Abstract
Objective:
To investigate the mechanism of apoptosis dependent on the Fas/FasL (Fas ligand) complex in the presence of N-nitrosodimethylamine (NDMA) in human leukocytes.
Methods:
Polymorphonuclear neutrophils (PMNs) and peripheral blood mononuclear cells (PBMCs) were isolated form whole blood by density centrifugation. The concentration of NDMA was assessed by cellular toxicity assay. Apoptotic cells were assessed with flow cytometry and the expression of pro- and antiapoptotic proteins was investigated by Western blotting in PMNs and PBMCs treated with NDMA and/or FasL.
Results:
PMNs showed a higher ratio of apoptotic cells than PBMCs after exposure to NDMA and/or FasL. Enhanced apoptosis was related to the increased expression of proapoptotic proteins in neutrophils following exposure to either NDMA or FasL. In PBMCs, the relation was observed after exposure to FasL only. PMNs and PBMCs incubated with NDMA and FasL simultaneously demonstrated the highest increase in protein expression.
Conclusions:
NDMA shows a stronger proapoptotic effect with PMNs than with PBMCs. The Fas/FasL complex, along with other proapoptotic proteins of the receptor (Fas, FADD) and mitochondrial pathway (Noxa, Puma, Bim), plays a key role in the induction of neutrophil apoptosis. Synergic effects of NDMA and FasL which lead to higher induction of apoptosis in PMNs than in PBMCs indicates a multistage and varied regulation of apoptosis in different populations of leukocytes.
Introduction
Human exposure to xenobiotics may significantly impact numerous organs and systems, including the immune system. Xenobiotics, such as N-nitrosodimethylamine (NDMA), are a significant group of adventitious agents. NDMA is the most toxic, carcinogenic, mutagenic, and teratogenic compound among the 300 known nitrosamines. 1 Adverse consequences of exposure to NDMA result both from the compound’s primary effects, causing acute toxicity, as well as from its metabolites (monoalkyl amines, active carbocation). The degree of toxic damage depends on the dosage. The organ which is the most vulnerable to NDMA is the liver. 2
The natural environment is widely contaminated with NDMA, mostly because of human economic and industrial activity, leading to continuous risk of exposure. 3 NDMA is found in food products such as meat cured with sodium nitrate as well as fresh and processed fish. Also, NDMA exposure may result from the consumption of beer, malt, powdered milk, and aged cheeses. Estimates show that an average European’s dietary ingestion of nitrosamines (mostly NDMA) ranges from 0.3 to 1 µg per day. 2
Health effects of adverse environmental factors are the subject of numerous studies. However, the impact of xenobiotics on leukocytes is rarely discussed in the medical literature. Our previous studies as well as research conducted by other authors indicated that NDMA has a multidimensional effect on the function, activity, and survival of human leukocytes. 4 –6
Apoptosis involves the receptor and mitochondrial pathways. The cascade of proapoptotic signals in the receptor pathway is induced by connecting the appropriate ligand with a death receptor of the tumor necrosis factor (TNF) superfamily. Our earlier research indicated that NDMA is involved in apoptosis induced by one of these receptors—DR5. We demonstrated that NDMA increases apoptosis in leukocytes via the DR5-dependent extrinsic pathway and Bax and Mcl-1 proteins of the intrinsic pathway. 4,7
Numerous authors have shown that there are Fas functional death receptors of the TNF superfamily (Apo-1, CD95) on the surface of leukocytes, which also demonstrate the expression of this receptor’s ligand (FasL). 8 –10 Fas/FasL-induced apoptosis seems to play a key role in the case of neutrophils ( polymorphonuclear neutrophils (PMNs)). These cells demonstrate the highest expression of the Fas receptor and FasL ligand among all phagocytic cells. 9
Following FasL connection with the C-terminal section of the Fas receptors death domain (DD), the same domain of the Fas-associated death domain (FADD) adaptor protein is also bound. The death effector domain (DED) on its N-terminus connects the tandem DED in the procaspase-8 initiator prodomain. The produced death-inducing signaling complex (DISC) comprises of a receptor, FADD adaptor protein, and procaspase-8. 11 Next, proteolytic activity turns procaspase-8 into caspase-8 (Figure 1). 12 –14 Cells producing significant amounts of active caspase-8, such as lymphocytes, also effectively activate caspase-3, -6, and -7, inducing apoptosis without the involvement of the mitochondrial pathway. 14 –16 It was also demonstrated, however, that Fas/FasL-induced apoptosis in neutrophils depends on mitochondria and occurs with increased expression of Bid protein which is activated by caspase-8. 17 The Bid proapoptotic proteins of the Bcl-2 family moves to mitochondria where they activate pro- and antiapoptotic BH3-only proteins such as Bim (Bcl-2-interacting mediator of cell death), Noxa (Noxae), and Puma (p53- upregulated modulator of apoptosis), leading to the activation of executor caspase-3 (Figure 1). 9,18,19

Proposed scheme of the Fas/FasL-induced apoptosis process mediated by the proteins of the receptor and mitochondrial pathway in neutrophils. FasL: Fas ligand.
