Abstract
Background:
Previous studies have demonstrated that platelet activation occurs in patients with aortic stenosis (AS). This study sought to evaluate the changes in hematologic and clinical parameters noted with the improvement in AS following transcatheter aortic valve implantation (TAVI) in patients with severe AS at high risk of surgery.
Patients and Methods:
The study included 33 patients who underwent TAVI. In addition to biochemical, clinical, and echocardiographic examinations, hematologic blood parameters were recorded before TAVI, at discharge, and at 1 and 4 months.
Results:
Mean platelet volume (MPV) showed a progressive decrease after TAVI. On echocardiography at 1 month, aortic valve area significantly increased, with significant decreases in peak and mean gradients. Progressive decreases were also noted in N-terminal proB-type natriuretic peptide levels.
Conclusion:
Our findings show that TAVI improves hemodynamic parameters of the valve with marked clinical and echocardiographic improvement, resulting in decreased platelet activation and MPV in patients with severe AS.
Introduction
As life expectancy increases, so does the number of patients with aortic stenosis (AS) owing to aging population. Transcatheter aortic valve implantation (TAVI) has emerged as an alternative to surgical aortic valve replacement for patients with severe symptomatic AS, who are considered to be at very high or prohibitive operative risk. 1,2
There are studies showing that there is an association between AS and mean platelet volume (MPV). 3 Evidence from several studies has shown that shear stresses in turbulent flow as a result of stenotic valves induce platelet activation. 4 –9 Mean platelet volume is a marker of platelet size and activation. Increased MPV reflects active and large platelets. Large platelets that contain more dense granules are metabolically and enzymatically more active than small ones and have a higher thrombotic potential. 10,11
The aim of this study was to evaluate follow-up clinical, echocardiographic, and hematologic parameters especially platelet activation after TAVI in patients having severe AS and high surgical risk. Hematologic parameters especially platelet activation have not been studied following TAVI.
Patients and Methods
Study Population
This study included 33 patients (15 males, 18 females; mean age 77.3 ± 5.9 years) undergoing TAVI between October 2010 and February 2012. All the patients had follow-up data at 1 month and at 4 months. In all, 27 patients received balloon-expandable Edwards SAPIEN (Edwards Lifesciences, Irvine, California) and 6 patients received self-expanding CoreValve (Medtronic, Minneapolis, Minnesota) prostheses by the transfemoral (n = 32) and transapical (n = 1) approaches.
All patients had severe AS and New York Heart Association (NYHA) class III or IV symptoms and were at high risk of surgery due to comorbidities such as chronic obstructive pulmonary disease, pulmonary hypertension, peripheral artery disease, or low ejection fraction. The decision for TAVI was rendered by a consensus at the heart team meeting. Preoperative risk was assessed on the basis of the European System for Cardiac Operative Risk Evaluation (EuroSCORE) or The Society of Thoracic Surgeons (STS) risk calculator systems. 12,13 In the absence of other contraindications to surgical valve replacement, high-risk status was defined as a logistic EuroSCORE of >20% or an STS score of >10%.
Hypertension was defined as the use of antihypertensive medications or detection of systolic blood pressure >140 mm Hg or diastolic blood pressure >90 mm Hg in at least 2 separate measurements. Diabetes mellitus was defined as a previous diagnosis with the use of diet or antidiabetic drugs or a fasting venous blood glucose level of 126 mg/dL on 2 occasions in previously untreated patients. Body mass index was calculated as weight in kilograms divided by the square of the height in meters. Contrast-induced nephropathy was defined as an increase in serum creatinine level ≥0.5 mg/dL or ≥25% from baseline (admission) within 72 hours of radiocontrast administration.
Exclusion criteria were as follows: a narrow or too wide annulus of the aortic valve (≤18 mm or ≥27 mm) on echocardiography, aortic valve area of more than 0.8 cm2, short distance between the main coronary artery and aortic valve (for Edwards SAPIEN valve <8 mm, CoreValve <14 mm), severe left ventricular systolic dysfunction (ejection fraction <20%), acute coronary syndrome, history of renal or liver disease, malignancy, hematologic disorders, acute or chronic infection, and life expectancy of less than 12 months due to noncardiac causes. The study was approved by the local ethics committee, and all patients gave informed consent.
