Abstract
This study aimed to assess the feasibility of using the plasma neutrophil gelatinase-associated lipocalin (NGAL) level at the time of presentation in the emergency department (ED) to predict acute kidney injury (AKI) and the long-term neurological outcomes of acute charcoal-burning carbon monoxide (CO) poisoning. This retrospective study included 260 patients who suffered acute charcoal-burning CO poisoning. The median plasma NGAL concentration at the time of presentation in the ED after acute charcoal-burning CO poisoning was 78 (54–115) ng/ml. The NGAL level was an independent predictor of AKI development and could be used to stratify the severity of AKI. However, the area under the receiver operating characteristic curve (AUC) of the predictive model for AKI that included both the plasma NGAL level and clinical parameters was comparable to that of the predictive model including only the clinical parameters. The plasma NGAL level at the time of presentation in the ED was an independent factor predicting long-term neurological outcomes in patients who did not develop AKI. In these patients, the plasma NGAL level significantly improved the predictive accuracy of the model when used in combination with clinical parameters. In contrast, the plasma NGAL level was not associated with long-term neurological outcomes in patients who developed AKI. Measurement of the plasma NGAL level at the time ED presentation might improve the prediction of long-term neurological outcomes in patients who do not develop AKI after acute charcoal-burning CO poisoning. However, it might not offer additional benefit for AKI prediction compared to previously used markers.
Keywords
Introduction
Acute kidney injury (AKI) is common among acutely ill patients worldwide and is associated with high morbidity and mortality and prolonged hospitalization. 1 Therefore, the timely diagnosis of AKI and implementation of preventative strategies have been emphasized as the most effective tools to improve outcomes. Neutrophil gelatinase-associated lipocalin (NGAL), a member of the lipocalin superfamily, was first described as a covalently bound protein present in neutrophils. NGAL is one of the most upregulated transcripts in the kidneys after ischemia, as well as septic and toxic AKI, in humans. 1 Indeed, plasma NGAL has been identified as one of most promising candidates for the diagnosis and monitoring of AKI. 2 However, the poor diagnostic performance of NGAL for AKI in patients with preeclampsia suggests that the diagnostic value of NGAL for AKI should be determined in individual clinical settings. 3 In acute carbon monoxide (CO) poisoning, the incidence rate of AKI ranges from 8.8% to 18%. 4 –6 AKI after acute CO poisoning is associated with neurological symptoms or signs remaining at discharge, the development of delayed neurologic sequelae (DNS), and in-hospital or 28-day mortality. 4,5 However, despite emerging results showing an association between AKI and outcomes of acute CO poisoning, 4,5 no study to date has investigated the diagnostic performance of NGAL for AKI after CO poisoning.
In addition, recently, the potential neuroprognostic value of NGAL was demonstrated in cardiac arrest studies. 7,8 Several studies have indicated that the plasma level of NGAL reflects inflammation and is not only restricted to the detection of AKI. 2,9 Both the potential of NGAL as a neuroprognostic predictor in cardiac arrest and the association between plasma NGAL and inflammation, which contributes to neuronal injury after CO poisoning, 10 highlight the need for research concerning the predictive performance of NGAL for long-term neurological outcomes of acute CO poisoning.
Therefore, we aimed to determine whether the plasma NGAL level at presentation enhanced the predictive accuracy for AKI during hospitalization and whether the plasma NGAL level can be used to stratify the severity of AKI after acute charcoal-burning CO poisoning. Furthermore, we investigated whether NGAL at presentation improved the predictive accuracy for long-term neurological outcomes of CO poisoning by charcoal burning.
Method
Study design and setting
This investigation was a retrospective observational study performed via chart review at a single academic tertiary care center. The study design was approved by the Institutional Review Board of Chonnam National University Hospital.
Participant selection
The inclusion criteria for this study were as follows: patients 18 years or older who presented to our emergency department (ED) with acute CO poisoning due to charcoal burning within 24 h between January 2014 and March 2017. Acute CO poisoning by charcoal burning was defined as a history of exposure to CO from charcoal burning and an initial COHb level exceeding 5% (10% in smokers) at our ED or the primary hospital.
