Abstract
The use of doxorubicin (DOX) as an antitumor therapeutic agent is limited due to its cardiotoxic effects. Metformin (Met) and sitagliptin (Sitg) are suggested to improve cardiac function. The present study aimed to determine the potential protective effects of Met and Sitg on DOX-induced cardiotoxicity. Rats were divided into six groups: groups I, II, and III received normal saline, Met, and Sitg, respectively. Groups IV, V, and VI received DOX only, Met + DOX, and Sitg + DOX, respectively. Heart tissue was used for biochemical assays which measured cardiac reduced glutathione (GSH), thiobarbituric acid reactive substances (TBARS), and tumor necrosis factor α (TNF-α). Serum creatinine kinase (CK) and lactate dehydrogenase (LDH) were also measured. The heart apex was prepared for histological (hematoxylin and eosin) and immunohistochemical examination. Intoxication of DOX was associated with a significant elevation in serum CK-MB and LDH, reduction in cardiac GSH, and increased TBARS and TNF-α compared to the controls. Administration of Met or Sitg to DOX-intoxicated rats suppressed serum CK-MB and LDH. Moreover, cardiac GSH was elevated with decreased TBARS and TNF-α. These results were confirmed by histological study. Met and Sitg caused inhibition of caspase 3 and upregulation of B-cell lymphoma 2 (Bcl-2) expression in DOX-intoxicated animals. Sitg was found to exert a significantly better protective effect compared to that of Met. It was concluded that Sitg might be more effective than Met in reducing myocardial injury in DOX-induced cardiotoxicity in rats.
Introduction
Doxorubicin (DOX) is an anthracycline antibiotic commonly used in the treatment of several types of tumors. 1 It has been used as an antitumor agent since the 1950s with documented efficacy. Its mechanism of action involves intercalation into the DNA helix 2 and inhibition of the topoisomerase II enzyme activity, leading to its antitumor effect. 3 Nevertheless, the therapeutic use of the drug is limited by the severe cardiotoxicity after acute, as well as cumulative, dose of DOX. 4 Irreversible cardiomyopathy resulting from the chronic use of DOX may lead to congestive heart failure. 5 Many factors contribute to the development of DOX cardiotoxicity. The upregulation of reactive oxygen species (ROS) generation with myocyte apoptosis is considered to be a crucial cause of this toxic effect. 6 Therefore, the exploration of new therapeutic agents to allow safe dose escalation with limited cardiotoxic effects of DOX is warranted.
Metformin (Met) is a biguanide widely used as the first-line therapy for patients with type 2 diabetes mellitus (T2DM). 7 Met has beneficial effects on glucose and lipid metabolism. The antihyperglycemic effect of Met appears to limit cardiovascular death in patients with T2DM. 8 It has been suggested that Met also has a direct cardioprotective effect, independent of its glucose-lowering activity. 9
Sitagliptin (Sitg), a dipeptidyl peptidase 4 (DPP4) inhibitor, has been used in the treatment of T2DM. 10 It exerts its antidiabetic effect in the blood through the inhibition of glucagon-like peptide 1 (GLP-1) degradation by DPP4 enzyme. 11 Recently, GLP-1 analogs are shown to be beneficial during ischemia–reperfusion (I/R) injury in both animal models and in clinical studies through the limitation of infarct size, improvement in cardiac function, and enhancement of myocardial glucose uptake. 12,13 Evidence regarding the cardioprotective effects of DPP4 inhibitors is scarce and controversial. DPP4 inhibitors have been shown to attenuate the infarct size and improve the left ventricular function during myocardial I/R injury. 14
The aim of the present study was to determine the potential protective effects of Met and Sitg on experimentally DOX-induced cardiotoxicity in a nondiabetic rat model. The study was carried out using biochemical and histological methods.
Materials and methods
Chemicals
DOX (Adriblastina W) was obtained from Pharmacia (Milan, Italy). Met (1,1-dimethyl biguanide hydrochloride) was purchased from Sigma-Aldrich (St. Louis, Missouri, USA). Sitg Januvia® was purchased from Merck and Co., Inc. (Whitehouse Station, New Jersey, USA).
Animals and experimental design
The rats were housed in conditions in accordance with the Helsinki declaration for animals and care. The present study was conducted on 48 adult male Wistar albino rats weighing 160–200 g. The rats were obtained from the animal house of the Faculty of Medicine, University of Alexandria, Egypt. The animals were kept under controlled conditions, fed a standard rat chow diet, and provided free access to food and water. All experimental procedures were approved and performed in compliance with the guidelines of the Local Ethical Committee of the Faculty of Medicine, University of Alexandria.
After 1 week of acclimatization, 24 animals were randomly divided into 3 groups, with 8 rats in each group: group I—control nonintoxicated rats received normal saline orally; group II—nonintoxicated rats were treated with Met (500 mg/kg body weight), 15 and Met was dissolved in normal saline to a final concentration of 100 mg/ml; group III—nonintoxicated rats were treated with Sitg (10 mg/kg body weight), 16 and Sitg was dissolved in normal saline to a final concentration of 2 mg/ml. The prepared drug solutions and the vehicle were administered as 5 ml/kg. Both drugs and the vehicle were administered once daily via oral gavage for 7 days beginning on day 1 of the experiment.
