are presented in Table 8 and Figure 1. On PND 80, prenatal exposure to different concentrations of BPA and its analogs BPB, BPF, and BPS in the area % of seminiferous tubule exhibited marked changes in the testis histology (figure 1). Significant reduction was observed in the area of seminiferous tubules in BPA 25 μg/L (p < 0.05) and BPA 50 μg/L (p < 0.001) when compared to the control group. Similarly, there was also significant reduction observed in the area % of seminiferous tubule of BPB 25 μg/L (p < 0.05) and BPB 50 μg/L (p < 0.01) when compared to the control. Significant reduction was also observed in the area of seminiferous tubules in BPF 25 μg/L (p < 0.01) and BPB 50 μg/L (p < 0.001) when compared to the control. Similarly, BPS 25 μg/L and BPS 50 μg/L caused significant reduction (p < 0.01 and 0.001) in the area of seminiferous tubules when compared to the control group.
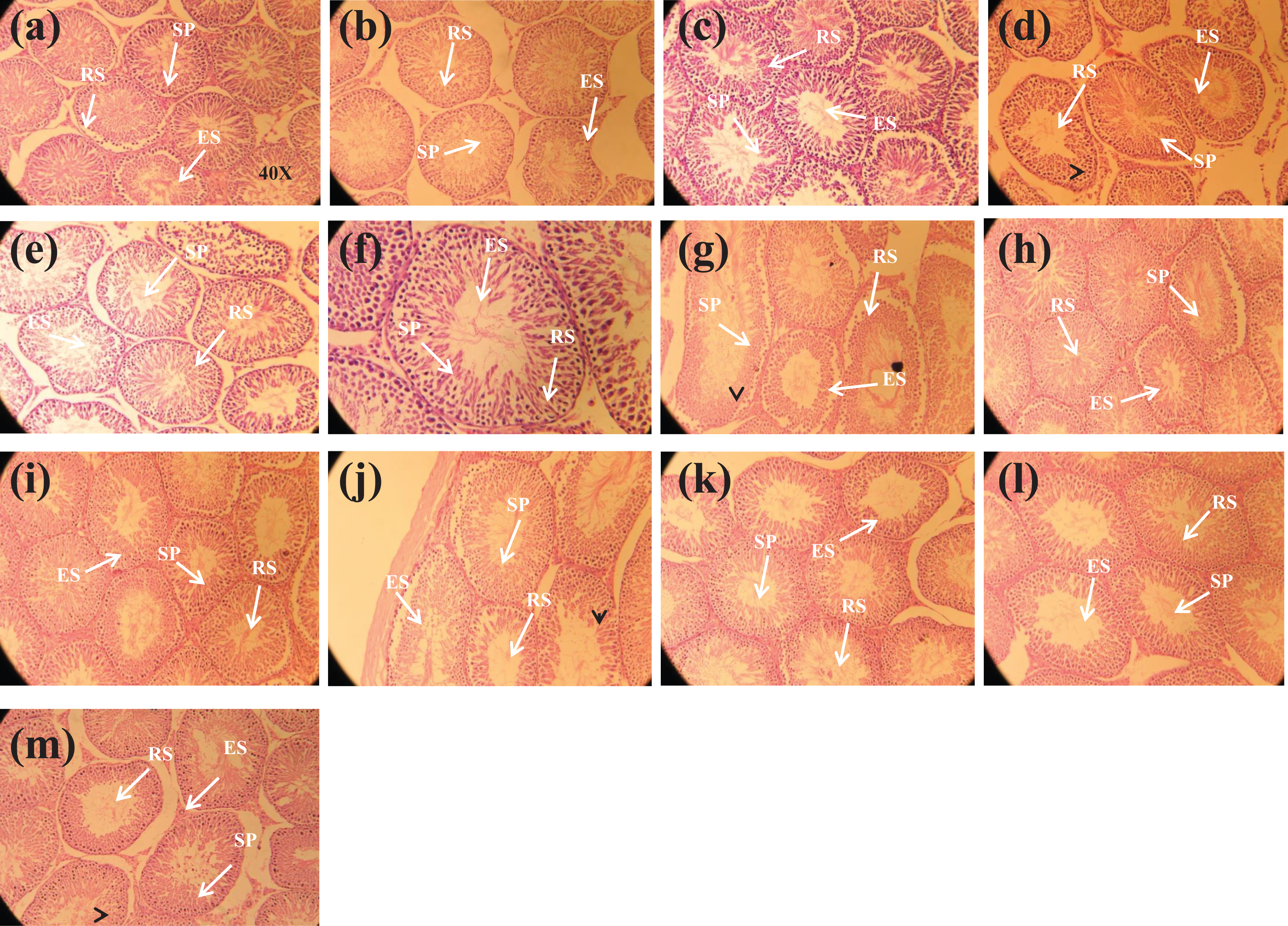
Photomicrograph from testicular tissue showing (a) control; having thick epithelium with SP, RS, ES, and filled lumen with sperm (b–d); BPA (5, 25, and 50 μg/L) treated groups presenting seminiferous tubules with epithelium (line without arrow head) and spermatids (white arrow); (e–g) BPB (5, 25, and 50 μg/L) treated groups presenting seminiferous tubules with epithelium (line without arrow head) and elongating spermatids (white arrow); (h–j) BPF (5, 25, and 50 μg/L) treated groups presenting seminiferous tubules with epithelium (line without arrow head) and elongating spermatids (white arrow); (k–m) BPS (5, 25, and 50 μg/L) treated groups presenting seminiferous tubules with epithelium (line without arrow head) and spermatids (white arrow). H&E (40×). SP: spermatogonia; RS: round spermatids; ES; elongated spermatids; BPA: bisphenol A; BPB: bisphenol B; BPF: bisphenol F; BPS: bisphenol S; H&E: hematoxylin and eosin.