Severity of AS, aortic valve structure, and the aortic root were evaluated by transthoracic and transeosophageal echocardiography (General Electric Vivid 7 GE Vingmed Ultrasound AS, Horten, Norway). Echocardiographic measurements were performed in the left lateral decubitus position according to the criteria of the American Society of Echocardiography. 14 Multislice computed tomography and angiography were performed for the assessment of aortic root-arch calcification, diameters of the femoral and iliac arteries, and calcifications and tortuocities. Coronary arteries were evaluated before the procedure through standard coronary angiography. Transfemoral or transapical approaches were used based on the condition and size of iliofemoral arteries and the degree of calcification. The valves and delivery systems used were of new generation, and patients were considered to be eligible for the transfemoral approach if their iliac and femoral arteries were at least 6 mm in diameter. The procedure was accomplished under general anesthesia in 18 patients and under mild sedation in 15 patients. Access was gained by a surgical cutdown in 20 patients and a percutaneous closure device (Prostar XL, Abbott Vascular, Redwood City, California) was used in 13 patients. For coronary revascularization, percutaneous coronary intervention was performed in 7 patients 3 weeks before the procedure and in 2 patients during the procedure.
Procedure
The TAVI procedure was performed in a sterile environment (catheterization laboratory) under general or local anesthesia. The femoral artery with a greater diameter and less tortuosity was selected. Two sheaths were placed in the contralateral femoral artery and femoral vein for placement of a pigtail catheter in the aorta and a pacemaker lead in the right ventricle, respectively. For a proper procedure, the balloon was predilated after passing the native valve with a straight-tip guide wire and an Amplatzer left guide catheter. During balloon predilatation, ventricular tachycardia was