Small dose-finding pilot studies with three animals for each dose were conducted for Met and Sitg. The drug dose that provided the best improvement in histological sections of the heart, after DOX intoxication, was chosen for each drug.
The remaining 24 rats were injected with DOX dissolved in normal saline (20 mg/kg intraperitoneally (i.p.)) divided equally on days 2 and 4 of the experiment. This dose is well documented to induce cardiotoxicity in rats.
17
DOX was dissolved to a final concentration of 4 mg/ml. The prepared solution was administered as 5 ml/kg. These rats were divided into three groups (
On the last day of the experiment, the animals were fasted for 8 h. On the eighth day, the rats were anesthetized, and blood samples were collected from the abdominal aortae of all animals, followed by separation of the sera. Blood glucose levels were measured using a blood glucose monitor (Accu-Checks, Roche, Germany), and the sera were used for the estimation of creatinine kinase-MB (CK-MB) and lactate dehydrogenase (LDH) activities.
The rats were then killed by decapitation. The chest of each animal was opened, and the heart was rapidly excised. The apex of the heart in each rat was obtained and prepared for histological and immunohistochemical examination. Apoptotic and antiapoptotic changes were evaluated by assessing the caspase 3 and B-cell lymphoma 2 (Bcl-2) using immunohistological examination. The remainder of the heart was homogenized (Biohom homogenizer) in 20 mM phosphate buffer (pH 7.4) containing 0.5 mM butylated hydroxytoluene to prevent sample oxidation. The homogenates were centrifuged at 3000 r/min at 4°C for 15 min. The supernatant of the homogenate was used for biochemical assays.
Biochemical studies
Estimation of CK-MB and LDH activities
CK-MB activity was estimated in the serum using a diagnostic kit (Sigma-Aldrich, St. Louis, Missouri, USA). The increase in absorbance at 340 nm was measured spectrophotometrically to calculate CK-MB level (U/l). LDH activity was detected by a colorimetric (450 nm) assay. Serum total LDH activity was calculated as units per liter. 18
Estimation of cardiac reduced glutathione
Reduced glutathione (GSH) was determined according to the method described by Ellman. 19 The procedure is based on the reaction of GSH with 5,5-dithiobis-2-nitrobenzoic acid, forming a product that has a maximal absorbance at 412 nm. The results are expressed as milligrams per gram tissue.
Estimation of cardiac thiobarbituric acid reactive substances
Cardiac homogenates were used for the determination of thiobarbituric acid reactive substances (TBARS), identified as the product of lipid peroxidation that reacts with thiobarbituric acid (TBA) to produce a pink-colored complex that can be measured spectrophotometrically at 532 nm, using tetramethoxypropane as a standard. 20 The concentration of TBA was calculated for each sample after reference to the standard curve and expressed as nanomoles per milligram protein.
Estimation of cardiac TNF-α level
TNF-α was measured by solid-phase sandwich enzyme-linked immunosorbent assay using two types of highly specific antibodies. Tetramethylbenzidine was used as chromogen. The strength of color measured at 450 nm was proportional to the quantities of rat TNF-α, which was expressed in picograms per milligram protein. 21
Estimation of total protein in cardiac homogenate
The total protein concentration was determined by the method of Lowry et al. 22 using bovine serum albumin as the standard.
Histological and immunohistochemical examination
Light microscopic study
Hematoxylin and Eosin stain
After the rats were killed, the heart apex, which is mostly formed by the left ventricle, was fixed in 10% formol saline and processed to get paraffin blocks. From these blocks, sections were cut and stained with hematoxylin and eosin (H&E) for microscopic examination. 23
Immunohistochemistry
Immunohistochemical procedures were conducted within the Pathology Department, Faculty of Medicine, Alexandria University.
Immunohistochemical staining for caspase 3
For detection of caspase 3-positive apoptotic cells in myocytes, paraffin sections were stained with rabbit anti-caspase 3
24
(1:1000 dilution; Fremont, California, USA). The poly-
The endogenous peroxidase activity was inactivated with 5% hydrogen peroxide in methanol for 15 min at 37°C. The slides were then placed in citrate buffer (pH 6.0) and microwaved for 25 min for antigen retrieval. Nonreactive staining was blocked using 1% horse serum in TBS (pH 7.4) for 3 min. Primary antibodies (rabbit anti-caspase 3) were applied overnight at 4°C, followed by incubation with biotinylated goat anti-rabbit immunoglobulin G (IgG) secondary antibodies (1:200 dilution; Biospa, Milano, Italy) and streptavidin peroxidase complex (Vectastain, Burlingame, California, USA). Bound antibodies were visualized using diaminobenzidine tetrahydrochloride solution (Biogenex, San Ramon, California, USA), followed by a hematoxylin counter stain. The positive reactions appeared brown in color.