Effect of different concentrations (5, 25, and 50 μg/L) of BPA and its analogs BPB, BPF, and BPS exposure from GD 1 to PND 1 on testis histology of male rats on PND 80.a

BPA: bisphenol A; BPB: bisphenol B; BPF: bisphenol F; BPS: bisphenol S; GD: day of gestation; PND: postnatal day.
aValues are presented as mean ± SEM.
bSignificance at p < 0.05 versus control.
cSignificance at p < 0.001 versus control.
dSignificance at p < 0.01 versus control.
Moreover, marked changes were also observed in the area % of the interstitial space on PND 80 prenatal exposure to different concentrations of BPA and its analogs BPB, BPF, and BPS in the histology of testis. Significant reduction was observed in the area % of interstitial space of BPA, BPB, BPF, and BPS 50 µg/L (p < 0.05) when compared to the control. However, other doses 5 and 25 μg/L of BPA and its analogs BPB, BPF, and BPS did not reduce area % of interstitial space as compared to the control.
Area of the lumen was also observed on PND 80 prenatal exposed rats to different concentrations of BPA and its analogs BPB, BPF, and BPS. Significant reduction (p < 0.001) was observed in the area of the lumen of exposed groups of BPA, BPB, BPF, and BPS 50 μg/L when compared to the control. On the PND 80, prenatal exposure to different concentrations (5 and 25 μg/L) of BPA and its analogs BPB, BPF, and BPS did not cause significant difference in the area % of epithelium in the testis histology.
Seminiferous tubules diameter in different treatment groups and control is presented in Table 8. Significant reduction was observed in the seminiferous tubules diameter in BPA, BPB, BPF, and BPS 50 μg/L (p < 0.05) when compared to the control. However, BPA and its analogs BPB, BPF, and BPS other doses (5 and 25 μg/L) did not reduce seminiferous tubule diameter as compared to the control.
