Abstract
A quantitative assessment of the genotoxicity of silver nanoparticles (AgNPs) ascribed to its transplacental transfer and tissue distribution in pregnant rats was carried out in this study. A single intravenous (i.v.) injection of AgNPs with a size range from 4.0 to 17.0 nm was administered to pregnant rats at a dose of 2 mg/kg b.w. on the 19th day of gestation. Five groups beside control, each of the five rats were euthanized after 10 min, 1, 6, 12, or 24 h, respectively. The accumulation of nanoparticles (NPs) in mother and fetal tissues was quantified by inductively coupled plasma optical emission spectroscopy, where the highest accumulation level was recorded in maternal blood (0.523 µg/ml) after 24 h of administration. AgNPs induced accumulation in spleen tissue higher than placenta and fetal tissue homogenates. The data showed significantly detected levels of 8-hydroxydeoxyguanosine in all collected samples from administered animals compared with untreated individuals. Level of 8-OHdG in amniotic fluid exhibited the greatest values followed by maternal spleen, kidneys, and liver, respectively. Investigation by transmission electron microscope showed that the transfer of AgNPs through placental wall caused indentation of nuclei, clumped chromatin, pyknotic nuclei, and focal necrotic areas, while AgNPs appeared mainly accumulated in the macrophages of the spleen. Therefore, the data assume that the genotoxicity studies of AgNPs must be recommended during a comprehensive assessment of the safety of novel types of NPs and nanomaterials. Additionally, exposure to AgNPs must be prevented or minimized during pregnancy or prenatal periods.
Introduction
Silver nanoparticles (AgNPs) are extensively used as active antimicrobial agents in cosmetic and hygienic supplies. 1 Additionally, AgNPs are used in medical tools that are placed in the body for long time. 2 Presently, it is used in dental materials 3 and is used in water purification 4 and biosensors. 5
In general, the antimicrobial activity of nanoparticles (NPs) is linked to their ability to modify the cellular permeability and yield reactive oxygen species (ROS). 6 Previously, it was recognized that AgNPs below 10 nm in size are able to infiltrate into cytoplasm and disturb cellular metabolism and constrain biochemical courses. 7 Human exposure to silver and silver-related composites mostly take place through different routes of exposure, including dermal, oral, and inhalation resulting in accumulation into secondary organ targets including liver, spleen, kidneys, and brain. 8 Also, the highest silver accumulation was shown previously to be in spleen and liver of mice administered 10, 40, and 100 nm AgNPs, irrespective of its coatings. 9 They postulated that the majority of the silver is generally filtered by the liver, while the remaining is distributed by the blood to organs which contain large numbers of macrophages such as spleen, liver, and lung that are responsible for elimination of foreign bodies from blood. This role of phagocytic cells in accumulation and clearance of NPs was also seen in other metal NPs such as gold nanoparticles (AuNPs). 10
It was also shown previously that AgNPs are able to induce oxidative stress and cause cell damage and apoptosis in human liver cells linked with the induction of apoptosis and increase of micronucleus formation and chromosomal damage. 11
The primary function of the placenta in all species is to promote selective gas exchange, transfer of immunoglobulins from mother to fetus, and secretion of hormones which are important for fetal growth and development besides transport of nutrients and waste products between mother and fetus. 12 It is well documented that the placenta is a highly susceptible target organ for drug- or chemical-induced adverse effects due to transplacental transfer processes. 13
As mentioned previously, NPs of most material are able to influence on infant’s organs as a result of their transfer via placenta or breast milk. 14 AgNPs were found significantly accumulated in mice fetus homogenates after intravenous (i.v.) administration. 15 Moreover, the impact of AgNPs on the proper functions of reproductive system had been verified in several bioassays. 16,17
As revealed previously, the free radicals and other ROS are constantly generated in vivo and cause oxidative damage to biomolecules such as nucleic acids, proteins, and lipids. Many of the engineered nanomaterials (ENMs) were found previously to cause chromosomal fragmentation, DNA strand breaks, oxidative DNA adducts, and alteration in gene expression profiles. 18,19 For example, titanium dioxide nanoparticles (TiO2NPs) showed genotoxic effects in mammalian cell system, 20 Copper oxide nanoparticles (CuONPs) induced DNA fragmentation, lipid peroxidation, and micronucleus formation in the same cells, 21 while zinc oxide nanoparticles (ZnONPs) exposure to cell line A549 showed the induction of DNA double strand breaks (DSBs) and increased intracellular ROS levels. 22
Several investigations documented that AgNPs have high affinity to induce genotoxic effects in different forms such as salmonella strains (OECD TG 471), 23 human peripheral blood cells, 24 mammalian cells, 25 human lung fibroblast cells (IMP-90), 26 and the Chinese hamster ovary cell line (CHO-K1). 27
Among numerous types of oxidative DNA damage, the formation of oxidized DNA base, 8-hydroxydeoxyguanosine (8-OHdG) was found previously induced during oxidative DNA damage-repair after AgNPs administration in vitro. 28 8-OHdG is an ubiquitous marker of oxidative stress. During the repair of DNA genotoxic damage in vivo by exonucleas, the resulting 8-OHdG is excreted into urine without further metabolism. 29
So, this study was designed to focus on the oxidative 8-OHdG formation by AgNPs attributing to their transplacental passage between mother and fetus and organ distribution after a single i.v. dose.