To further explore the cellular mechanisms NDMA effects, we studied the course of Fas/FasL-induced apoptosis in human leukocytes. We assessed the involvement of death receptors and mitochondrial pathway proteins by evaluating their expressions in the presence of NDMA and/or the FasL ligand, which initiate Fas/FasL-dependent apoptosis (Figure 1).
Methods
Material for research
The research material was composed of venous blood from 20 healthy volunteers (9 males and 11 females) aged 20–42 years. The donors were nonsmokers and did not consume alcohol for at least 48 h prior to blood donate. After the volunteers signed their consent, the blood samples were collected from the basilic vein, 8 ml of which were stored with ethylenediaminetetraacetic acid (EDTA), and 2 ml without any anticoagulant in order to extract serum. The study was approved by the Ethics Committee of the Medical University of Białystok (R-I-002/573/2013).
Isolation of polynuclear and mononuclear peripheral blood cells
The cell fractions containing neutrophils and mononuclear cells were isolated in a concentration gradient using Polymorphrep (Axis-Shield, Oslo, Norway). The obtained mononuclear and polynuclear cell populations were suspended in Hank’s fluid and medium discovered in Roswell Park Memorial Institute (RPMI)-1640, respectively, with a culture medium (BIOMED-Lublin, Poland) containing 7.4% (20/270 µl) of an autologous serum, 100 U/ml of penicillin, and 50 ng of streptomycin (Polfa Tarchomin S.A., Poland), leading to a density of 5 × 106 cells/ml. The cells exposed to NDMA in a concentration of 10 µg/ml (Sigma-Aldrich, Steinheim, Germany; CAS no. 62-75-9) and/or FasL in a concentration of 1 ng/ml, were incubated for 20 h on micro plates (Microtest III-Falcon; BD Biosciences, Bedford, USA) at a temperature of 37°C in an incubator with a maintained 5% CO2 flow (NUAIRE™, Plymouth, USA). The cytotoxic effect of NDMA was analyzed by using the tetrazolium-based colorimetric assay (MTT reduction assay).
Cellular toxicity assay
Cytotoxic effect of NDMA on neutrophils was analyzed by way of the tetrazolium-based colorimetric assay (thiazolyl blue tetrazolium bromide (MTT) reduction assay). The MTT assay is dependent upon the ability of mitochondrial dehydrogenase in living cells to reduce the yellow MTT to a blue formazan product. It has been shown that an increase in absorbance values correlates to an increase in cellular enzymatic activity and cell number. PMN and peripheral blood mononuclear cells (PBMCs) were plated in 96-well microplates at 1 × 106 cells/well in 200 μl od medium without NDMA or at increasing doses of the xenobiotic: 0.5, 1, 5, 10, 250, 1000, 2500, and 5000. After 2 h of incubation, 20 μl of stock MTT solution (5 mg/mL) was added to each culture well and plates were incubated for 1 h at 37°C in a 5% CO2 incubator. The plates were then centrifuged, the medium was aspirated, and 200 μl of dimethyl sulfoxide (DMSO) was added to lyse the cells and dissolve the MTT formazan crystals. The plates were mechanically agitated and absorbance was measured at 570 nm by using a UVN-340 ASYS Hitech GmbH microplate reader (Biogenet, Eugendorf, Austria). Experiments were performed in triplicate and the collected data are expressed as the percentage of MTT reduction compared to control cells (unexposed). Metabolic activity decrease of studied leukocytes was observed in cells incubated with NDMA at > 10 μg/ml concentration (Figure 2). This proved the cytotoxic effect of this compound.