Patients were excluded if (1) they were dependent on others for daily living before CO poisoning, (2) they had a history of kidney disease requiring renal replacement therapy (RRT) before CO poisoning, (3) they were transferred to another facility before the final outcome was determined, (4) they were treated with protocols that did not adhere to our hospital guidelines, (5) they experienced cardiac arrest related to CO poisoning before presentation to the ED, or (6) they were discharged against the doctor’s orders. In addition, patients poisoned with substances other than CO and pregnant patients as well as those who died due to reasons unrelated to complications of CO poisoning after discharge were excluded (Figure 1).

Algorithm for subject selection. CO: carbon monoxide; RRT: renal replacement therapy.
Our hospital treatment guidelines for acute CO poisoning recommend initiation of normobaric oxygen (NBO) or hyperbaric oxygen (HBO) therapy according to the presence of indications for HBO therapy. The indications for HBO for nonpregnant patients in our institution are the presence of any neurological deficit, including loss of consciousness and seizure, and a COHb level greater than 25%, regardless of symptoms. The details of the treatment guidelines were described at our previous studies. 11
Methods and measurements
Data including baseline characteristics, laboratory results including plasma NGAL level at presentation, and clinical course including complications during hospitalization were collected by reviewing the medical records of patients. The investigated complications during hospitalization included respiratory failure (defined as the need for MV support for any reason), hypotension (defined as the need for intravenous vasopressor infusion to maintain blood pressure), and AKI.
Outcomes
Development of AKI during hospitalization
AKI was defined and staged according to the kidney disease improving global outcomes classification. 12 The baseline plasma creatinine level was determined using one of three approaches 13 : (1) for patients with laboratory data in our hospital, the most recently recorded creatinine level was used; (2) for patients without laboratory data and for whom AKI was resolved during hospitalization, the lowest steady-state creatinine level after recovery from AKI was used; and (3) for patients without laboratory data and with sustained AKI during hospitalization, the first creatinine level obtained at the ED was used. Patients who were discharged or transferred to another hospital without AKI within 3 days after CO poisoning were classified as not having AKI because 95.8% of AKI cases after CO poisoning were detected at presentation. 4
Long-term neurological outcomes
The long-term neurological state was assessed by a structured phone interview with patients and family using the GOS at least 15 months after discharge. Long-term neurological outcomes were divided into good (GOS score of 4–5) outcomes, in which patients performed activities of daily life independently, and poor (GOS score of 1–3) outcomes, in which patients developed a dependency or died. Patients who were lost to follow-up were excluded from further statistical analysis. Because the predictive value of NGAL changed after adjustment for the presence of AKI 14 and because the majority of AKI cases after acute CO poisoning were diagnosed at presentation, 4 the neuroprognostic performance of NGAL was evaluated in subgroups with and without AKI to eliminate the effect of AKI on the performance of NGAL.
NGAL analysis and factors that influence the NGAL level
Plasma NGAL is routinely measured in patients who present to the ED because plasma NGAL can predict AKI in adult poisoning and because treatment of poisoning-related AKI potentially requires hemodialysis or RRT. 15 Venous blood was drawn at presentation in our ED, and plasma NGAL was measured in the ED laboratory with particle-enhanced turbidimetric immunoassay (The NGAL TestTM Reagent Kit BIOPORTO A/S, Hellerup, Denmark), which has a measurable range of 25–3000 ng/ml. The normal range of NGAL in our hospital is 30–135 ng/ml. The level of NGAL is influenced by baseline renal function, age, and sex. 1 Therefore, the baseline estimated glomerular filtration rate (eGFR), age, and sex were entered as confounding factors into all multivariate analyses in this study. The baseline eGFR was calculated using the baseline creatinine level and the Modification of Diet in Renal Disease Study equation. 16
Data analysis
