Abstract
Dibutyl phthalate (DBP), a ubiquitous environmental contaminant, has been reported to be involved in hepatic and renal tissue damage. However, the role of DBP in oxidative stress and in extracellular signal-regulated kinase (ERK1/2) pathways remains unclear. To investigate the underlying mechanism, Kunming (KM) mice received daily doses of combinations of 50 mg/kg DBP, 50 mg/kg vitamin E (VitE), and 1 mg/kg PD98059 for 28 consecutive days. Any changes in reactive oxygen species (ROS) and malondialdehyde (MDA) levels, as well as any histopathological alterations in tissues, were observed to assess oxidative stress. In addition, the levels of alanine aminotransferase, aspartate aminotransferase, and albumin in serum were used to evaluate liver function. The levels of creatinine and urea nitrogen in serum were measured to evaluate kidney function. We found that DBP significantly increased oxidative damage and the expression of phosphorylated ERK1/2. Furthermore, pretreatment with the ERK inhibitor PD98059 followed by the antioxidant VitE attenuated the levels of ROS, MDA, ERK1/2 phosphorylation, and DBP-mediated disorders, indicating that the oxidative stress and the ERK1/2 pathways are associated with DBP-induced hepatic and renal dysfunction in KM mice.
Keywords
Introduction
Phthalates (PAEs), a class of synthetic chemicals, are important plastic-modifying additives used in daily products, including some food packaging materials and medical appliances. 1,2 PAEs emit from these materials, which can lead to high PAE exposure to humans via medical treatment. 3,4 Among all PAEs, dibutyl phthalate (DBP) is a ubiquitous environmental contaminant in terms of deleterious health outcomes, such as developmental and/or reproductive disruption. 5
Epidemiological data suggest that DBP is responsible for adverse hepatic effects. 6,7 Animal studies have also shown that DBP is a reproductive and developmental toxin. 8 Although DBP does not accumulate in any organ shortly after administration, over the long term, it will be found primarily in the liver and kidney. Zeng et al. 9 showed that DBP can be found in the liver and kidney of rats. Furthermore, accumulating evidence from human studies has reported that DBP can enter the body and reach the liver via the circulatory system and biotransformation. 10,11 Therefore, the liver is more sensitive to DBP toxicity than is the kidney, another important target organ, which has a strong compensatory function and various detoxification functions. 8,12 DBP has been reported to be involved in hepatic and renal tissue damage. However, the molecular mechanism of DBP-induced liver and kidney damage still needs to be explored.
Some studies have shown that oxidative stress could explain the adverse changes observed in DBP-exposed rats and/or humans. 12,13 Oxidative stress results in the generation of reactive oxygen species (ROS), and overproduction of ROS can cause tissue injury and also activate kinases or inhibit phosphatases resulting in the stimulation of signaling pathways. 14 The extracellular signal-regulated kinases 1 and 2 (ERK1 and ERK2), often referred to collectively as ERK1/2, are members of the mitogen-activated protein kinase (MAPK) family of proteins observed in all eukaryotes. The ERK1/2 pathway is activated by diverse extracellular and intracellular stimuli, including various cellular stressors, such as ROS. 14 ROS can inhibit protein phosphatases, leading to the activation of the ERK1/2 signaling pathway, which has been shown to be a critical regulator of cell differentiation, cell physiology, and neuronal function. 15 Phosphorylation of ERK1/2 has also been implicated in a variety of pathological conditions in the damaged organs, including cancer and other diseases. 16 The ERK1/2 inhibitor, PD98059, protects the brain against cell death caused by ROS/ERK activation in a cardiac arrest rat model and also attenuates the liver and renal dysfunction caused by zymosan. 15,17 Thus, the oxidative stress pathway and ERK1/2 pathway may be a potential therapeutic strategy for DBP-mediated disorders.
In this study, we determined the levels of oxidative stress and the expression of phosphorylated ERK1/2 induced by DBP in liver and kidney tissues of Kunming (KM) mice. We looked at changes in ROS and malondialdehyde (MDA) levels and at histopathological alterations in liver and kidney tissues to assess oxidative stress. In addition, levels of alanine aminotransferase (ALT), aspartate aminotransferase (AST), and albumin (ALB) in serum were detected to evaluate liver function, and levels of creatinine (CREA) and urea nitrogen (UREA) in serum were measured to evaluate renal function. The antioxidant VitE and the ERK1/2 pathway inhibitor PD98059 were also used to demonstrate that the ERK1/2 pathway mediates the oxidative damage induced by DBP. Such changes may have implications for DBP-induced hepatic and renal dysfunction in mice via ROS/ERK1/2 activation.
Materials and methods
Ethics statement
All experimental procedures were approved by the Office of Scientific Research Management of Hubei University of Science and Technology (Xianning, China). The protocol followed in this study has been examined and approved by the Animal Ethics Committee of Hubei University of Science and Technology (Xianning, China).
Animal preparation
Male KM mice (22 ± 1 g) were obtained from Hubei Province Experimental Animal Center (Wuhan, China). All mice were housed under specific pathogen-free (SPF) conditions at 20–25°C with 50–70% humidity and a 12-h light/12-h dark cycle. A commercial diet and filtered water were provided ad libitum. Mice were quarantined for ≥5 days prior to the study. Eight mice were used in each group to minimize the number of experimental animals needed while simultaneously ensuring the statistical validity of the results.
Reagents and kits
DBP (>99%, CAS: 84-74-2), VitE (CAS: 59-02-9), 20,70-dichlorodihydrofluorescein (DCFH-DA), and 2-thiobarbituric acid (TBA) were purchased from Sigma Aldrich (St Louis, Missouri, USA). PD98059 (CAS: 167869-21-8) was purchased from MedChemExpress (Monmouth Junction, New Jersey, USA). Mouse kits for ALB, ALT, AST, CREA, and UREA were purchased from iCubio Medical Technology (Shenzhen, China). Mouse enzyme-linked immunosorbent assay (ELISA) kits for phosphorylated ERK1/2 were obtained from eBioscience (San Diego, California, USA). All other chemicals were of the highest grade commercially available or as indicated.
