Abstract
The cardiotoxic adverse effect of doxorubicin (DOX) is the major factor limiting its use. Recently, mesenchymal stem cells (MSCs) have been implicated in the preclinical studies of treatment of DOX-induced cardiotoxicity. The question is MSCs pretreated with platelet-rich plasma (PRP) have a better influence on DOX-induced cardiotoxicity compared to the influence of MSCs alone. Twenty-four Wistar rats were categorized into control, DOX-treated, MSC-treated, and PRP/MSC-treated groups. DOX was injected for two consecutive weeks. Light microscopic, biochemical markers (interleukin 10 (IL-10), tumor necrosis factor alpha (TNF-α), and creatine kinase-MB (CK-MB)), immunohistochemical (Bax, Bcl2, vascular endothelial growth factor (VEGF), and cardiac troponin-I (CT-I)), and oxidative/antioxidative markers (malondialdehyde (MDA)/superoxide dismutase (SOD)) were measured. Degenerative cardiac changes were detected in the DOX-treated group with complete loss of the architecture and coagulative necrosis. These changes were accompanied with the elevation of the serum level of CK-MB and loss of CT-I immunoreactivity. The major factors in the DOX-induced cardiotoxicity were the oxidative stress (elevated MDA/decreased SOD), inflammation (elevated TNF-α/decreased IL-10), and cardiac apoptosis (lower Bcl2, higher Bax, and lower Bcl2/Bax ratio). MSCs and PRP/MSCs attenuate DOX-induced cardiotoxicity. Better attenuation was observed in the PRP/MSC-treated group. PRP/MSC combination reduced greatly the MDA and TNF-α and increased IL-10, Bcl2/Bax ratio, and VEGF. PRP had no significant influence over the Bcl2, Bax, and SOD. In conclusion, DOX in its toxic dose induced myocardial injury. This destructive effect is related to oxidative stress, inflammation, and cardiac apoptosis. PRP/MSC possesses a better attenuation over the DOX-induced toxicity compared to MSC alone.
Introduction
Doxorubicin (DOX), an anthracycline antibiotic, is an effective antitumor drug used in the treatment of human malignancies. 1 It is commonly used in the treatment of leukemia, lymphoma, osteosarcomas, neuroblastoma, hepatoblastoma, breast cancer, Wilm’s tumor, and the tumors of the bile duct, endometrial tissue, and esophagus. 2 Its cardiotoxic adverse effect is the major factor limiting its use. 3 The exact mechanisms responsible for the DOX-induced cardiotoxicity are unidentified; oxidative stress appeared as a major factor in such toxicity. 4 Cardiac inflammation, mitochondrial dysregulation, and apoptosis also contribute to DOX-induced cardiotoxicity. 5,6
Two main strategies are used to reduce DOX-induced cardiotoxicity: structural modification and drug combination. 7 The former involves the synthesis of DOX analogs or preparation of novel DOX formulations and the latter involves the use of cardioprotective agents. 7 Preclinical studies have evaluated several cardioprotective adjuvants: dexrazoxane, mesna, β-receptor blockers, angiotensin receptor blockers, amifostine, leucovorin, and erythropoietin. 8 However, a few were approved in clinical trials, and much fewer were approved for clinical use. 7
Recently, mesenchymal stem cells (MSCs) have been implicated in the preclinical studies of treatment of DOX-induced cardiotoxicity. 9,10 Stem cells are clonogenic, self-renewing, and multipotent cells. 11 The sources of cardiac-specific stem cells include embryonic stem cells, bone marrow, or other tissues derived adult stem cells (hematopoietic stem cells and MSCs). 11,12 MSCs have several advantages: transdifferentiate into cardiomyocytes, easy to grow, capable to suppress immune rejection, and limit the inflammatory response. 13
Platelet-rich plasma (PRP) is a fraction of plasma with a higher number of platelets compared to the whole blood. 14 The potential therapeutic effect of PRP is due to various cytokines, growth factors such as platelet derived growth factors, transforming growth factors β1, insulin-like growth factors, platelet factor 4, fibroblast growth factor 2, and vascular endothelial growth factor (VEGF). 15,16 Those cytokines and growth factors recruit resident stem cells to the site of injury, where they are stimulated to secrete additional growth factors and anti-inflammatory cytokines. 17 In addition, growth factors stimulate cell proliferation and increase differentiation and extracellular matrix deposition. PRP plays a role in the enhancement of angiogenesis and stem cell proliferation. In addition, PRP helps in cell differentiation. 18 Platelets have also been found to have anti-inflammatory activity via inhibition of the nuclear factor-kB pathway. 19 Platelets circulate in an activated state in many disorders and form complexes with other inflammatory and immune cells. 20 The release rate of activated platelets applied topically to the inflamed cartilage in arthritis or skin ulcers in scleroderma may suppress the inflammation and facilitate tissue repair. 20
The aim of the present work was to compare the effect of MSCs pretreated with PRP versus the effect of MSCs alone on DOX-induced cardiotoxicity in the adult male albino rat. Their possible mechanisms of cardioprotection were also studied.