Material and methods
Chemicals
AgNPs colloids were obtained from Nano Lab, 6th of October City, Giza, Egypt, and suspended in 0.9% trisodium citrate. Nitric (HNO3; 69%) and hydrochloric acid (HCl; 30/34%) were supplied by S D Fine CHEM Limited (SDFEL) Company 315-317 T.V. Industrial Estate, 248 Worli Road, Mumbai-30, Maharashtra, India. Hydrogen peroxide (H2O2; 30%) was supplied by Central Drug House (P) Ltd., New Delhi-110002, India. Millique® ultrapure deionized water was used in all experimental procedures. Rat 8-OHdG competitive ELISA Kit (ER 1487; 96 T) was purchased from Bioassay Technology Lab: KORAIN BIOTECH Co. Ltd, China.
Characterization of AgNPs
The absorption optical spectra of AgNPs were recorded using UV-VIS spectrophotometer (LABOMED, UVS 2700, Los Angeles, CA, USA). All spectra were recorded in air at room temperature. On the other hand, the AgNPs in the 0.9% sodium citrate solution were filtered using filters coated with carbon, mounted on an electron microscope grid (200 mesh, Veco, Eerbeek, Holland), and visualized under a transmission electron microscope (JEOL, JEM-2100, 200KV, Japan). The diameters of AgNPs were measured at a magnification of 35,000×.
Experimental design
Healthy female Wister rats (average age 8 weeks old) were obtained from The Holding Company for Biological Products & Vaccines (VACSERA Co.), Giza- Egypt, allocated to plastic cages covered with metal grids with wood chips for bedding and allowed to acclimate for 2 weeks in the animal facility conditions of the Zoology Department, Faculty of Science, Tanta University, Egypt before divided into experimental groups. Target values for temperature and relative humidity were 22 ± 1°C and 55 ± 5% RH, respectively, and 12-h light/dark (L/D) cycle. The rats were given drinking water and normal experimental pelleted animal food ad libitum. Male rats obtained from the same animal provider were allowed to mate with females in the animal facility conditions. When pregnancy was confirmed by the presence of vaginal plug, the pregnant females were isolated from the other animals until experimentation and followed by the time-scheduled sacrifice. All animals were carefully observed daily and their body weights, food consumption, and water intakes were measured precisely every week from the day they were obtained, while body weights were measured daily after pregnancy to follow up any signs of toxicity or abnormality during the experiment. The Institutional Animal Care and Use Committee (IACUC) of the Faculty of Science, Tanta University approved the animal caring ethics (No.: IACUC-SCI-TU-003). The guidelines for animal care were followed according to the National Institutes of Health guide for the care and use of Laboratory animals (NIH Publications No. 8023, revised 1978).
Placental transfer study
Each pregnant rat was administered a single i.v. dose of 2 mg/kg b.w. which is equal to four times of the No Observed Adverse Effective Level (NOAEL) of AgNPs, on day 19 of gestation. 30 The females were euthanized after the following time intervals: 10 min, 1, 6, 12, and 24 h. At each time point, five treated animals and another five normal controls were euthanized to obtain desired samples from maternal blood and tissues such as liver, kidneys, spleen, placenta, besides fetuses, and amniotic fluids for AgNPs levels quantification, molecular, and electron microscopy (EM) detections. The samples were stored in −86°C until use.
AgNPs quantification
Silver (Ag+) metal was extracted from blood or tissues after 10 min, 1, 6, 12, 24 h using acid digestion process according to the method of Jimenez-Lamana et al. 31 with minor modifications using ultrasonic waves in a sonicator water bath instead of microwave digestion. Ag+ was measured in maternal blood, spleen, placenta, and fetal homogenate. One gram of each sample was digested with 5 ml of HNO3 in ultrasonic water bath until clear solution after adding 0.5 ml of HCl to precipitate Ag+. Two hundred microliters of H2O2 were added to improve the digestion process. The mixture was diluted, filtered, and completed to 30 m with Millipore ultrapure deionized H2O (Milli-Q® Merck Integral Water Purification System, Darmstadt, Germany).
Ag+ content was measured on inductively coupled plasma optical emission spectroscopy instrument (ICP-OES, optima 7000 Perkin Elmer, MA, USA). All measurements were performed on microwave plasma model 4200 MP-AES (Agilent, Santa Clara, CA, USA). An auto sampler was used to deliver samples into instrumental cyclonic spray chamber with mass flow controlled nebulizer gas flow as 0.65 L/min. The instrument was operated in a fast sequential mode and featured to cooled charged coupled device detector. Background and spectral interferences were easily corrected and accurately using PerkinElmer WinLab32™ software (MA, USA).
The limit of detection (LOD) of Ag was calculated as double the standard deviation of a series of measurements of a solution against the blank absorbance of 0.5 ppb. Working standards were used and quality assurance procedures and precautions were carried out to ensure reliability of the results. Samples were carefully handled to avoid contamination. A recovery experiment was carried out by spiked untreated tissues with 100 ng of AgNPs. The fortified samples were processed for analysis as described above. The average recoveries were 82.4% and 65.6% for blood and animal’s tissues. For placental transfer (%) was calculated as AgNPs concentration in fetus/applied dose × 100.
Electron microscopy
A complete histopathological examination was performed on spleen and placenta samples. Transmission electron microscopy (TEM) preparations were done for samples taken after 12 and 24 h of administration. After dissection of rats, the desired samples of the spleen and placenta were freshly kept in cold 4% phosphate-buffered glutaraldehyede (pH 7.4) for 24 h. For EM examination, the material was washed in Millonig’s phosphate buffer (pH 7.3) and then post fixed in 2% osmic acid in Millonig’s phosphate buffer at 4°C for 4 h. The specimens were then dehydrated in ascending series of ethyl alcohol and air dried. These specimens were blocked in epoxy resin and semi thin sections were stained by methylene blue. These sections were mounted on copper grids and viewed under electron microscope (JEM-2100, 200KV, Japan). The electron micrographs of selected areas of different regions were taken by an attached camera and enlarged as required.