Categorical variables are presented as frequencies, and continuous variables are presented as medians and interquartile ranges. For continuous variables, Student’s t-test or the Mann–Whitney U test for two-group comparisons and analysis of variance or the Kruskal–Wallis test and the Mann–Whitney U test with Bonferroni’s adjustment for three-group comparisons were used according to the normality of the data. Normality was tested using the Shapiro–Wilk test. For categorical variables, the χ2 test or Fisher’s exact test was used. To assess the predictive value of plasma NGAL for outcomes, univariate and multivariate logistic analyses were performed. The following variables, which have been suggested as prognostic factors in CO poisoning and can be easily evaluated at ED presentation, were tested in the univariate analysis: age, initial glasgow outcome scale (GCS), intentional exposure to CO, arterial HCO3 −, white blood cell (WBC) count, and C-reactive protein (CRP), creatinine and troponin I levels. 17 A multivariate binary logistic regression model (model 1) was established using significant univariate variables. Plasma NGAL, confounding factors for plasma NGAL, and significant univariate variables were entered into a second multivariate logistic regression model (model 2). For the multivariate logistic analysis, plasma NGAL was logarithmically transformed using the base logarithm of 2 because of the positively skewed and wide distribution of plasma NGAL. If two or more univariate variables retained in the multivariate binary or ordinary analysis were highly correlated in the linear regression analysis, one variable was removed to avoid collinearity. The multivariate analysis was performed using the entering method. Estimated odds ratios (ORs) and 95% confidence intervals (CIs) were calculated for all significant variables. The diagnostic performance of each regression model was assessed using receiver operating characteristic (ROC) curves and corresponding areas under the curve (AUCs). Predictive probabilities of the regression models were used for assessment of classification performance. To determine whether the addition of plasma NGAL significantly improved the performance of model 1, tests recommended by DeLong et al. 18 were performed. If NGAL improved the prognostic accuracy, the cutoff value of NGAL with 90% or 99% sensitivity and the optimal cutoff value of NGAL using the Youden index were selected. To investigate the value of NGAL for stratifying the severity of AKI, an ordinary logistic regression model was developed. A p-value <0.05 was considered statistically significant. All statistical analyses were performed using SPSS 21.0 (SPSS, Chicago, Illinois, USA) and Stata version 13.1 (StataCorp, College Station, Texas, USA).
Results
Table 1 describes the baseline characteristics of the 260 patients divided into tertiles based on plasma NGAL levels and presented at a mean of 1.8 h after exposure to CO at our ED. The initial COHb level and plasma NGAL level at presentation were 11.4 (6–20)% and 78 (54–115) ng/ml, respectively. Fifty-four (20.8%) patients had an elevated NGAL (NGAL > 135 ng/ml is the upper normal limit of NGAL in our hospital).
Baseline characteristics and clinical course of patients with acute charcoal-burning CO poisoning according to the level of plasma NGAL at presentation.

AKI: acute kidney injury; CO: carbon monoxide; DWI: diffusion weighted imaging; eGFR: estimated glomerular filtration rate; ED: emergency department; HBO: hyperbaric oxygen; NAGL: neutrophil gelatinase-associated lipocalin.
aThe duration of exposure to CO was available for 151 patients.
bTime interval from last CO exposure to arrival at ED.
cDiffusion weighted imaging was available for 163 patients.
dHaving an indication for hyperbaric oxygen therapy.
eBaseline eGFR.
fTime interval from arrival at ED to the conduction of the first hyperbaric oxygen treatment
gThe number of HBO sessions performed during the first 24 h after admission.
hThe need for mechanical ventilation regardless of reason (14 cases of problems related to the central nervous system (seizure or coma), 12 cases of pulmonary edema or aspiration, and 5 cases of severe hypotension and decreased mentality)
At presentation, the upper tertile group was older, had a longer exposure to CO, a lower GCS score, lower arterial HCO3 − levels, higher WBC counts and CRP and creatinine levels, and a higher prevalence of diffusion weighted imaging abnormality than did the middle and lower tertile groups.
During hospitalization, the upper tertile group required more HBO therapy sessions and demonstrated a higher prevalence of respiratory failure and AKI than did the other two groups.
Plasma NGAL at presentation and AKI
AKI occurred in 26.2% of patients, with only one requiring RRT during hospitalization. The group with AKI had a higher NGAL level at presentation than did the group without AKI (Figure 2). In the multivariate analysis of easily obtainable univariate predictors (model 1), arterial HCO3 −, CRP, and elevated troponin I were identified as independent predictors of AKI. In model 2, which included univariate predictors, plasma NGAL, and its confounding factors, plasma NGAL was identified as an independent predictor of AKI (adjusted OR 2.195 (95% CI: 1.244–1.032)) (Table 2). However, comparison of the AUCs for models 1 and 2 revealed no significant difference in the predictive accuracies of the models (AUC of 0.915 (0.874–0.947) in model 1 versus AUC of 0.934 (0.896–0.961) in model 2, p = 0.320).