Experimental protocol
The KM mice were divided randomly into eight groups of eight mice each (Figure 1). The groups were treated as follows: group A: control (saline), group B: 50 mg/kg/day DBP, group C: 50 mg/kg/day VitE, group D: 50 mg/kg/day DBP combined with 50 mg/kg/day VitE (DBP + VitE), group E: 1 mg/kg/day PD98059 (PD98059), group F: 50 mg/kg/day DBP plus pretreatment of 1 mg/kg/day PD98059 (DBP + PD98059), group G: mg/kg/day VitE combined with pretreatment of 1 mg/kg/day PD98059 (VitE + PD98059), and group H: 50 mg/kg/day DBP combined with 50 mg/kg/day VitE plus pretreatment of 1 mg/kg/day PD98059 (DBP + VitE + PD98059). DBP was dissolved in Tween 80 (DBP:Tween 80 is 1:1 in wt/wt) together with a saline solution to obtain 50 mg/kg. 8 The concentration of Tween 80 was previously demonstrated to be pharmacologically inert in in vivo experiments. 12 According to Ourique et al., 18 VitE was suspended in sterile saline to obtain 50 mg/kg. VitE acts as an antioxidant via intragastric administration. The ERK inhibitor PD98059 solution was prepared in normal saline containing 2% dimethyl sulfoxide (Fisher Scientific, Waltham, MA, USA) and was injected at 1 mg/kg for 30 min before other treatments. 19 Experimental mice received daily doses according to which group they had been assigned for 28 consecutive days. After 28 days, all mice were killed. The livers and kidneys were collected and processed according to the requirements of the tests described.
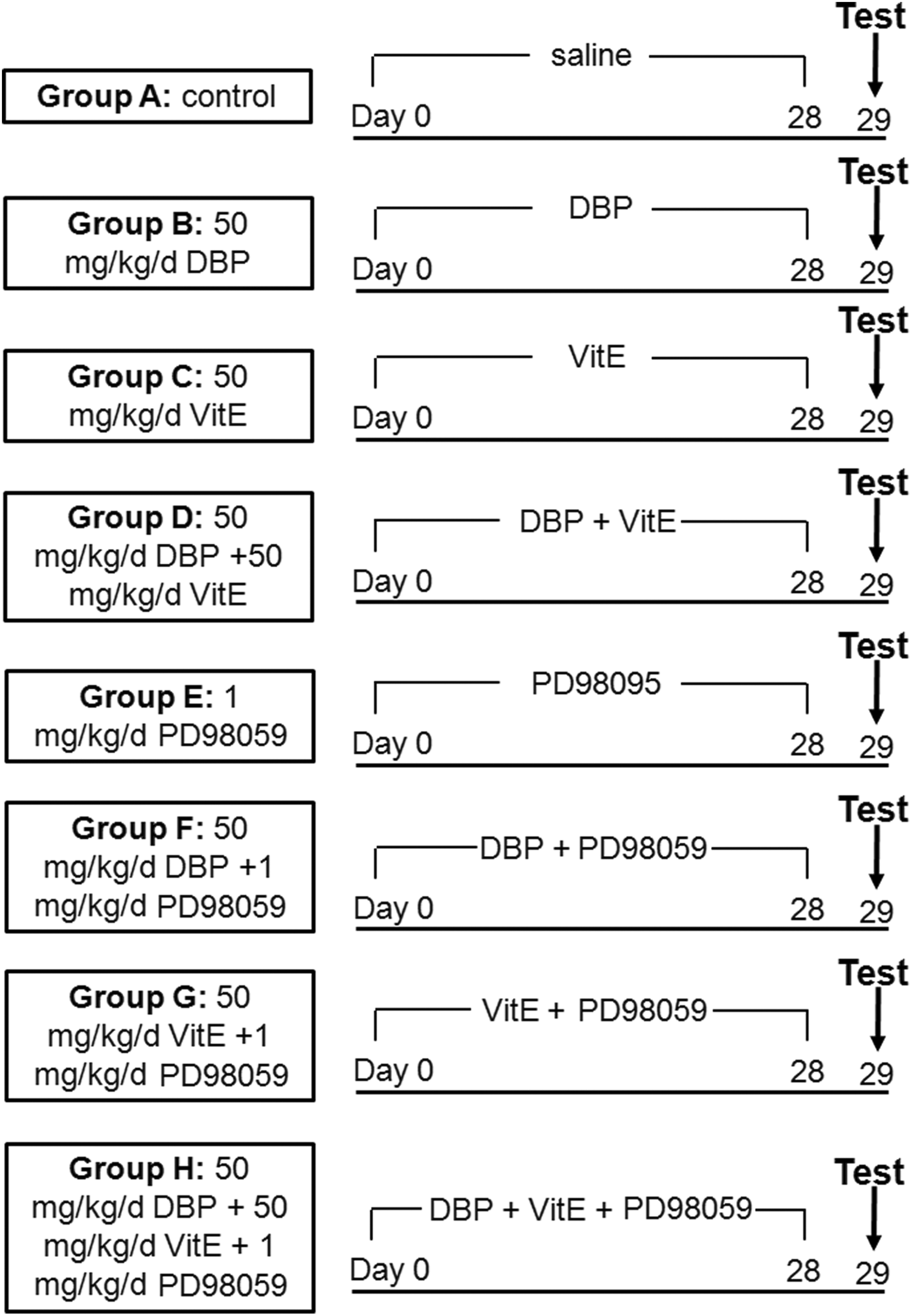
Experimental protocol.