8-OHdG quantification
Maternal blood, amniotic fluids, and tissue samples of treated females of 1, 6, 12, and 24 h time points were prepared after centrifugation at 2800–4500 × g for 20 min. In addition, tissues were homogenized with phosphate-buffered salinebuffer pH 7.4 and centrifuged as described above. Supernatant was subjected to ELISA protocol of rat 8-OHdG enzyme-linked immunosorbent assay kits, where an aliquot (40 µl) was added to microtiter plates which are pre-coated with 8-OHdG monoclonal antibodies, 10 µl 8-OHdG antibodies, and 50 µl of streptavidin-HRP. The plates were covered, shaken gently, and incubated at 37°C for 60 min. So, liquid was drained and the plates were washed five times with washing solution (30 s for each). Fifty microliters of chromogen solution A were added, followed by another volume of chromogen B, shaken gently and incubated for 10 min at 37°C. Then, 50 µl of stop solution was added and the intensity of the produced color was measured at 450 nm using a computer-controlled microplate reader (BioTek, 800 TS ELISA absorbance reader, VT, USA). Triplicate measurements were performed for each sample and the coefficient of variation was within 10% during analysis period. The standard curve was done versus the logarithmic of concentration. Finally, 8-OHdG levels were expressed as nanograms per milliliter or grams. The limits of quantification ranged from 0.05 to 20 ng/ml, while LOD was set as 0.027 ng/ml.
Statistical analysis
All data were presented as a means ± SE and subjected to analysis of variance. The statistical analysis was performed using the CoStat program. 32
Results
AgNPs characterization
Figure 1 represents the microstructural characterization of AgNPs. TEM images showed a majority of nearly spherical AgNPs with diameters ranging from 4.32 to 16.9 nm. These images correspond to a cross section of a polymeric matrix permeated with AgNPs. AgNPs formation was monitored by ultraviolet-visible (UV-VIS) spectrophotometer. The maximum absorbance was stated at λmax (420.0 nm) as illustrated in Figure 2 indicating Ag+.

Electron micrographs of TEM of AgNPs suspended in 0.9% trisodium citrate. Images were visualized on instrument (JEOL, JEM-2100, 200KV) at ×5000 and ×10000, respectively. TEM: transmission electron microscopy; AgNPs: silver nanoparticles.
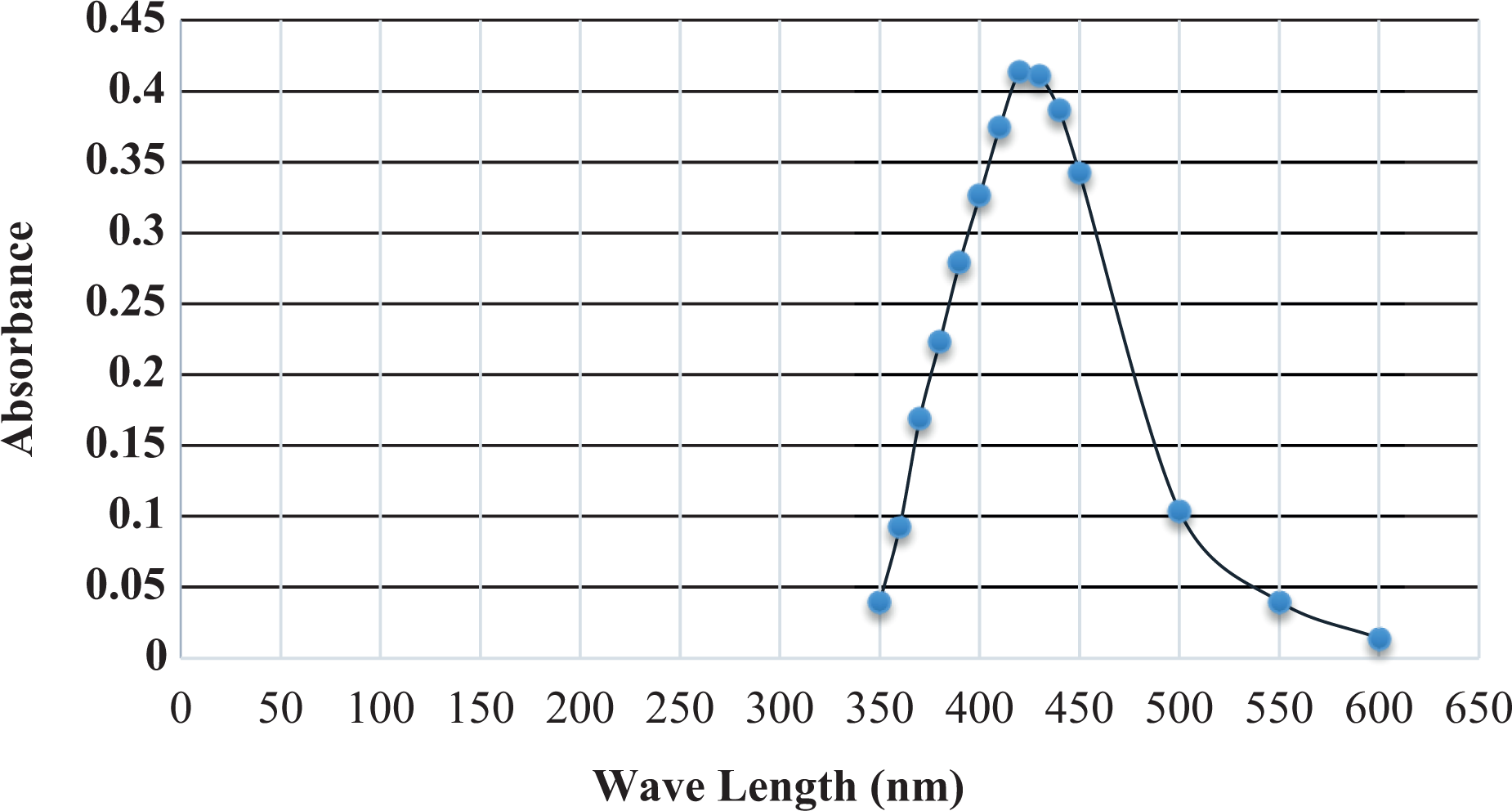
AgNPs examined on UV-VIS spectrometer against standard blank (Millique® ultrapure deionized water). Particles absorbance was maintained at range from 350 to 600 nm indicating silver. UV-VIS: ultraviolet-visible; AgNP: silver nanoparticle.