Immunohistochemical staining for Bcl-2
A monoclonal mouse anti-human Bcl-2 protein was used as the primary antibody for Bcl-2 immunohistochemical staining. 25 Deparaffinized tissue sections were placed in a glass jar filled with citrate buffer, at a pH of 6.0. Intrinsic peroxidase activity was inhibited by 0.3% hydrogen peroxide in methanol for 30 min, and nonspecific binding was blocked with normal goat serum. The primary antibody (1:200 dilution; Fremont, California, USA) was applied overnight at 4°C, followed by incubation with secondary antibody. The secondary antibody, peroxidase-conjugated goat anti-mouse IgG (Fremont, California, USA), was applied to the sections at a dilution of 1:500 for 40 min at room temperature. Bound antibodies were visualized using diaminobenzidine tetrahydrochloride (DAB (3,3′-diaminobenzidine)) solution, followed by a hematoxylin counter stain. The positive reactions appeared brown in color.
Pictures were taken of five fields from each group. Images were viewed and recorded using an Olympus microscope (Tokyo, Japan)—equipped with a Spot digital camera, using MATLAB software (Image J, the
Statistical analysis
Statistical analyses were performed by Graph Pad InStat software (Version 3.02). Data are presented as the mean ± SD, and analysis was performed using two-factor analysis of variance followed by Bonferroni’s multiple comparison test.
Results
Biochemical results
The results of the present study showed that injection of 20 mg of DOX by i.p. (group IV) was associated with a significant elevation in serum CK-MB and LDH compared to the control group (group I; Table 1). Cardiac GSH significantly decreased in association with increased cardiac TBARS in comparison to the control group (Table 2). Cardiac TNF-α was also significantly elevated in comparison to the control group (Table 3).
Effect of Met and Sitg on serum indicators of cardiac injury (CK-MB and LDH) induced by doxorubicin cardiotoxicity in rats (

DOX: doxorubicin; CK-MB: serum creatine kinase MB levels; LDH: lactate dehydrogenase; Met: metformin; Sitg: sitagliptin; NC: control.
a
b
c
Effect of Met and Sitg on parameters of cardiac oxidative damage (GSH and TBARS) induced by doxorubicin cardiotoxicity in rats (

DOX: doxorubicin; GSH: reduced glutathione; TBARS: thiobarbituric acid reactive substances; Met: metformin; Sitg: sitagliptin; NC: control.
a
b
c
Effect of Met and Sitg on cardiac inflammatory parameter TNF-α induced by doxorubicin cardiotoxicity in rats (

DOX: doxorubicin; TNF-α: tumor necrosis factor α; Met: metformin; Sitg: sitagliptin; NC: control.
a
b
c
Administration of Met (group II) or Sitg (group III) to nonintoxicated rats demonstrated insignificant differences in regard to all of the tested parameters in comparison to saline-administered nonintoxicated rats (group I).
Table 1 demonstrates that administration of Met or Sitg to DOX-intoxicated rats significantly suppressed serum CK-MB and LDH. Cardiac GSH was significantly elevated with decreased cardiac TBARS (Table 2). Cardiac TNF-α was also significantly suppressed by treatment with either Met or Sitg compared to the DOX-intoxicated group (Table 3).
Sitg was found to exert a significantly greater suppression of serum CK-MB and LDH in DOX-intoxicated rats relative to Met treatment (Table 1). The same trend was also noted in regard to cardiac TBARS and TNF-α (Tables 2 and 3, respectively). Sitg treatment also induced a significantly higher increase in cardiac GSH in intoxicated rats in comparison to Met treatment (Table 2).
Regarding blood glucose levels, the results of the present study demonstrated that the differences in the blood glucose levels among the tested groups were not statistically significant (Table 4).
Effect of Met and Sitg on blood glucose levels in rats (

DOX: doxorubicin; Met: metformin; Sitg: sitagliptin; NC: control.
Histological results
The heart sections of the rats treated with Met or Sitg (group II and group III, respectively) were practically indistinguishable from the sections of normal, nonintoxicated rats that received saline (group I). Examination revealed classical histological pattern of cardiac muscle fibers. The cardiac muscle fibers were running in different directions and exhibited pale eosinophilic sarcoplasm. The fibers showed oval, vesicular, centrally located nuclei, and faint striations. The interstitial spaces were observed as minimal slit-like areas between the branching and anastomosing fibers (Figure 1(a) and (b)).