Organ coefficients
The livers and kidneys of the mice were trimmed of extraneous tissues and weighed. The viscera coefficient (calculated as 100% × organ weight (g)/body weight (g)) was used as an indicator of organ-specific toxicity and general toxicity in the mice.
Histopathological examinations
The hepatic and renal tissues of the mice were isolated for the preparation of histopathological slides. All samples were incubated in fixative (4% paraformaldehyde) solution for 24 h at room temperature, following which the distal pieces were embedded in paraffin, sectioned into 10-μm slices, stained with hematoxylin and eosin (H&E) according to standard protocols, 12 and observed using a BX53 microscope (Olympus, Tokyo, Japan). The tissue sections were examined qualitatively by two experienced pathologists in a blinded manner.
Determination of liver and kidney function
The liver and kidney function of mice from different treatment groups were determined using an animal automatic biochemical analyzer (iMagic-V7 Automatic Analyzer, iCubio). The ALB, ALT, and AST levels in serum were measured to evaluate liver function. The CREA and UREA levels in serum were measured to evaluate renal function.
Preparation of tissue homogenates
The liver and kidney tissues from each mouse were placed in 10 mL/g ice-cold phosphate-buffered saline (pH 7.5) and homogenized using a glass homogenizer. The homogenates were then centrifuged at 10,000 × g for 10 min at 4°C, and the resultant supernatants were collected for later detection of ROS, MDA, and phosphorylated ERK1/2.
Detection of ROS and MDA
The levels of ROS in the supernatant were determined based on the reactions between ROS and the by-products of DCFH-DA. 12 The reaction mixture was allowed to incubate for 5 min in the dark, and the fluorescence was then quantified using a FLx800 fluorescence monitor (Bio-Tek Instruments, Winooski, Vermont, USA) with an excitation wavelength of 485 nm and an emission wavelength of 520 nm.
MDA assay was conducted, as described by Ma et al. 12 MDA can react with TBA to form a stable chromophoric product at high temperature (90–100°C), which can be quantified at wavelengths of 450, 532, and 600 nm. The MDA concentration was determined using the following standard curve equation: MDA content (μmol/L) = 6.45 × (OD532 – OD600) – 0.56 × OD450, where OD is the optical density. The results were normalized to the protein concentration measured by a Lowry assay for each sample and expressed as μmol/g protein.
Analysis of phosphorylated ERK1/2
The levels of phosphorylated ERK1/2 in hepatic and renal tissues were measured using commercial ELISA kits according to the manufacturer’s instructions. The sensitivity of the ELISA kits was 10 pg/mL for phosphorylated ERK1/2. The concentrations were determined in duplicate for each sample.
Statistical analyses
The values are presented as mean ± standard error of the mean. The differences between groups were analyzed for significance by performing a one-way analysis of variance combined with a Tukey test. A p value of <0.05 was considered statistically significant. Statistical graphs were generated using GraphPad Prism 5.02 (GraphPad Software Inc., La Jolla, California, USA).
Results
DBP-mediated liver and kidney damage
The viscera coefficient (Figure 2) tended to decrease with increasing DBP dosage. The 50 mg/kg/day DBP exposure group showed a significant decrease in the viscera coefficient (p < 0.01, p < 0.05) when compared with the vehicle control group. In the DBP + VitE + PD98059 group, the liver coefficient showed a significant increase when compared with the DBP exposure group (p < 0.05) (Figure 2(a)). Compared with the DBP + VitE group, the DBP + VitE + PD98059 group also showed a significant increase (p < 0.05) (Figure 2(a)). Although no statistical difference was noted for the renal tissues, the trend was the same as described above for liver tissues (Figure 2(b)). The results show that both VitE and PD98059 can reduce the decline in the organ coefficient mediated by DBP, with PD98059 having a more significant effect than VitE.

Organ coefficients of mice in the different treatment groups (n = 6). (a) Liver viscera coefficient and (b) kidney viscera coefficient. *p < 0.05, **p < 0.01: 50 mg/kg/day DBP group compared with the control; #p < 0.05: DBP + VitE group compared with the DBP + VitE + PD98059 group; &p < 0.05: 50 mg/kg/day DBP group compared with the DBP + VitE + PD98059 group. DBP: dibutyl phthalate; VitE: vitamin E.
From the H&E staining, we observed that the liver underwent alterations in response to DBP exposure (Figure 3(a)). In the control group, the liver tissue structure and the cell morphology were normal. No significant histopathological changes were observed in the VitE, PD98059, and VitE + PD98059 groups. In the 50 mg/kg/day DBP group, there was an obvious edema of the liver cells, the structure of the hepatic cord was unclear, and the hepatic sinusoid became narrow while the central vein was dilated. In the DBP + VitE, DBP + PD98059, and DBP + VitE + PD98059 groups, hepatocyte edema and sinusoidal narrowing were correspondingly alleviated.