The level of plasma NGAL at presentation according to the development of AKI, severity of AKI, and long-term neurological outcome. (a) The level of plasma NGAL at presentation was higher in the group with AKI than in the group without AKI (67.5 (52.3–91.2) ng/ml in the group without AKI versus 133.6 (78.5–214.5) ng/ml in the group with AKI; p < 0.001). (b) The plasma NGAL levels at presentation in the group without AKI, the group with stage 1 AKI, the group with stage 2 AKI, and the group with stage 3 AKI were 67.5 (52.3–91.2) ng/ml, 82.8 (63–140.4) ng/ml, 159.5 (86–225.9) ng/ml, and 229.8 (154.2–299.3) ng/ml, respectively. The plasma NGAL of the group with stage 2 or 3 AKI was higher than that of the group without AKI and that of the group with stage 1 AKI. (stage 0 vs stage 2; p < 0.001, stage 0 vs stage 3; p < 0.001, stage 1 vs stage 2; p = 0.002, stage 1 vs stage 3; p < 0.001). (c) In the subgroup that did not develop AKI during hospitalization, the patients with good long-term neurological outcomes had a higher level of plasma NGAL than the patients with poor long-term neurological outcomes (67 (52–89.2) ng/ml in patients with good neurological outcomes versus 153.4 (120.6–255.5) ng/ml in patients with poor neurological outcomes; p = 0.001). (d) In the subgroup with AKI, there was a significant difference in plasma NGAL at presentation between a good neurological outcome and poor neurological outcome (117.7 (62–183.4) ng/ml in patients with good neurological outcomes versus 156.3 (86–262.5) ng/ml in patients with poor neurological outcomes; p = 0.029). (a), (c), (d) *p < 0.05 compared with counterpart, **p < 0.01 compared with counterpart, ***p < 0.001 compared with counterpart. (b)***p<0.001 compared with the group without AKI, ## p<0.01 compared with stage 1 AKI, ### p<0.001 compared with stage 1 AKI. NAGL: neutrophil gelatinase-associated lipocalin; AKI: acute kidney injury.
Univariate and multivariate binary logistic regression for the development of AKI after acute charcoal-burning CO poisoning.
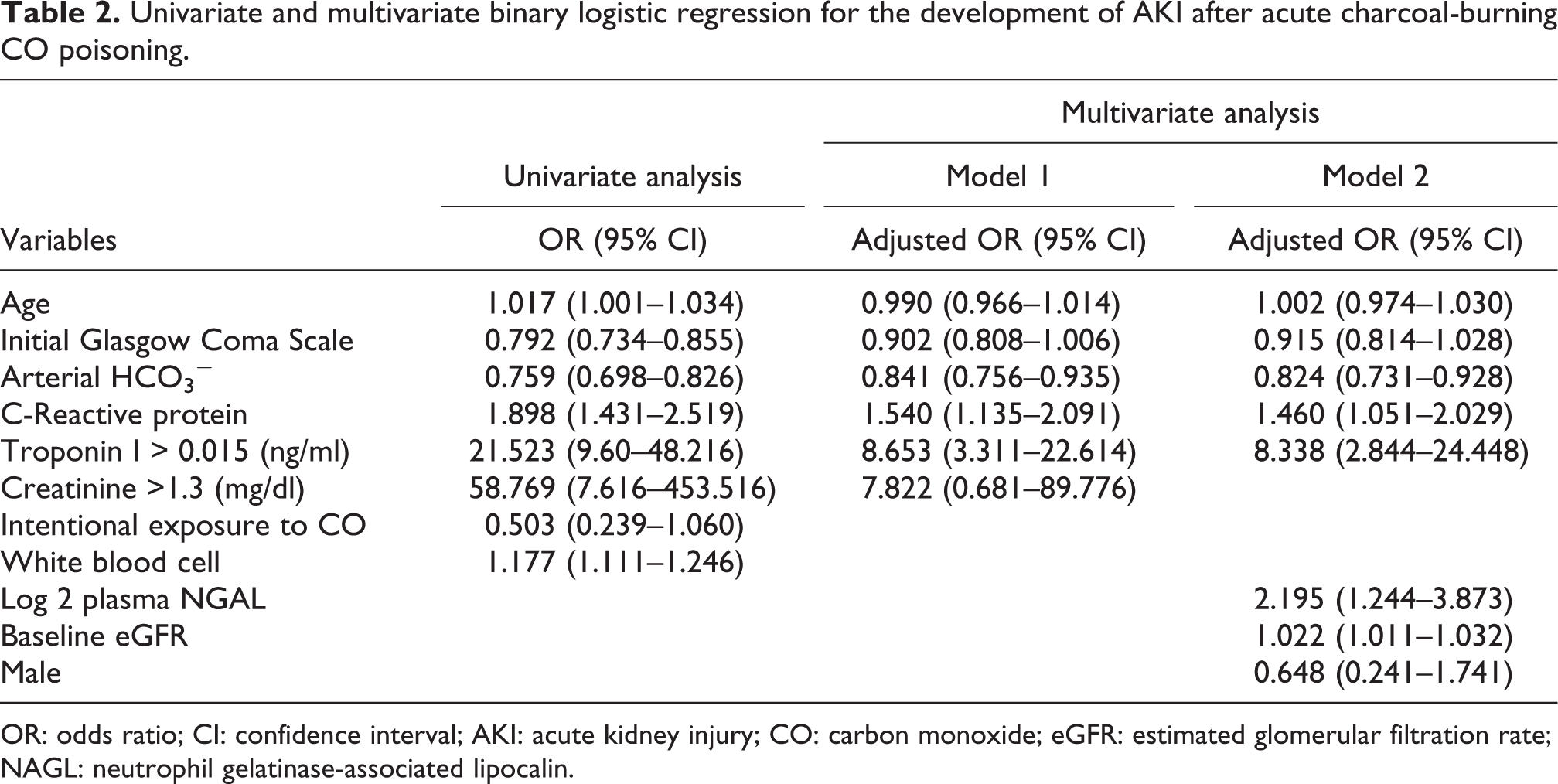
OR: odds ratio; CI: confidence interval; AKI: acute kidney injury; CO: carbon monoxide; eGFR: estimated glomerular filtration rate; NAGL: neutrophil gelatinase-associated lipocalin.