induced by rapid ventricular pacing, providing an optimal reduction in cardiac output by creating transient cardiac standstill. This was usually achieved at a heart rate of 200 bpm. The CoreValve or Edwards SAPIEN valves were then passed through the delivery systems and expanded at the level of the native valve. Rapid ventricular pacing was repeated at that stage in patients receiving an Edwards SAPIEN valve. Since the CoreValve device is self-expanding, there was no need to repeat rapid ventricular pacing at that stage. The aortic root and peripheral arteries were evaluated after the intervention by contrast injection into the aortic root and by peripheral angiography.
Blood Sampling
Blood samples were drawn from the antecubital vein between 08.00 and 10.00
Statistical Analysis
Statistical analysis was performed using the statistical software SPSS 17.0 for Windows (SSPS Inc, Chicago, Illinois). Data were expressed as mean ± standard deviation for continuous variables and as numbers with corresponding percentages for categorical variables. The “paired sample test” was used to compare the pre- and postprocedural results. A P value of less than .05 was considered to be significant.
Results
Baseline demographic, clinical, and procedure parameters are given in Table 1. All devices were properly positioned and valves were found to be properly functioning on postprocedural angiographic and echocardiographic evaluations. Permanent pacemaker implantation was required in 4 patients (3 CoreValve, 1 Edwards SAPIEN) due to atrioventricular conduction abnormalities. Peripheral arterial injuries occurred in 2 patients. Coronary artery occlusion occurred in a patient in the Edwards SAPIEN group. 15 No paravalvular aortic regurgitation of greater than grade +2 was observed. In all, 7 patients developed contrast nephropathy, 1 patient required temporary dialysis, and the remaining patients recovered with hydration therapy. No patients developed clinical stroke.
Demographic, Clinical, and Procedural Parameters of the Patients at Baseline (N = 33).a
