Abstract
It was reported that novel O,O′-diethyl-(S, S)-ethylenediamine-N,N′-di-2-(3-cyclohexyl) propanoate dihydrochloride (DE-EDCP) displayed in vitro antiproliferative activity on several human and mouse cancer cell lines, which was comparable to that of the prototypical anticancer drug cisplatin. In order to reveal its toxicity profile, acute and repeated-dose toxicity studies were performed in Naval Medical Research Institute (NMRI) Han mice. The intravenous LD50 values of DE-EDCP were found to be 95.3 and 101.3 mg/kg body weight in female and male mice, respectively. In the subacute toxicity study, DE-EDCP was administered intravenously at the doses of 15, 25, and 40 mg/kg/day for a period of 28 days. There were no adverse effects on general condition, growth, feed and water consumption, and hematological parameters. There was a significant increase in urea and alanine aminotransferase in female mice and aspartate aminotransferase and alkaline phosphatase in both genders in 40 mg/kg/day dose-treated group. The histopathological changes confined to the liver and kidney, but in other organs were not found. Satellite group revealed that changes in the kidney and liver were less pronounced, suggesting their reversibility. Interactions with DNA could also be of importance for understanding DE-EDCP toxic side effects. Hyperchromic effect obtained with ultraviolet–visible, suggested electrostatic interactions between DE-EDCP and calf thymus DNA. The toxicity testing of DE-EDCP was conducted to predict human outcomes.
Introduction
Cancer is one of the most prevalent diseases that kill millions of people every year. It is very difficult to treat it because organism doesn’t recognize cancer cells as foreign to the body. 1,2 Because of this reason, the most of drugs used in cancer treatment at the present time are cytotoxic drugs that interfere in some way with the operation of the cell’s DNA. They don’t make the difference between “normal” and cancer cells.
Over the last 50 years, several hundred thousand of natural and synthetic chemical compounds have been tested for their anticancer activity, but only a few of these are in clinical use today. 3 A main goal for scientists is to design new drugs that will be more selective for cancer cells, and thus have a smaller number of side effects.
The most famous anticancer drug, cisplatin, showed potent antitumor activity in the 1970s and it is still used in the treatment of many types of cancers, especially solid cancers. 4 –6 Unfortunately, it is widely known that it also has significant limitations in use owning to severe side effects like nephrotoxicity, ototoxicity, and neurotoxicity. 7,8 As a result of the great efforts to overcome this problem, many platinum (Pt) complexes were synthesized with hope of better pharmacological properties. 9 –11 Besides the metal-based anticancer agents, except cisplatin, only carboplatin and oxaliplatin are in worldwide clinical use. 12,13 Iproplatin and tetraplatin were abandoned due to various reasons, but the most promising candidate that completed phase III trials is lipophilic agent, satraplatin. 14,15
In the last 10 years, various Pt(II) and Pt(IV) complexes with N,N′-bidentate compounds were synthesized. Many of them showed antitumor activity, especially octahedral Pt(IV) complexes with cyclohexyl-functionalized ethylenediamine-N,N′-diacetate-type compound. There are several reports on the cytotoxicity of these compounds. 16 –19 Before them, many other compounds, classes of PtII (cisplatin analogues) and PtIV complexes (satraplatin analogues) with ethylenediamine-N,N′-diacetate (edda)-type ligands, were synthesized. 17,20 It was found that generally they are less efficient than cisplatin. Opposite them, PtIV complexes with esterifies edda-type ligands showed greater anticancer activity than corresponding PtII complexes and cisplatin, 21 –23 causing very rapid necrotic cell death in some cell lines.
In most cases, high cytotoxic action was accomplished thanks to Pt ions. 19,24 Generally, metal-based anticancer agents are active due to their metal centre, which is positively charged and thus binds to the negatively charged residues of proteins and nucleic acids and in this manner kill cancer cells. 25 However, some of the esters showed serious antitumor activity themselves. 19,24,26 Considering that metal ion also has positive contribution to the toxicity effect, organic compounds that lack metal and have incorporated ethylenediamine deserve special attention.
Recently, synthesized group of compounds belongs to symmetric diesters of dicyclohexyl-ethylenediamine dipropanoic acid, which do not contain metal, but showed a significant effect on some tumor cells. Investigation of cytotoxicity showed that O,O′-diethyl-(S, S)-ethylenediamine-N,N′-di-2-(3-cyclohexyl) propanoate dihydrochloride (DE-EDCP) exerted significant toxicity toward glioma, melanoma, fibrosarcoma, and human leukemic cell lines. 19,24 The cytotoxicity of DE-EDCP on leukemic cell lines was comparable to that of prototypical anticancer drug cisplatin. 24
DNA plays a major role in the life process because it carries heritage information and instructs the biological synthesis of proteins and enzyme through the process of replication and transcription of genetic information. DNA is quite often the main cellular target for studies with smaller molecules of biological importance like carcinogens, steroids, and several classes of drugs. The investigation of drug–DNA interactions is of current general interest and importance, 27 –29 especially for designing of new DNA-targeted drugs and screening of these in vitro. Absorption studies, together with cyclic voltammetry (CV) as a versatile technique to investigate redox activities during drug–DNA interactions, are considered the most common and elementary technique to explore the interaction studies of drug molecule with the DNA by observing the changes in the absorption spectrum of DNA. 30,31 The investigation of drug–DNA interaction can provide information of the structural features which determine the therapeutic affectivity of the drug. Interactions of DE-EDCP with DNA could also be of significance for clarifying the molecular mechanism of toxicity or side effect in vivo.