Cytotoxic activity of NDMA, evaluated by the MTT assay. The data present the average values of five independent experiments. NDMA: N-nitrosodimethylamine.
Flow cytometry apoptosis assessment
Apoptotic cells were assessed by flow cytometry using a ready-made kit (Apotest™-FITC; Nexins Research, Holland). PMNs and PBMCs fractions immediately after isolation and after 20-h incubation with and without the presence of NDMA were stained with FITC conjugated with annexin V and propidium iodide. The samples were analyzed with CellQest Analysis software (Becton Dickinson, Epics Coulter, USA). The results were presented in the form of the percentage of annexin-V-positive and propidium-iodide-positive cells in the case of late apoptosis and the percentage of annexin-V-positive and propidium-iodide-negative cells for early apoptosis.
Western blot analysis of protein expression
The PMNs and PBMCs fractions were lysed via sonification using Ultrasonic Processor Vibra Cell (Sonic, Newtown, USA) in the presence of a Protease Inhibitor Cocktail (Sigma-Aldrich, Steinheim, Germany). The obtained lysate was suspended in a Laemmli buffer (Bio-Rad Laboratories, Hercules, California, USA). Fifty micrograms of the protein were used for electrophoresis with SDS-PAGE gel Mini PROTEAN®Tetra Cell (Bio-Rad Laboratories). The separated protein fractions were transferred onto 0.2 μm pore-sized nitrocellulose membranes (Bio-Rad Laboratories). Next, the membranes were incubated at 4°C for 4 h with specific polyclonal antibodies: anti-FADD (R&D Systems, Minneapolis, USA), anti-cleaved Bid, anti-Noxa, anti-Bim, anti-Bcl-2 (Santa Cruz Biotechnology, Heidelberg, Germany) and monoclonal antibodies: anti-Fas, anti-Puma, anti-Mcl-1, anti-β-Actin (Santa Cruz Biotechnology). After washing with 0.1% tris buffered saline with Tween (TBS-T), the membrane was incubated at room temperature for 1 h with antibodies marked with alkaline phosphatase directed against IgG (Vector Laboratories, Burlingame, California, USA). Immunoreactive protein bands were obtained by adding 5-Bromo-4-chloro-3-indolyl phosphate/ nitro blue tetrazolium (BCIP/NBT) Liquid Substrate System (Sigma-Aldrich). The intensity of the bands was assessed using ImageJ software (public domain, Java-based image processing program developed at the National Institutes of Health, USA) and estimated by arbitrary units (AUs). The antibody against beta-actin (Santa Cruz Biotechnology), which detects the expression of beta-actin in cells lysates, was used for standardization.
Statistical analysis
The results were analyzed using Statistica version 9.1 (StatSoft, Inc., Tulsa, Oklahoma, USA). The normality of data distribution was examined using the Kolmogorov-Smirnov test. The data were not normally distributed therefore U-Mann-Whitney nonparametric tests were applied. Flow cytometry data were presented as median and ranges (minimum and maximum). The data in Figures 3 and 4 were presented as mean ± SD. Statistical significance level was set at p < 0.05.