When stratifying AKI based on severity, groups with stage 2 or stage 3 AKI had higher NGAL levels at presentation than did groups without AKI or with stage 1 AKI (Figure 2). In ordinary logistic regression analysis, an increase in plasma NGAL at presentation was independently associated with an increase in the odds of an advanced stage of AKI, with an adjusted OR of 2.802 (95% CI: 1.782–4.406; p < 0.001).
Plasma NGAL level at presentation and long-term neurologic outcomes
Long-term persistent neurological sequelae developed in 11.5% of cases assessed at 27 (22.5–29) months after discharge. Regardless of the development of AKI, the group with poor neurological outcomes had higher NGAL levels at presentation than did the group with good neurological outcomes (Figure 2, Supplementary material Figure S1).
Plasma NGAL had an adjusted OR of 10.683 (95% CI: 1.913–59.643) for long-term neurological outcome after adjustments for baseline eGFR, age, sex, and significant univariate factors for long-term neurological outcomes in patients without AKI after acute charcoal-burning CO poisoning (Table 3). The AUCs of model 2, including plasma NGAL and univariate predictors, were significantly greater than those of model 1, which applied only significant univariate factors (0.959 (0.919–0.982) in model 2 versus 0.919 (0.869–0.953), p = 0.033) (Figure 3). In contrast, plasma NGAL was not associated with long-term neurological outcome in patients with AKI after acute charcoal-burning CO poisoning (Table 4).
Univariate and multivariate binary logistic regression for the long-term neurological outcome in patents without AKI after acute charcoal-burning CO poisoning.

OR: odds ratio; CI: confidence interval; AKI: acute kidney injury; CO: carbon monoxide; eGFR: estimated glomerular filtration rate; NAGL: neutrophil gelatinase-associated lipocalin.

Comparison of the predictive accuracy for long-term neurological outcomes between models 1 and 2. The AUCs of model 2, including plasma NGAL and significant univariate factors, were significantly greater those that of model 1, which applied only significant univariate factors (0.959 (0.919–0.982) in model 2 versus 0.919 (0.869–0.953) in model 1, p value = 0.033). The variables, which have been suggested as prognostic factors in CO poisoning and which can be easily obtained at ED presentation, were tested in the univariate analysis. AUC: areas under the curve; CO: carbon monoxide; ED: emergency department; NAGL: neutrophil gelatinase-associated lipocalin.