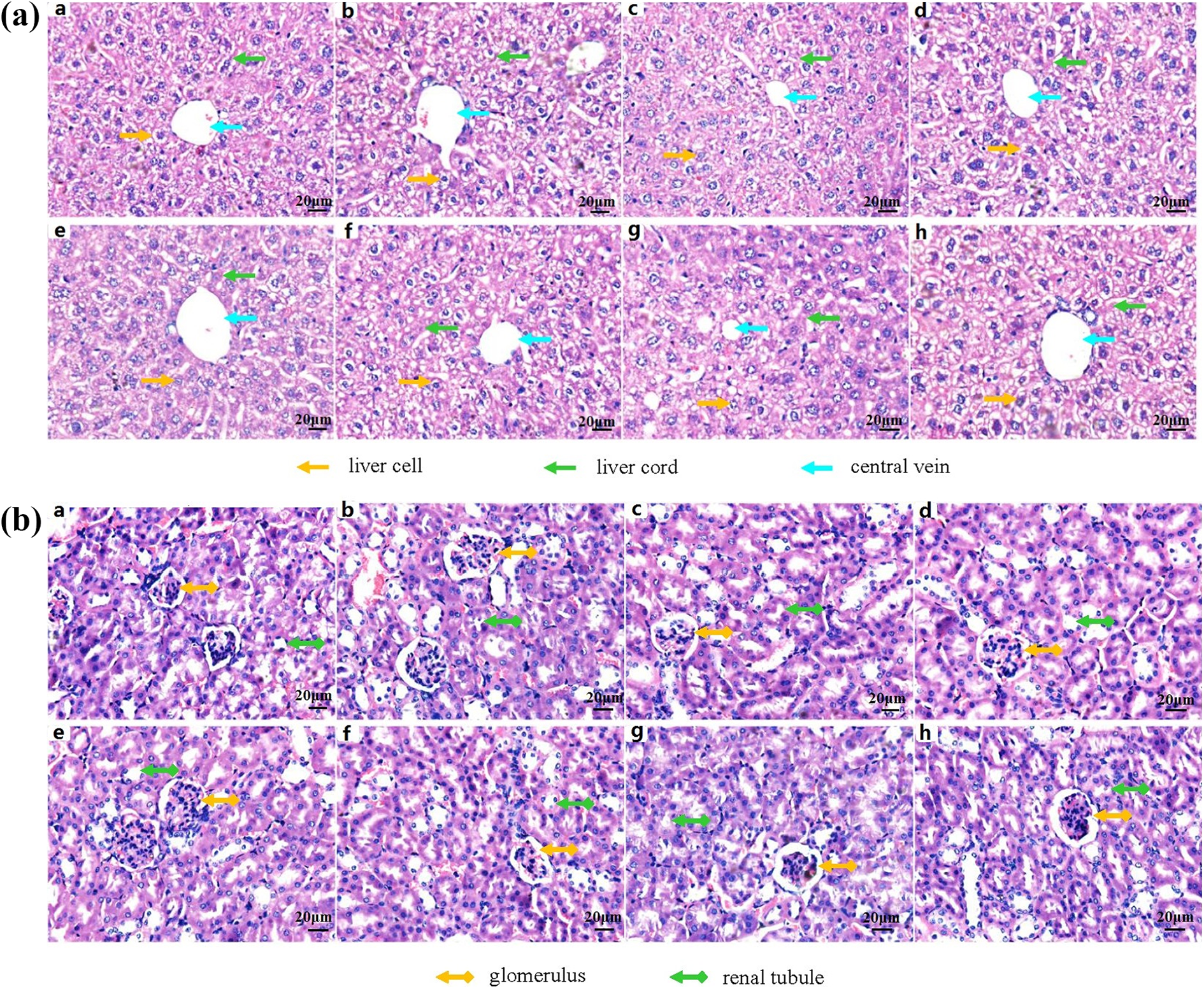
Representative histological images of liver and kidney slices (stained with H&E), Magnification, ×40. Row (a) in the liver slices: liver cell (yellow arrow), liver cord (green arrow), and central vein (blue arrow); row (b) in the kidney slices: glomerulus (yellow arrow) and renal tubule (green arrow). H&E: hematoxylin and eosin.
The kidney slices also showed damage caused by DBP (Figure 3(b)). The structures of the glomeruli and renal tubules in the control group were normal. Similar to the control group, the renal tissue structure and the cell morphology of the VitE, PD98059, and VitE + PD98059 groups were normal. However, histopathological changes in the kidney tissues of the 50 mg/kg/day DBP group were obvious. The glomerular capillary loops were lobulated, the renal tubular epithelial cells were edematous, and the lumen of the renal tubule was smaller. In the DBP + VitE and DBP + PD98059 groups, the glomerular cells were proliferated and the renal tubular epithelial cells were slightly edematous. The renal tubular epithelial cells in the DBP + VitE + PD98059 group were also slightly edematous.
DBP-promoted liver and kidney dysfunction
The ratio of AST to ALT in serum is a sensitive index of hepatocyte injury and is commonly used to indicate the extent of damage in hepatocytes. A ratio of AST/ALT >1 indicates that the liver damage is more serious. The AST/ALT ratios for the different treatment groups are shown in Figure 4(a). The AST/ALT ratio of the DBP group was significantly higher than that of the control group (p < 0.01). The ratio of AST/ALT of the DBP + PD98059 group and the DBP + VitE + PD98059 group was significantly lower than that of the DBP group (p < 0.05, p < 0.01). Compared with the DBP + VitE group, the AST/ALT ratio of the DBP + VitE + PD98059 group was significantly lower (p < 0.05). The results showed that VitE and PD98059 can alleviate the increases in AST and ALT levels induced by DBP and that PD98059 had a more significant effect on the reduction of AST and ALT in serum than VitE.

Liver dysfunction results for mice in the different treatment groups (n = 6). (a) AST/ALT ratio and (b) ALB content. **p < 0.01: 50 mg/kg/day DBP group compared with the control. #p < 0.05: DBP + VitE group compared with DBP + VitE + PD98059 group. &&p < 0.01: 50 mg/kg/day DBP group compared with the DBP + VitE + PD98059 group. DBP: dibutyl phthalate; VitE: vitamin E; ALT: alanine aminotransferase; AST: aspartate aminotransferase; ALB: albumin.
