Abstract
Heart failure (HF) is a leading cause of hospitalization across the world and is known to cause ill-health and heavy economic losses. In the present study, a rat model of isoproterenol (ISO, 85 mg/kg subcutaneously for two subsequent days) induced HF was developed. ISO induces HF by its direct effect, that is, rise in left ventricular end-diastolic pressure (mechanical) and indirectly by altering the baroreflex (neural), electrocardiography (electrical), and development of oxidative stress and hyperlipidemia (chemical). Fenofibrate, a hypolipidemic drug, which ameliorates myocardial energy metabolism was seen to improve the both ISO-induced oxidative stress and lipid profile and consequently improved Baroreflex Sensitivity (BRS), partial ventricular functions, and cardiac hypertrophy. Therefore, our result suggests that fenofibrate treatment protected the heart by alleviating the ISO-induced effects, that is, neural, mechanical, electrical, and chemical alterations.
Introduction
Cardiovascular diseases (CVDs) have an enormous impact on modern societies, as they cause high mortality and morbidity. According to World Health Organization (WHO), an estimated 17.7 million people have died from CVD in 2015, which represents 31% of all global deaths. 1 One of the major causes of CVD-related deaths is heart failure (HF), which is a clinical syndrome, characterized by the heart’s inability to pump blood efficiently. It acts as an end point medical state for most of the cardiovascular pathologies causing cardiac damage that subsequently leads to cardiac remodeling, which is the key pathophysiological process that ends in the development of pumping failure of the heart. 2
Animal models of HF are useful in understanding the basic mechanisms involved in the progression of HF on the basis of changes in myocardial structure and function. As the disease progresses, these models also give clue to the relevant pharmacological interventions required in HF. Among the variety of experimental models of HF, isoproterenol (ISO)-induced HF model is widely used as it is simple, inexpensive, and clinically representative of the pathophysiological changes seen in HF. Several clinical and experimental studies have revealed that arterial baroreflex is blunted in HF. Consequently, baroreflex is believed to play an important role in HF pathophysiology 3 –6 and, therefore, impairment of its function is considered as an important risk factor for cardiovascular mortality. 7 Baroreflex modulates changes in the neurohumoral drive of the heart and blood circulation, 8 –10 regulates arterial blood pressure (ABP) which is important for maintenance of adequate cardiac tissue perfusion, and balances the oxygen and nutrient demands necessary for proper cardiac metabolism. 11 –13 To maintain the continuous excitation–contraction coupling, the heart consumes more energy than any other organ. Normally this energy is generated in myocardium through mitochondrial oxidation of fatty acids and accounts for 60–90% of the total energy production. 14 Numerous experimental and clinical studies indicate that the HF is also characterized by a shift in the utilization of energy source, 15,16 that is, instead of utilizing fatty acid myocardium starts using glucose as an energy source. This results in impaired mitochondrial function and reduction in the production of energy, leading to worsening the heart function, promoting ventricular hypertrophy, and subsequently aggravating the HF. 17,18 The shift in the utilization of energy during cardiac metabolism occurs mainly due to decreased expression of the peroxisome proliferator-activated receptor alpha (PPAR-α) in the mitochondria of myocardial cells. 14 Therefore, therapies like fenofibrate, which helps in improving the energy utilization during cardiac metabolism, constitute an interesting therapeutic intervention. 19 Fenofibrate is a highly selective and efficient PPAR-α ligand, which is known to reduce the levels of low-density lipoprotein (LDL), cholesterol, and triglycerides (TG) and to elevate the levels of high-density lipoprotein (HDL). 20 Earlier studies have indicated a cardioprotective role of fenofibrate as it is seen to promote fatty acid oxidation in the mitochondria and improve myocardial energy metabolism. 21 Fenofibrate also possesses the capacity for numerous pleiotropic activities as its action on the heart is seen to be antioxidant, anti-inflammatory, and antifibrotic actions, which ameliorate cardiac functions. 22,23
Despite increasing evidences, validating the beneficial effects of fenofibrate on the improvement of cardiac energy metabolism on animal models of CVD and CVD patients, little is known about its effect on the baroreflex sensitivity. Interestingly, so far, the previous studies have not correlated the improvement of cardiac energy metabolism, baroreflex sensitivity, ventricular hypertrophy, and ventricular dysfunction in ISO-induced HF. Therefore, in the present study, to assess the role of myocardial energy metabolism and to evaluate its effect on (a) baroreflex sensitivity, (b) ventricular hypertrophy, and (c) ventricular dysfunction in ISO model of HF in Wistar rats, fenofibrate was used.