From available literature, no work has been done on the toxicity profiles of DE-EDCP. The objective of the present study is to evaluate the acute and subacute toxicity profiles of DE-EDCP in mice. In this study, we studied the DE-EDCP-DNA interaction in combination with CV and especially ultraviolet–visible (UV-Vis) spectrophotometry. This is aimed at generating data on the level of safety of DE-EDCP injected intravenously.
Materials and methods
Good laboratory practice
This study was performed according to the regulations and standards: National-Serbian (The Law on the Experimental Animal Treatment) and European (Directive 2010/63/EU; European Convection for the Protection of Vertebrate Animals used for Experimental and other Scientific Purposes). The experiment was approved by the Animal Ethics Committee – Galenika a.d. (permission no. 3/12). All experiments were performed in compliance with all laws of the Republic of Serbia.
Synthesis of DE-EDCP
DE-EDCP was synthesized by following an already described procedure. 19 It is a white powder, soluble in water, m.p.: 208°C; proton nuclear magnetic resonance (200 MHz, DMSO-d 6) δ/ppm: 0.89 (m, C7, 4 H), 1.26 (m, C5, C6, 8 H; CH3CH2OOC–, 6 H), 1.72 (m, –CH2Cy; C4, 2 H; C5, 4 H; C6, 4 H), 3.96 (s, CH3CH2OOC–, 4 H), 4.05 (m, –NH2CH2CH2NH2–, 4 H), 4.25 (m, –OOCCHNH2–, 2 H), 10.09 ppm (m, –NH2CH2CH2NH2–, 4 H); carbon-13 nuclear magnetic resonance (50 MHz, DMSO-d 6) δ/ppm: 14.1 CH3CH2OOC–), 25.6 (C7), 25.6 (C4), 31.8 (C6), 33.4 (C5), 36.9 (C3), 57.3 (C8), 62.3 (C2; CH3CH2OOC–), 169.3 ppm (C1); IR (ATR, cm−1): 2900–2500, 1739, 1450, 1215, 802; MS (LC–MS, 4000 V) m/z: 425 ([M-2HCl+H]+, 45.01%). Calculated mass fractions of elements, w/%, for C24H46O4N2Cl2·0.5 H2O (M r = 497.54) are C 56.90, H 9.35, and N 5.53 and found C 56.40, H 8.92, and N 5.50. Its structural formula is shown in Figure 1.

Structural formula of DE-EDCP. DE-EDCP: O,O′-diethyl-(S, S)-ethylenediamine-N,N′-di-2-(3-cyclohexyl) propanoate dihydrochloride.
Preparation of DE-EDCP solution
For all toxicity studies, a stock solution of DE-EDCP (17.5 mg/mL) was prepared in sterile saline (0.9% sodium chloride). Dilutions in saline were made to permit the intravenous (i.v.) administration of increasing doses of DE-EDCP in suitable volume, up to a maximum of 0.2 mL. 32 The solutions filtered through a sterile filter (pore size: 0.20 mm), and the respective dose administered intravenously within 30 min after dissolution.
Animals
Male and female NMRI Han mice, 5–6 weeks old, average weight 18–22 g, were used in the study. The females were nulliparous and non-pregnant. The animals were obtained from the vivarium of Galenika a.d., Center for Biomedical Research, Belgrade, Serbia. They were housed in vivarium of Galenika, in sterilized polypropylene cages, in a room with controlled temperature of 22 ± 2°C and relative humidity of 60 ± 5% under a 12-h light/12-h dark cycle. Each cage contained three mice of the same sex. Prior to commencement of study, all mice have been acclimated to laboratory conditions for 7 days and evaluated for weight gain and any gross signs of disease or injury. They were fed with commercial pellet food for small rodents (Veterinarski zavod, Subotica, Serbia) and water ad libitum. Beddings were used after sterilization by autoclaving and changed on a weekly basis. All efforts have been made to minimize the number of animals used and their suffering.
Acute toxicity study
Four groups of six mice per sex per group were i.v. given through the tail vein a single dose of DE-EDCP at 25, 50, 100, and 150 mg/kg body weight (bw), while the control group received an equivalent quantity of normal saline solution only. The animals were fasted for 16 h before administration. Following administration, the animals were observed individually for the appearance of signs of toxicity at 30 min, 2 h, 4 h, and 6 h for the first 24 h. Thereafter, observation was continued daily for a total of 14 days. The visual observations included changes in the skin and fur, eyes, ears, and mucous membranes and also respiratory, circulatory, autonomic, and central nervous system as well as somatomotor activity. Observation parameters also included tremor, convulsions, salivation, diarrhoea, lethargy, sleep, and coma. The bws were recorded before the dose administered, on days 4, 8, and 14. In addition, the intake of food and water behavior was monitored.
At the end of the test, all survived mice were anesthetized with ketamine/xylazine (100/10 mg/kg i.p.) followed by cervical dislocation. The animals that died during the experiment and all the surviving ones were subjected to a gross necropsy. The internal organs, including brain, heart, lungs, liver, spleen, pancreas, stomach, intestines, kidneys, adrenals, and sex organs, were grossly examined. The numbers of dead mice in each sex per group were counted and the median lethal dose (LD50) was calculated using the graphical method of Miller and Tainter. 33
Subacute toxicity study
Experimental design
Three treated groups of 12 mice (six males and six females) i.v. received DE-EDCP at the doses of 15, 25, and 40 mg/kg bw once daily for 28 days, but the control group received the same volume of sterilized saline solution. All mice were observed for the appearance of signs of toxicity or behavioral alterations once daily during the experimental period. Individual animal bws were collected before administration of DE-EDCP and every third day as well as on the last day before killing. Food (g) and water (mL) consumption per group was recorded daily. At the end of the experiment (1 day after the last dose, on 29th day), after an overnight fast, blood was collected by cardiac puncture under ketamine/xylazine anesthesia (100/10 mg/kg i.p.) for hematological and serum biochemical analysis. After the blood collection, the animals were killed by cervical dislocation and vital organs were taken for histological examination.