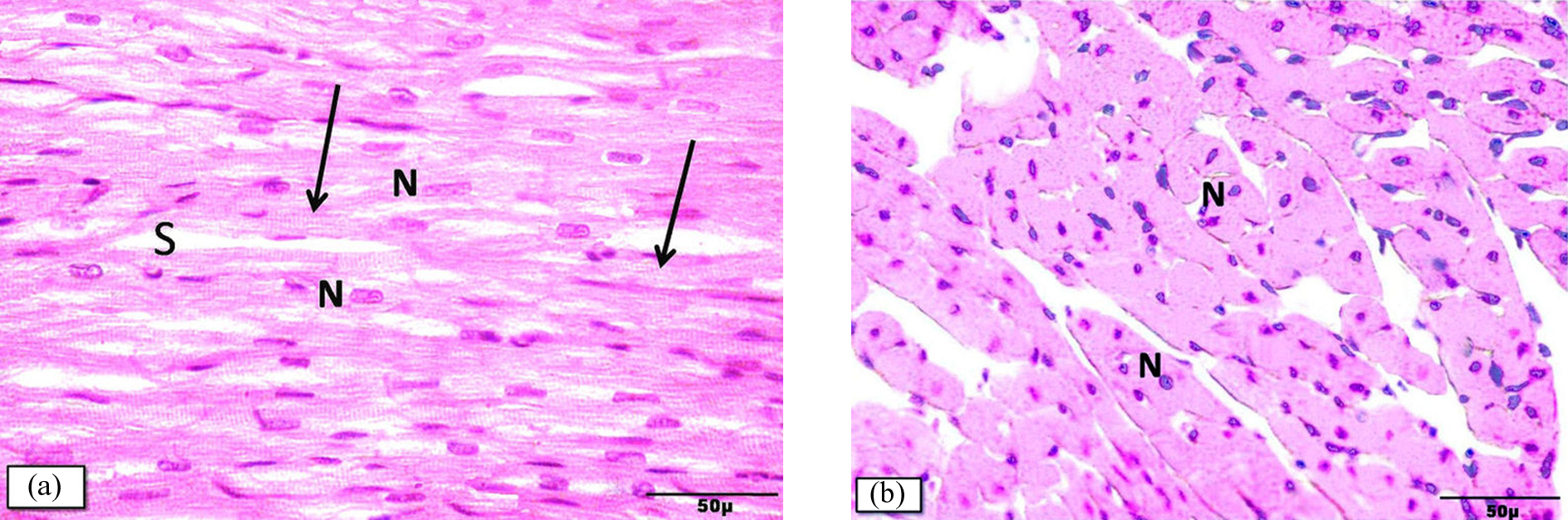
Cardiac muscle fibers of groups I, II, and III showing (
The heart sections of DOX-intoxicated rats showed histological changes in muscle fibers, with areas of degeneration and interrupted muscle fibers, wide interstitial spaces (Figure 2(a) and (b)), and interstitial (Figure 2(c)) and perivascular (Figure 2(a) and (c)) inflammatory cellular infiltration. Some muscle fibers appeared with vacuolated sarcoplasm (Figure 3(a)), while others showed hypereosinophilia (Figure 3(b)). Some of the nuclei of cardiac muscle fibers were dark and small (Figures 2(b) and 3(a) and (b)).
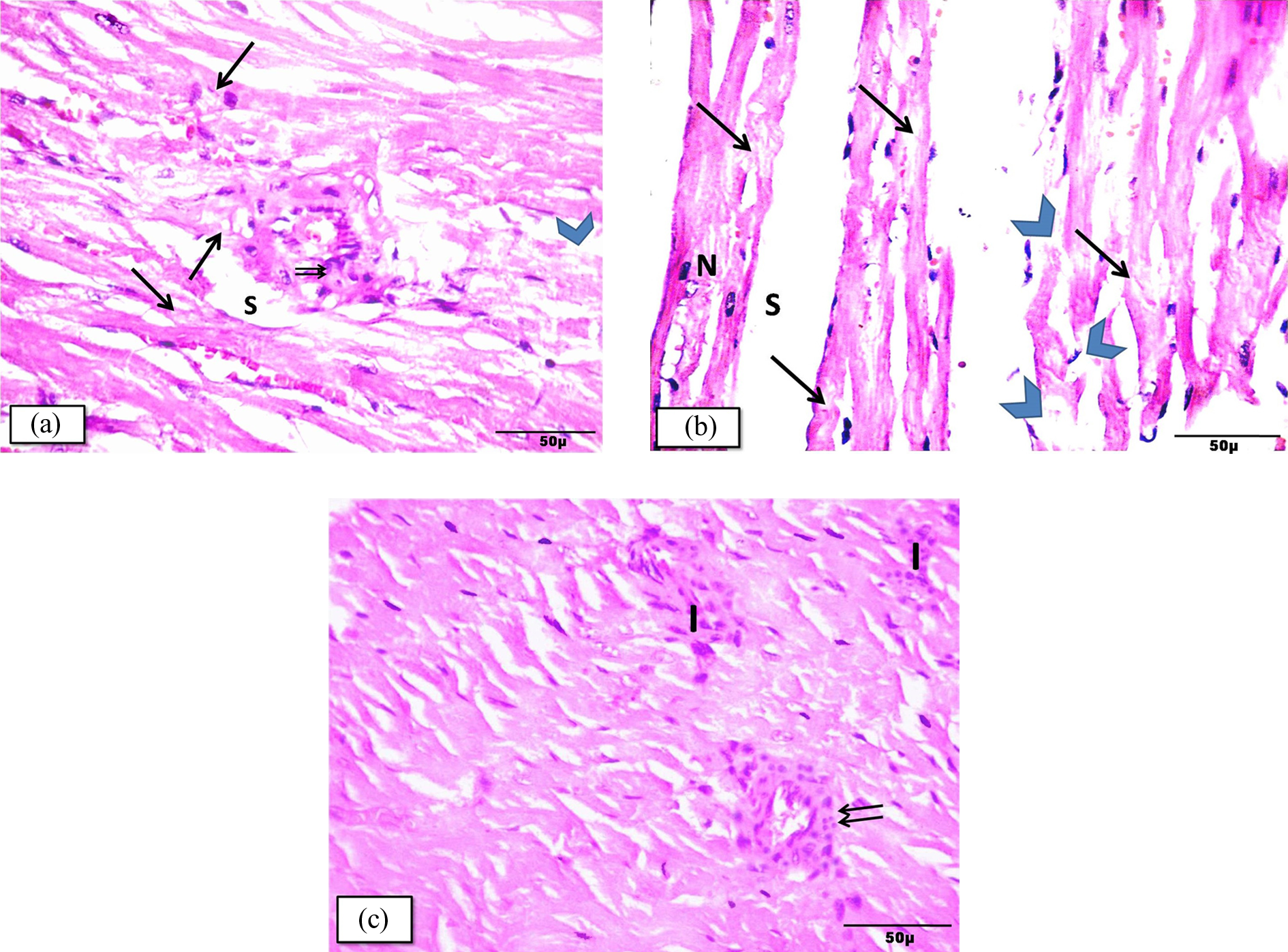
Photomicrographs of cardiac muscle fibers of group IV (DOX) illustrating (a) and (b) focal areas of myocytic degeneration (arrow) and interrupted muscle fibers (arrow head). Some fibers exhibit deeply stained nuclei (N) in (b). Wide interstitial spaces (S) and perivascular cellular infiltration (↑↑) were also observed. (c) Interstitial (I) and perivascular cellular infiltration (↑↑) are seen. H&E staining. Mic. Mag. ×400. H&E: hematoxylin and eosin.

Photomicrographs of the same group illustrating (a) obliquely and transversely cut muscle fibers with excessively vacuolated sarcoplasm (arrow head) and deeply stained nuclei (N). (b) Some myofibers show areas of hypereosinophilic sarcoplasm (arrow) and deeply stained nuclei (N). H&E stain. Mic. Mag. ×400. H&E: hematoxylin and eosin.
Treatment with Met (group V) ameliorated DOX-induced myocardial alterations, deduced by the observation of many fibers retaining their normal appearance (Figure 4(a)), less widening of interstitial space (Figure 4(b)), and moderate interstitial cellular infiltration (Figure 4(c)) was found. However, myocytic degeneration and deeply stained nuclei remained in localized areas (Figure 