Abstract
Although Bisphenol A (BPA) is still used in consumer products, concerns about its toxicity led to the adoption of structurally related replacement products such as Bisphenol F (BPF) and Bisphenol S (BPS). Unfortunately, comparing the biological responses to BPA and BPA substitutes
Introduction
Bisphenol A (BPA, CAS 80-05-7) is present in a wide range of consumer products including electronics, toys, medical equipment, paper receipts, and food containers. 1 The acute toxicity of BPA is low, as rat oral LD50 estimates range from 3.25 to 5.0 g/kg bodyweight (bw). 2 In rodent repeated exposure studies performed according to standardized regulatory guidelines, BPA toxicity is predominantly observed above 100 mg/kg bw/day, while the no-observed-adverse-effect level (NOAEL) is estimated at 5 mg/kg bw/day. 3 Additionally, BPA’s effects in standardized regulatory uterotrophic and Hershberger assays also proved subtle and equivocal.4,5
Contrastingly, hypothesis-driven studies focusing on various endocrine-related endpoints reported effects of BPA in the low/sub microgram per kg bw range, in both males and females. 6 These discrepancies between regulatory-based and exploratory studies were alternatively attributed to differences in rodent strains and exposure protocols, different sources of bisphenol contamination, variable phytoestrogen levels in animal feed, and non-monotonic dose–response curves.7-9 These conflicting observations fuelled debates on the toxicity of BPA3,10,11 and more generally on the health risk assessment of endocrine disruptors.12,13 Faced with such contradictory evidence, regulators in North America and Europe adopted measures to limit the use of BPA, notably in baby bottles. 1
Bisphenol F (BPF, CAS 620-92-8) and Bisphenol S (BPS, CAS 80-09-1) are two prominent BPA substitutes. Although BPF and BPS levels are increasing in environmental and human samples,14-19 information on their
As observed for BPA, regulatory and hypothesis-driven studies also reached diverging conclusions on the impacts and potencies of BPF and BPS. Hypothesis-driven
The comparison of BPA, BPF, and BPS
Materials and Methods
Materials
Bisphenol A (Cat# 239658, Lot# MKCD7508, >99.0% purity), Bisphenol F (Cat# B47006, Lot# 05712ME, 99.3% purity), Bisphenol S (Cat# 103039, Lot# MKCF0795, 99.3% purity), EE (Cat# E4876, Lot# MKBK0176V, 100% purity), and sodium carboxymethyl cellulose (average MW ∼250,000, degree of substitution 0.9, Cat# 419303, Lot# MKCB9856) were purchased from Sigma-Aldrich Canada (Oakville, ON, Canada). Unless otherwise specified, additional chemicals and reagents were also purchased from Sigma-Aldrich Canada.
Animal Treatment
The animal exposure protocol based on OECD Test Guideline 407
42
complied with the Canadian Council on Animal Care guidelines and was approved by Health Canada’s Institutional Animal Care Committee (HCO-ACC protocol# 2018-001). Approximately 6-week-old male Fischer rats (Charles River Laboratories, St. Constant, QC, Canada) were allowed to acclimate to the facility and trained for gavage for 12 to 16 days. Rats were housed singly in vented Allentown NexGen Rat 900 polysulfone cages with water bottles of the same material fitted with stainless steel dispenser caps (Allentown Inc. Allentown, NJ, USA). They were provided Teklad Aspen Sani-Chips Bedding (quality assurance report date 11/08/18, Envigo, East Millstone, NJ, USA), with metal shelter, 100% Aspen Chew Stick (Lomir Biomedical, Notre-Dame-de-l'Île-Perrot, QC, Canada), Rodent Nesting Sheets (Cat# K3510, Lot# 219252, Bio-Serv, Flemington, NJ, USA), and Bed-r’Nest puck (Cat# BRN4SR, The Andersons, Inc. Maumee, OH, USA). The room was maintained at 22 ± 1°C and 50 ± 10% humidity on a 12 hour light cycle (6:00 AM to 6:00 PM). Rats had
Fischer rats were selected for this investigation, as their reported sensitivity to estrogens and endocrine disruptors43,44 may facilitate the comparison of BPA, BPF, and BPS endocrine effects. A preliminary 14-day pilot study assessing rat bodyweight gains, liver enzymatic activities, and a limited panel of clinical chemistry parameters was performed to identify BPA, BPF, and BPS doses affecting these endpoints without causing overt signs of toxicity. These equipotent bisphenol doses were then decreased by a constant factor over five levels, ranging from the doses typically assessed in regulatory studies to the doses assessed in more hypothesis-driven investigations. Due to the low bisphenol doses administered and to the inherent variability of serum hormone levels in males, 45 nine rats were allocated per treatment group.
Using random numbers generated in Excel, 153 rats (127 ± 16 g, approximately 8 weeks old) were randomly divided across 17 treatment groups (9 rats per treatment group). A similar approach was used to determine the locations of the rat cages on the racks and the order of necropsy, using a randomized block design (9 blocks). 46 Staff involved in both the animal exposure phase and the assessment of endpoints were not blinded to the treatment groups, in order to implement precautionary measures to prevent cross-contamination. Control vehicle and EE treatment groups were always weighed and dosed first, and bisphenol-treated rats were dosed from the lowest to the highest doses, changing gloves between BPA, BPF, and BPS to avoid cross-contamination. Weekly cage changes were done in an animal transfer station with filtered air flow to further limit contamination.
Rats were gavaged for 28 consecutive days with BPA (0.05, 0.5, 5, 50, or 500 mg/kg bw/day), BPF (0.035, 0.35, 3.5, 35, or 350 mg/kg bw/day), BPS (0.03, 0.3, 3, 30, or 300 mg/kg bw/day), or EE (50 μg/kg bw/day) resuspended in a weight-adjusted volume of 0.3% sodium carboxymethyl cellulose in water (5 mL/kg bw). Rats from the control group received the same weight-adjusted volume of dosing vehicle. Stock solutions of BPA, BPF, and BPS stored at 4°C were extensively stirred and sonicated before preparing the working solutions weekly. An ethinylestradiol stock solution in 100% ethanol stored at −20°C was diluted 5000 times in aqueous 0.3% sodium carboxymethyl cellulose and briefly mixed to generate a new dosing solution daily. Dosing solutions were continuously stirred while dosing rats. Starting on the second week of exposure, a few rats from the 500 mg BPA/kg treatment group showed mild signs of dehydration. They were provided HydroGel (Cat# 70-01-5022, Lot# 091818, ClearH2O, Westbrook, ME, USA) until the dehydration resolved.
On the 21st day of dosing, rats were transferred to metabolic cages with a solid platform to collect urine and feces for 24 h. At 9:00 PM on the night preceding the final necropsy, rats were fasted. The following morning, they were anaesthetized using isoflurane and blood was withdrawn from the abdominal aorta for hematology and serum clinical chemistry analyses. Brain, heart, liver, spleen, kidneys, adrenals, thymus, testes, epididymis, prostate, and seminal glands were excised and weighed. A piece of liver (approximately 1.5 g) was excised, homogenized in 2.5 volume of ice-cold 0.05 M Tris/0.15% KCl buffer, pH 7.4 and centrifuged at 10,000 g for 20 min at 4°C to obtain the S9 fraction. Sera and liver S9 fractions were kept at −80°C until analysis.
Quantification of Urinary BPA, BPF, and BPS
BPA, BPF, and BPS quantification protocols were based on Method 6301.04 from the Centers for Disease Control and Prevention. Analytical bisphenols and isotopically labeled bisphenols for calibration, spiking, and standard curves were purchased from Cambridge Isotope Laboratories (Tewksbury, MA, USA) and included BPA (Cat# ULM-7106-1.2), 13C12-BPA (Cat# CLM-4325-1.2), BPF (Cat# ULM-9827-1.2), 13C12-BPF (Cat# CLM-9866-1.2), BPS (Cat# ULM-9320-1.2), and 13C12-BPS (Cat# CLM-9319-1.2). Unless otherwise stated, Optima grade solvents and reagents for liquid chromatography–mass spectrometry (LC-MS) were purchased from Thermo Fischer Scientific (Nepean, ON, Canada).