The satellite group (six males and six females) was given DE-EDCP at the dose of 40 mg/kg/day for 28 days and kept further for another 14 days in order to detect the reversible or delayed occurrence of toxic effects. A concurrent control group was also used. Daily visual observations were made and recorded systematically similar to those performed as for treated groups. At the end of the experiment, on the 43th day from the beginning of the study, all mice were anesthetized with ketamine/xylazine (100/10 mg/kg i.p.), killed, and subjected to a detailed gross pathology, and organ weights were recorded.
Hematological parameters
Blood samples were collected by cardiac puncture into sterile tubes with anticoagulant Ethylenediaminetetraacetic Acid (EDTA), (Merck KgaA, Darmstadt, Germany) for hematological tests. Hematological parameters, measured by an automated analyzer (ABX Pentra XL 80, Horiba ABX diagnostics), were red blood cell count (RBC), white blood cell (WBC) count, hemoglobin (HGB) concentration, hematocrit, mean corpuscular volume, mean corpuscular hemoglobin (MCH), MCH concentration, red cell distribution width, platelet count (PLT), mean platelet volume, and the percentage of differentiated WBCs, including neutrophils (NEUT), lymphocytes (LYMPH), and monocytes (MONO).
Biochemical parameters
Blood samples were collected by cardiac puncture into sterile without anticoagulant tubes for biochemical tests. Blood samples were centrifuged at 5000 r/min for 15 min and serum was separated. Biochemical parameters were measured by automatic Architect ci8200-integrated serum/plasma analyzer system (Abbott Laboratories, Illinois, USA). Renal and hepatic functions were evaluated by measuring the concentrations of urea, creatinine, uric acid, aspartate aminotransferase (AST), alkaline phosphatase (ALP), total bilirubin, and alanine aminotransferase (ALT).
Observation of gross pathology (macroscopic examination)
All mice were killed by cervical dislocation at the end of the experiment and complete necropsy was performed. The criteria of the gross pathological examination were based on position, shape, size, color, and consistency of the organs.
Histopathological studies
At the time of necropsy, organs, such as brain, heart, lungs, liver, spleen, pancreas, stomach, intestines, and kidneys, were removed, weighted, and collected for microscopic examination. They were fixed with 10% neutral-buffered formalin solution and embedded in paraffin. 34 Sections of 5-µm thicknesses were stained with hematoxylin and eosin (H&E), examined for histopathological changes under a light microscope (Olympus Bx60; Olympus, Tokyo, Japan).
DNA binding studies
DNA from calf thymus was purchased from Sigma Aldrich (Saint Louis, MO, USA).
UV-Vis study
The absorption titration measurements were performed using a Thermo Scientific spectrophotometer from 800 nm to 200 nm. The DNA concentration was fixed at 20 ppm in phosphate buffer solution (PBS) at pH 7, and titration was done with different aliquots of DE-EDCP standard solution to make concentrations of 0, 20, 40, 60, 80, and 100 ppm. After preparing DE-EDCP–DNA solutions, they were allowed to incubate for 5 min at room temperature before the absorption spectra were recorded.
CV interaction study
CV measurements were done in the same concentration range 0–100 ppm of the DE-EDCP presented in the PBS of 20 ppm of DNA. The voltammetric measurements were performed using a potentiostat/galvanostat (CH Instruments, Austin, USA) controlled by the corresponding electrochemical software. The cell (5 mL) consisted of a three-electrode system, a boron-doped diamond electrode (Windsor Scientific, UK; doping level of 1000 ppm) as a working electrode, a silver (Ag)/silver chloride (AgCl; saturated potassium chloride (KCl)) as a reference electrode, and a Pt wire as a counter electrode. All potentials reported in this article are referred versus the Ag/AgCl (saturated KCl) reference electrode. Also, all experiments were done at ambient temperature after 5 min of incubation period.
Statistical analysis
Data were expressed as mean ± standard deviation unless otherwise stated. Statistical analyses were performed using SPSS v.22 (SPSS, Chicago, Illinois, USA). The difference between experimental and control groups was separately evaluated in males and females. The homogeneity of variance and normality distribution were tested with Levene’s and Kolmogorov–Smirnov tests, respectively. The independent Student’s t-test was used to compare any significant difference between two groups. One-way analysis of variance followed by Dunnett’s post hoc test was used for statistical comparison between control- and various treated groups. The p value of 0.05 or less was considered to be statistically significant.
Results
Acute toxicity study
The acute toxicity of DE-EDCP was studied in adult NMRI Han mice. LD50 values were calculated by probit analysis within 95% confidence limits. The percentage mortality values are plotted against log doses (Figure 2) and then the dose corresponding to probit 5, that is, 50% was determined, and the results are shown in Table 1. LD50 were 95.3 and 101.3 mg/kg bw for female and male mice, respectively.