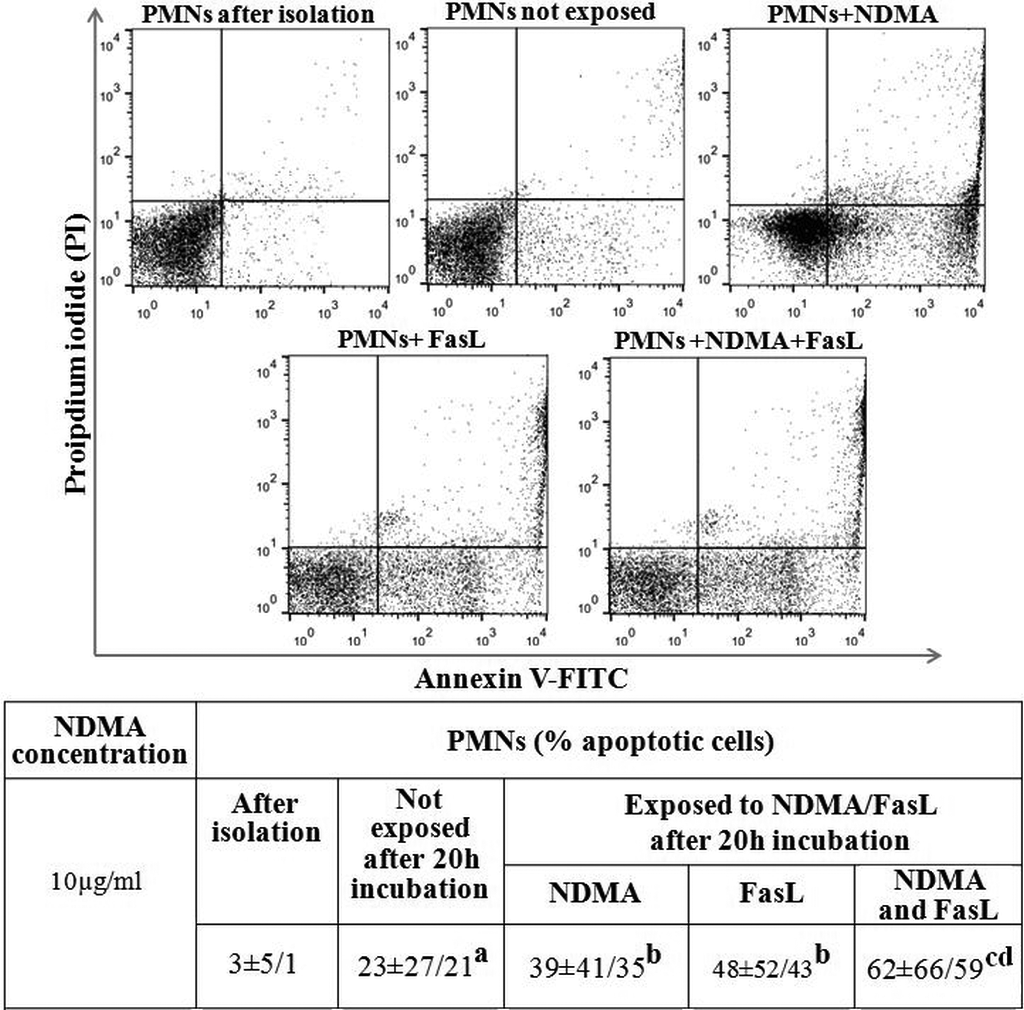
Percentage of apoptotic PMNs presented as median ± maximum and minimum values. The figure illustrates the apoptotic cells immediately after isolation, after 20 h incubation, exposed to NDMA (10 µg/ml), FasL (1 ng/ml), NDMA + FasL and not exposed. aStatistical difference between the percentage of apoptotic cells immediately after isolation and unexposed apoptotic cells after 20 h incubation (p < 0.05). bStatistical difference between percentage of apoptotic cells exposed to NDMA or FasL and unexposed apoptotic cells (p < 0.05). cStatistical difference between the percentage of apoptotic cells exposed to NDMA and FasL simultaneously and the percentage of unexposed apoptotic cells (p < 0.05). dStatistical difference between the percentage of apoptotic cells exposed to NDMA and FasL simultaneously and cells exposed to NDMA or FasL (p < 0.05). PMN: polymorphonuclear neutrophils; NDMA: N-nitrosodimethylamine; FasL: Fas ligand.