Seminiferous tubule epithelial height in the testis histology showed significant increase in the treated group of BPA 50 μg/L (p < 0.05) when compared to the control. Epithelial height in the seminiferous tubules increased significantly (p < 0.01) in BPB 50 μg/L treated group as compared to the control. Similarly, BPF treatment caused significant (p < 0.05) increase in 50 μg/L treated group when compared to the control group. Likewise, BPS 50 μg/L also increased (p < 0.01) seminiferous tubules epithelial height when compared to the control group (Table 8).
Caput and Cauda epididymis histology of exposure to BPA and its analogs BPB, BPF, and BPS after PND 80
On PND 80, prenatal exposure to different concentrations of BPA and its analogs BPB, BPF, and BPS exhibited no marked changes in the caput epididymis histology presented in Table 9 and figure 2 and 3. Histology of caput epididymis was evaluated and diameter of lumen and tubules, height of epithelial, lumen, and epithelial percentage were not significantly different in the exposed groups as compared to the control.
Effect of different concentrations (5, 25, and 50 μg/L) of BPA and its analogs BPB, BPF, and BPS exposure from GD 1 to PND 1 on caput epididymis histology of male rats on PND 80.
BPA: bisphenol A; BPB: bisphenol B; BPF: bisphenol F; BPS: bisphenol S; GD: day of gestation; PND: postnatal day.
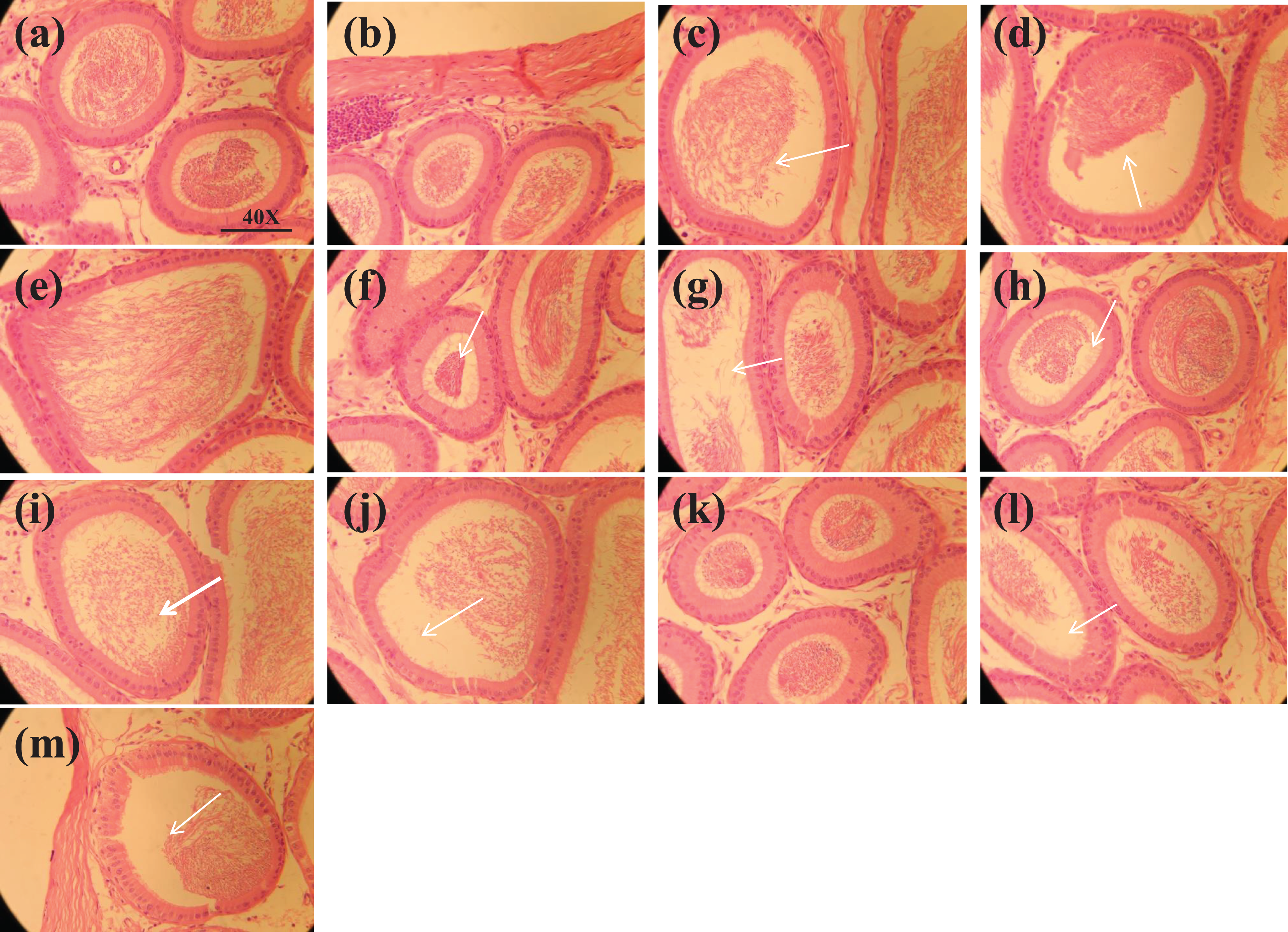
Photomicrograph of caput epididymis tissue showing (a) control; with compact arrangement of caput tubules with sperm filled lumen; (b–d) BPA (5, 25, and 50 μg/L) exposed groups presenting seminiferous tubules with less number of sperm in the lumen or tubules with empty lumen (arrow). Similarly, (e–g) BPB (5, 25, and 50 μg/L) exposed groups showing less number of sperms in the lumen or tubules with the empty lumen (arrow). (h–j) PBF (5, 25, and 50 μg/L) exposed groups, presenting seminiferous tubules with less number of sperm in the lumen (arrow) and exposed group showing less number of sperms lumen (arrow). Likewise, (k–m) BPS (5, 25, and 50 μg/L) exposed groups showing caput tubules with less number of sperms in the lumen and exposed group presenting less number of sperms and empty lumen. H&E (40×). BPA: bisphenol A; BPB: bisphenol B; BPF: bisphenol F; BPS: bisphenol S; H&E: hematoxylin and eosin.