The lower levels of ALB in serum reflect chronic liver damage. The changes in the ALB content for the different treatment groups are shown in Figure 4(b). The ALB levels in the serum of the DBP group were significantly lower than that in the control group (p < 0.01). The ALB content in the DBP + PD98059 group and the DBP + VitE + PD98059 group was significantly higher than that of the DBP group (p < 0.05, p < 0.01). Compared with the DBP + VitE group, the ALB levels in the DBP + VitE + PD98059 group were significantly higher (p < 0.05). The results showed that VitE and PD98059 can attenuate the reduction of ALB caused by DBP and that PD98059 had a more significant effect on the increase of ALB in serum than VitE.
CREA is an important indicator of renal function. A noticeable increase in CREA levels in serum indicates glomerular filtration dysfunction. The changes in CREA levels of the different treatment groups are shown in Figure 5(a). The CREA levels in the DBP group were significantly higher than those in the control group (p < 0.01). The CREA content in the DBP + PD98059 group and the DBP + VitE + PD98059 group was significantly lower than that of the DBP group (p < 0.05, p < 0.01). Compared with the DBP + VitE group, the CREA levels in the DBP + VitE + PD98059 group were significantly lower (p < 0.05). These results indicate that VitE and PD98059 can attenuate the increase of CREA levels mediated by DBP and that PD98059 had a more significant effect on the decline of CREA in serum than VitE.
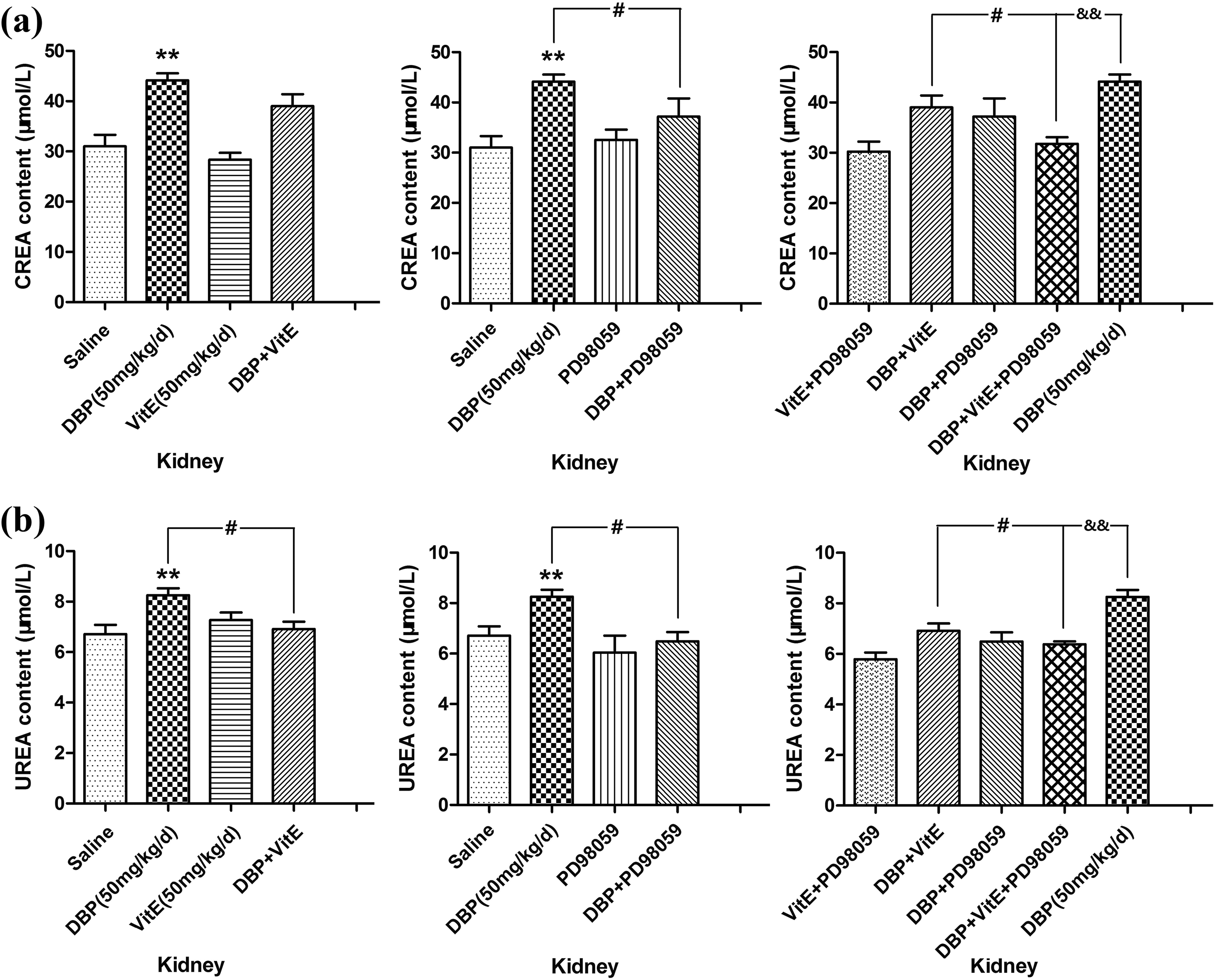
Kidney dysfunction results for mice in the different treatment groups (n = 6). (a) CREA content and (b) UREA content. **p < 0.01: 50 mg/kg/day DBP group compared with the control. #p < 0.05: DBP + VitE group compared with the DBP group. #p < 0.05: DBP + VitE group compared with the DBP + VitE + PD98059 group. &&p<0.01: 50 mg/kg/day DBP group compared with the DBP + VitE + PD98059 group. DBP: dibutyl phthalate; VitE: vitamin E; CREA: creatinine; UREA: urea nitrogen.