Percentage of apoptotic PBMCs presented as median ± maximum and minimum values. The figure illustrates the apoptotic cells immediately after isolation, after 20 h incubation, exposed to NDMA (10 µg/ml), FasL (1 ng/ml), NDMA + FasL, and not exposed. aStatistical difference between the percentage of apoptotic cells immediately after isolation and unexposed apoptotic cells after 20 h incubation (p < 0.05). bStatistical difference between percentage of apoptotic cells exposed to NDMA or FasL and unexposed apoptotic cells (p < 0.05). cStatistical difference between the percentage of apoptotic cells exposed to NDMA and FasL simultaneously and the percentage of unexposed apoptotic cells (p < 0.05). dStatistical difference between the percentage of apoptotic cells exposed to NDMA and FasL simultaneously and cells exposed to NDMA or FasL (p < 0.05). PBMCs: peripheral blood mononuclear cells; PMN: polymorphonuclear neutrophils; NDMA: N-nitrosodimethylamine; FasL: Fas ligand.
Results
Apoptosis of neutrophils and mononuclear cells assessed with flow cytometry after incubation with NDMA and/or FasL
The ratio of apoptotic cells increased after exposition to NDMA or FasL in neutrophils and in comparison, to unexposed cells. The highest ratio of apoptotic PMNs was observed in the case of simultaneous exposition to NDMA and FasL (Figure 3).
The percentage of apoptotic mononuclear cells after incubation with NDMA or FasL was higher than in unstimulated cells. The highest number of apoptotic PBMCs was observed after exposition to NDMA and FasL simultaneously (Figure 4).
Expression of receptor signaling pathway proteins assessed with Western blot analysis
Fas receptor
Neutrophils demonstrated a reduced expression of Fas after incubation with NDMA as compared to unexposed cells. Exposition of neutrophils to FasL and to NDMA and FasL simultaneously led to increased expression of the Fas receptor, compared to unexposed cells or cells exposed to NDMA only (Figure 5).

Western blot analysis of Fas, FADD, and tBid proteins in PMNs and PBMCs after 20 h incubation exposed to NDMA (10 µg/ml), FasL (1 ng/ml), NDMA + FasL, and not exposed. aStatistical difference between not exposed and exposed cells (p < 0.05), bStatistical difference between cells exposed to NDMA and cells exposed to FasL (p < 0.05), cStatistical difference between cells exposed to NDMA and FasL simultaneously and cells exposed to NDMA (p < 0.05), dStatistical difference between cells exposed to NDMA and FasL simultaneously and cells exposed to FasL (p < 0.05). eStatistical difference between PMNs and PBMCs (p < 0.05). PBMCs: peripheral blood mononuclear cells; PMN: polymorphonuclear neutrophils; NDMA: N-nitrosodimethylamine; FasL: Fas ligand.
No changes in expression were observed in mononuclear cells exposed to NDMA, although the cells indicated increased expression of Fas after incubation with FasL and NDMA + FasL, in comparison to unexposed cells. Fas expression was significantly higher in neutrophils in comparison to mononuclear cells (Figure 5).
FADD protein
Increased expression, comparable with exposition to NDMA, of the FADD adaptor protein was observed in PMNs and PBMCs after incubating the cells with FasL and NDMA and FasL simultaneously (Figure 5).
tBid protein
Reduced expression of truncated Bid (tBid) was observed in neutrophils exposed to NDMA in comparison to unexposed cells. However, the expression of this protein in neutrophils increased after they were exposed to FasL and was greater than the expression observed in PMNs incubated with NDMA. Highest expression was observed in neutrophils incubated with FasL + NDMA when compared with unexposed cells and cells exposed to NDMA or FasL separately (Figure 5).
In mononuclear cells, the highest expression of tBid was observed when the cells were exposed to FasL (Figure 3). After incubating PBMCs with NDMA + FasL, the cells showed reduced expression of tBid in comparison to FasL exposed cells and higher expression than in unstimulated PBMCs or those exposed to NDMA only. In comparison to the relevant mononuclear cells, the expression of the studied protein was lower in neutrophils exposed to FasL but higher in cells exposed to NDMA + FasL (Figure 5).
Expression of mitochondrial signaling pathway proteins assessed with Western blot analysis
Noxa protein
Expression of Noxa increased when PMNs were exposed to NDMA, FasL, and NDMA + FasL. Noxa expression was significantly higher when the cells were exposed to NDMA only, in comparison to cells incubated with FasL and NDMA + FasL. After incubation with FasL, the expression of the studied protein was higher compared with unexposed cells but lower than in PMNs incubated with NDMA or NDMA + FasL (Figure 6).