Materials and methods
Wistar rats (300–350 g) were obtained from the animal house of Jamia Hamdard, Delhi. The care and use of laboratory animals were in accordance with the recommendations by the National Accreditation Board of Testing and Calibration Laboratories. All experimental protocols were approved by the Institutional Animal Ethical Committee, Jamia Hamdard, New Delhi, India, and experiments were performed according to the guidelines of the Committee for the Purpose of Control and Supervision of Experiments on Animals, Government of India. During the experimental study period, the rats were housed at constant room temperature, humidity, and light cycle (12-h light:12-h dark) with free access to tap water and were fed with commercial standard chow ad libitum.
Grouping of animals
Fifty rats were divided into five groups, each group having 10 rats. In group I (control group), rats were maintained on standard chow and water, and no treatment was given. In group II (vehicle group), rats were given vehicle, namely carboxymethyl cellulose solution. In group III (fenofibrate per se group), normal rats were administered fenofibrate (100 mg/kg/day,) orally by gavage for 14 days along with normal diet and water. In group IV (ISO group), rats were subcutaneously administered with ISO hydrochloride (85 mg/kg/day) for 2 consecutive days and were fed with normal water and food for 14 days. In group V (fenofibrate treatment group), rats were subcutaneously administered with ISO (85 mg/kg/day) for 2 consecutive days. After the ISO administration, fenofibrate (100 mg/kg) was given orally by gavage once a day for 14 days along with normal diet and water.
Induction of heart failure
ISO was dissolved in freshly prepared normal saline (sodium chloride 0.9% w/v) and injected subcutaneously in rats (85 mg/kg) daily for 2 consecutive days (day 0 and day 1) and rats were monitored for change in weight, food, and water intake for 14 days 24,25 after which the experiments were conducted on 15th day.
Measurement of electrocardiography
At 15th day of the experimental period, the rats of all the groups were anesthetized using urethane (1 g/kg, intraperitoneally (i.p.)) and the dermal layer of their both front paws and hind legs was connected to the electrocardiography (ECG) leads, which were then connected to the PowerLab Data Acquisition System (Chart 5.4.2, AD Instruments, Australia), and then, ECG was recorded. 26
Measurement of hemodynamic parameters and ventricular functions
To measure the ABP and heart rate (HR), femoral artery of the anesthetized animal was cannulated by a polyethylene catheter attached to a pressure transducer (MLT0699-DC-06A) in conjunction with a PowerLab Data Acquisition System (Chart v.8.1.8, AD Instruments).
To evaluate the left ventricular cardiac function, the right carotid artery was cannulated and the catheter was pushed inside the heart until it was placed in the left ventricle so that the left ventricular pressure (LVP) could be recorded. The maximum and minimum rates of LVP (LV (dP/dt)max and LV (dP/dt)min) were recorded online by differentiating LVP. Left ventricular end-diastolic pressure (LVEDP) and maximal rate of rise of LVP divided by LVP (LV(dP/dt)max/LVP) were determined offline from the LVP data.
Measurement of baroreflex sensitivity
Baroreflex sensitivity was assessed as previously described 27 by administering increasing doses of vasoconstrictor phenylephrine (20–40 mg/mL/kg) and vasodilator sodium nitroprusside (20–40 mg/mL/kg) as bolus injections through a venous catheter. The resultant changes in HR at corresponding rise or fall in systolic blood pressure (SBP) were measured at different time intervals. The relationship between an increase in SBP or tachycardia evoked by phenylephrine and associated bradycardia or decrease in SBP evoked by sodium nitroprusside was assessed by regression analysis for individual animals. The regression coefficient (slope of the regression line), expressed as beats per minute per millimeter of mercury (beats/min/mmHg), was taken as an index of baroreflex sensitivity measurement.