Graph showing log doses versus probit from Table 1 for the calculation of i.v. median lethal dose of DE-EDCP in (a) female NMRI Han mice (median lethal dose, LD50, using probit analysis is 95.3 mg/kg) and (b) male NMRI Han mice (median lethal dose LD50 using probit analysis is 101.3 mg/kg). i.v.: intravenous; DE-EDCP: O,O′-diethyl-(S, S)-ethylenediamine-N,N′-di-2-(3-cyclohexyl) propanoate dihydrochloride.
Determination of LD50 values by Miller and Tainter method of DE-EDCP in mice.

DE-EDCP: O,O′-diethyl-(S, S)-ethylenediamine-N,N′-di-2-(3-cyclohexyl) propanoate dihydrochloride; i.v.: intravenous.
a Corrected formula: for the 0% dead: 100(0.25/n). For the 100% dead: 100[(n-0.25)/n], where n is the number of animals in each group (n = 6).
After i.v. injection, 100% death was recorded for all the animals that received 150 mg/kg bw of DE-EDCP, while 33.3 and 16.7% death, respectively, for female and male mice that received 100 mg/kg bw of DE-EDCP died within 4 days. There was no death among the animals that received 25 and 50 mg/kg bw of DE-EDCP.
The i.v. injection of DE-EDCP up to 50 mg/kg bw did not cause any death in mice and there were no signs of obvious behavioral and physical adverse effects. There were no observed statistically significant differences in survival between the male and female mice.
The clinical sign observed at higher doses (>100 mg/kg bw) 6 h after treatment was accelerated breathing. This was followed by decreased activity, reduced activity on the external stimulus, loss of appetite with consequent reduction in feeding, cachexia, dehydration, coma, and death within 12 h to 4 days depending on the dosage. Shaky movement and unsteady gait (ataxia) were also observed in majority of the dosed animals. No diarrheic feces were observed during the experiments.
Gross examination of all organs of acute death mice revealed swollen and congested liver, with irregular light areas indicating the fatty changes, pronounced blood vessels and spotted bleeding in the pancreas, and slightly lighter kidneys compared to the control group.
The remaining female and male mice in the 100 mg/kg bw dose group survived 14 days post dose. A statistically significant decrease in final bw gain in this group compared to control group was noted (p < 0.05). During the study period, it could be seen scratching marks on their skin and hyperactivity. The liver and kidney organ alterations in the form of light discolored areas and changes in hardness were considered to be biologically significant. No gross abnormalities in size, color, and consistency throughout the experiment in any other organ compared with those of the controls were noticed.
Subacute toxicity study
For the subacute toxicity study, the mice were given daily i.v. injections of 15, 25, and 40 mg/kg bw of DE-EDCP for 28 days (treated groups).
Mortality and clinical signs
The treated mice (female and male) were found to be healthy with no differences being noted with respect to the control group. There were no changes observed in their behavior and locomotor activity. There was the absence of any visual sign of intoxication during the 28-day period. As well, in the satellite group, no reversibility or persistence of any toxic effects was observed. There were no changes observed in morphological characteristics of skin, eyes, nose, ears, and fur. All animals showed normal nutritional status.
Effect of DE-EDCP on food and water intake and bw
Food and water were freely available and consumption was checked daily. Daily food and water consumption of DE-EDCP-treated groups was found to be insignificant in both sexes when compared to the control mice (p > 0.05). Details of food and water consumption in all treated, satellite, and control groups were considered in Table 2.
Daily food and water consumption of mice treated with DE-EDCP.a,b

DE-EDCP: O,O′-diethyl-(S, S)-ethylenediamine-N,N′-di-2-(3-cyclohexyl) propanoate dihydrochloride; SD: standard deviation.
a Values are expressed as mean ± SD, n = 6 mice/sex/group.
b No significant difference from the control group at p > 0.05.
Decrease in bw gain is one of the indicators of adverse effect. Nonsignificant (p > 0.05) increase in bw was observed in all treated and satellite groups compared with the control group. Bws of female and male mice in treating group with 40 mg/kg bw of DE-EDCP recorded over a period of 28 days and compared with control group are shown in Figure 3, respectively. No significant differences in bw were observed between satellite and control groups (p > 0.05) during the study period. Hence, we can assume that DE-EDCP in concentration up to 40 mg/kg bw does not have a general toxic effect and influence animal appetite and, therefore not affect the normal growth of mice.

Effects of DE-EDCP (40 mg/kg bw) on the bws in the subacute toxicity testing (treated group and control group): (a) female mice and (b) male mice. The values are expressed as mean ± SD (n = 6 animals/group). DE-EDCP: O,O′-diethyl-(S, S)-ethylenediamine-N,N′-di-2-(3-cyclohexyl) propanoate dihydrochloride; bw: body weight; SD: standard deviation.
Hematological parameters
The effects of subacute administration of DE-EDCP on hematological parameters are presented in Table 3. Most hematology measures (HGB, total RBCs, RBCs distribution width, total WBCs, NEUT, LYMPH, MONO, and PLT) in both sexes of mice treated with 40 mg/kg bw and satellite groups were not significantly different from the control group (p > 0.05). Hematological analysis also showed no significant change in treatment groups (15 and 25 mg/kg bw) as compared to the control group.