Bisphenol deconjugation was carried out in a final reaction volume of 400 μL containing 100 μL of sample (rat urine, appropriately diluted in water), 100 μL of isotopically labeled bisphenol standards (in methanol at 125 ppb each), 100 μL of deconjugation standards (4-methylumbelliferyl sulfate (Sigma-Aldrich, Cat# M733), 4-methylumbelliferyl-β-D-glucuronide (Sigma-Aldrich, Cat# M9130) and 13C4-4-methylumbelliferone (Cambridge Isotope Laboratories, Cat# CLM-6023-S) in methanol at 250 ppb each), and 100 μL 1.0 M ammonium acetate buffer (pH 5.0) containing 0.45 U/mL
Solid phase extractions were performed using Oasis HLB 6 cc/200 mg cartridges (Waters, Milford, MA, USA). Samples were added to the HLB cartridges (preconditioned with acetone, followed by methanol and then water). The cartridges were washed with 12 mL of Optima grade water and then dried completely under vacuum. Samples were eluted with 6 mL of methanol followed by 6 mL of acetone. Afterwards, eluates were transferred to a TurboVap II (Biotage, Uppsala, Sweden) and evaporated to dryness under a gentle nitrogen stream in a water bath set at 45°C. Samples were reconstituted in 250 μL methanol.
Samples were analyzed on a Waters ACQUITY Ultra Performance LC (coupled to a TSQ Quantum Ultra mass spectrometer (Thermo Fischer Scientific)). Five μL of reconstituted sample were injected and separation was performed at 30°C on an ACQUITY UPLC BEH C18 column (1.7 µm, 2.1 mm × 50 mm, Waters) fitted with a VanGuard Pre-Column (2.1 × 5 mm). The mobile phase consisted of (A) 5% methanol in Optima water and (B) 100% methanol. The analytes were eluted using a linear gradient going from 5 to 100% methanol in 7 minutes. Four minutes were allowed for equilibration between sample injections. Flow rate was set at 0.25 mL/min. The mass spectrometer detector was operated in multiple reaction monitoring using electrospray ionization in negative mode (ESI-). The capillary temperature was set at 350°C and the collision gas pressure at 1.5 mTorr. The precursor ion -> product ion (m/z) transitions monitored for quantification (and confirmation) were 227.00 -> 211.20 (133.26) for BPA, 199.00 -> 93.3 (105.4) for BPF, and 248.80 -> 108.20 (92.20) for BPS.
Method blanks and standard calibration curves (7 concentrations, 1-200 ppb) were run for every set of samples. Quality control criteria for standard curves were a R2 > 0.995 and a calculated concentration of the calibration standards within 30% of the expected value. Deconjugation was considered complete with a 4-UMB/4-UMB (IS) area count ratio above 0.4. Recovery for spiked control samples was considered satisfactory between 70% and 130%. Limits of detection and limits of quantification were 0.70 ppb and 0.94 ppb for BPA, 0.73 ppb and 1.02 ppb for BPF, and 0.32 ppb and 0.63 ppb for BPS, respectively. As the lowest calibration standard used for quantification was 1 ppb, the method quantification limits for BPA and BPS were 1.00 ppb and 1.02 ppb for BPF.
Urinary Bisphenol Excretion in Male Rats Exposed to BPA, BPF, or BPS. Urine 24 h Volumes, Creatinine Levels, Specific Gravities, and Specific Gravity-Corrected BPA, BPF, and BPS Concentrations Are Provided.
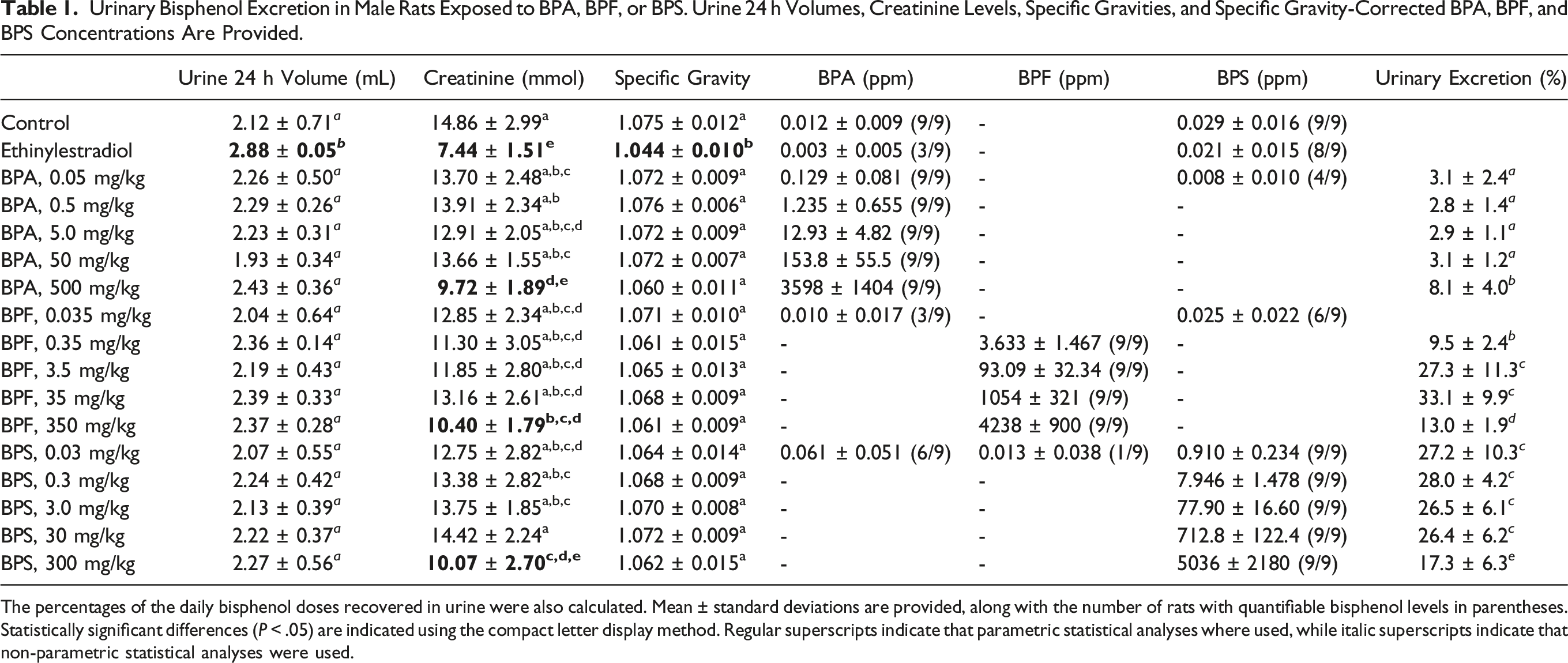
The percentages of the daily bisphenol doses recovered in urine were also calculated. Mean ± standard deviations are provided, along with the number of rats with quantifiable bisphenol levels in parentheses. Statistically significant differences (
Hematology, Clinical Chemistry, and Liver Enzymatic Activities
Hematological parameters (white blood cells, red blood cells, hemoglobin, hematocrit, mean corpuscular volume, mean corpuscular hemoglobin, mean corpuscular hemoglobin concentration, platelet, mean platelet volume, platelet distribution width, platelet large cell ratio, reticulocytes, neutrophils, lymphocytes, monocytes, eosinophils, and basophils) were measured using a Sysmex XT-2000iV (IDEXX Laboratories, Westbrook, MN, USA). Serum clinical chemistry (total protein, albumin, creatinine, blood urea nitrogen, bilirubin, calcium, inorganic phosphate, cholesterol, triglycerides and alkaline phosphatase, alanine aminotransferase, aspartate aminotransferase activities, and gamma-glutamyl transferase) and urine creatinine levels were assessed using an ABX Pentra 400 clinical analyzer (Horiba ABX Inc., Irvine, CA, USA). Adrenocorticotropic hormone, luteinizing hormone, follicle stimulating hormone, thyroid stimulating hormone and prolactin were measured in duplicate using a MILLIPLEX assay (Cat# RPTMAG-86K-04) from MilliporeSigma (Sigma-Aldrich Canada). Serum estradiol levels were measured in duplicate using an ELISA (Cat# 55-ESTR-E01) from ALPCO (Salem, NH, USA) according to the manufacturer’s instructions. Gonadotropin-releasing hormone and corticosterone levels were measured in duplicate by ELISAs (Cat# EKC39138 and EKU03465) from Biomatik (Wilmington, DE, USA). Ethoxyresorufin-O-deethylase (EROD), benzyloxyresorufin-O-dealkylase (BROD), and pentoxyresorufin-O-dealkylase (PROD) activities were assessed in liver S9 extracts as previously described. 48