Cardiac hypertrophy
At the end of each experiment, the whole heart was excised from rats and washed in normal saline several times to ensure there is no blood inside it. The heart was dried, and the weight of whole heart was measured for calculating the ratio of heart weight to body weight to evaluate cardiac hypertrophy.
Biochemical estimations
To determine the effect of HF and also the effect of fenofibrate on lipid profile, the total cholesterol (TC) level, TG, HDL cholesterol (HDL-C), LDL cholesterol (LDL-C), very low-density lipoprotein cholesterol (VLDL-C) and levels of myocardial injury marker creatine kinase-Muscle/Brain (CK-MB) were measured in blood serum by using appropriate commercially available kits.
Oxidative stress
Lipid peroxidation
Elevated levels of malondialdehyde (MDA) were used as an indicator of oxidative stress. MDA is a stable product of lipid peroxidation and was estimated in the present study by reaction with thiobarbituric acid. 28
NADPH oxidase expression
The processed heart tissues were preserved in the 10% paraformaldehyde overnight followed by dehydration in 30%, 20%, and 10% sucrose solution successively up to 3 days and were fixed after that in formaldehyde fixative until immunochemical staining. Then, 5- to 15-µm-thick sections of paraffin-embedded tissues were cut using grading-type Lieca microtome and boiled in 0.1 M citrate buffer (pH 6.0) for 5 min for antigen retrieval process and then incubated in 0.3% hydrogen peroxide in methanol, followed by incubation in blocking buffer containing 0.1 M phosphate-buffered saline, 0.04% Triton X-100 and 10% normal goat serum. Sections were incubated in antibodies and anti-NADPH oxidase (1:100, ABCAM, Cambridge, MA, USA) for overnight at 4°C. After rinsing in buffer, sections were processed using a three-layer peroxidase staining kit (Thermo Scientific System, Waltham, MA, USA). The peroxides complex was visualized with 3,3-diaminobenzidine (DAB Plus substrate, Thermo Fisher Scientific, USA). The slides were counterstained with hematoxylin for 5 s. Slides were cleaned in Xylene, gradually dehydrated with ethanol medium, and then photographed under Olympus microscope BX51 (Hamburg, Germany).
Histopathology
The heart was fixed in 10% formalin solution for histopathological analysis. Formalin-fixed tissues (left ventricle) were embedded in paraffin, sectioned, and stained with hematoxylin and eosin (H&E). The sections were examined under light microscope, and photomicrographs were taken.
Statistical analysis
The results were presented as mean ± standard error of the mean (SEM). All data were analyzed by analysis of variance, followed by Tukey’s multiple comparison tests for analysis between the groups. The value of p < 0.05 was considered as statistically significant.
Results
Body weight
Animals from all the groups exhibited no change in body weight.
Cardiac hypertrophy
The heart weight to body weight ratio (HW/BW) in control and ISO rats were 2.98 ± 0.18 and 4.05 ± 0.21, respectively. The increase in HW/BW ratio in ISO-treated rats is indicative of myocardial hypertrophy developed on ISO administration. The treatment with fenofibrate was effective in improving the hypertrophy induced by ISO (Figure 1). Fenofibrate per se and vehicle group did not show any effect on the HW/BW ratio when compared with control rats.