Photomicrograph of cauda epididymis tissue showing (a) control; with compact arrangement of cauda tubules with sperm filled lumen; (b–d) BPA (5, 25, and 50 μg/L) exposed groups presenting cauda tubules with less sperm in the lumen. Similarly, (e–g) BPB (5, 25, and 50 μg/L) exposed groups, presenting cauda tubules with less sperm in the lumen. Likewise, (h–j) BPF (5, 25, and 50 μg/L) exposed group presenting cauda tubules with less sperm in the lumen. In the same way, (k–m) BPS (5, 25, and 50 μg/L) exposed group presenting cauda tubules with less sperm in the lumen. H&E (40×). BPA: bisphenol A; BPB: bisphenol B; BPF: bisphenol F; BPS: bisphenol S; H&E: hematoxylin and eosin.
Cauda epididymis histology of different treatment groups of BPB, BPF, and BPS and control is presented in Table 10. There was also no significant difference observed in the area covered by epithelium and lumen in the histology of cauda epididymis when compared to the control. Maternal exposure to different doses of BPA and its analogs BPB, BPF, and BPS did not have any significant difference in the tubular diameter, lumen diameter, and epithelial height after exposure.
Effect of different concentrations (5, 25, and 50 μg/L) of BPA and its analogs BPB, BPF, and BPS exposure from GD 1 to PND 1 on cauda epididymis histology of male rats on PND 80.

BPA: bisphenol A; BPB: bisphenol B; BPF: bisphenol F; BPS: bisphenol S; GD: day of gestation; PND: postnatal day.
Effects of different concentrations (5, 25, and 50 μg/L) of BPA, BPB, BPF, and BPS exposure from GD 1 to PND 1 on plasma hormones concentrations in male rats on PND 80
Plasma testosterone concentrations in different treatment groups of BPA and its analogs BPB, BPF, and BPS and control are presented in Table 11. Testosterone concentrations reduced significantly in BPA, BPB, BPF, and BPS 50 μg/L (p < 0.05) when compared to the control. On the other hand, BPA, BPB, BPF, and BPS low concentrations (5 and 25 μg/L) did not have any significant difference in the plasma testosterone concentrations as compared to the control.