AgNPs accumulation
The data of NPs concentrations in maternal blood, fetuses, spleen as well as placenta after i.v. administration in pregnant rats are presented in Table 1. The accumulation levels of AgNPs in maternal blood accounted for 0.120 µg/ml after 10 min, while recorded the highest value (0.523 µg/ml) after 24 h of administration. However, AgNPs accumulation in maternal spleen tissues was greater than that in other tissues. The descending order of AgNPs accumulation in spleen was as follows: 1.625, 1.659, 0.079, 0.048, and 0.025 µg/g and in placenta was 0.32, 1.26, 0.29, 0.51, and 0.30 µg/g after 10 min, 1, 12, 6, and 24 h, respectively.
Tissue distribution of AgNPs in pregnant rats administered a single i.v. injection of AgNPs at different time intervals.a,b

ND: not detected; AgNP: silver nanoparticle; i.v.: intravenous.
aEach value represents the mean of five treated animals ± SE.
bPlacental transfer (%) = AgNPs concentration in fetus/applied dose × 100.
c,dDifferent letters indicate significant difference at p < 0.05 levels.
The data for the accumulation of AgNPs in the fetuses’ homogenates showed that the AgNPs levels were not detected in controls, then time dependently increased reaching a peak of 0.116 μg/g at the 6 h time point. Afterwards, it was declined to 0.043 μg/g after 12 h, then to 0.029 μg/g after 24 h. The placental transfer of AgNPs into fetus relative to the applied dose also revealed a peak at 6 h (0.031μg/g), then a sharp decline at 12 and 24 h (Table 1). As expected, the spleen exhibited the highest AgNPs accumulation levels at all the time points except after 24 h.
8-OHdG quantification
Levels of 8-OHdG in maternal blood and fetuses are presented in Table 2 and Figure 3. The data show significantly detected values in all samples after AgNPs administration. In maternal blood, AgNPs induced 8-OHdG formation at time points after administration. No significant differences were observed in DNA adduct levels after 1 and 12 h, while the highest induction was found after 24 h accounting for 16.80 ng/ml, and the least value was 2.73 ng/ml after 6 h. In case of fetal homogenate, the least value of 8-OHdG formation exhibited 1.96 ng/g after 12 h of administration. Other organs of pregnant rats showed different levels of 8-OHdG formation at different time points after i.v. administration (Figure 4). 