Histopathology
All tissues except testes were fixed in 10% neutral buffered formalin solution for at least 24 h before processing and hematoxylin and eosin staining. Testes were fixed in Modified Davidson Solution for 48 h and then transferred to 10% neutral buffered formalin before processing and periodic acid-Schiff and hematoxylin staining. Qualitative assessments of 5 micron sections from the pituitary gland, trachea, lung, esophagus, thyroid, thymus, mammary glands stomach, liver, heart, pancreas, spleen, duodenum, jejunum, ileum, proximal colon, distal colon, adrenal glands, kidney, bladder, mesenteric and axillary lymph nodes, skeletal muscle, testis, prostate, seminal vesicle, and coagulating gland were performed by an experienced pathologist using available guidelines from INHAND (International Harmonization of Nomenclature and Diagnostic Criteria for Lesions in Rats and Mice) as previously described. 48
Statistical Analysis
All rats reached the end of the exposure period and were included in statistical analyses. However, due to technical issues, a reduced number of samples per treatment group were assessed for serum creatinine (6 samples), estradiol (6-7 samples), and corticosterone (5-9 samples). Shapiro–Wilk’s and Brown–Forsythe’s tests were first used to assess data normality and homogeneity of variance. Datasets (or log-transformed datasets) satisfying these assumptions were then analysed by one-way ANOVA followed by the Student-Newman-Keuls post-hoc test. Datasets that failed to meet these assumptions were analyzed using the non-parametric Kruskal–Wallis ANOVA followed by the Student–Newman–Keuls method (or Dunn’s post-hoc test if treatment group sizes were unequal). Differences between treatment groups were considered statistically significant for
Results
Quantification of Urinary Bisphenols
Urine samples were collected on the 21st day of exposure to assess BPA, BPF, and BPS excretion. Intriguingly, lower than usually reported 24-hour urine volumes49,50 and higher than expected urine specific gravities 51 were consistently observed across rats from all treatment groups (Table 1). These observations may be due to the addition of a solid platform to the wire mesh bottom of metabolic cages, which has been shown to result in increased food consumption and lower, more stable urine volume. 50 The highest BPA, BPF, and BPS doses all resulted in similar decreases in urine creatinine levels, a phenomenon also observed in the EE treatment group. Although a good correlation (R2 = 0.85) was observed between urine creatinine levels and specific gravities, the latter presented statistically significant difference from control group values only for the EE treatment group. Hence, specific gravity was used to normalize the urinary bisphenol concentrations presented in Table 1.
Higher urine specific gravities (indicative of higher metabolite concentrations) may explain why matrix effects (not observed during method development with more diluted urine samples collected from metabolic cages without a solid platform) prevented the quantification of BPF in the control, EE, and lowest bisphenol dose treatment groups. Significant BPA (12 ± 9 ppb) and BPS (29 ± 16 ppb) contaminations were observed in all vehicle control urine samples (Table 1). These concentrations were about an order of magnitude below the urinary levels measured at the lowest BPA and BPS doses administered. BPA and BPS contaminations were also detected in the urine of 3/9 to 8/9 rats from the EE and lowest bisphenol dose treatment groups (Table 1). Cross-contamination could not be assessed at higher bisphenol doses, as quantification of the administered bisphenols required substantial dilution of urine samples.