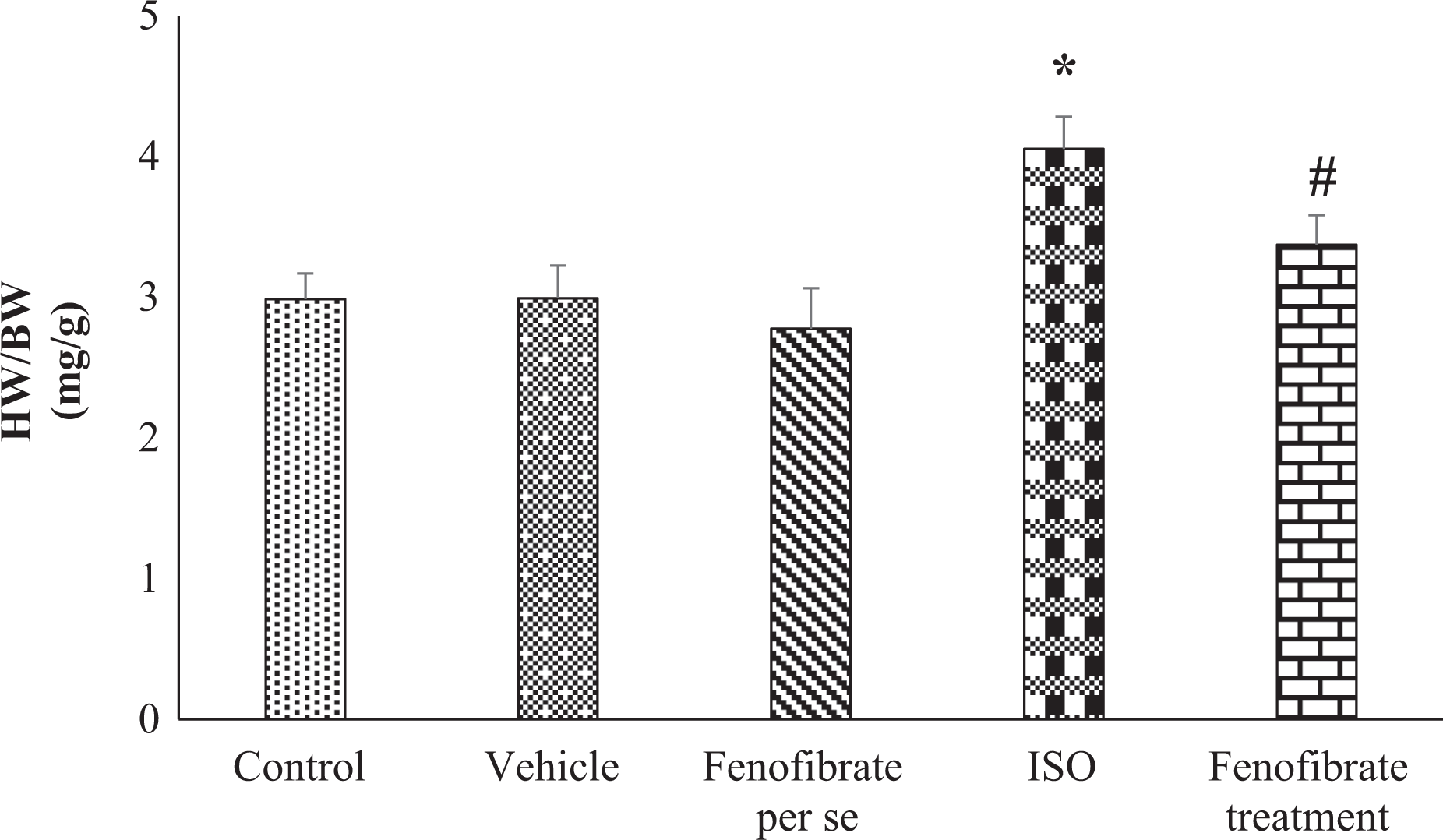
Heart weight to body weight (HW/BW) (mg/g) ratio. Results represent mean ± SEM of seven animals per group. Results obtained are significantly different from the control group (*p < 0.05) and ISO (#p < 0.05), respectively. SEM: standard error of the mean; ISO: isoproterenol.
Arterial blood pressure and heart rate
ISO-administered rats displayed a significant drop in SBP, diastolic blood pressure (DBP), mean arterial pressure (MAP), and HR in comparison to control rats (Table 1). The treatment with fenofibrate significantly improved the arterial pressure parameters, namely SBP, DBP, and MAP in comparison to control rats and HR in comparison with ISO-treated rats. However, the SBP, DBP, and HR in fenofibrate per se and vehicle group were similar to the control group.