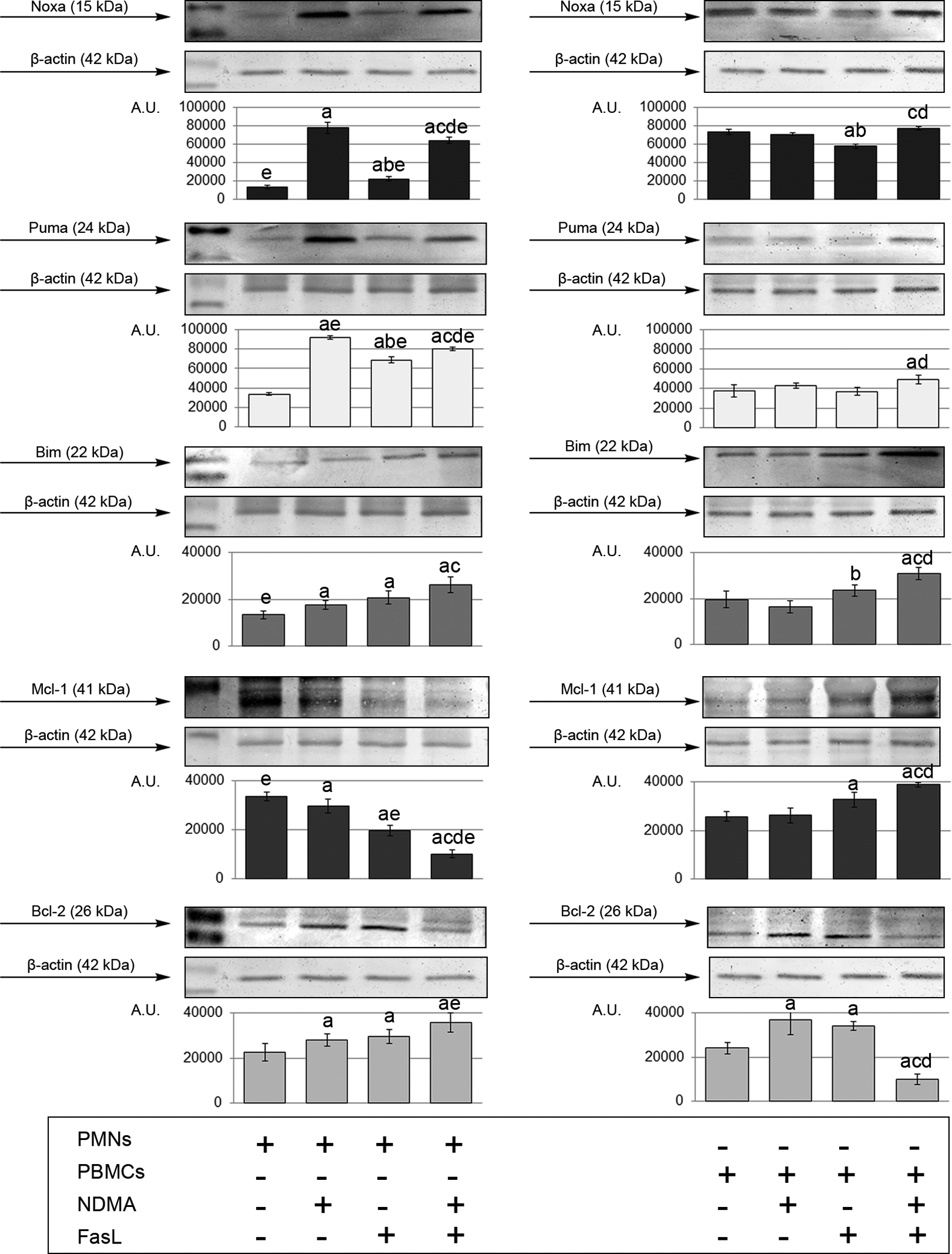
Western blot analysis of Noxa, Puma, Bim, Mcl-1, and Bcl-2 proteins in PMN and PBMCs after 20 h incubation exposed to NDMA (10 µg/ml), FasL (1 ng/ml), NDMA + FasL, and not exposed. aStatistical difference between not exposed and exposed cells (p < 0.05), bStatistical difference between cells exposed to NDMA and cells exposed to FasL (p < 0.05), cStatistical difference between cells exposed to NDMA and FasL simultaneously and cells exposed to NDMA (p < 0.05), dStatistical difference between cells exposed to NDMA and FasL simultaneously and cells exposed to FasL (p < 0.05). eStatistical difference between PMNs and PBMCs (p < 0.05). PBMCs: peripheral blood mononuclear cells; PMN: polymorphonuclear neutrophils; NDMA: N-nitrosodimethylamine; FasL: Fas ligand.