Rat housing and feed (and plasticware) were ruled out as the main sources of bisphenol contamination, as three out of four control rat urine samples from a previous study performed in the same facility 48 presented BPA, BPF, and BPS levels below quantification thresholds (1 ppb), while the fourth sample presented a BPA concentration of 2.1 ppb. Quantification of bisphenols in a 2.5 mL volume of analytical grade water that passed through the collection funnel of a metabolic cage washed after the last urine sample collection revealed BPA and BPF concentrations of 3.4 ppb and 12 ppb, respectively. These results suggest that bisphenol traces remaining in metabolic cages washed between sample collection may have leached into subsequent urine samples. However, this explanation alone doesn’t account for the BPA and BPS contamination levels measured in rat urine samples. While gloves were changed between BPA, BPF, and BPS gavages, they were not changed when rats were placed in metabolic cages. It is therefore possible that some contamination arose from fur-to-fur transfer and subsequent reabsorption through grooming. Although little information is available on the potential contribution of this contamination route, rats were handled in a similar way for regular cage changes, and hence such fur-to-fur bisphenol transfer may have occurred on a weekly basis. Unfortunately, the sources of bisphenol cross-contamination observed in the current study could not be conclusively determined.
Weekly Food and Water Consumption of Male Rats Exposed to BPA, BPF, BPS, or EE.
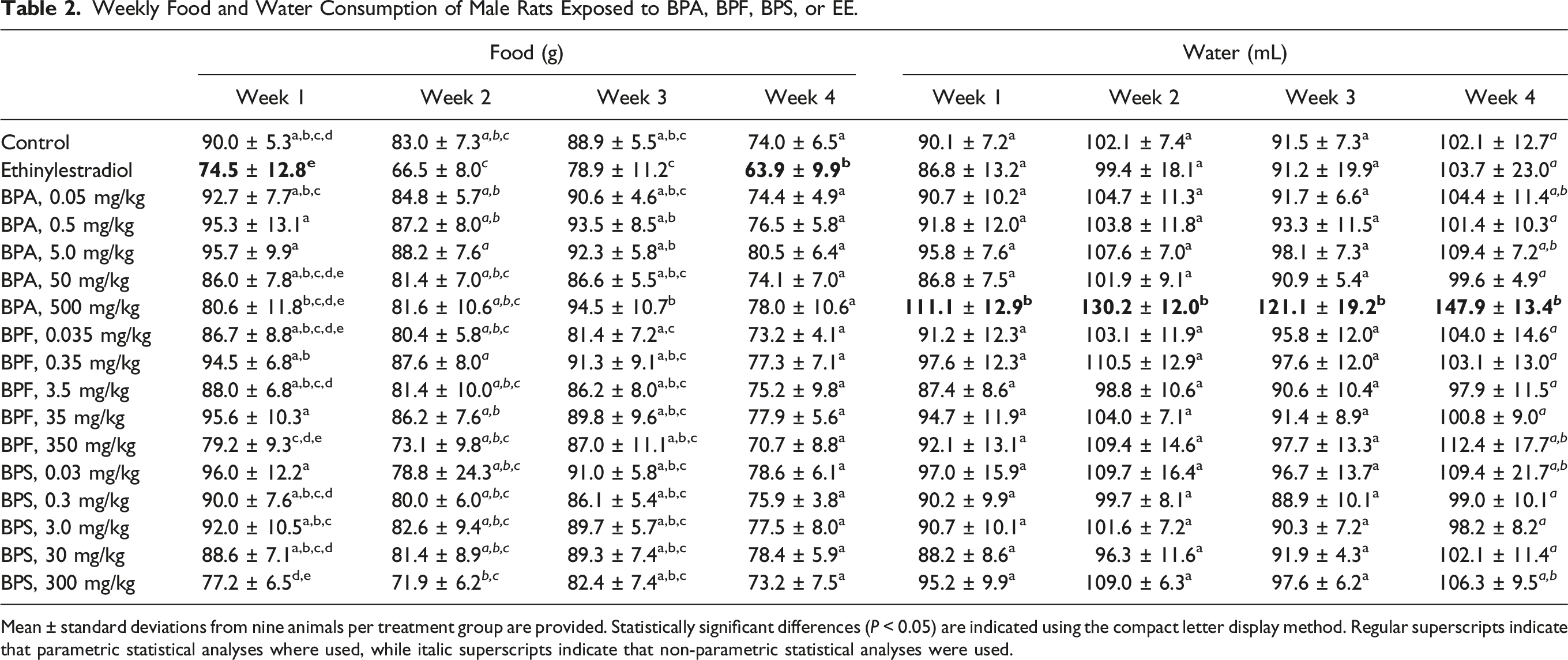
Mean ± standard deviations from nine animals per treatment group are provided. Statistically significant differences (
Rat Health, Bodyweight Gains, and Organ Weights
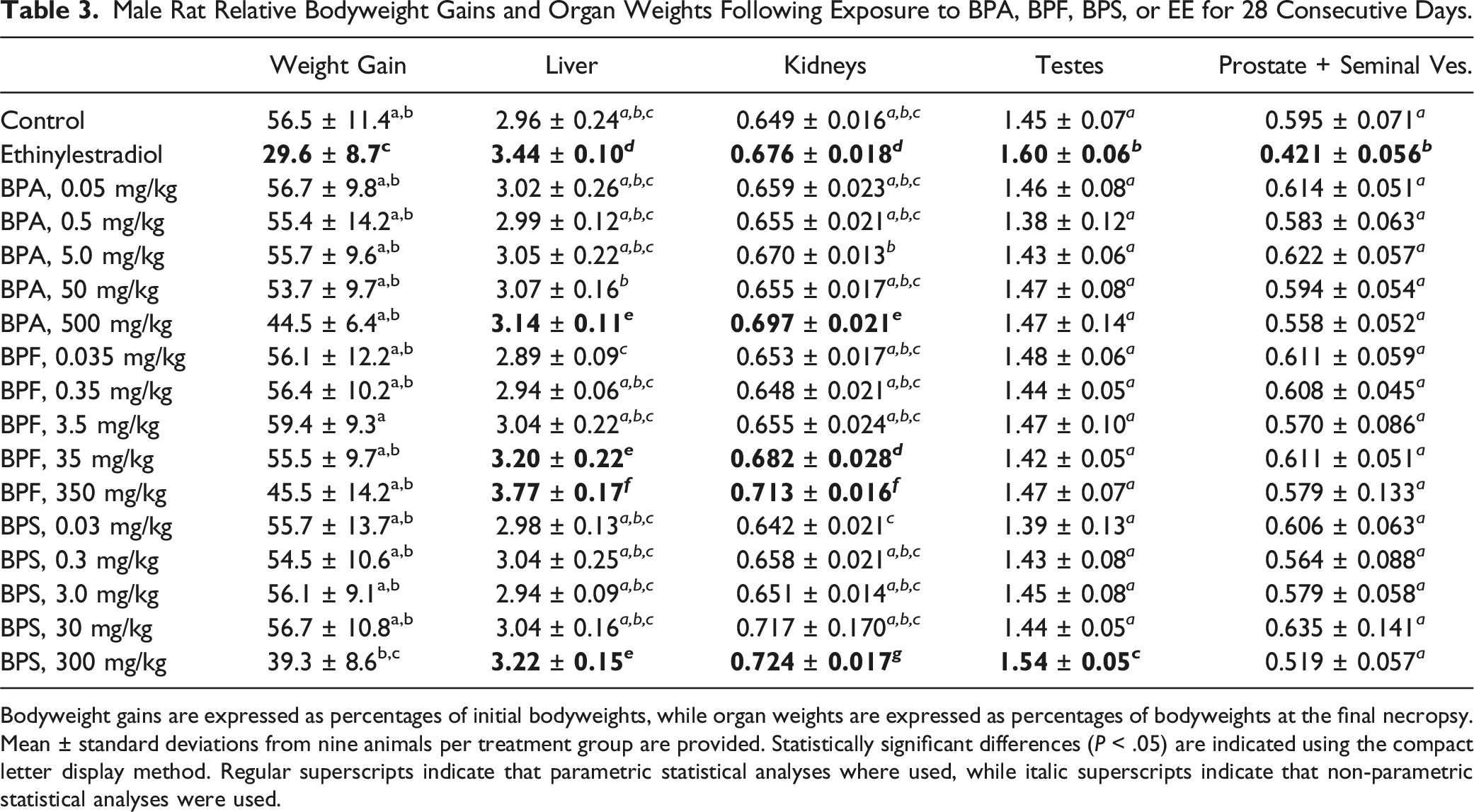
Beside the sporadic observation of porphyrin around the eyes and nose of a small number of rats exposed to the highest BPA, BPF, and BPS doses, bisphenol administration by gavage for 28 consecutive days did not result in overt behavioral signs of distress or toxicity. Analysis of growth curves (expressed as percent weight gains over initial bodyweights) by repeated measures two-way ANOVA revealed significant effects of treatment group and exposure day, and a significant interaction between treatment group and exposure day. Significant differences from the control rat growth curves were observed for the EE and 300 mg BPS/kg treatment groups (Figure 1), while significant decreases in food consumption were observed only for the EE-treated rats (Table 2). Significantly decreased total relative bodyweight gains over the whole exposure period were limited to the EE treatment group, although total relative weight gains in the 300 mg BPS/kg treatment group were not statistically different from those of the EE-treated rats (Table 3). Although rats exposed to the 500 mg BPA/kg dose consistently drank significantly more water than control rats throughout the exposure period (Table 2), this treatment group was also the only one where a few rats exhibited mild signs of dehydration (that resolved quickly upon providing HydroGel). Significantly increased liver and kidneys relative weights compared to the control group were observed at the highest BPA, BPF, and BPS doses (Table 3). The 300 mg BPS/kg treatment group also affected testes relative weight, while the only significant differences from control group values observed at lower bisphenol doses were the increased liver and kidneys relative weights noted in the 35 mg BPF/kg treatment group. EE-treated rats presented statistically different relative weights for liver, kidneys, testes, and prostate plus seminal vesicles (Table 3). Relative weights of brain, thymus, thyroid, heart, spleen, adrenals, and epididymides in bisphenol- and EE-treated rats were not significantly different from control group values (Table S1). Growth curves (expressed as percent weight gains over initial bodyweights) of male rats exposed to BPA, BPF, BPS, or EE. Errors bars represent standard deviations and are provided for the control group and the two treatment groups presenting statistically different growth curves, according to repeated measures two-way ANOVA ( Male Rat Relative Bodyweight Gains and Organ Weights Following Exposure to BPA, BPF, BPS, or EE for 28 Consecutive Days. Bodyweight gains are expressed as percentages of initial bodyweights, while organ weights are expressed as percentages of bodyweights at the final necropsy. Mean ± standard deviations from nine animals per treatment group are provided. Statistically significant differences (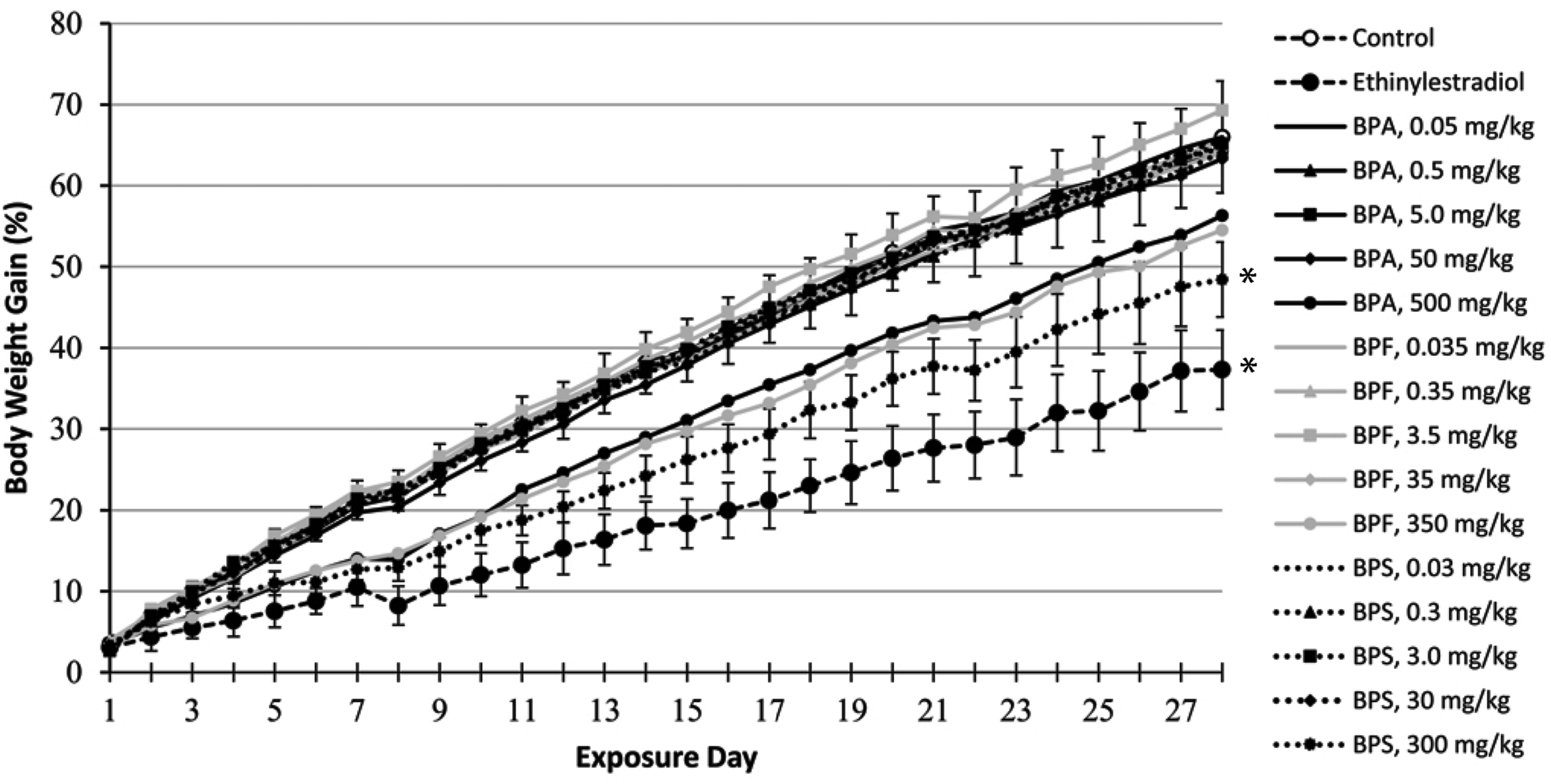
Hematology, Clinical Chemistry, and Liver Enzymatic Induction
Male Rat Hematology and Clinical Chemistry Parameters Following Exposure to BPA, BPF, BPS, or EE for 28 Consecutive Days.