4(b)) and localized areas of sarcoplasmic hypereosinophilia are detected (Figure 4(c)). Notably, treatment with Sitg prevented the cardiotoxic effects of DOX as demonstrated by the nearly normal myocardial appearance (Figure 5(a)), although the foci of myocytic degeneration were still detected (Figure 5(b)).

Photomicrographs of the myocardium of group V (DOX + Met) showing (a) normal appearing myofibers with central oval nuclei (N) and narrow interstitial space (S). (b) and (c) Some fibers exhibit deeply stained nuclei (N) and focal areas of myocytic degeneration (arrow head) and others with hypereosinophilic sarcoplasm (arrow). Note the interstitial cellular infiltration (I) in (c). H&E stain. Mic. Mag. ×400. S: interstitial space; H&E: hematoxylin and eosin; DOX: doxorubicin; Met: metformin.

Photomicrographs of the myocardium of group VI (DOX + sitagliptin) revealing (a) nearly normal appearing muscle fibers with central oval nuclei (N) and narrow interstitial spaces (S). (b) Myofibers showing foci with myocytic degeneration (arrow head). H&E stain. Mic. Mag. ×400. H&E: hematoxylin and eosin; N: nucleus; DOX: doxorubicin; Met: metformin.
The current immunohistochemical study further examined effects of DOX-mediated caspase 3 activation on myocardial apoptosis and the changes in expression of Bcl-2. No significant differences in the expression of caspase 3 and Bcl-2 in groups I, II, and III were noted (Figures 6(a) and 7(a) and Table 5). Myocardial apoptosis was significantly increased in DOX-intoxicated rats (group IV) compared to the control, as shown by a significant increase in caspase 3 activity with intense immune reactivity brown-colored staining (Figure 6(b) and Table 5). DOX simultaneously suppressed the activity of Bcl-2 (Figure 7(b) and Table 5) compared with the rats in control group I.