Effect of DE-EDCP on hematological parameters in treating and satellite groups compared with those of the control group.a–d

DE-EDCP: O,O′-diethyl-(S, S)-ethylenediamine-N,N′-di-2-(3-cyclohexyl) propanoate dihydrochloride; RBC: red blood cell count; WBC: white blood cell count; HGB: hemoglobin; HCT: hematocrit; MCV: mean corpuscular volume; MCH: mean corpuscular HGB; MCHC: mean corpuscular HGB concentration; RDW: red cell distribution width; PLT: platelet count; MPV: mean platelet volume; LYMPH: lymphocytes; MONO: monocytes; NEUT: neutrophils; SD: standard deviation.
a Values are expressed as mean ± SD, n = 6 mice/sex/group.
b A treated group: mice were treated with DE-EDCP at 40 mg/kg/day for 28 days.
c A satellite group was treated with DE-EDCP at 40 mg/kg/day for 28 days followed by no treatment for 14 days.
d There is no significant difference from the control group at p > 0.05.
Clinical biochemistry
Serum samples of animals in all treated and satellite groups were analyzed and compared at each end point with the controls. Table 4 presents the effects of i.v. administration of 40 mg/kg bw of DE-EDCP on serum biochemical parameters in both sexes of mice. A significant (p < 0.05) increase in urea and ALT levels in female mice and in AST and ALP levels in both genders was recorded in only 40 mg/kg bw treated group compared to those of the control group. In the male mice of satellite group, analysis showed only significant increase in AST levels. Other parameters did not show significant differences from the control group (p > 0.05).
Biochemical parameters induced by 40 mg/kg bw dose of DE-EDCP in treating and satellite groups compared with those of the control group.a–c

DE-EDCP: O,O′-diethyl-(S, S)-ethylenediamine-N,N′-di-2-(3-cyclohexyl) propanoate dihydrochloride; AST: aspartate aminotransferase; ALP: alkaline phosphatase; ALT: alanine aminotransferase; bw: body weight; SD: standard deviation.
a Values are expressed as mean ± SD, n= 6 mice/sex/group.
b A treated group: mice were treated with DE-EDCP at 40 mg/kg/day for 28 days.
c A satellite group was treated with DE-EDCP at 40 mg/kg/day for 28 days followed by no treatment for 14 days.
d p < 0.05 indicates significant variance in comparison with control group.
e p < 0.05 indicates significant variance in comparison with control group.
f p < 0.05 indicates significant variance in comparison with satellite group.
No statistically significant differences in serum biochemical parameters in the groups receiving 15 and 25 mg/kg bw of DE-EDCP as compared with the control group were noted.
Relative organ weights and gross pathology
No significant changes (p > 0.05) of relative organ weights of the vital organs were observed in the all treated and satellite groups compared with the control group at necropsy.
Changes in color, size, shape, hardness of organs, hypertrophy, atrophy, necrosis, and hemorrhage are a first-hand indication of toxicity of some chemical compound. The macroscopic examinations of the organs of mice treated with various doses of DE-EDCP did not show any of these changes in comparison with organs of control group.
Histopathological examination (H&E stain)
The microscopic examination revealed that none of the organs in the male and female groups treated with 15 and 25 mg/kg bw of DE-EDCP showed any alteration in cell structure or any unfavorable effects when viewed under the light microscope using multiple magnification powers.
The histopathological findings of high-dose (40 mg/kg bw) group exhibit microscopic changes in the kidneys tissue and liver tissue in the majority of both the female- and male-treated mice when compared to the control groups. Light microscopic examination of sections of kidney and liver of the control group showed a normal histology (Figure 4(a) and (b)).
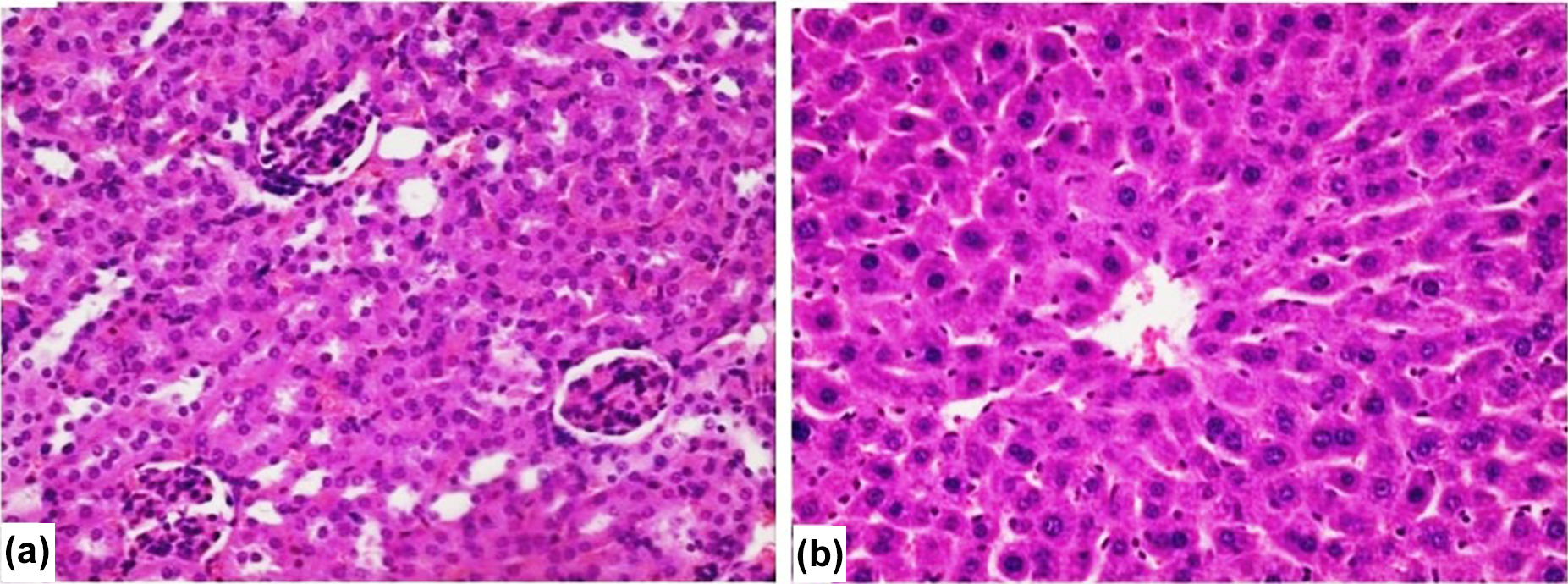
Photomicrographs of mouse kidney (a) and liver (b) structure of control group, H&E stain. (a) The normal structure of the kidney with glomerulus and proximal convoluted tubules around them. (b) The normal structure of the liver with hexagonal-shaped lobules with a central vein in the middle and trabecular arrangement of hepatocytes ((a) and (b) ×400). H&E: hematoxylin and eosin.