Effect of different concentrations (5, 25, and 50 μg/L) of BPA and its analogies BPB, BPF, and BPS exposure from GD 1 to PD 1 on plasma testosterone and estrogen concentrations in male rats on PD 80.a

BPA: bisphenol A; BPB: bisphenol B; BPF: bisphenol F; BPS: bisphenol S; GD: day of gestation; PND: postnatal day; LH: luteinizing hormone; FSH: follicle stimulating hormone.
aValues are presented as mean ± SEM.
bSignificance at p < 0.05 versus control.
cSignificance at p < 0.001 versus control.
dSignificance at p < 0.01 versus control.
Plasma estradiol after PND 80 of exposure showed significant increase in BPA, BPB, BPF, and BPS 50 μg/L (p < 0.001) as compared to the control. However, other doses (5 and 25 µg/L) of BPA and its analogs BPB, BPF, and BPS did not reduce plasma estradiol as compared to the control.
Plasma LH concentrations in different treatment groups and control are presented in Table 11. Significant reduction in LH concentrations was observed in BPA, BPB, BPF, and BPS 50 µg/L (p < 0.001) treated groups when compared to the control. However, there was no significant difference observed in the other treated groups of BPA and its analogs BPB, BPF, and BPS in comparison to the control.
Plasma FSH concentrations in the different treated groups of BPA and its analogs BPB, BPF, and BPS are presented in Table 11. Significant reduction in BPA, BPB, BPF, and BPS 50 μg/L (p < 0.01) was observed when compared to the control. However, BPA, BPB, BPF, and BPS 5 and 25 μg/L did not affect plasma FSH concentrations when compared to the control group. Total number of different cells population present in the seminiferous tubules of rats testis is presented in Table 12. Significant reduction in the number of spermatogonia was observed in the group exposed with the highest doses of BPA, BPB, BPF, and BPS 50 μg/L (p < 0.05) when compared with the control. However, BPA, BPB, BPF, and BPS 5 and 25 μg/L treated groups did not significantly reduce number of spermatogonia as compared to the control.
Effect of different concentrations (5, 25, and 50 μg/L) of BPA and its analogs BPB, BPF, and BPS exposure from GD 1 to PND 1 on the number of different cell types in the testis and sperm number and motility in male rats on PND 80.a

aValues are presented as mean ± SEM.
bSignificance at p < 0.05 versus control.
cSignificance at p < 0.01 versus control.
Effects of different concentrations (5, 25, and 50 μg/L) of BPA, BPB, BPF, and BPS exposure from GD 1 to PND 1 on the number of different cell types in the testis and sperm number and motility in male rats on PND 80
In the number of spermatocytes, significant reduction was observed in the highest dose treated groups of BPA, BPB, BPF, and BPS 50 μg/L (p < 0.05) when compared to the control. On the other hand, the other doses 5 and 25 μg/L of BPA, BPB, BPF, and BPS did not reduce number of spermatocytes as compared to the control.
Number of spermatids in different treatment groups and control is presented in Table 12. Significant reduction was observed in the highest dose groups of BPA, BPB, BPF, and BPS 50 μg/L (p < 0.01) when compared to the control. However, there was no significant difference observed in BPA, BPB, BPF, and BPS 5 and 25 μg/L treated groups when compared to the control.
Exposure to different concentration of BPA and its analogs BPB, BPF, and BPS did not cause any significant reduction in the percentage of motile sperm as presented in Table 12. However, highest concentrations of BPA (50 μg/L) caused significant reduction (p < 0.05) on the percentage motile sperm when compare to the control. Significant reduction was observed in BPB 50 μg/L (p < 0.01) when compared to the control. Motile sperm percentage was reduced significantly (p < 0.05 and 0.01) in BPF 25 and 50 μg/L as compared to the control. On the other hand, PBS 25 and 50 μg/L significantly reduced (p < 0.05 and 0.01) percentage of motile sperms when comparison was done with the control.