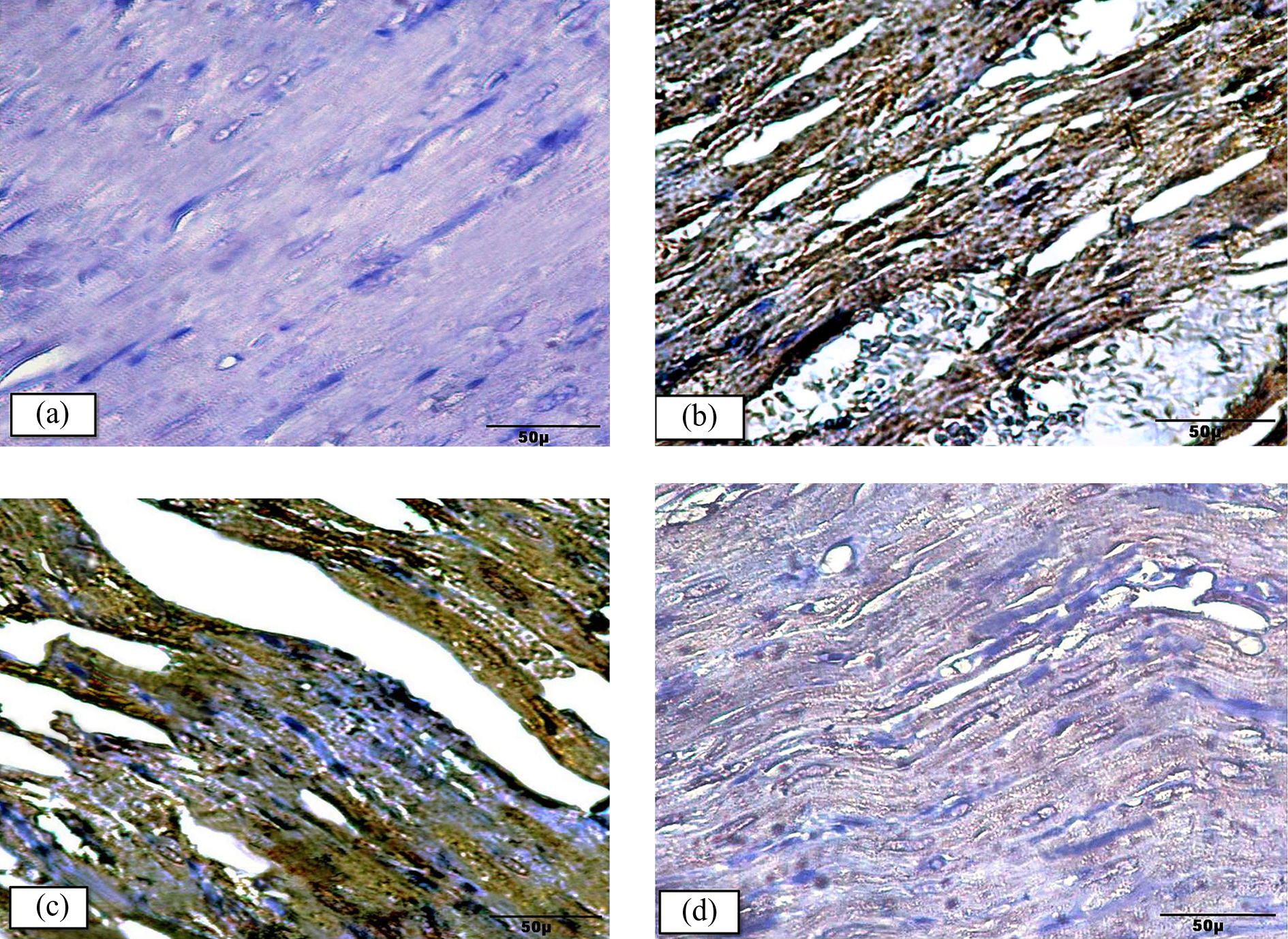
Representative photomicrographs demonstrating immunohistochemical expression of caspase 3 in rat myocardium: (a) Groups I, II, and III: There is no evidence of positive immunoreactivity. (b) Group IV (DOX) shows dark brown color of positive immunoreactivity in cardiac myocytes. (c) Group V (DOX + Met.): mild reduction in the immunoreactivity in the myocytes. (d) Group VI (DOX + sitg): marked reduction in immunoreactivity. Anti-caspase 3. Mic Mag. ×400. DOX: doxorubicin; Met: metformin; Sitg: sitagliptin.

Representative photomicrographs of immunohistochemical expression of Bcl-2 in rat myocardium demonstrating: (a) Groups I, II, III: no evidence of positive immunoreactivity. (b) Group IV (DOX): no evidence of positive immunoreactivity. (c) Group V (DOX + Met.): dark brown positive immunoreactivity. (d) Group VI (DOX + sitg): evident dark brown positive immunoreactivity. Anti Bcl2. Mic. Mag. ×400. DOX: doxorubicin; Met: metformin; Sitg: sitagliptin; Bcl-2: B-cell lymphoma 2.