Materials and methods
Animals
Wistar male albino rats were obtained from the Animal House, Faculty of Medicine, Cairo University. Male rats were only used to avoid any potential sex differences if any.
At the beginning of the study, the rats’ weight was 203 ± 20 g. The rats were housed in 12-h dark and 12-h light manner with food and water available ad libitum. The general toxicological data were recorded including the motility, food and water consumption, and health status. The body weight of each rat was recorded at the beginning and at the end of the experiment. The study was approved by the Ethics Committee, Faculty of Medicine, Cairo University (534/2018). The study was performed according to the ethical standards of the National Institutes of Health guide for the care and use of Laboratory Animals (8th edition, revised in 2011).
Preparation of PRP
A 10-mL volume of autologous blood was obtained from the tail vein of the six rats that comprised the PRP/MSC-treated group. The blood was drawn into vacuum tubes containing 10% sodium citrate. The PRP was prepared according to a double centrifugation protocol. 21 During the first centrifugation, the tubes were centrifuged at 160 G for 20 min at room temperature (RT) resulting in two constituents, serum component (SEC) in the upper fraction and blood cell component (BCC) in the lower fraction. The blood cell elements were performed using a laboratory centrifuge (Beckman Centrifuge; Beckman Coulter, Brea, California, USA). During the second centrifugation, a mark was made 6 mm below the line that separated the BCC from the SEC. All content above this point was pipetted and transferred to another 5-mL vacuum tube without anticoagulant to increase the total amount of platelets collected for the second centrifugation. Then, the sample was centrifuged again at 400 G for 15 min resulting in two constituents: SEC and PRP. The PRP was separated from the SEC. The PRP was kept without packed cells at RT for no longer than 6 h. 22
Isolation, propagation, and identification of bone marrow-derived MSCs from rats
Rat bone marrow-derived MSCs (BM-MSCs) were harvested from 12 2- to 4-month-old (200–250 g) male Wistar rats. The rats were killed after the administration of sodium pentobarbital intraperitoneally at a dose of 30 mg/kg. The femora and tibiae were carefully cleaned. The tip of each bone was cut with a bone cutter. Bone marrow cells were flushed from the medullary cavities of the femora and tibiae and disaggregated into a single-cell suspension by sequential passage through a 23-gauge needle. This single cell suspension was cultured in 15 mL of MSC-specific medium; minimum essential medium supplemented with 10% fetal bovine serum and penicillin/streptomycin in T-75 culture flasks at 37°C in 5% carbon dioxide incubator. Three to 4 days later, non-adherent cells were removed by changing the medium. After 10 days in culture, adherent cells formed homogenous fibroblast-like colonies. When MSCs became confluent (80–90%), adherent cells were passaged with trypsin (0.25%) by incubating for 10 min. Three passages were measured to obtain the optimal number of MSCs before transplantation. The de-adhered cells after trypsin treatment were collected in a 15-mL falcon tube and centrifuged at 480 × g for 5 min and these cells were counted and tested for viability by trypan blue. 13
Flow cytometry analysis of cell surface markers
The choice of immunophenotyping was established on the International Society for Cellular Therapy proposal. 23 Briefly, the adherent MSCs were characterized by fluorescence-activated cell sorting (FACS) with antibodies against CD34, CD45, CD90, and CD105 (Beckman Coulter, NE15106) and the non-adherent hematopoietic cells were washed off. The cells were incubated at RT for 30 min with monoclonal antibodies CD34, CD45, CD90, and CD105 labeled with fluorescein isothiocyanate and analyzed on the flow cytometer (FACS Caliber, BD Bioscience, San Jose, California, USA).
Test materials
DOX was obtained from Samarth Life Sciences Private Limited, Mumbai, Maharashtra, India. DOX was dissolved in sterile 0.9% saline solution and was injected 2.5 mg/kg intracardiac in six equal injections at 48 h intervals for two consecutive weeks to achieve a total dose of 15 mg/kg BW, which is well documented to achieve cardiotoxicity. 24
MSCs were injected into the rat tail vein as a single dose of 1 mL (106 cells).