UREA is another principal indicator of renal function. The level of UREA in serum indirectly reflects glomerular filtration dysfunction. The levels of UREA in serum for the different treatment groups are shown in Figure 5(b). The UREA levels of the DBP group were significantly higher than those of the control group (p < 0.01). The UREA content in the DBP + VitE group, DBP + PD98059 group, and DBP + VitE + PD98059 group was significantly lower than that of the DBP group (p < 0.05, p < 0.05, p < 0.01). Compared with the DBP + VitE group, the UREA levels of the DBP + VitE + PD98059 group were significantly lower (p < 0.05). These results show that VitE and PD98059 can reduce the elevation of UREA induced by DBP and that PD98059 had a more significant attenuating effect on UREA in serum than VitE.
ROS generation and lipoperoxidation injury
ROS is one of the most important biomarkers of oxidative stress, and its generation provides evidence of oxidative damage. The changes in the ROS content for the different treatment groups are shown in Figure 6. The ROS content in the cells of the DBP group was significantly higher than that in the control group (p < 0.01). The ROS content in the DBP + PD98059 group and the DBP + VitE + PD98059 group was significantly lower than that of the DBP group (p < 0.05, p < 0.01), and compared with the DBP + VitE group, the ROS content of the DBP + VitE + PD98059 group was significantly lower (p < 0.05). The results showed that VitE and PD98059 can block the increase of ROS caused by DBP and that PD98059 had a more significant attenuating effect on intracellular ROS than VitE.
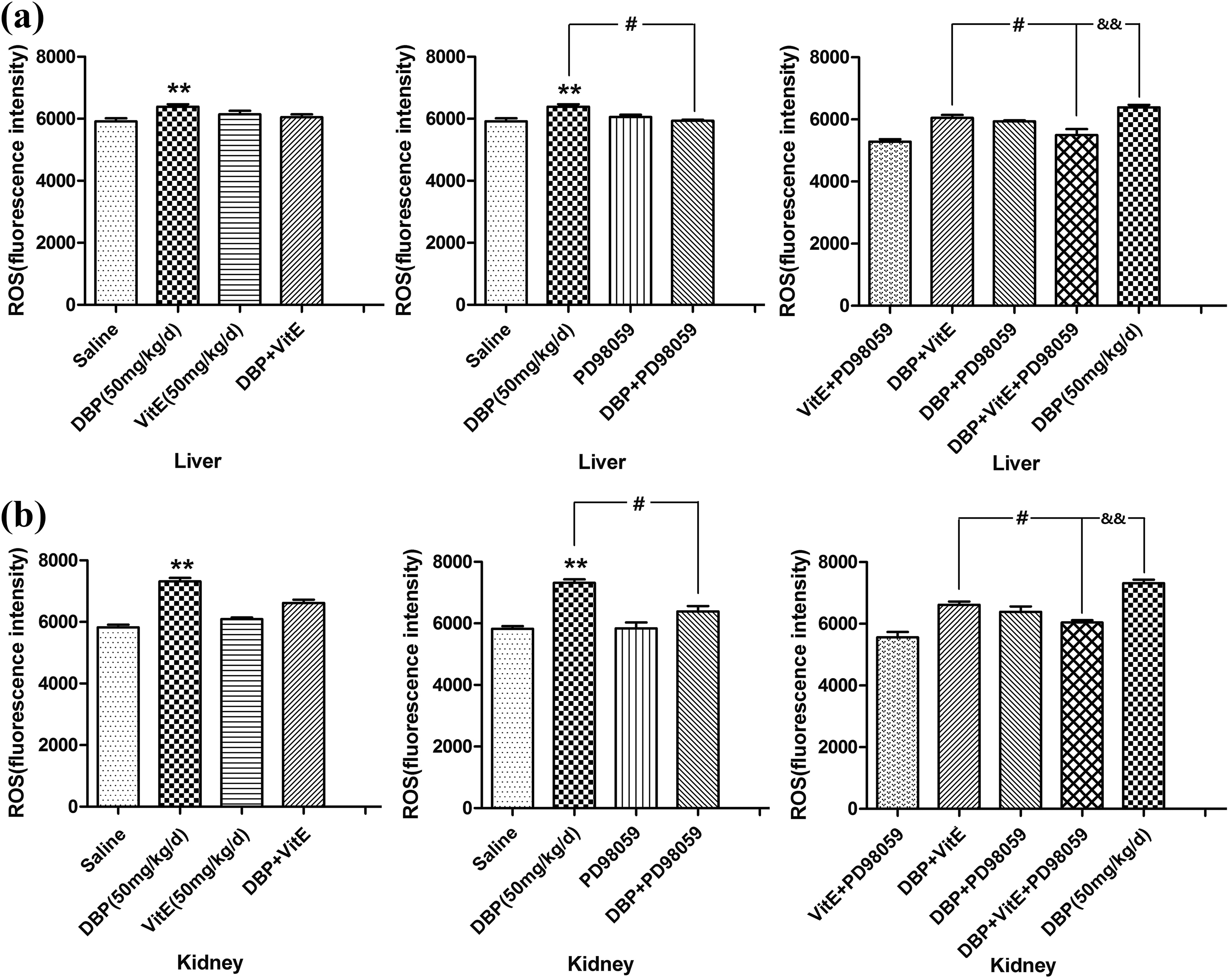
Intracellular ROS content of liver and kidney tissues of mice in the different treatment groups (n = 6). (a) Intracellular ROS content of the liver and (b) intracellular ROS content of the kidney. **p < 0.01: 50 mg/kg/day DBP group compared with the control. #p < 0.05: DBP + VitE group compared with the DBP + VitE + PD98059 group. &&p < 0.01: 50 mg/kg/day DBP group compared with the DBP + VitE + PD98059 group. DBP: dibutyl phthalate; VitE: vitamin E; ROS: reactive oxygen species.