Discussion
EDCs are synthetic or natural compounds which alter the endocrine functions often through mimicking or blocking the endogenous hormones. 71 Plasticizers and pesticides are often the main sources of these synthetic EDCs. The action of these EDCs on the endocrine system has resulted in the developmental deficits in many invertebrates and mammals. 72 –74 Exposure in early life to EDCs appear to have more severe effects and endocrine disturbances also persist through later life. 44,75,76 In the present study, we investigated the possible effects of BPA and its analogs BPB, BPF, and BPS different concentrations in drinking water in the prenatal development of rats.
Endocrine disruptors research has shown that persistent exposure to low dose of EDCs leads to disturbed molecular, cellular, and physiological functions in many organs inside the body. 6,77,78 The present study shows that exposure to different levels of BPA and its analogs BPB, BPF, and BPS impact the animals through postnatal growth and sexual maturation. In the development of the normal reproductive system in the prenatal life, there is a great role of pituitary and hypothalamus. Hormones from both pituitary and hypothalamus lead into the normal development of the reproductive system and any abnormality in the levels of hormones at the developmental stage can lead to abnormal development or poor reproductive efficiency. 79 –81 Gonadotropin-releasing hormone (GnRH) in this regard has a great role in the regulation of spermatogenesis and testosterone secretion in the testis. 82,83 For the normal onset of puberty, GnRH plays an important role and it has great physiological function in the adult animals. 84 GnRH is furthered controlled by another important member of hormone in the hypothalamus known as kisspeptin which regulates the normal pulsatile secretion of GnRH. 85 GnRH pulse also play an important role in the regulation of estrogen receptor which control the onset of puberty and ovulation in mammals. Abnormal levels of estrogen due to EDCs lead to abnormal development in rats. 85,86 Exposure to EDCs in rodents during the developmental stages has been observed to be associated with disturbed reproductive functions at puberty. 87 EDCs exposure in human has been observed to induce alterations in the normal development of reproductive organs. 88,89 BPA and its analogs BPB, BPF, and BPS different concentrations were investigated and its effects were analyzed in the rats through water exposure by prenatal exposure in the offspring reproductive system. 19,44,90 –97
Previously, it has been observed that animals prenatally exposed to BPA were observed in weight gain in the offspring and obesity with adult stages of life. 17,98 –102 In the current study, we observed increased BW by prenatal exposure to BPA, BPB, BPF, and BPS in pups at PND 1 until adult. There was significant difference observed in the BW gain of the animals exposed to high concentrations of BPA, BPB, BPF, and BPS in comparison to the control in the current study which is also in line with different previous studies. 76,93,103,104
Development of reproductive organs and its physical examination has shown toxic effects of EDCs (BPA and its analogs) on the development of the reproductive system in rodents as AGD in animals (the distance between anus and genitals which reflect the state of development of reproductive system in rodents and mammals). 105 –109 In our current study, there was no significant difference observed in the AGD of male rats exposed to different concentrations of BPA and its analogs as BPB, BPF, and BPS as compared to the control. There have been previously similar studies where animals have been exposed to different concentrations of BPA and some of its analogs in which they have not shown any difference in the anogenital area. 105,110,111 Similarly, NR is also considered an important marker for the altered androgens at the time of development. 98,106,112 This can give an indication of an abnormal reproductive system at the time of puberty. 113 Parameters as such are these days considered necessary for the detection of adverse effects of any EDCs exposure. 113,114 There was no significant difference observed in the NR of male rats which can be because of the concentrations of different bisphenols used for this study.
It has also been observed that BPA and some of its analogs are also involved in alteration of the structure of the antioxidant enzymes by inducing toxicity in different organs of the reproductive system. 22,115 Different in vitro and in vivo studies have shown BPA and some of its analogs exposure induce antioxidant enzymes activity, increasing ROS concentrations, reduce DSP, and alteration in the tubules of seminiferous tubules. 22,97,115,116 Different sperm parameters were observed in the present study showing significant reduction in the percentage of motile sperms in the higher dose groups of BPA and its analogs BPB, BPF, and BPS.