MSCs pretreated PRP were injected into the rat tail vein two components separately: 1-mL MSCs and 0.5-mL PRP, respectively.
Compliance with ethical standard
The experimental design was in accordance with the guidelines of the Institutional Animal Care, Faculty of Medicine, Cairo University. The work was complied with the ARRIVE guidelines and was carried out in accordance with the U.K. Animals (Scientific Procedures) Act, 1986 and associated guidelines, EU Directive 2010/63/EU for animal experiments.
Experimental design
Twenty-four Wistar rats were randomly categorized into four groups: control group, DOX-treated group, MSC-treated group (the rats received BM-MSCs concomitant with DOX), and PRP/MSC-treated group (the rats received the BM-MSCs pretreated with PRP concomitant with DOX). The sample size was based according to the resource equation method. 25,26 All rats were killed on the 17th day.
Tissue sampling
At the end of the experiment, the rats were euthanized by putting each rat in a conical flask for a few minutes with a piece of cotton that was soaked in 1.9% ether. Blood samples were taken by cardiac puncture and allowed to clot. The serum samples were used for biochemical analyses.
The heart was immediately removed and washed in ice-cold glass slides, homogenized separately in 10 volumes (w/v) of 0.1-M phosphate buffer, pH 7.4, using a Polytron homogenizer (Merry Zhao Laizhou Keda Chemical Machinery Co., Ltd.) for 1 min. The homogenates were centrifuged at 4000 r/min for 20 min and refrigerated at 4°C. The supernatant were used for histochemical assay.
The heart was dissected and fixed immediately in 10% formalin saline. Paraffin blocks were prepared and four successive sections of 5 µm thickness were prepared from each specimen.
Light microscopic study
Three sections were obtained from each specimen. The sections were stained with hematoxylin and eosin (H&E) and Masson’s trichrome methods. The technique of Masson’s trichrome stain was done according to Sheehan and Hrapchak. 27 The stained sections were examined and photographed using a Canon digital camera (Canon, Tokyo, Japan) connected to IBM computer system. The evaluation of all section was carried out in a blinded manner by a pathologist who was unaware of treatment groups.
Immunohistochemical study
The immunohistochemical studies were carried out according to Wang et al. 28 The cardiac tissue samples from all the groups were fixed in 10% formalin for 48 h and the paraffin blocks were prepared. Each sample was cut into 5-μm thick sections and taken onto poly-lysine-coated slides, air-dried overnight at RT, incubated at 60°C for 20 min, dewaxed in xylene, and rehydrated using different descending concentrations of ethanol. Later, the samples were boiled for 10–20 min in antigen retrieval solution (0.1 M citric acid, 0.1 M sodium citrate buffer solution, pH 6), cooled at RT for 20 min, and washed twice in Phosphate-Buffered Saline (PBS).
The cardiac sections from each paraffin block were incubated with anti-rabbit Bax monoclonal antibody (E63), (ab32503), anti-rabbit Bcl2 monoclonal antibody (E17; ab32124), anti-VEGF receptor 1 antibody (Y103; ab32152), and anti-cardiac troponin-I (CT-I) antibody (1C11; ab47003; Abcam, Cambridge, UK) at 4°C overnight. The sections were maintained at RT for 30 min and rinsed with PBS (pH 7.4) three times, each for 5 min. After removal of PBS, the sections were incubated with anti-rabbit Immunoglobulin G (IgG) (ab205718) at 37°C for an hour and a half and rinsed three times with PBS (pH 7.4), each for 5 min.
Biochemical assay
Tumor necrosis factor alpha and interleukin 10: The serum level of the inflammatory markers tumor necrosis factor alpha (TNF-α) and interleukin 10 (IL-10) was assayed by the commercially enzyme-linked immunosorbent assay kits supplied by Biopsies, China, according to manufacturer instructions.
Creatine kinase-MB: The serum level of creatine kinase-MB (CK-MB; mg/dL), a marker enzyme for myocardial injury, was determined by the kit method supplied by Agappe Diagnostics Ltd, Kerala, India. The CK-MB was measured in the presence of an antibody to CK-M monomer. This antibody entirely inhibited the activity of CK-MM. The CK-MB activity values were derived based on the absorption of Nicotinamide adenine dinucleotide phosphate (NADPH) at 340 nm. 29
Histochemical assay (oxidative/antioxidative markers)
Protein concentration in the homogenates that were prepared as described in the following was assessed according to the Biuret method. 30 One milliliter of the homogenate was mixed with three of biuret reagent which gives a blue colored complex and incubated for 10 min at 37°C. Absorbance was taken at 540 nm.