8-OHdG levels in amniotic fluid recorded the highest values followed by maternal spleen, kidney, and liver, respectively. Moreover, the 8-OHdG induction reached the highest levels after 24 h followed by 1 h, while the least level was recorded after 6 h of administration compared with untreated group.
8-OHdG levels in pregnant rats at different time intervals after a single i.v. injection of AgNPs at time 0 h.a

ND: not detected; 8-OHdG: 8-hydroxydeoxyguanosine; i.v.: intravenous.
aEach value is the mean of three replicates ± SE.
b,c,dDifferent letters indicate significant difference at p < 0.05 levels.
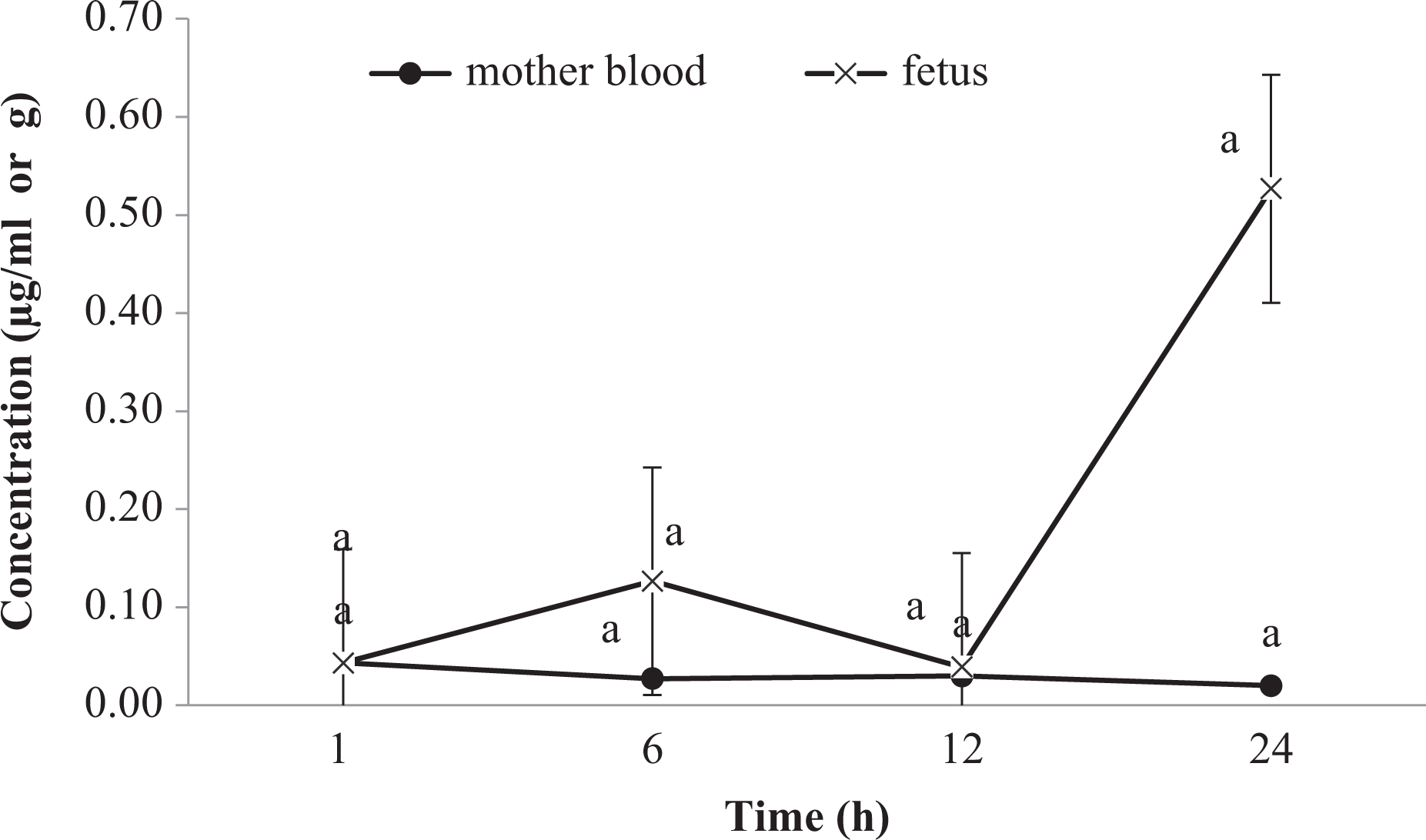
AgNP levels (µg/ml or g) in either maternal blood or fetus after i.v. administration of a single AgNP dose at different time intervals. Vertical bars indicate standard error (n = 5 replicates). Values with common superscripts are significantly different at p < 0.05. AgNP: silver nanoparticle; i.v.: intravenous.

The induction of 8-OHdG in different organs of pregnant rats after i.v. administration of a single AgNP dose. Vertical bars indicate standard error (n = 5 replicates). Values with common superscripts are significantly different at p < 0.05. AgNP: silver nanoparticle; i.v.: intravenous; 8-OHDG: 8-hydroxydeoxyguanosine.
Ultrastructure of spleen and placenta
Spleen
In rats treated with AgNPs, the spleen showed ultrastructural observation confined to increasing of intracellular spacing between the cells, destructed endoplasmic reticulum which formed rounded shapes besides appearance of increased numbers of lysosomal and multivesicular bodies with electron dense precipitates inside its matrix as compared with control group. Additionally, AgNPs caused obvious damage to the cells forming cytoplasmic vacuolization. Macrophages filled with AgNPs were also observed, while spleen sections of untreated group, showed normal components (Figure 5). The ultrastructural changes were more evident in the spleen of the 24 h time point than that of the 12 h time point.