SBP, DBP, HR, MAP measurements.a

SBP: systolic blood pressure; DBP: diastolic blood pressure; HR: heart rate; MAP: mean arterial pressure; ISO: isoproterenol; SEM: standard error of the mean.
aResults represent the mean ± SEM of seven animals per group. Results obtained are significantly different from control group (*p < 0.05) and ISO (#p < 0.05) respectively.
Left ventricular functions
Left ventricular functions were significantly altered in the ISO group. LVEDP was markedly increased (Figure 2(a)), whereas maximum and minimum rates of developed LVP (LV (dP/dT)max and LV(dP/dt)min) (Figure 2(b) and (c)) and myocardial contractility index (LV(dP/dTmax/LVP)) (Figure 2(d)) were decreased significantly in ISO rats in comparison with the control rats. In rats with ISO-induced HF, treatment with fenofibrate significantly improved the ISO-induced depression in LV(dP/dT)max, LV(dP/dt)min, and LV(dP/dTmax/LVP). The ISO-induced increase in LVEDP was significantly improved in the fenofibrate-treated group in comparison to ISO group. However, complete restoration was not observed. No statistically significant difference was noted in group II (vehicle group) and group III (fenofibrate per se) left ventricular functions when compared with the control group.

Left ventricular functions: (a) LVEDP, (b) LV (dP/dt)max maximal rate of rise of LVP, (c) LV (dP/dt)min minimal rate of rise of LVP, and (d) LV (dP/dt)max/LVP maximal rate of rise of LVP divided by LVP. Results represent the mean ± SEM of seven animals per group. Results obtained are significantly different from the control group (*p < 0.05) and ISO (#p < 0.05) respectively. LVEDP: left ventricular end diastolic pressure; LVP: left ventricular pressure; SEM: standard error of the mean; ISO: isoproterenol.
Baroreflex sensitivity
ISO-treated rats exhibited a significant reduction in baroreflex sensitivity to phenylephrine and sodium nitroprusside in comparison to the control group. Treatment of ISO rats with fenofibrate significantly enhanced baroreflex sensitivity (Figure 3(a) and (b)). Fenofibrate per se and vehicle group showed no significant change in comparison to the control group.

Baroreflex sensitivity to (a) phenylephrine and (b) sodium nitroprusside. Results represent the mean ± SEM of seven animals per group. Results obtained are significantly different from the control group (*p < 0.05) and ISO (#p < 0.05), respectively. SEM: standard error of the mean; ISO: isoproterenol.
ECG parameters
In the present study, ISO-treated rats had significantly extended QT and PR interval along with depressed QRS segment and elevated ST segment in comparison to the control rats. These changes are indicative of ISO-induced electrical alterations in HF rats. Fenofibrate treatment significantly improved the ECG abnormalities suggesting its cardioprotective efficacy (Table 2). No significant change was observed in fenofibrate per se (group III) and vehicle group (group II) in comparison to the control group.
ECG parameters: ST height, QRS interval, PR interval, and QT interval.a

ISO: isoproterenol; SEM: standard error of the mean.
aResults represent the mean ± SEM of seven animals per group. Results obtained are significantly different from control group (*p < 0.05) and ISO (#p < 0.05) respectively.
Serum marker of cardiac injury
In comparison to the control rats, ISO-administered rats exhibited a significant rise in the activity of serum myocardial injury marker enzyme CK-MB. Treatment with fenofibrate in the ISO group was seen to significantly improve the alterations of serum CK-MB. However, fenofibrate treatment did not restore the CK-MB to baseline levels (Figure 4(a)). Fenofibrate per se and vehicle group did not show any significant changes in CK-MB serum levels in comparison with the control groups.

(a) CK-MB serum levels and (b) serum MDA levels. Results represent the mean ± SEM of seven animals per group. Results obtained are significantly different from the control group (*p < 0.05) and ISO (#p < 0.05), respectively. MDA: malondialdehyde; SEM: standard error of the mean; ISO: isoproterenol; CK-MB: creatine kinase-MB.