MDA is the end product of lipid peroxidation caused by excessive production of ROS. In accordance with increasing ROS levels, an increase in MDA levels was found in the liver and kidney tissues (Figure 7). The MDA levels in cells of the DBP group were significantly higher than that in the control group (p < 0.01). The MDA content in the DBP + PD98059 group and the DBP + VitE + PD98059 group was significantly lower than that of the DBP group (p < 0.05, p < 0.01). Compared with the DBP + VitE group, the MDA content of the DBP + VitE + PD98059 group was significantly lower (p < 0.05). The results showed that VitE and PD98059 can decrease the enhancement of MDA induced by DBP and that PD98059 had a more significant attenuating effect on intracellular MDA than VitE.

Intracellular MDA content of liver and kidney tissues of mice in the different treatment groups (n = 6). (a) Intracellular MDA content of the liver and (b) intracellular MDA content of the kidney. **p < 0.01: 50 mg/kg/day DBP group compared with the control. #p < 0.05: DBP + VitE group compared with the DBP + VitE + PD98059 group. &&p < 0.01: 50 mg/kg/day DBP group compared with the DBP + VitE + PD98059 group. DBP: dibutyl phthalate; VitE: vitamin E; MDA: malondialdehyde.
DBP-promoted phosphorylated ERK1/2 levels
The changes in phosphorylated ERK1/2 levels for the different treatment groups are shown in Figure 8. The phosphorylated ERK1/2 levels in the cells of the DBP group were significantly higher than the control group in both hepatic and renal tissues (p < 0.05). The phosphorylated ERK1/2 levels in hepatic and renal tissues of the DBP + VitE + PD98059 group were significantly lower than those of the DBP group (p < 0.01). The phosphorylated ERK1/2 levels in the liver tissues of the DBP + PD98059 group and the DBP + VitE + PD98059 group were significantly lower than that of the DBP group (p < 0.05, p < 0.01). While compared with the DBP + VitE group, the phosphorylated ERK1/2 levels in the liver tissues of the DBP + VitE + PD98059 group were significantly lower (p < 0.05) (Figure 8(a)). In the kidney, the phosphorylated ERK1/2 levels in the DBP + VitE group and the DBP + PD98059 group were significantly lower than that of the DBP group (p < 0.05, p < 0.01) (Figure 8(b)). The results also showed that VitE and PD98059 can attenuate the upregulation of phosphorylated ERK1/2 levels caused by DBP and that PD98059 had a more significant attenuating effect on phosphorylated ERK1/2 levels in the liver than VitE.

Intracellular phosphorylated ERK1/2 level of liver and kidney tissues of mice in the different treatment groups (n = 6). (a) Intracellular phosphorylated ERK1/2 level of the liver and (b) intracellular phosphorylated ERK1/2 level of the kidney. **p<0.01: 50 mg/kg/day DBP group compared with the control. #p < 0.05: DBP + VitE group compared with the DBP + VitE + PD98059 group. &&p < 0.01: 50 mg/kg/day DBP group compared with the DBP + VitE + PD98059 group. DBP: dibutyl phthalate; VitE: vitamin E; ERK1/2: extracellular signal-regulated kinase.
Discussion
It has been reported that DBP, frequently inhaled at a rate of 0.5 mg/kg/person/day, can be hydrolyzed by lipase in the intestinal tract. Studies have also found that DBP released from polyvinyl chloride blood bags can accumulate in many organs of the human body as a result of blood transfusion. 20 Epidemiological studies have shown that DBP and its metabolites can be detected in human blood and urine and that the residual DBP can seriously affect the human reproductive, immune, and other systems. 10,11 DBP is a PAE ester that can induce peroxisome proliferation, and the exposure to DBP can increase the peroxisome proliferation-activated receptors (PPARs) in mice and humans. 21,22 DBP is also an exogenous exposure, which can stimulate oxidative stress and activate MAPK cascades as an oxide. 23 In this study, we found that the oxidative stress and the ERK1/2 pathways are involved in DBP-induced liver and kidney damage in mice. DBP significantly increased oxidative damage and the expression of phosphorylated ERK1/2. In addition, we found that pretreatment with the ERK inhibitor PD98059, combined with antioxidant VitE, attenuated ROS, MDA, ERK1/2 phosphorylation, and DBP-mediated disorders, indicating that the ROS/ERK1/2 pathway is responsible for DBP-mediated liver and kidney damage.
After DBP exposure, liver cells with obvious edemas were found. The structure of hepatic cells including the hepatic cord, the hepatic sinusoid, and the central vein was changed. Kidney slices also showed signs of damage induced by DBP exposure. Extreme edemas of epithelial cells in the glomeruli were also observed. Glomerular capillary loops were lobulated, and the lumen of renal tubules became smaller. At this stage, the organs have impaired integrity and function. By contrast, administering VitE and/or pretreating DBP-treated mice with PD98059 tended to protect against injury to these two organs.
The visible tissue damage clearly showed the increased risk of liver and kidney dysfunction induced by DBP. ALT is mainly present in the cytoplasm of liver cells, and AST exists in the cytoplasm and mitochondria of hepatocytes. Normal cells do not release these enzymes into the blood due to the undamaged and intact cell membrane. When hepatocytes are severely damaged, leading to increased cell degeneration or cell membrane permeability, the ALT and AST contained in the hepatocyte will be released into the bloodstream, resulting in higher levels of ALT and AST in serum. The ratio of AST/ALT is a commonly used clinical metric to reflect hepatocyte damage. 24 In the case of cirrhosis of the liver in a person, the ratio can reach 1.44 with chronic active hepatitis, and the ratio is often higher than normal. An AST/ALT ratio >1 in serum indicates severe liver damage. 24 In general, ALB made by the liver acts as a nutrient cell and maintains intravascular osmotic pressure in the body. When the liver is damaged, the production of ALB is reduced, and the degree to which it decreases is in parallel with the severity of hepatitis. Studies have shown that ALB levels in serum decreased in patients with chronic and severe hepatitis and cirrhosis. 24,25 In our study, the AST/ALT ratio in the serum of the DBP group increased significantly, while the ALB levels decreased significantly, indicating that DBP can cause severe damage to mouse hepatocytes. But results from the DBP + VitE + PD98059 group showed that the AST/ALT ratio increased to a lesser extent, and the ALB level decreased more, compared with that in the DBP group.