The main histopathological kidney change was parenchymatous degeneration of the proximal convoluted tubules near the glomeruli, which is characterized by increasing water content in the epithelial cells that line these tubules. It results in a weaker H&E staining of these cells, less visible intercellular borders and wrinkled apical surface, as well as in narrow lumens (Figure 5(a) to (c)). This histopathological change was found in kidney tissue of all 12 animals (12/12) of 40 mg/kg bw group. Also, infiltration of LYMPH and plasma cells around the collecting ducts was found in kidney tissue of 4 animals (4/12) of the same group (Figure 5(d)). No significant differences in kidney changes were observed between male and female mice (p > 0.05).

Photomicrographs of rat kidney structure from 40 mg/kg bw of DE-EDCP-treated group, H&E stain. (a) to (c) Parenchymatous degeneration of the narrow lumens proximal convoluted tubules near the glomeruli characterized by weaker H&E staining of the epithelial cells, less visible intercellular borders, and wrinkled apical surface. (d) Infiltration of LYMPH and plasma cell around the collecting ducts ((a) to (d) ×400). DE-EDCP: O,O′-diethyl-(S, S)-ethylenediamine-N,N′-di-2-(3-cyclohexyl) propanoate dihydrochloride; bw: body weight; H&E: hematoxylin and eosin; LYMPH: lymphocytes.
The liver tissue histopathological changes were microvesicular fatty changes found in 9 animals (9/12) of the treated group (Figure 6(a)), focal hepatocyte necrosis with neutrophil infiltration found in 8 animals (8/12; Figure 6(b)), LYMPH and plasma cell infiltration of portal space found in 6 animals (6/12) (Figure 6(c)), and sporadic congestion found in only 1 female mice (1/12; Figure 6(d)). Similarly, no significant (p > 0.05) histopathological changes were observed in the liver tissue of the female mice when compared to the male mice.

Photomicrographs of rat liver structure from 40 mg/kg bw of DE-EDCP-treated group, H&E stain. (a) Microvesicular fatty changes of hepatocytes. (b) Focal hepatocyte necrosis with neutrophil infiltration. (c) LYMPH and plasma cell infiltration of portal space. (d) Portal triad vein congestion ((a) to (d) ×400). DE-EDCP: O,O′-diethyl-(S, S)-ethylenediamine-N,N′-di-2-(3-cyclohexyl) propanoate dihydrochloride; bw: body weight; H&E: hematoxylin and eosin; LYMPH: lymphocytes.
The microscopic examination revealed that all other organs from the treated female and male mice did not show any alteration in cell structure or any unfavorable effects when viewed under the light microscope compared to controls.
In the satellite group of both sexes, the changes in kidney and liver were less pronounced, suggesting their reversibility. Parenchymatous degeneration of the proximal convoluted tubules was found in 3 (3/12) animals of satellite group, but their lumens were less narrow compared with the 40 mg/kg bw group (Figure 7(a)). Infiltration of LYMPH and plasma cells around the collecting ducts was found in only 1 animal (1/12) of the same group.

Photomicrographs of rat kidney (a) and liver (b) structure of satellite group, H&E stain. (a) Less pronounced parenchymatous degeneration of the proximal convoluted tubules with less narrow lumens and several normal tubules. (b) Rare microvesicular fatty changes and LYMPH and plasma cell infiltration of portal space ((a) and (b) 400×). H&E: hematoxylin and eosin; LYMPH: lymphocytes.
The microscopic liver findings of satellite group showed discrete and rare microvesicular fatty changes at 3 animals (3/12), and LYMPH and plasma cell infiltration of portal space found in 2 animals (2/12; Figure 7(b)), without focal hepatocyte necrosis and congestion.
Also, other organs (brain, lungs, heart, spleen, pancreas, stomach, and intestines) did not show any treatment-related histopathological changes. There was no inflammation, necrosis, hemorrhage, or cellular abnormalities (deposits, degeneration, vacuoles, etc.).
The UV-Vis monitoring of the DE-EDCP—DNA interaction was carried out by varying the concentration of DE-EDCP with a fixed concentration of calf thymus DNA. The UV absorption spectra show maxima at around 260 nm. Corresponding spectra are presented in Figure 8. Obviously, the increase of the concentration of DE-EDCP is followed by an increase of the absorbance maximum at selected wavelengths, finally resulting in hyperchromic effect.