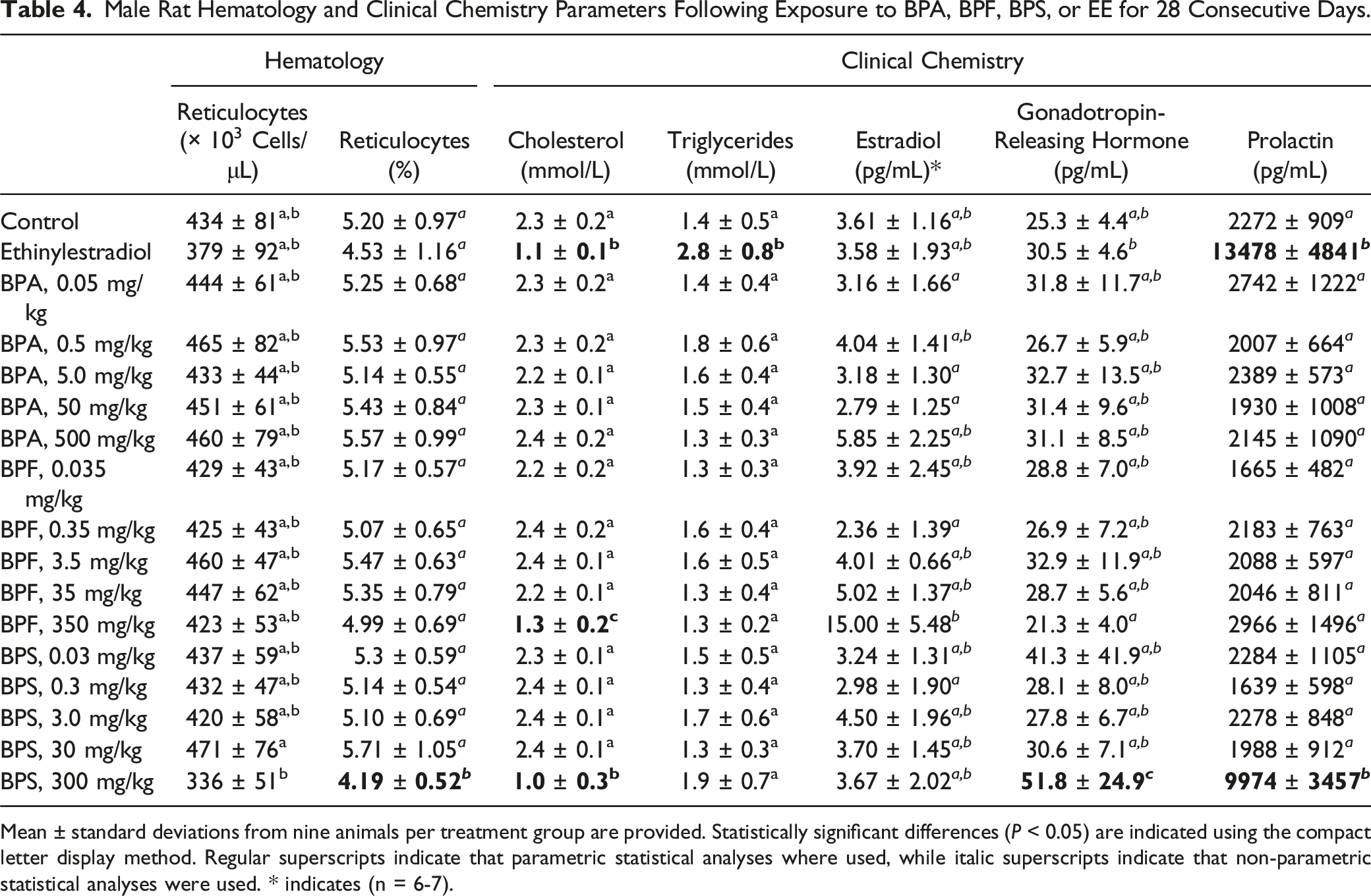
Mean ± standard deviations from nine animals per treatment group are provided. Statistically significant differences (
Hepatic Phase I Xenobiotic-Metabolizing Enzyme Activities in Male Rats Following Exposure to BPA, BPF, BPS, and EE for 28 Consecutive Days.