CREA is filtered by the kidney and is hardly absorbed by the renal tubules. The concentration of CREA in serum is mainly dependent on glomerular filtration in the presence of a stable exogenous CREA intake. 26 Usually, CREA levels in serum accurately reflect any renal parenchyma damage with higher levels than normal indicating kidney damage. UREA is the main end product of mammalian protein metabolism, which is synthesized through the ornithine cycle in the liver and excreted mainly by the kidney. Because the molecular weight of UREA is small and it is very soluble, the concentration of UREA in cerebrospinal fluid, serous cavity effusion, saliva, and sweat is basically the same. The concentration of UREA in serum is mainly affected by renal function. A healthy kidney excretes UREA very efficiently, whereas a weak UREA excretion ability might indicate renal damage. According to our results, the CREA and UREA levels in serum of the DBP group increased sharply, suggesting that DBP can cause serious injury to renal cells of mice, but these values were significantly attenuated after the administration of VitE and pretreatment with PD98059.
The histopathological observations proved that these toxic reactions are associated with oxidative damage. In this article, we confirmed that DBP could be expected to dramatically increase ROS levels, resulting in further tissue damage through the overproduction of ROS. ROS damage to lipids, as shown by the significantly increased levels of MDA in the livers and kidneys of mice, may be responsible for the collapse of cells that we observed in the histological examinations. Moreover, MDA can lead to the loss of cellular homeostasis due to the changes in the cell membrane. 27 This probably explains the liver and kidney dysfunction. In addition, ROS, acting as upstream signaling molecules, have been reported to be involved in signal transduction pathways and can induce apoptosis or necrosis, which ultimately result in pathological changes and lead to organ dysfunction. 22,28 However, treatment with VitE exerted antioxidant effects as evidenced by the downregulation of ROS and MDA, and even when PD98059 was added in the DBP group, the values of ROS and MDA were significantly lower. This is because PD98059 can decrease ROS production by inhibiting glutamate toxicity. 29 Our results show that ROS is involved in the liver and kidney dysfunction induced by DBP, and both VitE and PD98059 play a protective role by blocking the production of ROS.
The existing literature has documented that ERK1/2 levels are distributed in liver and kidney tissues. 30 Some studies have indicated that the overactivation of ERK1/2 can be found in liver cancer cases and that ERK1/2 pathway can also be involved in renal gluconeogenesis inhibition. 31,32 The ERK signaling pathway is a series of signal junctions and is also a downstream event of oxidative stress. Lee et al. 33 found that oxidative stress-induced apoptosis is mediated by ERK1/2 phosphorylation. Phosphorylation-activated ERK1/2 translocated from the cytoplasm to the nucleus mediates the activation of downstream transcription factors, such as nuclear factor kappa B and activator protein 1. These factors, in turn, regulate cell proliferation, differentiation, and apoptosis, leading to biological function changes. 34 It has recently been found that PAEs, acting as extracellular signals, can activate the p38 MAPK to cause corresponding posttranscriptional biological effects. 22 Other studies have also reported that ERK1/2 is activated by di-2-ethylhexyl phthalate (DEHP), a homologue of DBP, and is involved in DEHP-induced hepatotoxicity. 22 Thus, DBP-induced liver and kidney dysfunction is likely to be related to the activation of the ERK1/2 pathway. Our results showed that the overall levels of phosphorylated ERK1/2 are concomitant with the enhancement of oxidative stress based on the simultaneous increase seen in the DBP group. By contrast, after administering VitE and pretreating with PD98059, the phosphorylated ERK1/2 levels in liver and kidney tissues of mice exposed to DBP was significantly lower, and the degree of damage to the liver and kidney is also observed. This suggests that VitE and PD98059 can reduce the phosphorylated ERK1/2 levels to protect against damage of liver and kidney tissues.
Although the activation of PPARs mediated by PAEs has been recognized as a mechanism of hepatic and renal toxicity caused by PAEs, there has been a lack of awareness of PAE-induced human liver and kidney damage in the past. 12 In fact, the concentrations of PAEs and their metabolites detected in vivo were much lower than those required to activate PPARs in vitro, suggesting that PAEs-induced hepatic and renal toxicity in humans might not be mediated by PPARs. Instead, the nondependent regulatory mechanism of PPARs may take part in this process. In view of this, we conclude that the oxidative stress and the ERK1/2 pathways are involved in DBP-induced liver and kidney dysfunction. Moreover, the participatory mechanism of cellular signaling pathways on PAE-induced hepatic and renal toxicity in humans remains to be further and comprehensively studied.
Conclusions
This work exhibits that DBP induces liver and kidney damage via ROS/ERK1/2 pathway in mice and that the ERK inhibitor PD98059 combined with the antioxidant VitE reduced the DBP-mediated disorders. We also found that DBP exposure resulted in overproduction of ROS, which leads to oxidative stress in liver and kidney cells or tissues. Oxidative stress, in turn, activates the ERK1/2 pathway, which causes further liver and kidney dysfunction.
Footnotes
Author contributions
LC and JL contributed equally to this work.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of following financial support for the research, authorship, and/or publication of this article. This research was supported by the Science and Technology Research Project of the Education Department of Hubei Province (B2017182).