Adsorption spectra of 20 ppm DNA in presence, increasing amounts of DE-EDCP from 20 ppm to 100 ppm. Inset figure presents CV voltammograms for the same conditions. DE-EDCP: O,O′-diethyl-(S, S)-ethylenediamine-N,N′-di-2-(3-cyclohexyl) propanoate dihydrochloride; CV: cyclic voltammetry.
Representative cyclic voltammograms obtained after successive addition of DE-EDCP in the solution containing 20 ppm of DNA are shown as an inset of Figure 8. It is noticeable that increases of the DE-EDCP concentration are connected with the increase of the peak current at a potential of around 1.3 V, which is attributed to the oxidation of adenine base.
Discussion
According to literature data, compound DE-EDCP tested in vitro was very effective against four tumor cell lines: C6 and U251 glioma, L929 fibrosarcoma, and B16 melanoma. 19 Its antitumoral activity was comparable or superior to the antitumor action of cisplatin. Compared with cisplatin activity, it showed less toxicity toward nonmalignant counterparts.
As results of in vitro antitumor potential of DE-EDCP exhibited the high cytotoxic activity, the aim of this study was to investigate its in vivo behavior, that is, the acute and subacute toxicity in laboratory NMRI Han mice.
Acute toxicity studies of DE-EDCP were done as per OECD guidelines. According to the OECD, the preferred rodent species for such a study is a rat, although other rodent species may be used. In this i.v. acute toxicity study, the NMRI Han mice were employed to observe the toxicity effects of DE-EDCP. Since the available literatures suggest that the females are a little bit more sensitive in such studies, the OECD recommends the usage of female rats. However, in our experiment, animals of both sexes were used. The i.v. route of DE-EDCP administration was employed because oral administration of DE-EDCP is limited by problems related to its physicochemical properties, including instability and rapid metabolism. 35
The results of the acute study provide useful preliminary toxicity data to determine appropriate dose levels for repeated-dose toxicity studies as well as for determining possible target organs to be examined, more closely in toxicity studies of a longer duration. Determination of LD50 is a first step which provides information on health hazards likely to arise from short-term exposure to drugs. For the acute toxicity study, it was observed that the LD50 values of DE-EDCP in female and male mice were found to be 95.3 and 101.3 mg/kg bw intravenously, respectively. In line with the objectives of such study, this work has shown that mice are susceptible to the DE-EDCP and the organs most affected were liver, pancreas, and kidneys. The results of acute toxicity also show that DE-EDCP does not cause any higher toxic effect on the liver and kidney tissues at low (25 mg/kg bw) and moderate (50 mg/kg bw) doses, but could have any deleterious effects on those tissues in higher doses (>100 mg/kg bw).
In terms of toxicity, DE-EDCP is categorized into class, namely, moderately toxic. 36 According to this classification, the moderately toxic compound exhibits lethal doses of between 20 mg/kg bw and 700 mg/kg bw.
The subacute toxicity studies are aimed at finding out the toxic effect of a drug on prolonged exposure. It also gives the valuable information about the delayed effect that might be the result of cumulative effect of chemicals. This study is designed to understand the level of safe usage of DE-EDCP. Detailed clinical observations and pathology examinations were conducted.
The subacute toxicity study was carried out with doses 15, 25, and 40 mg/kg bw of DE-EDCP administrated i.v. to both sexes of mice once daily for 28 days. These doses were taken for the study because acute toxicity studies have shown that up to this concentration, there were no changes in bw, food and water consumption, and hematological parameters when compared with the control group. Changes in bw have been used as an indicator of adverse effects of drugs and chemicals.
An additional satellite group treated with 40 mg/kg bw of DE-EDCP was used for observation of reversibility, persistence or delayed occurrence of systemic toxic effects, and recovery from toxic effects, for at 14 days after treatment. This group was treated and housed under the conditions identical to those of the animals from treated groups. The satellite group in this study was i.v. administered with the DE-EDCP at a daily dose of 40 mg/kg/day for 28 days and no further treatment for the following 14 days before the termination of the study.
Administration of DE-EDCP for 28 days produced no clinical signs for toxicity or mortality in either sex. The food intake and water intake were showing similar pattern in all the groups, including control and three treatment groups as well as satellite group. No abnormal deviations were observed.
The hematopoietic system is one of the most sensitive systems to assess the toxicity of drugs in humans and animals. 37 The present study indicated that there was no significant difference in any of hematological parameters between the control group and 15, 25, and 40 mg/kg bw treated groups as well as satellite group, indicating that these doses of DE-EDCP didn’t have any significant effect on the circulating blood cells or on their production, suggesting its very low hematotoxicity.
The liver is the main organ in the detoxification and metabolism of chemicals which may impair its regular function due to xenobiotic modification in detoxification processes. ALT and AST are important enzymes present in hepatocytes and usually help to detect chronic liver diseases by monitoring their concentrations. 38 Any damage to the liver results in elevations of both ALT and AST in the blood. ALT, a better bioindicator of liver injury than AST, found in serum is taken as a first sign of cell and liver damage. 36,39 It catalyzes the conversion of alanine to pyruvate and glutamate. 40 There were significant changes (p < 0.05) in the serum levels of AST and ALT in the 40 mg/kg bw dose group. We observed an increase of serum AST in both genders and serum ALT in female mice compared to control. These results suggest that this dose of DE-EDCP altered the hepatic function and may be harmful to the liver what is confirmed by pathohistology findings of focal hepatocyte necrosis and microvesicular fatty changes primarily, as well as LYMPH and plasma cell infiltration of portal space. However, the results of biochemical analysis in satellite group showed that, except AST in male, all other parameters are at a normal level. This fact indicates that some of the processes are reversible. At a dose of 40 mg/kg bw of DE-EDCP, there was no significant difference in total bilirubin between treated and control groups.