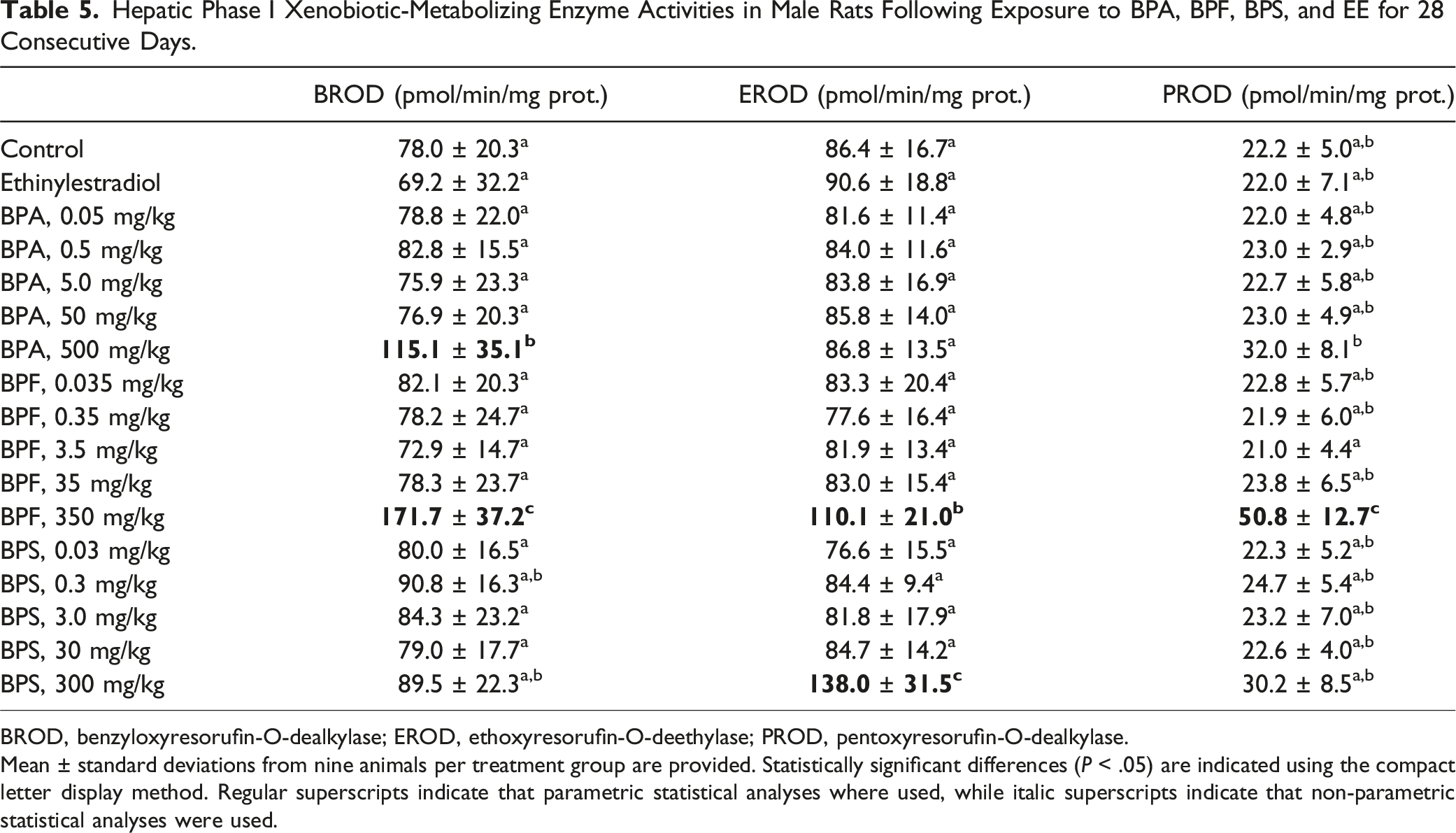
BROD, benzyloxyresorufin-O-dealkylase; EROD, ethoxyresorufin-O-deethylase; PROD, pentoxyresorufin-O-dealkylase.
Mean ± standard deviations from nine animals per treatment group are provided. Statistically significant differences (
Histopathology
A few gross tissue lesions were observed at the terminal necropsy. Two rats (from the 350 mg BPF/kg and 300 mg BPS/kg treatment groups) presented clearly enlarged cecum, while three rats (from the 0.5 mg BPA/kg, 500 mg BPA/kg, and 0.03 mg BPS/kg treatment groups) presented one noticeably smaller testis with a capsule rich in blood vessels, while the other testis was of normal size and appearance. Histopathological evaluation of the heart, lungs, trachea, stomach, intestines, bladder, spleen, skeletal muscle, adrenal, thyroid, pituitary and thymus glands, and axillary and mesenteric lymph nodes did not reveal any noticeable treatment-related effect. Minimal hepatic and pancreatic background lesions were observed at comparable frequencies in the vehicle control and bisphenol treatment groups (Table S5).
Most Salient Histological Observations in Male Rat Tissues Following Exposure to BPA, BPF, BPS, or EE for 28 Consecutive Days.
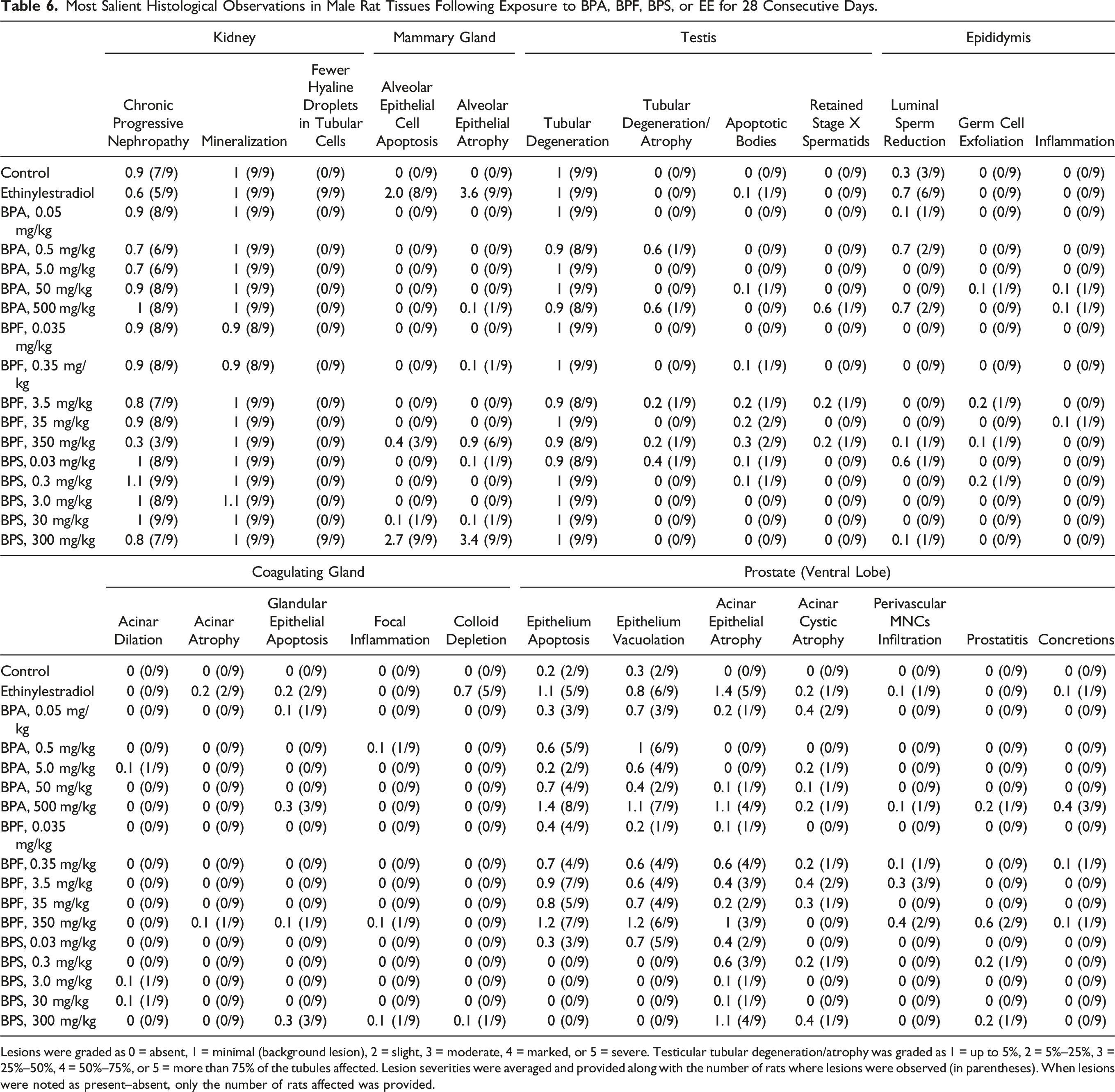
Lesions were graded as 0 = absent, 1 = minimal (background lesion), 2 = slight, 3 = moderate, 4 = marked, or 5 = severe. Testicular tubular degeneration/atrophy was graded as 1 = up to 5%, 2 = 5%–25%, 3 = 25%–50%, 4 = 50%–75%, or 5 = more than 75% of the tubules affected. Lesion severities were averaged and provided along with the number of rats where lesions were observed (in parentheses). When lesions were noted as present–absent, only the number of rats affected was provided.