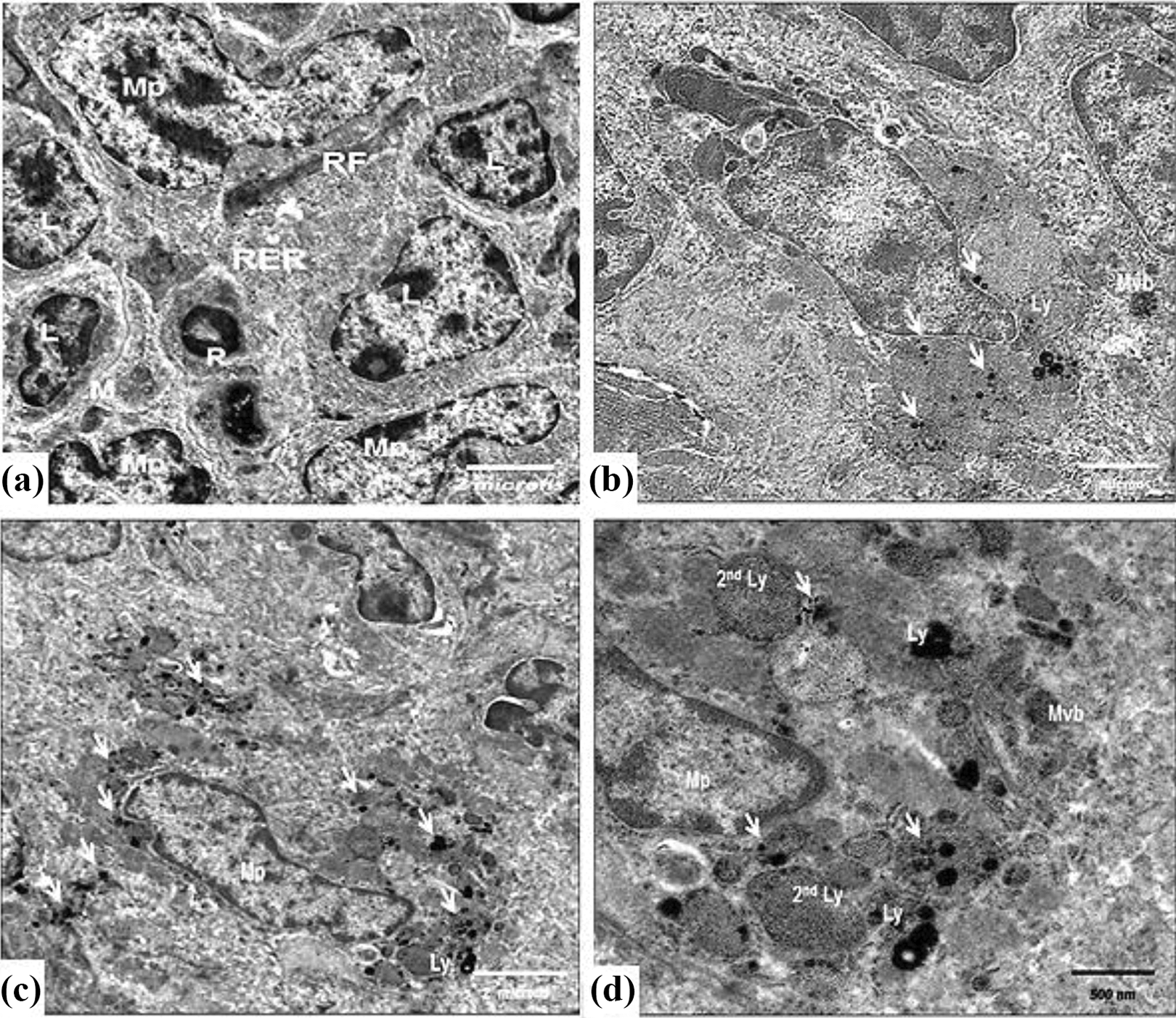
Electron micrographs of rat spleen: (a) Control white pulp with numerous normal lymphocytes (L), macrophage (Mp), reticulocytes (R), reticular fibers (RF), rough endoplasmic reticulum (RER), and mitochondria (M), ×8780; (b) a spleen from a rat treated with AgNPs after 12 h showing macrophages filled with electron dense AgNPs (arrows), lysosomal bodies (Ly), and multivesicular body (Mvb) with electron dense particles, ×8780; (c) a spleen from a rat treated with AgNPs after 24 h showing macrophages with aggregate of electron dense AgNPs (arrows) and lysosome (Ly), ×8780; (d) a magnified portion of (c). Note rounded shaped RER and destructed mitochondria, ×14600. AgNP: silver nanoparticle.
Placenta
To further demonstrate the tissue distribution of AgNPs, its effect was examined in the placental tissues by TEM after 12 and 24 h of administration. AgNPs were transferred through the placental barriers from mother’s blood to fetus, so they are clearly observed in placental cells particularly the trophoblasts during ultrastructure investigation. The transfer of AgNPs caused obvious focal necrotic areas, apoptotic changes as indicated by clumped chromatin, indentation of nuclear membranes, round shaped endoplasmic reticulum, and destructed mitochondria (Figure 6).
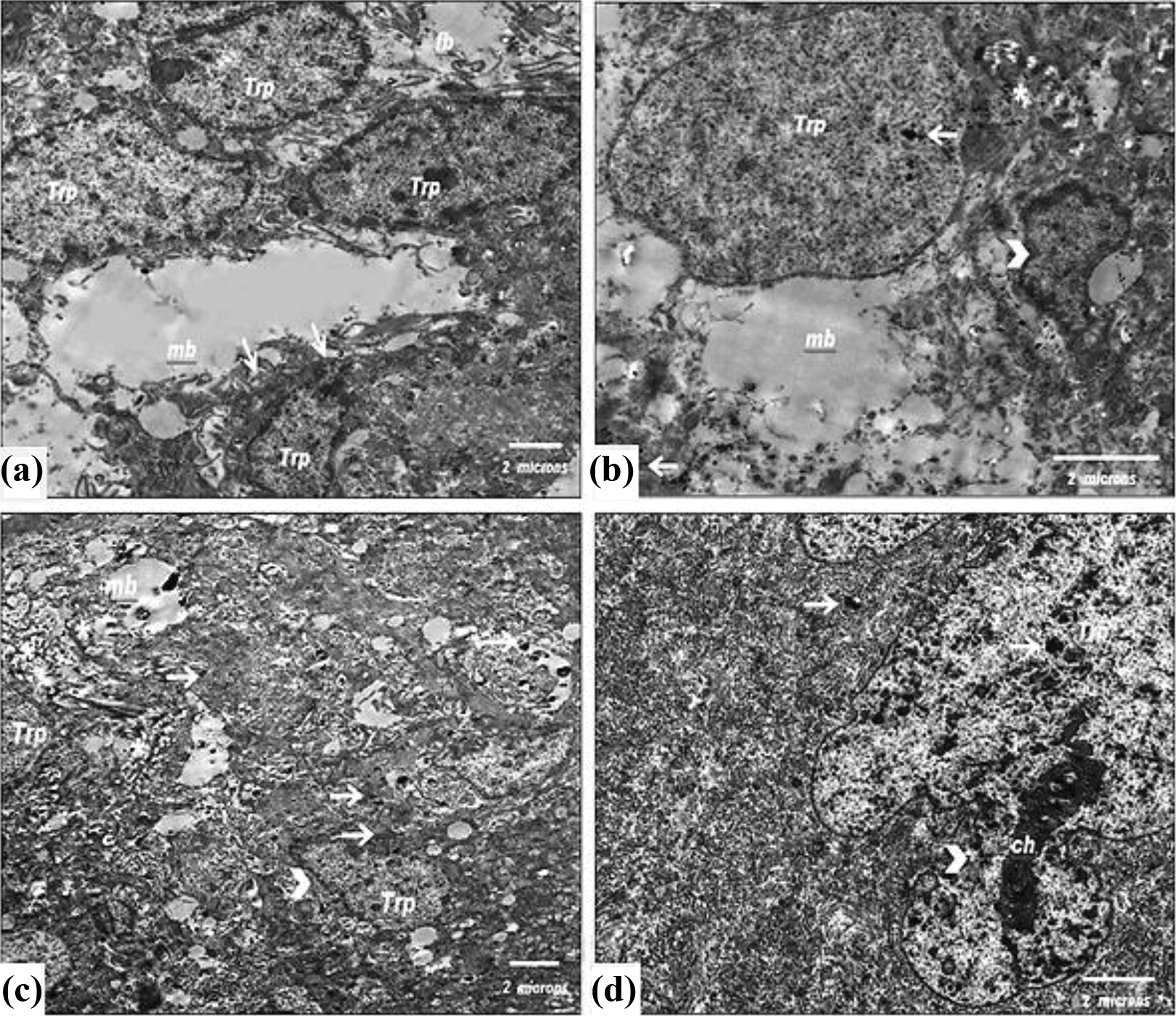
Electron micrographs of: (a) Ultrastructure of a labyrinth zone of control placenta containing trophoblasts (Trp) and the layer between fetal blood (fb) and maternal blood (mb). Note projections from Trp to mb (arrows), ×8780; (b) placenta after 12 h of dosage showing a Trp containing AgNP deposits (arrows), some necrotic areas in cytoplasm (asterisks) and indentation nuclear margins and clumped chromatin (arrow heads), ×11700; (c) labyrinth zone after 24 h of dosage showing Trp containing AgNPs depositions (arrows), irregular nuclear margins (arrow head), and focal necrotic areas (asterisks), ×5860; (d) labyrinth zone after 24 h of dosage showing Trp with necrotic properties such as chromatin condensation (ch), pyknosis, and irregular nuclear margins (arrow heads), ×8780. AgNPs: silver nanoparticles.