Lipid peroxidation
A marked elevation in serum MDA levels was observed in rats treated with ISO when compared to the control rats. Treatment with fenofibrate showed a significant decrease in MDA levels in comparison to ISO-treated rats (Figure 4(b)). However, fenofibrate did not completely restore the MDA levels as seen in the control rats. No significant change was observed in fenofibrate per se and vehicle group in comparison to the control group.
NADPH oxidase expression by immunohistochemistry
The immunohistochemistry study of the heart tissue in ISO-treated group showed enhanced brown color indicating heightened expression of NADPH oxidase (Figure 5(d)) as compared to the control group while fenofibrate treatment attenuated NADPH oxidase expression (Figure 5(e)). Similar to the control group, fenofibrate per se and vehicle group showed immunonegative staining.

Photomicrographs of NADPH oxidase immunostained heart sections of (a) control group, (b) vehicle group, (c) fenofibrate per se, (d) ISO group showing wide patches of strong immune-positive reactions in the inflammatory cells and surrounding myocardium (arrow), and (e) fenofibrate treatment group showing normal weak immune reaction in myocardium. ISO: isoproterenol.
Serum lipid profile
An increase in serum concentration of TC, TG, LDL-C, and VLDL-C along with the decrease in HDL-C was observed in ISO-treated group in comparison to the control group, indicating that ISO adversely affects the serum lipid profile. Treatment with fenofibrate was seen to significantly improve the ISO-induced increase in TC, TG, LDL-C, VLDL-C and decrease in HDL. However, recovery was not complete. No significant change was observed in fenofibrate per se group and vehicle group in comparison to the control group (Table 3).
TC, TG, HDL-C, LDL-C, VLDL-C measurements.a
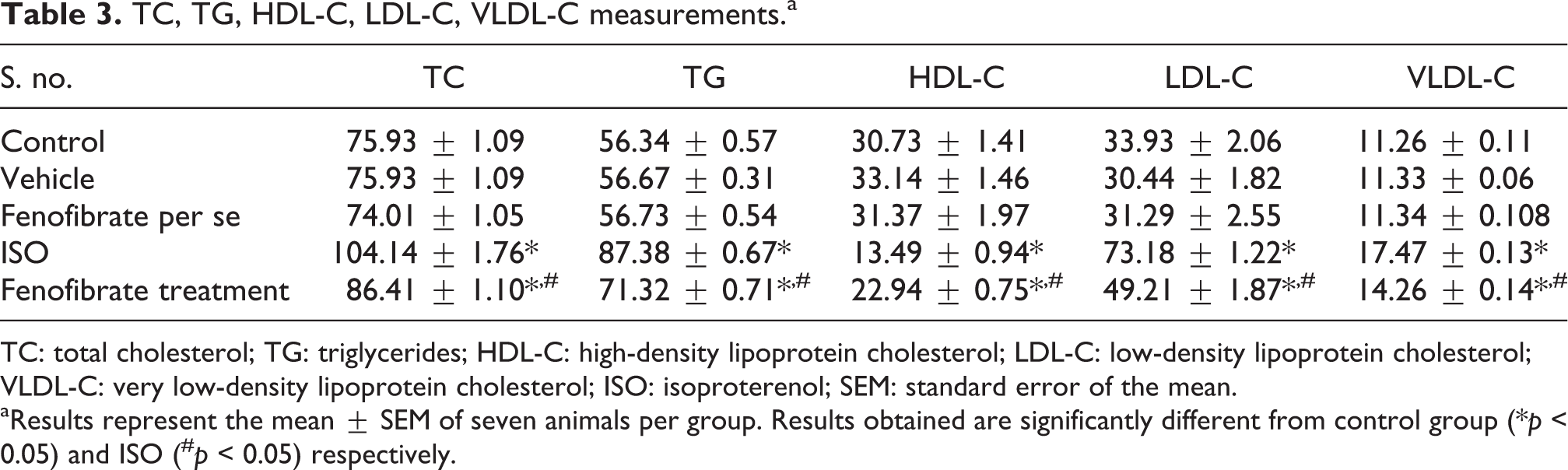
TC: total cholesterol; TG: triglycerides; HDL-C: high-density lipoprotein cholesterol; LDL-C: low-density lipoprotein cholesterol; VLDL-C: very low-density lipoprotein cholesterol; ISO: isoproterenol; SEM: standard error of the mean.
aResults represent the mean ± SEM of seven animals per group. Results obtained are significantly different from control group (*p < 0.05) and ISO (#p < 0.05) respectively.