Malondialdehyde, an oxidative stress marker, was measured according to Tipple and Rogers. 31 The cardiac specimen was homogenized in 1 mL of PBS, pH 7.0 with a micropestle in a microtube; 20% trichloric acid solution was added to the thyroid tissue homogenate to precipitate the protein and centrifuged; 0.8% thiobarbituric acid solution was added to the supernatants. After boiling for 10 min in a water bath, the absorbance was measured at 405 nm using a commercial kit (Biodiagnostic, Cairo, Egypt). The concentration of malondialdehyde (MDA) was calculated using the standard curve and expressed per milligram of protein.
Superoxide dismutase (SOD) activity in tissue homogenate was measured (using a commercial kit purchased from Biodiagnostic, Cairo, Egypt) according to Weydert and Cullen 32 by the inhibition of nitroblue tetrazolium reduction by O2-generated by the xanthine/xanthine oxidase system. One unit is the amount of SOD that inhibits the rate of formazan dye formation by 50%.
Morphometric measurements
Image analysis was performed using the software Leica Quin 500, Germany. It was used to measure the areas percent of collagen fibers in Masson’s trichrome stain as well as positive Bax and Bcl2 immunostaining reaction in a standard measuring frame using a magnification 400× by light microscopy transferred to the monitor’s screen. The area percent values for each group were obtained from five different fields from each slide.
Statistical analysis
Statistical analysis was performed using statistical package for the social sciences statistical software (SPSS) version 21.0 (IBM Corporation, Somers, New York, USA). The data were expressed as means ± standard deviation. All parameters were tested by Kolmogorov–Smirnov test for normality. Statistical evaluation was done using one-way analysis of variance followed by Bonferroni pairwise comparisons. Significance was considered when the p-value was ≤ 0.05. The percentage of increase or decrease (difference) of all study parameters were calculated per the following formula: Percentage of difference = (Mean difference value between two groups)/(Value of the compared group) × 100.
Results
The general toxicological profile and mortality
The DOX-treated rats looked sicker, weaker, and lethargic when compared with the other treated animals. The urine was pink colored on the day of DOX administration. Two rats died in this group (on the 12th and 13th days). No mortality was observed in the other groups (Table 1).
Comparison of the body weight among the different groups at the end of study.a

DOX: doxorubicin; MSC: mesenchymal stem cell; PRP: platelet-rich plasma; SD: standard deviation; BW: Body weight.
aThe mean weight ± SD at the beginning of the study was 203 ± 20 g, minimum 195 g, 210 g.
bStatistically significant.
At the beginning of the study, the rats’ weight was 203 ± 20 g. Weight loss was observed in the DOX-treated groups. At the end of the experiment, the mean body weight of the DOX-treated group was 43% lower than that of the control group. The weight of the MSC-treated and PRP/MSC-treated groups were 7 and 14.9% higher than the weight of the DOX-treated group. Compared to that of the MSC-treated group, the mean body weight of the PRP/MSC-treated group was 7% higher.
Characterization of the MSCs
The MSCs were CD90+, CD105+, while they were CD34− and CD45− (Figure 1).

Immunophenotyping of the BM-MSCs by flow cytometry. The cells were CD90+ and CD105+, while they were CD34− and CD45−. BM-MSC: bone marrow-derived mesenchymal stem cell.
Detection of the MSCs in the heart
The capability of injected stem cells to heal the heart depends upon the effective delivery of injected cells in the cardiac tissue. The stem cells were labeled with red fluorescent protein (Figure 2).

MSCs are labeled with red fluorescent protein (arrows). (a) Control rat; (b) DOX-treated rat, (c) MSC-treated rat, and (d) PRP/MSC-treated rat. Total magnification ×400. MSC: mesenchymal stem cell; DOX: doxorubicin; PRP: platelet-rich plasma.
The morphology of heart assessed by light microscopy
The structure of heart as revealed by H&E staining: The control group presented normal cardiac morphology (Figure 3(a)). Degenerative changes were detected in the DOX-treated group including cytoplasmic vacuolization (6/6), pyknotic cardiac nuclei (4/6), apoptotic myocardial cells (4/6), wavy cardiac muscle fibers (2/6), degenerated cardiac muscle fibers with complete loss of the architecture (2/6), and coagulative necrosis (1/6). The lumen of the arterioles were thickened with massive blood extravasation (3/6). Inflammatory cellular infiltrate was also observed (2/6; Figure 3(b) to (f)).