Discussion
The data obtained from the AgNPs tissue accumulation presented in this study after a single i.v. administration dose (2 mg/kg b.w) to pregnant rats indicate that AgNPs are rapidly absorbed from the dosing site and distributed from blood into tissues and organs as evidenced by the detection of AgNPs concentration levels in all analyzed samples. The detection of AgNPs in placental barriers exhibited the highest values after 1 and 12 h, respectively. The present data confirmed directly the feasibility of AgNPs to transfer via the placenta reaching fetuses. Such transport of various types of NPs has been previously postulated as a potential source of risks among fetus and newborn development. 33 This aspect is in agreement with previous data obtained by Melnik et al., 14 where AgNPs were identified in the fetuses of all pregnant rats in amounts significantly exceeded the detection limits after oral administration of labeled AgNPs. To our knowledge, there are a few studies concerning the placental transfer of NPs in experimental animals. However, differently sized silica and TiO2NPs showed accumulation of fluorescently labeled 70 nm size silica and 143 nm size TiO2NPs in placenta after 24 h of i.v. injection. 34 AuNPs sized 1.4 and 18 nm induced placental transfer in female rats after i.v. injection. Their uptake in placenta was 3 and 0.02% for 1.4 and 18 nm NPs, respectively. 35 In another investigation, silica NPs of a bigger size of 300 or 1000 nm were not observed in placenta or fetuses. 34
Among transplacental mechanisms, previous studies indicated that the high transport of NPs depends on particles size. 36 The threshold of NP penetration through the barrier was found to be between 143 and 300 nm for silica NPs in mice 34 and between 80 and 240 nm for polystyrene NPs in humans. 37 However, many authors indicated that the glomerular filtration threshold for NPs is between 6 and 8 nm 38,39 which is lower than that for placental barriers. The trophoblastic cells of the placenta are interconnected and form a continuous layer of syncytium, minimizing NPs interception between the cells. 40 Regarding kinetic pattern of AgNPs, the present data indicate that the NPs proportions in fetuses were less than those found in placental tissues. This organ has an active and selective role in transfer mechanisms. On this basis, placental transfer would conform essentially to that across semi-permeable membrane and be determined by physical characteristics such as the thickness on either side of the membrane perfusion pressure (maternal blood pressure) or placental flow. The value of diffusion constant depends on other parameters e.g. molecular size, ionic dissociation and lipid solubility. It would be therefore expected that lipid soluble substances passage across the placenta would be accelerated as compared with those that are less fat soluble. 41
In this regard, the phenomenon is associated with that documented here in the spleen tissue accumulation of AgNPs compared with those found in fetal tissues as well as indicated by EM interpretation of spleen sections, where its uptake was found to be higher than those of placenta. These findings also are in agreement with that obtained by Tan et al., 42 where negatively charged particles are capable to penetrate endothelial vessel cells, while the positively charged NPs were proved to be more greatly filtrated in kidneys or other related tissues.
In the present study, the concentrations of NPs were different regarding time in different organs, blood, and amniotic fluid. It is well-known that some organs are more prone to Ag+ deposition than others. 42 As stated previously, 43 Ag+ ions displayed deposition in rat’s organs following oral administration of 9 mg/kg/day for 28 day as follows: small intestine > stomach > liver > kidney > lung > muscles > brain, respectively. However, administration pattern and particle size of metals may change the model of kinetics, especially distribution system. In the present study, the accumulation of AgNPs in some organs, for example, spleen and placenta were found greater than others, this could be explained as the existence of an abundant pool of proteins able to absorb Ag+, especially when metallothionein (MT) pool is saturated. It is very likely to be responsible in cysteine-rich organs for potent Ag+ binding capability. 44 They confirmed the localization of Ag-MT in rat kidney cortex using autofluorescent signals and immunohistochemistry after injection of silver acetate. This concept explains the role of clearance process and redistribution of Ag+ into blood circulation ending to deposition in organs as seen in the present work after the last administration period (24 h).