No statistically significant differences in liver function parameters were noted in the treatment groups with 15 and 25 mg/kg/day of DE-EDCP.
Renal function was evaluated by urea, creatinine, and uric acid serum levels. Urea and creatinine are sensitive markers of kidney alterations, especially when these markers are increased concomitantly. Creatinine is known as a good indicator of renal function. Rise in creatinine means there is an obvious damage to functional nephrons. 41 In the female treatment group with 40 mg/kg bw, serum urea was slightly elevated compared with the control group, but in a satellite group, its level was decreased to the normal value.
Also, ALP is one of the biomarkers for detecting renal tubular injury. The high enzyme activity of ALP was found in the cells of proximal convoluted tubules. 42 The increased activity of this biomarker, detected in serum shortly after tubular injury, indicated its release from proximal tubular cells and suggests predicting acute renal failure. 43 An increase in ALP may also be a sign of some primary bone (osteoporosis, osteosarcoma, bone metastasis, Paget disease) or diseases with bone damages (hyperparathyroidism) as well as liver diseases (hepatitis, cirrhosis, hepatocellular carcinoma). 44 All of these diseases were excluded at first by gross examination of those organs and after that by their histopathological analysis. According to all these, increased by ALP value indicates renal tubular injury.
In our study, ALP was elevated in both genders which received the dose of 40 mg/kg bw but fact that in satellite group, this parameter decreased to normal range, suggests reversible nature of these changes. This is also in accordance with histopathological findings of tubular injury caused by parenchymatous degeneration of the proximal convoluted tubules. The kidney function parameters (creatinine and uric acid) did not reveal any relevant changes following the administration of DE-EDCP. Insignificant difference (p > 0.05) between serum biochemistry of control and the female and male treatment groups with 15 and 25 mg/kg/day may be an indication of the inability of DE-EDCP to change biochemical parameters investigated. These results correlate well with the findings from histopathological examination of the kidneys and liver. The results of the histopathological assessment showed no significant histopathological change in the internal, especially vital, organs in any of the treatment groups with DE-EDCP at the doses of 15 and 25 mg/kg/day.
In this study, we monitored changes in adsorption properties of DNA after the addition of DE-EDCP. According to obtained results, it can be concluded that hyperchromic effect obtained during the addition of DE-EDCP in a solution containing a constant amount of DNA is followed by an increase in absorbance of DNA, which occurs upon DNA denaturation. It is known that two strands of DNA are held together by stacking interaction and hydrogen bonds. However, after the addition of DE-EDCP as a denaturing agent, these interactions are disrupted. This kind of interactions of DE-EDCP-DNA can be explained as electrostatic binding or partial uncoiling of the helix structure as the absorbance of single-stranded DNA is around 40% higher than double-stranded DNA in the same concentration. Similar results are observed with CV measurements. Under used experimental conditions, one well-defined peak was observed in solution containing only DNA at a potential of >1.30 V which is attributed to the oxidation of DNA bases. A constant increase of peak current can be explained by the increase of its concentration due to loss of the helix structure of nature DNA. In CV studies, the higher the current increase was noted as well as slight shifts in the spectral profile obtained by UV-Vis technique in the blue region which is dominant characteristics of such processes. 45
Conclusion
In conclusion, this study presented the results of the acute and subacute toxicity of DE-EDCP, a compound with antiproliferative activity in vitro on different tumor cell lines. Single i.v. administrations of DE-EDCP 25–150 mg/kg bw induced dose-dependent increase in general behavioral abnormalities in mice. The mortality rate also increased with increasing dosage. The median lethal dose, LD50, in female and male mice was found to be 95.3 and 101.3 mg/kg bw, respectively. Daily single i.v. doses of DE-EDCP 15, 25, and 40 mg/kg bw were observed to be well tolerated behaviorally after 28 days of dosing and induced no significant changes in body and organ weights of mice. Further, a mild, reversible, rise in the levels of some serum biochemical parameters (ALP, AST, and ALT) and histopathological changes in liver tissue and kidney tissue were noted at 40 mg/kg bw dose of DE-EDCP. In the male mice of satellite group, analysis showed only significant increase of AST levels. Other parameters did not show significant differences from the control group (p > 0.05). Since there were no signs of toxicity with respect to biochemical parameters, and gross and histopathological examinations noted in satellite group, it can be inferred that the DE-EDCP will not produce delayed onset of toxicity.
Conformational changes and partial uncoiling in the DNA structure, due to DE-EDCP–DNA interaction, were shown using two different techniques, electrochemical and spectroscopic. Hyperchromic shifts together with increases of the peak currents indicate electrostatic mode of interaction, which is often followed with DNA denaturation.
However, further chronic toxicity, genotoxicity, and reproductive toxicity studies are necessary to reevaluate these results.
Footnotes
Acknowledgements
The authors are thankful to Dr Vesna Piperski, Galenika a.d., Batajnicki drum bb, Belgrade reported on the histopathological examination of the tissues. The authors are further grateful to Božidar Lalović and Ljubinka Vasilijević, Galenika a.d., Batajnicki drum bb, Belgrade, for technical assistance and valuable comments.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Ministry of Education, Science and Technological Development, Republic of Serbia (grant numbers III45015 and 172035].