Cardiac muscle morphology in the control and DOX-treated groups. (a) Control rat: a transversely cut cardiac muscle fibers (*), a central vesicular nucleus (N), connective tissue septa (CT), and fibroblasts (F). (b) to (f) DOX-treated rats; (b): inflammatory cellular infiltrations (arrows) and coagulative necrosis (arrowheads). (c) Dilated congested blood vessels (bv), cytoplasmic vacuolations, apoptosis (*), and pyknotic cardiac nucleus (arrow). (d) Cytoplasmic vacuolations (arrows), thick lumen arteriole (Ar) with massive blood extravasation (*). (e) Wavy cardiac muscle fibers (w), degenerated cardiac muscle fibers (D), apoptosis (*), and thick lumen arteriole (Ar). (f) Completely loss of the myocardial architecture with marked degeneration (D), cytoplasmic vacuolations (arrowheads), apoptosis (*), and dilated congested arteriole with a thickened lumen (Ar). Hematoxylin and eosin staining. Scale bars 50 µm, total magnification ×400. DOX: doxorubicin.
The cardiac muscle morphology greatly improved in the MSC-treated and PRP/MSC-treated groups being better in the PRP/MSC-treated group. Small areas of cardiac degeneration and wavy cardiac muscle fibers were detected in the MSC-treated group (Figure 4(a) to (d)).

Cardiac muscle morphology in the MSC-treated and PRP/MSC-treated groups. (a) and (b) MSC-treated rats; normal cardiac area (N) and degenerated cardiac area (D). Not blood vessel (bv) and wavy cardiac muscle fibers (W). (c) and (d) PRP/MSC-treated rats; nearly normal cardiac muscle architecture. Not a small area of cardiac degeneration (D) and pyknotic cardiac nuclei (arrowheads). Hematoxylin and eosin staining. Scale bars 50 µm, total magnification ×400. MSC: mesenchymal stem cell; PRP: platelet-rich plasma.
The content of connective tissue fibers: Qualitative and quantitative study: Minimal distribution of collagen fibers was observed in the control group (Figure 5(a)). Increased contribution of collagen fibers was observed in the DOX-treated group (810% higher than that of the control group; Figure 5(b), Table 2). This increase was ameliorated in the MSC- and PRP/MSC-treated groups (52 and 68.9% lower than that of the DOX-treated group; Figure 5(c) and (d); Table 2). A nonsignificant difference was detected between the MSC- and PRP/MSC-treated groups (Table 2).

Collagen in the cardiac muscles of the different studied groups. (a) Minimal distribution of collagen fibers in a control rat. (b) Increase distribution of collagen fibers in a DOX-treated rat. (c) and (d) Minimal distribution of collagen fibers in the MSC-treated and PRP/MSC-treated rats. Masson’s trichrome staining, scale bars 50 µm, total magnification ×400. DOX: doxorubicin; MSC: mesenchymal stem cell; PRP: platelet-rich plasma.
Comparison of the Bcl2, Bax, Bcl2/Bax ratio, and area % of collagen fibers among the different studied groups.

PRP: platelet-rich plasma; DOX: doxorubicin; MSC: mesenchymal stem cell; SD: standard deviation.
aStatistically significant.
The immunoreactivity in the heart: Qualitative and quantitative study
The Bcl2 of the DOX-treated group was 69.2% lower than that of the control group. The Bcl2 of the MSC- and PRP/MSC-treated groups was 75 and 125% higher than that of the DOX-treated group. A nonsignificant difference was detected between the MSC- and PRP/MSC-treated groups (Figure 6(a) to (d); Table 2).

Bcl2 immunoreactivity (arrows) in the different studied groups. (a) The reaction in a control rat. (b) The reaction decreased in a DOX-treated rat. (c) and (d) Relative increase in the reaction in the MSC-treated and PRP/MSC-treated rats. The immunohistochemical visualization of Bcl2 was performed as described in “Materials and methods” section, scale bars 50 µm, magnification ×400. DOX: doxorubicin; MSC: mesenchymal stem cell; PRP: platelet-rich plasma.
The Bax of the DOX-treated group was 266% higher than that of the control group. The Bax of the MSC- and PRP/MSC-treated groups was 50 and 65.9% lower than that of the DOX-treated group. A nonsignificant difference was detected between the MSC- and PRP/MSC-treated groups (Figure 7(a) to (d); Table 2).

Bax immunoreactivity (arrows) in the different studied groups. (a) The reaction in a control rat. (b) The reaction increased in a DOX-treated rat. (c) and (d) The reaction in the MSC-treated and PRP/MSC-treated rats. The immunohistochemical visualization of Bcl2 was performed as described in “Materials and methods” section, scale bars 50 µm, magnification ×400. DOX: doxorubicin; MSC: mesenchymal stem cell; PRP: platelet-rich plasma.