In this study, the potential effects of AgNPs after a single i.v. injection to pregnant rats revealed significant formation of 8-OHdG in some samples, especially in maternal spleen, amniotic fluids, and kidneys. The last period (24 h) of administration showed a significant DNA adducts level as represented in Figure 4. The present findings indicate that AgNPs have the feasibility to induce DNA oxidation followed their rat’s body distribution compared with untreated group. Moreover, the compatibility between their accumulation in organs and DNA damage are associated with NPs characterization. Other literature have shown that NPs penetration through tissue’s membrane barriers depend on particle’s size and chemical composition of the surface coating of NPs, 36 where their results indicated that surface properties of NPs may explain the interaction with biomolecules and the distribution in the organs. In the present study, AgNPs were dispersed in citrate under ultrasonic waves before injection, where stabilizing of the layers prevents particles aggregation, enabling NPs to remain suspended in the water column. Citrate salt creates an electrostatic barrier to aggregation, where it competes with oxygen for surface sites and then decreases the rate of oxidation. 45
As mentioned previously, 46 AgNPs can enter the mitochondria and nucleus. Their localization in these cell components may induce disorders in cellular functions. 47 The localization of AgNPs in mitochondria and nucleus imposes a good marker among NPs genotoxic effects on organisms. This issue is in compatibility with those documented here in spleen and placenta sections investigated under TEM (Figures 5 and 6). This induction exactly depends on the ability of AgNPs to liberate Ag+ which can induce cell-specific, localized cellular toxicity as suggested by the Trojan horse theory. 46 Additionally, their ability to form ROS is considered the main pathway for NPs genotoxic effects and other risks. Increased ROS level is related to a series of events leading to membrane damage, DNA and protein damage, and apoptosis or necrosis. 48
Interestingly, the maternal blood showed high concentration of Ag+ after 24 h. This could be explained due to the fact that the clearance process could contribute to a redistribution of Ag+ in the body. This needs more verification. Also importantly, the relatively high concentration of oxidized DNA in the fetus despite a lower Ag+ concentration as compared to the mother’s levels could be explained due to the differences in the pharmacokinetic xenobiotic metabolism between young and adults and on the differences in molecular bases of drug metabolism between fetuses and adults. 49 Dramatic changes were previously found in mouse liver xenobiotic metabolizing enzymes and expression of transporter gene (XMETS) between fetus, neonate, and adults revealing differences in life stage response to drugs. 50
A number of previous studies employing various kinds of cultured cells and animal models suggested that both genotoxicity and apoptosis are important mechanisms for AgNPs-induced toxicity. For example, AgNPs induced DNA breakage in some cell lines using DNA comet assay 51 and DNA double-stranded breaks in treated human hepatoma cells 52 and mouse embryonic fibroblasts and stem cells. 53 In research on gene expression using an oxidative stress and antioxidant defense polymerase chain reaction array, the expressions of 17 of the 59 genes on the arrays were altered in cells treated with AgNPs. These genes are involved in producing ROS, antioxidants, and oxygen transporter and in oxidative stress responses and DNA repair. In that context, 5-nm AgNPs were mutagenic in mouse lymphoma cells due to the induction of oxidative stress. 54
On the other hand, it was suggested previously that metallic NPs enter cells; they might not induce DNA damage themselves, but instead corrode over time, releasing metallic ions that could induce genotoxicity. 55,56 In HepG2 cells exposed to 14–20 µg/ml of ZnONPs for 12 h, ROS triggered a decrease in mitochondrial membrane potential and an increase in the ratio of Bax/Bcl-2 leading to mitochondria-mediated pathway involved in apoptosis. 57 Singh et al. 58 showed that manganese oxide nanoparticles (MnO2NPs; 45 nm) significantly induced DNA damage in peripheral blood leukocytes and micronuclei of rats at a dosage of 1000 mg/kg b.w. However in the present work, the genotoxic effect of AgNPs was evident in both mother and their fetuses by oxidized DNA as measured by ELISA technique. The main known molecular mechanism of NPs-induced DNA damage is considered to be the induction of oxidative stress resulting from ROS generation. Thus, a wide range of NPs including AgNPs is capable to generate ROS and induce oxidative stress as well as DNA damage. 59 The generation of ROS and oxidative injury is thought to play a significant role in many of the observed biological responses to NPs. The size, surface area, and surface chemistry (e.g. reactive groups) of particular NPs are thought to play a role in the generation of ROS. Moreover, oxidative attack on the DNA may result in mutagenic structures such as 8-OHdG, which induces the instability of repetitive sequences. 60 The chemical reactions that bring about such mutations are based on the formation of highly reactive and short-lived hydroxyl radicals in close proximity to DNA. 61 In addition to their damaging effects on cellular proteins, lipid, and DNA, an increased level of ROS triggers the cell to respond by inducing pro inflammatory signaling cascades, ultimately inducing apoptosis. 59
Conclusion
The data of the present study demonstrate that AgNPs induced DNA damage in pregnant rats and embryos. The genotoxicity of AgNPs seems to be due to the generation of ROS that is considered as the major source of spontaneous DNA damage. AgNPs accumulation depends on their size and surface coating NPs. These findings might indicate that oxidized DNA (8-OHdG) is considered an important biomarker in genotoxicity evaluation of NPs. Moreover, when NPs transferred from exposed mothers to fetuses, they will then be distributed through their tissues and may induce adverse effects more than those realized in mother organs. Therefore, genotoxicological studies of NPs must be recommended during a comprehensive assessment of the safety of novel types of NPs and ENMs. Additionally, AgNPs uptake must be prevented or minimized during pregnancy and prenatal periods.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of following financial support for the research, authorship, and/or publication of this article: This study was supported by a grant from the Research Project Unit of Tanta University (project ID: 5-15-5; 4th cycle, 2015).