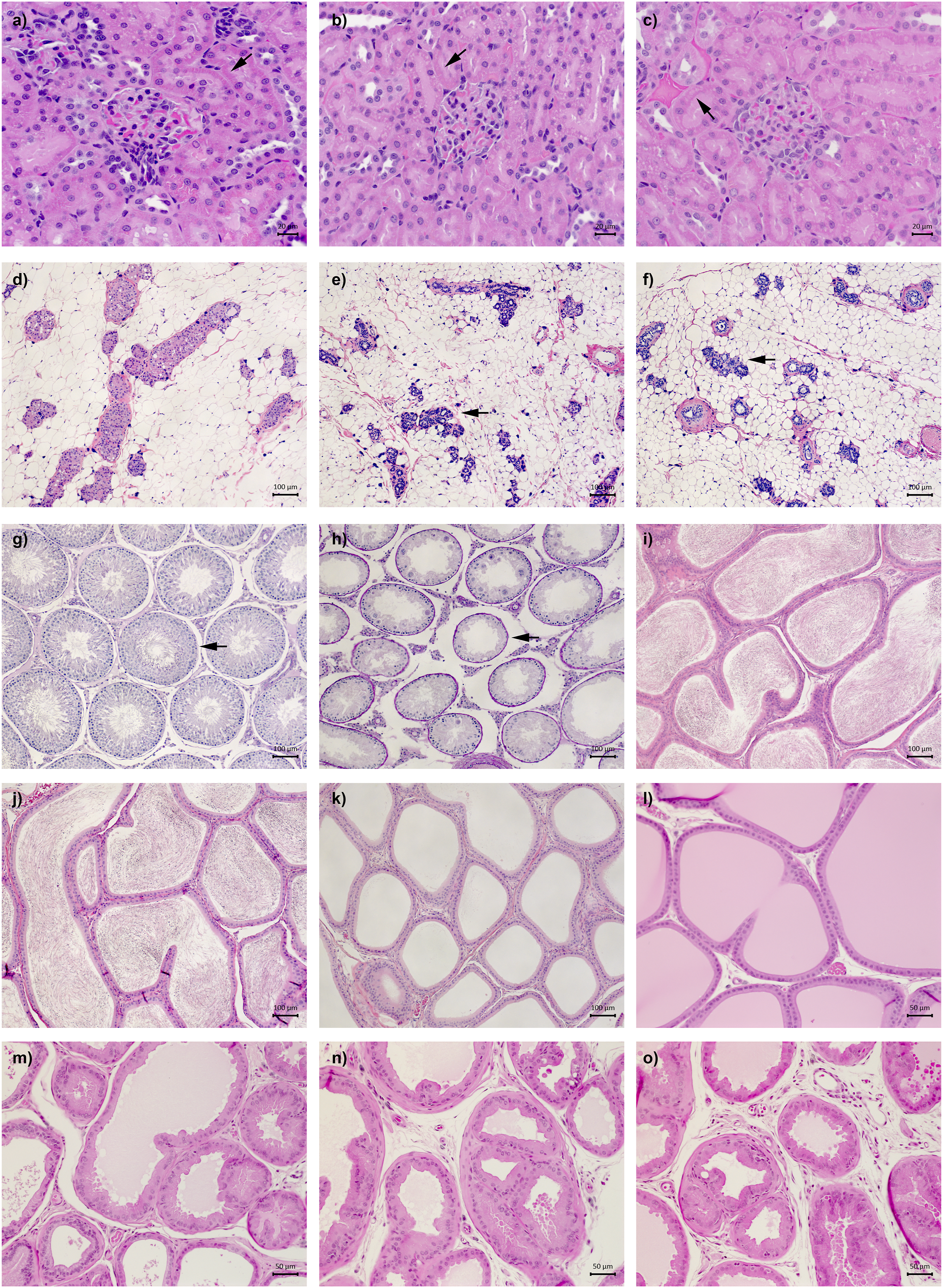
Illustrations of the most salient histological features observed in male rats exposed to BPA, BPF, BPS, or EE for 28 consecutive days. Kidney sections from the (a) control, (b) EE, and (c) 300 mg BPS/kg treatment groups. The arrows point to eosin-stained protein granules in p1 and p2 tubular epithelial cell cytoplasm. Mammary gland sections from the (d) control, (e) EE, and (f) 300 mg BPS/kg treatment groups. The arrows point to examples of moderate to marked acinar cell apoptosis and lobule atrophy. Testis sections from the (g) control and (h) 500 mg BPA/kg treatment groups. The arrows point to a representative site of minimal tubular degeneration in control testis, and to severe tubular atrophy and stage X spermatid retention observed exclusively in the smaller testis of the only rat from the 500 mg BPA/kg treatment group presenting testes of markedly different sizes. Epididymis sections from the (i) control, (j) EE, and (k) 500 mg BPA/kg treatment groups illustrating the minimal luminal sperm reduction observed in over half of EE-treated rat and the severe luminal sperm reduction observed in the only rat from the 500 mg BPA/kg treatment group presenting testes of markedly different sizes. Prostate sections from the (l) control, (m) EE, (n) 500 mg BPA/kg, and (o) 350 mg BPF/kg treatment groups. Obvious differences from control prostate histology were noted in the EE, 500 mg BPA/kg, and 350 mg BPF/kg treatment groups, as slight to severe epithelial cell atrophy, vacuolation, and apoptosis were frequently observed.
Moderate to marked mammary gland lobule atrophy and mild to moderate levels of alveolar epithelial cell apoptotic bodies were observed in at least 8/9 EE-treated rats (Table 6, Figure 2(d)–(f)). Similarly, all rats from the 300 mg BPS/kg treatment group presented moderate to severe lobule atrophy accompanied by slight to marked apoptosis. Minimal to slight lobule atrophy was noted in 6/9 rats and minimal to slight apoptosis in 3/9 rats from the 350 mg BPF/kg treatment group.
In the testes, minimal background lesions such as tubular vacuolation and apoptosis of germ cells (captured under the term tubular degeneration in Table 6) were observed in at least 8/9 rats in all treatment groups. Cases of marked to severe tubular degeneration/atrophy (defined by the presence of germ cell exfoliation and depletion, increased number of apoptotic, degenerating or multinucleated germ cells, and increased number of Sertoli-only tubules) were limited to the smaller testis from the three rats presenting testes of markedly different sizes (Table 6, Figure 2(g) and (h)). In the epididymis, minimal luminal sperm reduction was observed in 6/9 EE-treated rats, compared to 3/9 vehicle control rats (Table 6, Figure 2(i) and (j)). Severe epididymis luminal sperm reduction was limited to the three rats presenting one noticeably smaller testis (Table 6, Figure 2(k)). Additional minimal to slight perturbations were noted in at most 1/9 rats scattered across the BPA, BPF, and PBS treatment groups. In the seminal vesicles, the six minimal cases of histological perturbations observed in bisphenol-treated rats were scattered across five treatment groups (Table S5). In the coagulating gland, exposure to EE notably resulted in minimal to slight colloid depletion in 5/9 rats (Table 6). Interestingly, beside the two cases of minimal epithelial cell apoptosis observed in the coagulating gland of EE-treated rats, seven of the eight remaining cases were observed in the high-dose bisphenol treatment groups.
In the prostate ventral lobe, the majority EE-treated rats presented minimal to marked acinar epithelial atrophy (5/9), minimal to marked epithelium apoptosis (5/9), and minimal to slight epithelium vacuolation (6/9). The effects of bisphenols on prostate histology shared similarities with those of EE (Table 6, Figure 2(l)–(o)). Minimal to moderate acinar epithelial atrophy was frequently observed in BPA-, BPF-, and BPS-treated rats, affecting 3/9 to 4/9 rats at the highest doses. Intriguingly, while minimal to marked epithelial cell apoptosis and vacuolation were also observed in 6/9 to 8/9 rats exposed to the highest BPA and BPF doses (and in 2/9 control rats), they were not observed at the highest BPS dose. Although these bisphenol-induced histological perturbations were generally more frequent and pronounced at the highest doses, they were also frequently observed at lower doses. Additionally, four of the five cases of mild to moderate prostatitis were observed in the high-dose BPA, BPF, and BPS treatment groups, while three of the six minimal to slight concretion cases (small hyaline masses of degenerated cells that develop into compact concretions) were observed at the highest BPA dose.
Discussion
Although few substances have been studied as extensively as BPA, the toxicity of this widely used chemical is still the subject of intense debates. Unfortunately, the ambitious CLARITY-BPA consortium set up between US Food and Drug Administration and academic scientists to settle this question is unlikely to reach a definitive consensus, as disagreements emerged over the rat strain used, study design, data analysis, and results interpretation.