The Bcl2/Bax ratio of the DOX-treated group was 90% lower than that of the control group. The ratio in the MSC- and PRP/MSC-treated groups was 200 and 500% higher than the ratio of the DOX-treated group. Compared to that of the MSC-treated group, the ratio of the PRP/MSC-treated group was 100% higher (Table 2).
The CT-I of the DOX-treated group was 43.4% lower than that of the control group. Weakly positive and negative staining areas detected in this group indicating the presence myocardial infarction (MI). The CT-I of the MSC- and PRP/MSC-treated groups was 44.5 and 57.6% higher than that of the DOX-treated group. A nonsignificant difference was detected between the MSC- and PRP/MSC-treated groups (Figure 8(a) to (d); Table 3).

CT-I immunostaining in the different studied groups. (a) A strong positive immunostaining (arrows) in a control rat. (b) A weak positive immunostaining (arrowheads) and negative staining (*) indicating the presence of MI and cellular damage in a DOX-treated rat. (c) Areas with weakly positive immunostaining (arrowheads) and areas with strongly positive immunostaining in an MSC-treated rat. (d) A strong positive immunostaining (arrows) in the PRP/MSC-treated rats. The immunohistochemical visualization of CT-I was performed as described in “Materials and methods,” section, scale bars 50 µm, magnification ×400. CT-I: cardiac troponin-I; MI: myocardial infarction; DOX: doxorubicin; MSC: mesenchymal stem cell; PRP: platelet-rich plasma.
Comparison of the VEGF and CT-I among the different studied groups.

VEGF: vascular endothelial growth factor; CT-I: cardiac troponin-I; DOX: doxorubicin; MSC: mesenchymal stem cell; PRP: platelet-rich plasma; SD: standard deviation.
aStatistically significant.
The immunostaining of VEGF was weakly positive in the control and DOX-treated groups. The reaction increased in the MSC-treated group. In the PRP/MSC-treated group, the reaction markedly elevated (37-, 20-, and 7.6-folds higher than the control, DOX, and MSC-treated groups; Figure 9(a) to (d); Table 3).

VEGF immunostaining reaction (arrows) in the different studied groups. (a) and (b) A weak positive immunostaining in the control and DOX-treated rats. (c) A moderate positive immunostaining in an MSC-treated rat. (d) A strong positive immunostaining in the PRP/MSC-treated rats. The immunohistochemical visualization of VEGF was performed as described in “Materials and methods,” scale bars 50 µm, magnification ×400. VEGF: vascular endothelial growth factor; PRP: platelet-rich plasma; MSC: mesenchymal stem cell. DOX: doxorubicin.
The biochemical results
The CK-MB of the DOX-treated group was 155% higher than that of the control group. The enzyme level of the MSC- and PRP/MSC-treated groups was 38.6 and 49.3% lower than the enzyme level of the DOX-treated group. Compared to that of the MSC-treated group, the enzyme level of the PRP/MSC-treated group was 17.4% lower (Table 4).
Comparison of the CK-MB, TNF-α, and IL-10 among the different groups.

CK-MB: creatine kinase-MB; TNF-α: tumor necrosis factor alpha; IL-10: interleukin 10; DOX: doxorubicin; MSC: mesenchymal stem cell; PRP: platelet-rich plasma; SD: standard deviation.
aStatistically significant.
The TNF-α of the DOX-treated group was 525% higher than that of the control group. The level in the MSC- and PRP/MSC-treated groups was 32.7 and 55.6% lower than that of the DOX-treated group. Compared to that of the MSC-treated group, the level of TNF-α of the PRP/MSC-treated group was 34% lower.
The IL-10 level of the DOX-treated group was 50.3% lower than that of the control group. The level in the MSC- and PRP/MSC-treated groups was 44 and 77.5% higher than the level of the DOX-treated group. Compared to that of the MSC-treated group, the IL-10 level of the PRP/MSC-treated group was 23.3% higher.
The histochemical results (oxidative/antioxidative markers)
The MDA of the DOX-treated group was 671% higher than that of the control group. The level in the MSC- and PRP/MSC-treated groups was 48.3 and 58.4% lower than that of the DOX-treated group. Compared to that of the MSC-treated group, the level of MDA of the PRP/MSC-treated group was 19.5% lower (Table 5).
Comparison of the tissue oxidative/antioxidative markers among the different groups.