Univariate and multivariate logistic regression for the long-term neurological outcome in patients with AKI after charcoal-burning CO poisoning.

OR: odds ratio; CI: confidence interval; AKI: acute kidney injury; CO: carbon monoxide; eGFR: estimated glomerular filtration rate; NAGL: neutrophil gelatinase-associated lipocalin.
The optimal cutoff value of NGAL for long-term neurological outcome in patients without AKI was 112.4 ng/ml, with a sensitivity of 87.5% and a specificity of 88.6% (Table 5).
The optimal cutoff value of plasma NGAL and the cutoff value of plasma NGAL with 90% or 99% sensitivity for predicting the long-term neurological outcome in the subgroup without AKI.

AKI: acute kidney injury; NAGL: neutrophil gelatinase-associated lipocalin.
Discussion
This study demonstrated the following. (1) Although plasma NGAL was an independent predictor of AKI and was associated with increased severity of AKI, its combination with clinical parameters did not significantly improve the prediction accuracy for AKI. (2) Plasma NGAL significantly increased the accuracy of risk stratification for long-term neurological outcomes only in patients without AKI, but not in patients with AKI, after acute charcoal-burning CO poisoning.
Our results are consistent with studies highlighting plasma NGAL as an independent predictor of AKI in different clinical settings. The elevation of plasma NGAL results from renal injury (reduced glomerular filtration) or can be of extrarenal origin. 19 NGAL is stored in the granules of peripheral circulating mature neutrophils. 20 A strong correlation between plasma NGAL, interleukin (IL)-6, and IL-10 was found in septic animals. 21 Increased plasma NGAL levels have been reported in patients with systemic inflammatory conditions such as inflammatory bowel disease 22 and systemic inflammatory response syndrome. 23 NGAL is expressed at steady, low levels in various cell types, such as cells of the uterus, prostate, salivary gland, lung, and stomach. 1 Several experimental studies have demonstrated that increased NGAL mRNA expression by other distant organs, mainly the liver and lungs, substantially contributes to plasma NGAL accumulation in AKI. 24 Systemic inflammation or other organ injuries related to CO might contribute to the increase in plasma NGAL of extrarenal origin after charcoal-burning CO poisoning. In this study, 21 patients who did not experience AKI during hospitalization had an increased plasma NGAL level (range: 138–620 ng/ml) ng/ml) at presentation that was higher than the normal upper limit (135 ng/ml). Indeed, due to the combination of NGAL levels of both renal and extrarenal origin, the diagnostic value of plasma NGAL for the prediction of AKI might be restricted and fail to improve the predictive accuracy for AKI in acute charcoal-burning CO poisoning. However, the mechanism underlying the increase in plasma NGAL at presentation after charcoal-burning CO poisoning in our study remains elusive. Further prospective studies measuring other kidney biomarkers, such as kidney injury molecule-1 and tissue inhibitor of metalloproteinases-2, are needed to reveal the source of plasma NGAL after charcoal-burning CO poisoning.
An increased plasma NGAL level was associated with increased severity of AKI in this study. Plasma NGAL appears to be a useful prognostic tool for the initiation of RRT or progression to a more severe stage of AKI or chronic kidney disease (CKD). 25,26 However, because only one patient with AKI required RRT and because no patient progressed to CKD, we could not investigate the predictive value of NGAL in terms of the need for RRT.