There were also fewer sperms observed in the caput and corpus epididymis and the number of DSP was also reduced in the epididymis in some of the BPA, BPB, BPF, and BPS treated groups. The current study results are in accordance with some of the previous studies where prenatal exposure to BPA leads into arrest in the number of spermatogonial cells in the testicular tissues and less number of elongated sperms were observed in the seminiferous tubules. 117 –121 It can be stated from the results of the present study that BPA analogs BPB, BPF, and BPS not only lead into the reduction in the number of sperm but also lead into disturbance in the cauda and caput epididymis environment by reducing the viability and motility of sperms present.
Hormones play an important role in the initiation of puberty. In the concentrations of hormones like testosterone, progesterone, LH, and FSH significant differences were observed in animals exposed to different concentrations of BPA and its analogs BPB, BPF, and BPS. There have been several studies which have shown the same effect on the concentrations of different hormones after exposure to BPA or some of its analogs. 17,24,76,90,122 There was significant difference observed in the hormone levels of these animals exposed to low and high concentrations of BPA and its analogs. Which in other words suggest that considerably low dose of BPA and its analogs also bring considerable change on the development of many systems in the prenatal period. 24,44,90
In the sperm parameters, there was reduction observed in the DSP of the different groups of animals exposed to different concentrations of BPA and its analogs as BPB, BPF, and BPS. The comparable difference was observed in the number of sperms in the caput/corpus of epididymis in the exposed groups which was also observed in some previous studies. 122 –125 Other sperm parameters were also different in the treated groups of BPA and its analogs which are in relation with some previous studies. 77,126,127
Moreover, reduced hormonal levels were observed along with reduced number of sperm in the epididymis in the groups exposed to BPA and its analogs BPB, BPF, and BPS which are in accordance with the different previous studies were BPA and its analogs were observed with the similar results. 12,100,128 –131 The present study results regarding the hormonal levels are also in accordance with multiple studies with BPA and some of its analogs which reduced the concentrations of FSH supporting the histological alterations in the testis. 17,22,23,128,132,133
Histological results of testis revealed significant change in the morphology of testicular cells which can be because of estrogen receptors in these organs which play critical role in the spermatogenesis. Previously, there have been studies which have shown that exposure to different concentrations of BPA and its analogs in the prenatal life altered different hormones levels. 17,22,28,81,97,134 –136
The current study demonstrates that maternal exposure to 5–50 μg/L of BPA and its analogs BPB, BPF, and BPS at different concentrations during gestation and lactation period produces adverse effects on the development of male rat reproductive system. BPA and its analogs BPB, BPF, and BPS at different low concentrations prenatally exposed to male rats have potentially hazardous effects on spermatogenesis and lead to oxidative stress in the reproductive organs of male rats by not only reducing the DSP but also altering seminiferous tubule epithelium.
Conclusion
In conclusion, the results of the present study indicate the interactions between EDCs (BPA and its analogs) and reproductive system. However, BPA and its analogs BPB, BPF, and BPS showed effects on sexual development recognizing that bispheonols exposure to mother during pregnancy may induce reproductive toxicity in the male offspring. Here, we concluded that low concentrations of BPA and its analogs may have effect on the different organs and sexual development of adult male rats. Pregnancy and lactation are both critical periods for reproductive development in the offspring. Exposure to BPA and its analogs BPB, BPF, and BPS during this period will interfere with the development and function of reproductive systems. Our study implied that higher concentrations of BPA and its analogs BPB, BPF, and BPS may be the leading cause of abnormal reproductive functions related to BPA analogs toxicties in the early development in male rats.
Footnotes
Acknowledgements
This piece of work was fully funded by the Department of Animal Sciences, Quaid-i-Azam University, Islamabad, Pakistan.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.