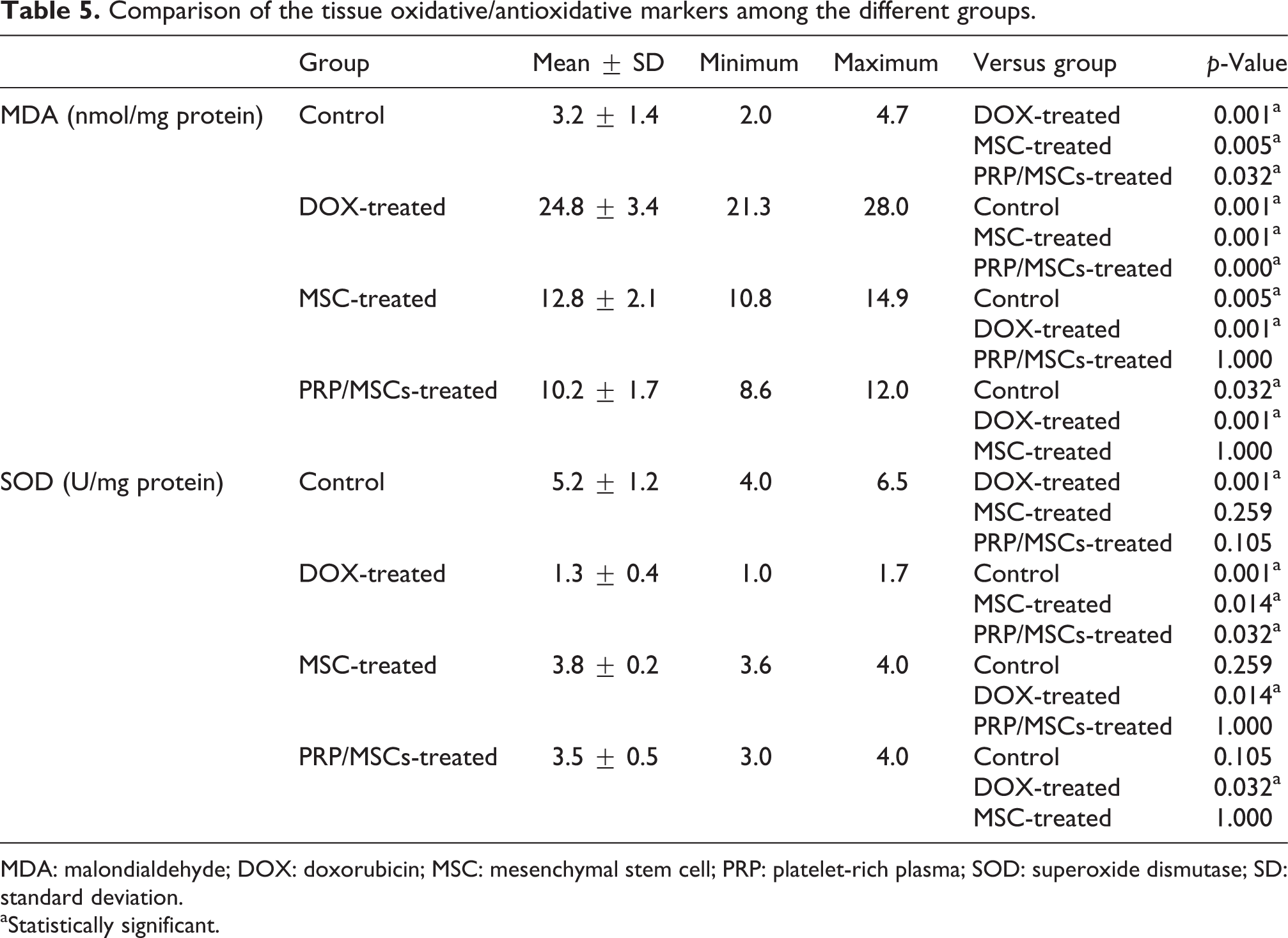
MDA: malondialdehyde; DOX: doxorubicin; MSC: mesenchymal stem cell; PRP: platelet-rich plasma; SOD: superoxide dismutase; SD: standard deviation.
aStatistically significant.
The SOD level of the DOX-treated group was 75% lower than that of the control group. The level in the MSC- and PRP/MSC-treated groups was 192 and 169% higher than the level of the DOX-treated group. A nonsignificant difference was observed between the MSC- and PRP/MSC-treated groups.