Few studies have evaluated the neuroprognostic value of NGAL; moreover, existing studies have been conducted only in settings of adult cardiac arrest. 7,8 It has been reported that serum NGAL measurements at 72 h (but not at admission and at 24, 48 h) after cardiac arrest are associated with the cerebral performance category (CPC) scale at the 6-month evaluation based on univariate analysis; however, the authors did not perform multivariate analysis. 7 Although NGAL is not a brain-specific marker, plasma NGAL at presentation significantly improved the diagnostic accuracy for long-term neurological outcome after acute charcoal-burning CO poisoning in the group without AKI. The neuroprognostic role of NGAL in patients without AKI might be explained by the association between plasma NGAL and inflammation, which is a pathological mechanism of neurological sequelae related to CO poisoning. 10 The notion that NGAL can reflect inflammation in patients without AKI is supported by a study in which the leukocyte count and CRP level were the main determinants of plasma NGAL in patients with a normal eGFR after ST-elevation myocardial infarction. 9 In healthy individuals, plasma NGAL levels were correlated with CRP and IL-10 but not with plasma Cr or eGFR. 2 Consistent with that finding, NGAL was positively correlated with the level of CRP (r = 0.343, p < 0.001) and WBC count (r = 0.275, p < 0.001) at presentation in patients without AKI in this study. Another plausible explanation for the neuroprognostic value of NGAL is the inhibitory effect of NGAL on neuronal survival. NGAL is normally expressed in the brain and is upregulated after induction of the peripheral inflammatory response. 27 Increased cerebral NGAL expression sensitizes neurons to cell death and attenuates Akt phosphorylation, which constitutes a key pathway involved in hippocampal neuroplasticity. 28 The AUC of NGAL at presentation was 0.839 (excellent performance for predicting long-term neurological outcome), and the sensitivity and specificity of the optimal cutoff value for long-term neurologic outcome were acceptable. However, because the predictive values of diagnostic tests vary according to disease prevalence, 29 these cutoff values of plasma NGAL might not be applicable to other populations with, for example, a different prevalence of poor long-term neurological sequelae related to CO poisoning from different CO gas sources or different treatment protocols for CO poisoning.
In contrast, despite the significant differences in NGAL related to neurological outcomes in the subgroup with AKI, NGAL was not identified as an independent predictive factor, and stage 3 AKI remained an independent factor for neurological outcome. The dominant role of eGFR rather than inflammation in determining the plasma NGAL level in the setting of AKI 9 might decrease the relevance of NGAL to the long-term neurological outcome. Additionally, enrollment of a low number of patients in the subgroup with AKI might explain the poor predictive performance of NGAL. Studies including a large number of patients with AKI are thus needed to determine the neuroprognostic role of NGAL.
This study has several limitations. First, a potential confounder that influences NGAL levels may exist, even after adjustments for baseline renal function, age, and sex. NGAL levels can be influenced by cancer or various pathological lesions in the liver or lung. 30 Second, the predictive value of NGAL can vary because of different NGAL assays, varying diagnostic criteria for the clinical endpoints of AKI and neurological outcome, and heterogeneous study populations (sex and age). 31 These factors impede the generalizability of this study. The results obtained in this study should thus be interpreted cautiously based on the various abovementioned factors. Third, neurological outcome assessments were not performed at the same fixed time point in each patient. Because neurological sequelae related to CO poisoning can improve over time, 32 the classification of poor outcomes can be overestimated. However, the time interval from acute CO poisoning to the assessment of neurological outcomes did not differ according to outcome (27 (23–29) months for good long-term neurological outcome versus 26 (22–29.5) months for poor long-term neurological outcome, p = 0.491). In addition, the potential for improvement in patents with a GOS of 2 and 3 is likely to be low because they were assessed at least 2 years after CO poisoning (at 25 (24–34.3) months for a GOS of 2 and at 29 (27–30) months for a GOS of 3). Neurological outcomes in patients with good outcomes were also assessed at least 15 months after CO poisoning. However, neurological deterioration at more than 15 months after CO poisoning is not related to CO poisoning. Fourth, plasma NGAL is a dynamic parameter; yet we evaluated the predictive value of NGAL at admission. Future studies should reveal the optimal time point at which to measure plasma NGAL for the prediction of outcomes after charcoal-burning CO poisoning.
Conclusions
Plasma NGAL at presentation is an independent predictor of long-term neurological outcome and might significantly improve predictive accuracy when used in combination with clinical parameters in patients without AKI after acute charcoal-burning CO poisoning. We recommend the inclusion of this easy and practical test for risk stratification among patients with acute charcoal-burning CO poisoning to permit further validation of this potentially valuable test.
Despite the significant association of plasma NGAL at presentation with the development and severity of AKI, the combination of plasma NGAL with clinical parameters does not significantly enhance the diagnostic accuracy for AKI after acute charcoal-burning CO poisoning.
Supplemental material
Supplement_1 - Predictive value of plasma neutrophil gelatinase-associated lipocalin in acute charcoal-burning carbon monoxide poisoning
Supplement_1 for Predictive value of plasma neutrophil gelatinase-associated lipocalin in acute charcoal-burning carbon monoxide poisoning by JM Moon, BJ Chun, MH Shin and YS Cho in Human & Experimental Toxicology
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by a Grant (CRI 18031-1) Chonnam National University Hospital Biomedical Research Institute.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