Mononuclear cells demonstrated reduced expression of the protein after incubation with FasL, as compared to unexposed cells and cells treated with NDMA. On the other hand, exposition of PBMCs to NDMA and FasL simultaneously resulted in increased expression of Noxa, compared to cells exposed to NDMA and FasL separately. Expression of this protein in unexposed neutrophils and neutrophils treated with FasL and FasL + NDMA was lower than in mononuclear cells (Figure 6).
Puma protein
The research found increased expression of Puma protein in neutrophils after the cells were incubated with NDMA, FasL, and the two factors simultaneously. The expression of the protein was lower in PMNs exposed to FasL than in PMNs exposed to NDMA. Simultaneous exposition of neutrophils to NDMA and FasL resulted in decreased expression of Puma compared to cells exposed to NDMA and higher expression in comparison to cells exposed to FasL (Figure 6).
Mononuclear cells demonstrated increased expression of Puma protein only after incubation with NDMA and FasL simultaneously, as compared to unexposed cells and cells incubated with FasL only. Expression of Puma in PMNs treated with NDMA, FasL, and both factors simultaneously was significantly higher than in PBMCs (Figure 6).
Bim protein
Exposition of PMNS to NDMA, FasL and both factors simultaneously produced increased Bim expression. The presence of NDMA + FasL caused elevated expression of the studied protein in comparison to cells incubated with NDMA only (Figure 6).
In mononuclear cells, the expression of Bim was higher after incubation with FasL, compared to cells incubated with NDMA. Exposition of PBMCs to NDMA and FasL simultaneously increased the expression of Bim protein, compared to unexposed cells and cells exposed to NDMA and FasL separately. Expression of Bin in unexposed neutrophils was lower than in mononuclear cells (Figure 6).
Mcl-1 protein
The expression of antiapoptotic Mcl-1 protein in neutrophils decreased after the cells were exposed to NDMA, FasL, and NDMA + FasL compared to unexposed cells. Lowest expression of the protein was observed in neutrophils incubated with NDMA and FasL simultaneously (Figure 6).
In mononuclear cells, exposition to FasL and NDMA + FasL increased the expression of Mcl-1. The lowest expression of the studied protein was observed in PBMCs incubated with NDMA and FasL simultaneously. Mcl-1 expression in PMNs exposed to FasL and NDMA + FasL was lower than in respective PBMC samples (Figure 6).
Bcl-2 protein
Exposition of PMNS to NDMA, FasL, and NDMA + FasL resulted in a statistically significant increase of expression of antiapoptotic Bcl-2 (Figure 6).
In PBMCs, elevated expression of Bcl-2 was observed after the cells were incubated with NDMA or FasL, compared to unexposed cells. Incubation of PBMCs with NDMA + FasL resulted in a significant reduction in Blc-2 expression as compared with unexposed cells or cells exposed to NDMA or FasL. Expression of Bcl-2 in neutrophils treated with NDMA + FasL was lower than in mononuclear cells (Figure 6).
Discussion
The presented findings indicate that the apoptogenic effects of NDMA have a significantly larger impact on neutrophils than on mononuclear cells. Wider effects of NDMA on PMNs most likely depend on the Fas/FasL complex. This observation is confirmed in research conducted by Gillette-Ferguson and Sidman, which showed that the coexpression of Fas and FasL in neutrophils guarantees the high level of susceptibility of these cells to apoptosis induced by Fas connecting with FasL, contrary to mononuclear cells, such as T-lymphocytes, which require additional, prior activation. 20
The combined effect of NDMA and FasL on the expression of mitochondrial apoptosis pathway proteins is also interesting. It was found that the simultaneous presence of the two factors results in the lowest expression of the antiapoptotic Mcl-1 protein in neutrophils, which enhances apoptosis in these cells.