Discussion
The DOX in its toxic dose induced MI. The presence of coagulative necrosis with inflammatory cells in the DOX-treated group is a definitive microscopic finding of MI. 33,34 These observed changes were accompanied with the elevation of the serum level of CK-MB and loss of CT-I immunoreactivity. The CK-MB and CT-I are released by the damaged cardiac tissue. 7 The CT-I is more specific in diagnosing myocardial injury than that of CK-MB 35 and is used as a marker for myocardial injury because it is localized to and specific to myocardium. 33 The loss of CT-I immunoreactivity with the lack of cross-striations of the cardiac muscle fibers present in the DOX-treated group determined that areas of definite infarction consistently present. 36 The negative staining for CT-I indicates that CT-I has been released by the tissue and into the bloodstream, thus staining negative for its absence. 37 Both MSCs and PRP/MSCs ameliorate such cardiac injury. Better amelioration was observed in the PRP/MSC-treated group.
DOX administration was accompanied by a high mortality (2/6). Contrary, the other studied groups showed no mortalities. The existence of such high mortality mostly is based on the previously observed MI. The complete loss of the cardiac architecture may be the other possible cause. Many authors reported the existence of mortality with the use of DOX. 38,39
The oxidant marker MDA was elevated, while the antioxidant marker SOD was decreased in the all DOX-treated groups. Oxidative stress is defined as the shift in the balance between oxidants and antioxidants in the favor of oxidants. 40 Oxidative stress has been approved to be a major factor in the DOX-induced cardiotoxicity. 4,41,42 The heart is sensitive to oxidative damage due to plenty of mitochondria, the site of basal reactive oxygen species (ROS) generation, and the inherent low antioxidant defenses. 43 Much improvement of both markers was observed in the MSC- and PRP/MSC-treated groups. MSCs were reported to alleviate the elevated tissue oxidative stress by reducing the ROS-induced apoptosis and modifying the redox microenvironment by direct autocrine and paracrine effects. 44 Despite the presence of significant MDA difference between the MSC- and PRP/MSC-treated groups, a nonsignificant SOD difference was observed between both groups.
Inflammation contributes to DOX-induced cardiotoxicity. 5,6 To assess inflammation, we studied the TNF-α as a pro-inflammatory cytokine and IL-10 as an anti-inflammatory cytokine. Compared to the control group, the TNF-α level was highly elevated, while the IL-10 level decreased in the DOX-treated group. Much improvement of both markers was observed in the MSC- and PRP/MSC-treated groups; however, they were better in the PRP/MSC-treated group. The better anti-inflammatory finding in the PRP/MSC-treated group is attributed to the PRP being a rich source of growth factors, interleukins, and other cytokines. 45,46
Cardiac apoptosis also contributes to DOX-induced cardiotoxicity. 5,6 The framework of the apoptotic signal pathway appears to be several proapoptotic signals which eventually converge into a common mechanism driven by caspases. 47 The caspases mechanism is negatively regulated by the Bcl2 family. 48 The Bcl2 family is classified into three subfamilies: a subfamily including Bcl2, a subfamily including Bax and Bak, and a subfamily including Bik and Bid. 49 The Bcl2 exerts antiapoptotic activity, while Bax exerts proapoptotic activity. 49 The ratio between antiapoptotic and proapoptotic members of the Bcl-2 family defines the liability of the cell to apoptosis. 49 The lower Bcl2, the higher Bax with the lower Bcl2/Bax ratio in the DOX-treated group confirm the role of apoptosis in the DOX cardiotoxicity. The MSCs and PRP/MSCs ameliorate the apoptosis in the DOX cardiotoxicity through their reversal of the abovementioned parameters. It seems that the upper hand of such amelioration is related to the MSCs, as there was a nonsignificant difference of the Bcl2 and Bax when the MSC- and PRP/MSC-treated groups compared to each other. However, the Bcl2/Bax ratio was 100% higher in the PRP/MSC-treated group.
The attenuation effect of the PRP/MSCs upon the DOX-induced cardiotoxicity was superior to the attenuation effect of MSCs alone. Qureshi et al. identified a total of 1507 unique proteins in platelets upon analysis of 10 independent human samples. 50 The PRP is a pool of biologically active autologous components which can be contributors to tissue healing and regeneration. 51 In addition, many growth factors, including the VEGF, were found within the PRP. 52 The VEGF of the PRP/MSC-treated group was markedly elevated compared to the other groups. Such factor activates the PI3K/Akt signaling pathway 53 ; the crucial cell survival pathway in cardiomyocytes. 54
As documented in the literature, MSCs can affect numerous organs; however, no event of thrombosis or organ damage was detected during the study.
In conclusion, DOX in its toxic dose induced myocardial injury and MI. This destructive effect is related to oxidative stress, inflammation, and cardiac apoptosis. PRP/MSC combination possesses better attenuation over the DOX-induced toxicity compared to MSC alone.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