Histological architecture in heart tissue
Microscopic examination of the normal control heart showed regularly arranged myocardial fibers with no inflammation or necrosis or hypertrophy. Rats from the HF group showed widespread myocardial structural disorder, such as fibrosis, leukocyte infiltration along with increased edematous intramuscular space (Figure 6(d)). Fenofibrate treatment showed a partial reversal of the progression of fibrosis in the heart (Figure 6(e)). The rat heart from fenofibrate per se and vehicle group showed normal histology as the myocardial integrity appeared to be intact, and there was no evidence of inflammation or infarction.

Photomicrographs showing histopathology of heart (H&E, ×20) (a) control group showing NMC, (b) vehicle group showing NMC, (c) fenofibrate per se group showing NMC, (d) ISO-administered group showing wide spread damage (LI, leucocyte infiltration; O, edema, V, vacuolization, HCM, hypertrophied cardiomyocytes), and (e) fenofibrate treatment group showing almost normal myocardium. NMC: normal cardiomyocytes. H&E: hematoxylin and eosin; ISO: isoproterenol.
Discussion
In the present study, the Wistar rat model of HF, developed by administration of ISO, was seen to cause neural (decreased BRS), mechanical (reduction in ventricular function), chemical (oxidative stress and increase in serum levels of TC, TGs, HDL-C, LDL-C), electrical (abnormality in ECG), and structural (cardiac hypertrophy) alterations. Fenofibrate treatment in rats with HF decreased hyperlipidemia and oxidative stress resulting in partial restoration of BRS (neural), leading to improvement in ventricular function (mechanical), ECG (electrical), and cardiac hypertrophy (structural).
In the ISO-induced HF model, subcutaneous administration of the β-adrenoreceptor agonist ISO causes direct and indirect effects on the heart. The ISO directly affects the heart by causing overstimulation of cardiac adrenergic receptors, which results in an increase in the cyclic adenosine monophosphate (cAMP) levels, a second messenger that causes extrusion of calcium (Ca2+) ions from the sarcoplasmic reticulum and also the phosphorylation of the voltage-dependent calcium channels in the cardiomyocytes. This results in intracellular calcium overload that leads to excessive myocardial contraction. 29 This excessive overpumping of the heart causes disruption in myocardial energy metabolism. 30 In the present study, ISO-induced excessive myocardial contraction results in impaired ventricular function, as evidenced by increased inotropic (LV (dP/dT)max: index of myocardial contraction), lusitropic (LV (dP/dT)min: index of myocardial relaxation), decreased myocardial contractility (LV (dP/dT)max/LVP: the maximal rate of rise of LVP divided by LV systolic pressure), and increased LVEDP (indicator of increased preload or incomplete emptying of the left ventricle), indicating the detrimental effects that are caused due to overstimulation of cardiac β-adrenoreceptors following ISO administration.
ISO also induced cardiac injury (increased CK-MB) and cardiac hypertrophy (increased HW/BW ratio) similar to earlier reports. 24,31 ISO pronounced abnormality in ECG is well characterized in our study, as evident by elevation of ST segment, prolongation of QT interval, and attenuation of QRS interval and PR interval.