Effect of Met and Sitg on apoptotic parameters (caspase 3 and Bcl-2) induced by doxorubicin cardiotoxicity in rats (
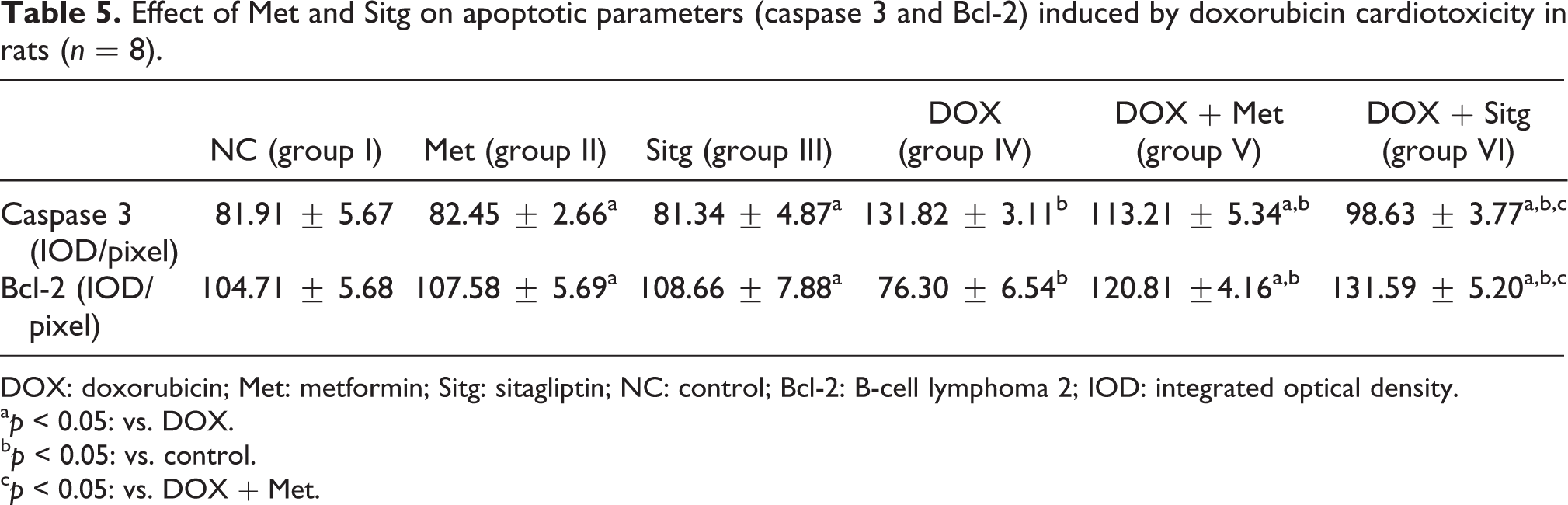
DOX: doxorubicin; Met: metformin; Sitg: sitagliptin; NC: control; Bcl-2: B-cell lymphoma 2; IOD: integrated optical density.
a
b
c
Treatment with Met (group V) or Sitg (group VI) significantly decreased caspase 3 activity (Figure 6(c) and (d), respectively). Bcl-2 activity in the same groups significantly increased in comparison to rats in the DOX-intoxicated group (Figure 7(c) and (d), respectively). Furthermore, it appears that Sitg treatment of DOX-intoxicated animals significantly improved myocardial apoptosis in comparison to Met treatment as shown in Table 5.
Discussion
DOX is an effective antineoplastic agent that is commonly used to treat various types of cancers such as ovarian, thyroid, gastric, and breast cancers. 1 However, its use has been restricted due to the dose-dependent cardiotoxicity that may proceed to irreversible heart failure. 5 A nondiabetic DOX intoxication rat model was used in the present study to explore the cardioprotective potential of Met and Sitg and to investigate the possible mechanisms of such an effect. The current study showed that injection of 20 mg of DOX was associated with acute cardiotoxicity as shown by a significant elevation in serum CK-MB and LDH.
It is generally accepted that measurement of the enzyme marker, LDH, alone is of little value in the assessment of myocardial injury. 26 Serum total CK activity and CK-MB concentration increased in parallel following myocardial injury. 27 Serum CK-MB is considerably more specific to myocardial damage than serum total CK. 28 The combined assessment of LDH and CK-MB may improve the tissue specificity of these markers. The significance of these biochemical parameters as indicators of cardiac injury in DOX cardiotoxicity has been previously proved by Boghdady. 29 The DOX-induced cardiotoxicity is further supported by the present histologic study, which showed massive changes in myocardial tissues with interstitial and perivascular inflammatory cellular infiltration and degenerative changes in the cardiac muscle fibers.
Although several mechanisms have been proposed to describe DOX-induced cardiotoxicity, these mechanisms are not fully understood. Among these mechanisms, generation of doxorubicinol metabolites, 30 mitochondrial disruption, alteration of cellular energy, and initiation of apoptosis cascades are some of the proposed mechanisms. Oxidative damage to cellular components is believed to be a major factor in DOX-induced toxicity. 6
The increase in oxidative stress may result from increased production of ROS, a relative deficit in the endogenous antioxidant defenses, or both. When antioxidant defenses are impaired or overcome, oxidative stress may produce lipid peroxidation. 31 In the present study, DOX increased the formation of cardiac lipid peroxidation, measured as TBARS, with depletion of cardiac GSH suggesting an exaggerated cardiac oxidative stress.
TNF is a cytokine generated by activated dendritic cells, T cells, and macrophages. TNF-α is a multifunctional cytokine that has been implicated in inflammation, cellular survival, growth, differentiation, and apoptosis. 32 A direct correlation has been found between serum TNF-α levels and the severity and progression of heart failure. 33
In the present study, increased cellular infiltration due to intoxication with DOX was associated with increased production of the proinflammatory cytokine TNF-α. The observation of DOX-induced TNF-α synthesis is consistent with previous studies. 34,35 Furthermore, TNF-α can increase myocardial oxidative stress through recruitment of leukocytes to inflammatory sites, with enhancement of ROS generation. 36,37 This may explain the increase in both TNF-α and oxidative stress in DOX-induced myocardial toxicity.