43
Hence, the main goal of the current study was not to add to the already abundant (and contentious) literature on BPA toxicity but to directly compare biological responses to BPA, BPF, and BPS
To confirm male Fischer rat responsiveness to estrogens and provide a positive control group for the comparison of BPA, BPF, and BPS estrogenic activities, additional rats were exposed to EE. Food consumption and bodyweight gains were significantly decreased in male Fischer rats exposed to 50 μg EE/kg bw/day (Tables 2 and 3, Figure 1), an effect that was only reported at 200 μg EE/kg bw/day in male Sprague-Dawley rats exposed according to the same OECD Test Guidelines. 54 Although prostate and seminal vesicle relative weights were perturbed at 50 μg EE/kg in both Fischer (Table 3) and Sprague-Dawley rats, 54 atrophic and apoptotic histological changes observed in Fischer rats prostate and mammary glands at 50 μg EE/kg (Table 6, Figure 2) were observed only at 200 μg EE/kg in Sprague-Dawley rats. 54 Serum pituitary and steroid hormones in the current study presented greater variabilities than anticipated, 45 which may explain why among the expected effects of estrogen supplementation,45,55,56 only prolactin levels were significantly affected (Table 4). However, alterations of mammary glands and accessory sex organs histology, which are considered the best integrated measures of hormonal perturbation,45,57 were also observed (Table 6, Figure 2). Perturbations of serum cholesterol and triglyceride levels reported following male rat estrogen supplementation 58 were observed in the current study (Table 4). Overall, these observations suggest that Fischer rats are more responsive to estrogens than the Sprague-Dawley rats used in the CLARITY-BPA studies11,44 but not as sensitive as other murine models. 9
The observation of bisphenol contamination in control rat urine samples (Table 1) may explain why increased bodyweight gains reported following rodent exposures to low bisphenol doses59,60 were not observed in the current study (Figure 1 and Table 3). At the terminal necropsy, two rats exposed to the highest BPF and BPS doses presented a noticeably enlarged cecum. Interestingly, bisphenols have been shown to affect gut contractility 61 and cecum distension. 26 In rodents, large cecum contents can be expulsed in response corticotropin-releasing hormone (CRH). 62 Although CRH was not measured in the current study, adrenocorticotropic hormone and corticosterone released in response to CRH 63 were not significantly affected (Table S4). Additional gross observations at the terminal necropsy were limited to three rats (from the 0.5 mg BPA/kg, 500 mg BPA/kg, and 0.03 mg BPS/kg treatment groups) presenting testes of noticeably dissimilar sizes. Although perturbations of testicular histology and spermatogenesis have been reported following bisphenol exposures, 64 observations of testis and epididymis lesions above the slight severity grade were limited to these three smaller testes in the current study (Table 6, Figure 2(g)–(k)).
Rats from the 500 mg BPA/kg treatment group drank considerably more water than control rats throughout the exposure period (Table 2). To our knowledge, this observation has not been reported in previous studies exposing rats to high BPA doses. Intriguingly, this increased water consumption was also accompanied by the observation of mild, sporadic dehydration affecting a few rats. Although rats from the 500 mg BPA/kg treatment group presented significantly increased relative kidney weights and decreased urinary creatinine levels (Tables 1 and 3), these changes are unlikely to explain these observations, as they were also observed at the highest BPF and BPS doses. Serum parameters which may have provided additional clues on kidney dysfunction (such as creatinine and BUN) were also unremarkable in the 500 mg BPA/kg treatment group (Table S3), while kidney histological features in this treatment group were indistinguishable from the vehicle control group (Table 6).
The only salient perturbations of kidney histology were noted in the EE and 300 mg BPS/kg treatment groups. All rats from these treatment groups presented a lower density of hyaline droplets in the proximal convoluted tubular cytoplasm (Table 6), a feature typically observed in female and castrated male rats. 65 The main component of these eosinophilic hyaline droplets is alpha-2u globulin, a protein synthesized under hormonal control in the liver.65,66 These observations further confirm the inhibitory effect of estrogens on alpha-2u globulin synthesis in the male liver 67 and identified an endocrine-related endpoint exclusively affected by BPS.
Significantly higher liver relative weights (Table 3) and phase I xenobiotic-metabolizing enzymatic activities (Table 5) were observed at the highest BPA, BPF, and BPS doses. Interestingly, while BPF induced BROD, EROD, and PROD activities, BPA only induced BROD while BPS only induced EROD, suggesting the activation and involvement of different nuclear receptors. 68 However, these BPA, BPF, and BPS exposures did not meaningfully affect liver histology (Table S5) or serum biomarkers of hepatic injury (Table S3). The only indication of altered liver function in the serum were the low cholesterol levels observed at the highest BPF and BPS doses (Table 4).
Although perturbations of serum hormone levels in adult male rats exposed to BPA were reported by multiple investigators, 64 no such effects were observed in the current study (Tables 4 and S4). However, significantly higher serum gonadotropin–releasing hormone and prolactin levels were observed at the highest BPS dose. Interestingly, BPS- and EE-treated rats which presented similar hyperprolactinemia (Table 4) also exhibited roughly comparable mammary gland acinar cell apoptosis and lobule atrophy (Table 6, Figure 2(d)–(f)). Although the increased estradiol concentrations following exposure to the highest BPF dose presented a large standard deviation and did not reach the threshold for statistical significance (Table 4), this treatment group also resulted in milder perturbations of the same mammary gland histological endpoints (Table 6). Overall, these observations are compatible with an important role for prolactin in the modulation of the male mammary gland, 69 and with a feminization of the male rat mammary glands characterized by a shift from a lobuloalveolar to a tubuloalveolar organization 70 in BPF- and BPS-treated rats.
While exposure to BPA failed to significantly affect serum hormone levels and mammary gland histology, other tissues offered clues on its potential endocrine effects. In the coagulating gland, minimal glandular epithelial apoptosis was observed in a third of the rats exposed to the highest BPA and BPS doses, a rate roughly comparable to the one observed in EE-treated rats (Table 6). In the prostate, exposure to the highest BPA dose resulted in the observation of acinar atrophy and epithelium vacuolation and apoptosis at a frequency and severity roughly comparable to the EE treatment group (Table 6, Figure 2(l)–(o)). Interestingly, while comparable prostate acinar atrophy was also observed at the highest BPF and BPS doses, epithelium vacuolation and apoptosis noted in most rats exposed to the highest BPA and BPF doses were not observed at the highest BPS dose.
In summary, while bisphenol cross-contamination prevented any conclusion regarding the effects of BPA, BPF, and BPS at lower doses, this investigation still allowed the direct comparison of their impacts at higher doses. Although adult male rat exposure only covers a small fraction of the potential effects of bisphenols across life stages and sexes, this investigation nevertheless uncovered intriguing differences between the impacts of BPA, BPF, and BPS. While the endocrine effects of BPA, BPF, and BPS all shared similarities with those of ethinylestradiol, they also presented noticeable differences across serum hormones and endocrine-responsive tissues. Overall, these observations suggest that BPA, BPF, and BPS may interfere with endocrine functions through slightly different molecular mechanisms.
Supplemental Material
Supplemental Material - Direct Comparison of the Impacts of Bisphenol A, Bisphenol F, and Bisphenol S in a Male Rat 28-Day Oral Exposure Study
Supplemental Material for Direct Comparison of the Impacts of Bisphenol A, Bisphenol F, and Bisphenol S in a Male Rat 28-Day Oral Exposure Study by Guillaume Pelletier, Gen Sheng Wang, Adam Wawrzynczak, Marc Rigden, Rocio Aranda-Rodriguez, and Don Caldwell in International Journal of Toxicology
Footnotes
Acknowledgements
The authors want to thank Mike Wade and Shamika Wickramasuriya for their helpful comments on this manuscript.
Author Contributions
G.P. conceived and secured funding for the experiments, coordinated experimental work and contributed to data acquisition, performed data analysis and interpretation, and drafted the manuscript; M.R. contributed to experimental work and data acquisition; A.W. and R.A.-R. contributed to analytical chemistry data acquisition; G.S.W. and D.C. contributed to histopathological assessment. All authors contributed to data interpretation, critically reviewed the manuscript, and approved the final version.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by the Chemicals Management Plan from the Government of Canada.
Ethical Statement
The animal exposure protocol was approved by Health Canada’s Institutional Animal Care Committee (HCO-ACC protocol# 2018-001) prior to the initiation of the study.
Data Availability Statement
Raw data are available on request.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