Apart from the direct effects on the adrenergic system and disturbed cardiac metabolism, catecholamines also exert an indirect effect on the cardiovascular system by producing oxidized products that cause oxidative stress. 32 Oxidative stress is a hallmark of various CVDs that causes cellular dysfunction and death. A number of studies have found a strong association between the development of HF and increased production of reactive oxygen species (ROS). 24,33 Our study also showed the increased generation of ROS as indicated by elevated MDA levels due to increased cardiac NADPH oxidase expression in ISO-induced HF. Increased lipid peroxides due to ISO treatment may also affect the mitochondrial and cytoplasmic membranes causing more severe oxidative damage to the heart and consequently releasing more lipid peroxides into circulation. 34
Increased oxidative stress observed in ISO-treated rats modifies hemodynamic parameters and the autonomic control functions of the heart. In the present study, reduction in arterial pressure values (SBP, DBP, and MAP) along with decreased HR indicates deranged sympathetic and parasympathetic inputs to the heart. The linear regression analysis of reflex changes in SBP in ISO rats indicates depressed BRS suggesting impaired autonomic control functions. In normal heart stimulation of adrenergic receptor in the heart causes increase in SBP and reflex decrease in HR by activating baroreceptors. In our study, decrease in HR despite decreased SBP in the ISO-treated rats observed indicates reflex alterations by the baroreceptors. Previous animal model studies indicate that ROS is directly involved in the inhibition of firing of arterial baroreceptor. 35 Disruption of cardiac energy metabolism, which results in an increase in circulating lipids and oxidative stress, has also been considered as a triggering factor for reduced BRS. In our study, ISO administration induced a significant elevation in serum levels of TC, TGs, LDL-C, and VLDL-C in Wistar rats. Hyperlipidemia in CVDs occurs due to lipid accumulation in myocardium through the activation of cAMP-protein kinase cascade promoting phosphorylation of hormone-sensitive lipase. 36,37,38
The shift in the substrates for myocardial energy metabolism, that is, change from fatty acids to glucose results in insufficient energy production, serum lipid alteration, and marked an increase in intracellular lipid accumulation that leads to lipotoxic cardiomyopathy 39 and further aggravates HF. 40 The previous study by Li et al. established that fenofibrate increases the fatty acid β oxidation enzymes by activating PPAR-α, promoting fatty acid oxidation in the mitochondria, and regulating myocardial energy metabolism. 21 In the present study, fenofibrate significantly attenuated the ISO-induced hyperlipidemia in Wistar rats. Oxidative stress was also decreased in the fenofibrate-treated animal, as indicated by the decrease in NADPH oxidase expression and reduced concentration of lipid peroxidation products (MDA), suggesting the antioxidant property of fenofibrate which is in accordance with earlier studies. 41,42 This antioxidant potential of fenofibrate may be partly due to its effects on lipoprotein metabolism.
In a rat model of HF, treatment with fenofibrate also improved the baroreflex sensitivity, suggesting that oxidative stress and hyperlipidemia are the instigating factors for blunting of BRS. In a prior study, we have reported that aspirin restores normal baroreflex function in hypercholesterolemic rats by its antioxidative and hypolipidemic action. 4 Fenofibrate also significantly improved the ISO-induced alterations in hemodynamic parameters (SBP, DBP, MAP, and HR) almost near to that in control group rats. Fenofibrate also restored the myocardial contractility and left ventricular function, as evidenced by a significant restoration of LV(dP/dtmax), LV(dP/dtmin), and LV(dP/dtmax)/LVP and correction of elevated LVEDP subsequent to ISO challenge. In hypertrophied heart, impaired cardiac metabolism is often observed. 43 In the present study, fenofibrate improved the ISO pronounced cardiac hypertrophy in Wistar rats, which might be due to its ability to improve myocardial energy metabolism, which is also evident by improved lipid profile and decreased oxidative stress. Thus, fenofibrate significantly improved the ISO-induced altered ECG pattern, ventricular functions, BRS, and histopathological changes. 44
However, CK-MB levels were not restored back to normal levels in fenofibrate-treated rats, suggesting that fenofibrate is not able to protect the heart from the direct damage caused due to ISO administration.
Conclusion
In conclusion, our study for the first time demonstrates the ability of fenofibrate to ameliorate the baroreflex sensitivity along with ventricular functions, ECG alterations, and ventricular hypertrophy in ISO-induced HF in Wistar rats.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