The presented results also show a significant involvement of Noxa and Puma proteins in the apoptosis of neutrophils exposed to NDMA, which is consistent with the observations of Gautam et al., who demonstrated the importance of these proteins in the induction of apoptosis in neutrophils. 21 Both Noxa and Puma belong to the group of BH3-only proteins which to directly bind and activate proapoptotic Bax and Bak proteins or bind indirectly with antiapoptotic proteins such as Bcl-2 or Mcl-1 so that they do not interfere with the activity of proteins from the Bax subfamily. 22,23 This allows Bax and Bak to integrate in the outer mitochondrial membrane, form pores and release apoptogenic factors which contribute to the activation of caspase-9 and -3. This results in the enhancement of signals from the receptor pathway. 24,25 Also, there seems to be no direct relation between the Fas/FasL complex and the activation of Noxa and Puma.
The involvement of Noxa in enhancing apoptotic signaling in neutrophils may be connected with the decreased activity of Mcl-1 in these cells. According to a derepression model, Noxa causes the Mcl-1 to heterodimerize and indirectly inhibits its antiapoptotic action. 26 The Bim protein demonstrates a different type of interaction with Noxa. The research presented by Kirschnek et al. proved that Noxa and Bim work synergistically and interdependently. 27 Increased expression of Bim observed after exposure to NDMA and FasL, coupled with intense activation of Noxa, is likely to be significant in transmitting apoptotic signals in neutrophils. However, the absence of changes in the expression of Noxa, Puma, and Bim observed when mononuclear cells were exposed to NDMA indicates a different mechanism of apoptosis induction than in neutrophils.
The synergistic effects of FasL and NDMA were also observed in the case of the antiapoptotic Bcl-2 protein. However, the potential inhibition of apoptotic signaling may not have special importance in neutrophil apoptosis due to the status of Bcl-2. Moulding et al. showed that its role in the programmed death of neutrophils is not as significant as in other cells. 28 This is because neutrophils show low expression of Bcl-2 in exchange for a higher expression of antiapoptotic proteins such as Mcl-1, A1, or Bcl-xl. 29
The results obtained in the study demonstrate that there is a collaboration between NDMA and FasL in strengthening the tBid activated mitochondrial apoptosis pathway. Because neutrophils produce relatively small amounts of caspase-8, the course of apoptosis—in this case, type 2 apoptosis—depends on the Bid protein (BH3 interacting domain death antagonist), a link between the receptor and mitochondrial pathways. 30 The activity of Bid is connected with the ability of active caspas-8 to proteolytically split the cytosol Bid into tBid. The key role of the Bid protein in neutrophils apoptosis induced by Fas/FasL complex was confirmed by Geering et al. They proved that neutrophils derived from the bid knockout mice were partially protected against apoptosis induced by Fas/FasL complex. 31
It may be concluded that neutrophils are more susceptible to proapoptotic effects of NDMA. The Fas/FasL complex plays, as well as proapoptotic proteins of the receptor (Fas, FADD) and mitochondrial pathway (Noxa, Puma, Bim) play a key role in the induction of neutrophil apoptosis. The demonstrated synergic effect of NDMA and FasL, which lead to stronger induction of apoptosis in PMNs rather than in PBMCs indicates a multistage and varied regulation of apoptosis in the studied leukocytes.
The observed changes may lead to reduced bioavailability and activity of leukocytes, especially neutrophils. People exposed to NDMA may present reduced recruitment and decreased phagocytic activity, resulting in weakened internal killing mechanisms and impaired first line of immune response. 32,33 Exposure to NDMA also affects mononuclear cells, which may lead to the impairment of specific cellular and humoral mechanisms. Clinical manifestations of these changes may include recurring infections caused by various pathogens and impaired antitumor response. 34
Further research, stretching across addition apoptotic proteins would make it possible to identify the interactions between particular proteins and determine their role in apoptosis induction in human leukocytes exposed to NDMA.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