Extrinsic, intrinsic, and perforin pathways of apoptosis all end at a common point, leading to the final stage of programmed cell death. Caspase 3 functions as an essential effector that ultimately causes the morphological and biochemical changes observed in apoptotic cells.
38
The extrinsic pathway is activated by external signals, such as TNF-α.
39
ROS also activates caspase 3 together with the release of cytochrome
Although the proteolytic cascade of caspases represents a central point in the apoptotic response, its initiation is tightly regulated, primarily by Bcl-2 family proteins among other factors. 42 Bcl-2 proteins are involved in the control of apoptosis upstream of caspase 3 activation and of any other irreversible cellular damage. 43 Increased Bcl-2 expression was found to inhibit the apoptotic cascade. The downregulation of Bcl-2 expression observed in the current study left the apoptotic effects of caspase 3 unopposed and resulted in an exaggeration of the DOX toxic effects. These results are in agreement with the findings of Yang et al. 44
Novel strategies to prevent the development of cardiotoxicity and to increase the intrinsic tolerance of myocytes to DOX are needed. In light of what we know about DOX intoxication, we investigated the effects of Met and Sitg on DOX-induced cardiotoxicity in a rat model.
The results of the current study showed that Met treatment significantly reduces serum levels of LDH and CK-MB in DOX-intoxicated rats. This demonstrates that Met is capable of decreasing myocardial injury induced by DOX intoxication. These biochemical changes were associated with amelioration of DOX-induced myocardial injury observed by microscopic examination. Nevertheless, treatment with Met was not associated with complete improvement in the deleterious effects of DOX intoxication. The heart sections still showed wide interstitial spaces, moderate inflammatory cellular infiltration, and mild degenerative changes in myocardial muscles.
Met improved myocardial oxidative stress as shown by increased cardiac GSH level and decreased myocardial lipid peroxidation measured as TBARS. This is in agreement with the findings of Ashour et al. 15
DOX treatment also reduced inflammatory cellular infiltration and the formation of proinflammatory cytokines including TNF-α in heart tissues as shown in the present study. Arai et al. 45 also demonstrated that Met attenuates inflammatory responses, at least in part, by suppressing the production of TNF.
In the present study, Met treatment reduced expression of caspase 3 in heart sections from DOX-intoxicated rats. This may be explained by the decreased expression of TNF-α and the amelioration of oxidative stress by Met treatment. Similarly, Met was found to inhibit the expression of caspase 3 in a gentamicin-induced apoptosis model. 46
The present data also showed increase in the expression of the antiapoptotic Bcl-2 marker by Met treatment of DOX-intoxicated rats. In accordance with the present study, Ullah et al. 47 reported an enhancement in the expression of antiapoptotic Bcl-2 by Met treatment.
The decreased apoptotic and improved antiapoptotic parameters suggest an antiapoptotic effect of Met in this rat model. Therefore, Met exerts cardioprotective effects that may be due to its direct beneficial effects on cellular and mitochondrial function.
Although Met was originally used as an antidiabetic agent, its cardioprotective effects are independent of its insulin-sensitizing effects because the results of the current study revealed insignificant alterations in the blood glucose levels during Met treatment.
DPP4 is the first step in the breakdown of native Glucagon-like peptide-1 (7–36) amide. Inhibition of DPP4 became an attractive therapeutic target in T2DM because such agents can prolong the insulinotropic effects of GLP-1 (7–36). 11
DPP4 also cleaves multiple peptides, many of which have been demonstrated to possess cardiovascular actions. 48
In the present study, the Sitg protective effects against DOX-induced myocardial injury were evidenced by decreased serum levels of CK-MB and LDH. This was supported by our histological results, which showed marked improvement in myocardial architectures, with the exception of focal degenerative changes. These results were in accordance with the previous findings of Chang et al. 49
The present data also demonstrate that Sitg possesses an antioxidant effect in DOX-intoxicated rats. This effect was documented by increased GSH production, together with the marked reduction in the expression of the lipid peroxidation marker TBARS.
Analysis of the results of the present study revealed a probable anti-inflammatory property for Sitg as indicated by a decrease in TNF-α production in DOX-intoxicated rats. This effect is almost similar to that exerted by linagliptin, another DPP-4 inhibitor, as reported by Kröller-Schön et al. 50 In the same line, anagliptin, another DPP-4 inhibitor, has been found to have the same effect on TNF-α production. 51
The present study also demonstrated that Sitg treatment of DOX-induced cardiotoxicity clearly showed antiapoptotic effects, including a significant decrease in caspase 3 expression and augmentation of Bcl-2 expression. Previous studies have stated similar observations as a result of the immunohistochemical analyses in the present study. 50,52
Although these drugs were originally used as antidiabetic agents, their cardiprotective effects appear to not be related to their secretagogue effects because the results of the current study revealed insignificant alterations in the blood glucose levels.
Comparison of the effects of Met and Sitg on DOX-induced cardiotoxicity revealed higher levels of protection by Sitg treatment in all of the parameters tested. Understanding whether this may be translated into clinical benefit for patients treated with DOX requires further analysis. However, our finding may pave the way for new strategies to increase the tolerance of patients to treatment with this important antineoplastic agent.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