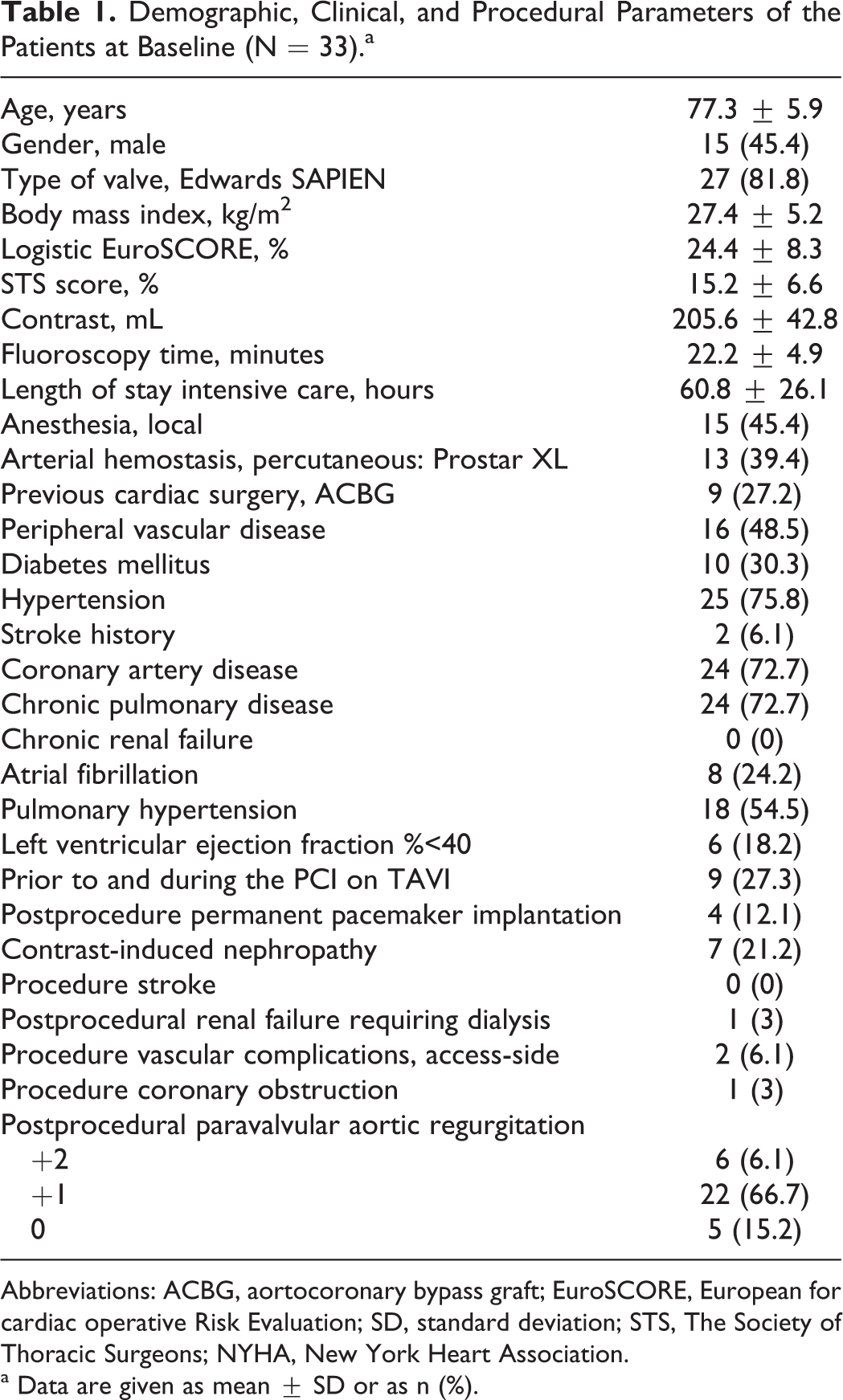
Abbreviations: ACBG, aortocoronary bypass graft; EuroSCORE, European for cardiac operative Risk Evaluation; SD, standard deviation; STS, The Society of Thoracic Surgeons; NYHA, New York Heart Association.
a Data are given as mean ± SD or as n (%).
Table 2 shows hematologic blood parameters. Compared to the baseline MPV (8.8 ± 1.1 fL), significant and progressive decreases were seen in MPV values at discharge (8.1 ± 1.2 fL), at 1 month (8.0 ± 1.2 fL), and at 4 months (7.9 ± 1.0 fL; Figure 1). Hemoglobin levels were significantly lower than baseline after the procedure, but there was a steady increase after discharge. Due to anemia, red blood cell distribution width was significantly higher at 1 month; however, it decreased at 4 months in association with the improvement of anemia. Basophil values were found to be lower at 4 months. No significant changes were observed in other hematologic parameters.
Hematologic Parameters (N = 33).a
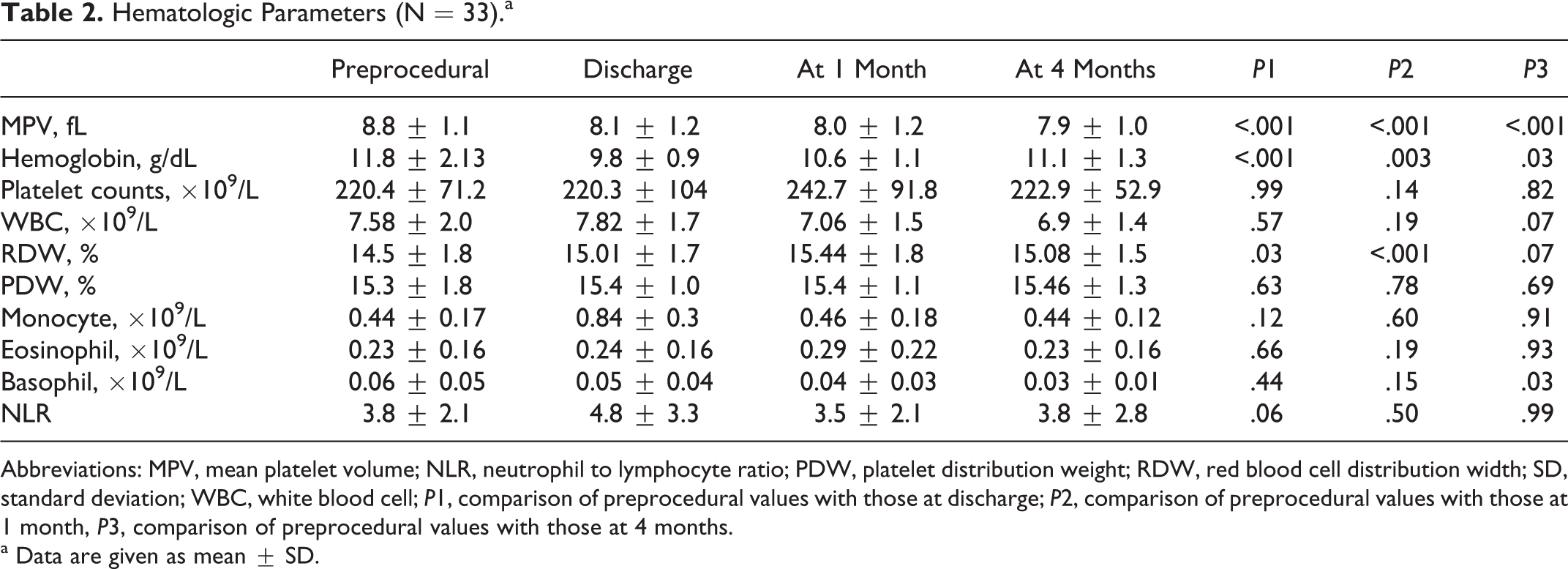
Abbreviations: MPV, mean platelet volume; NLR, neutrophil to lymphocyte ratio; PDW, platelet distribution weight; RDW, red blood cell distribution width; SD, standard deviation; WBC, white blood cell; P1, comparison of preprocedural values with those at discharge; P2, comparison of preprocedural values with those at 1 month, P3, comparison of preprocedural values with those at 4 months.
a Data are given as mean ± SD.
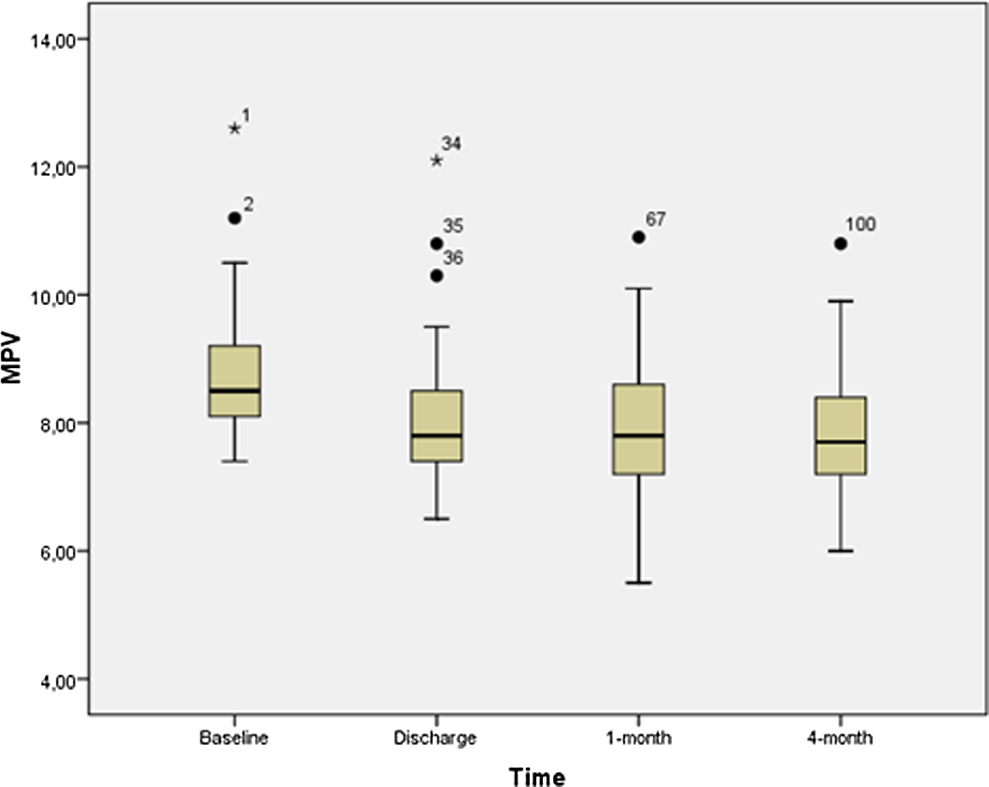
The follow-up of mean platelet volume (MPV) levels after transcatheter aortic valve implantation (TAVI).
Changes in hemodynamic and clinical parameters are summarized in Table 3. At 1-month echocardiographic follow-up, both aortic valve area and left ventricular ejection fraction significantly increased, with significant decreases in the transvalvular (peak systolic and mean) gradients. At 4-month follow-up, the overall mean NYHA functional class score decreased significantly.
Hemodynamic and Clinical Parameters (N = 33).a
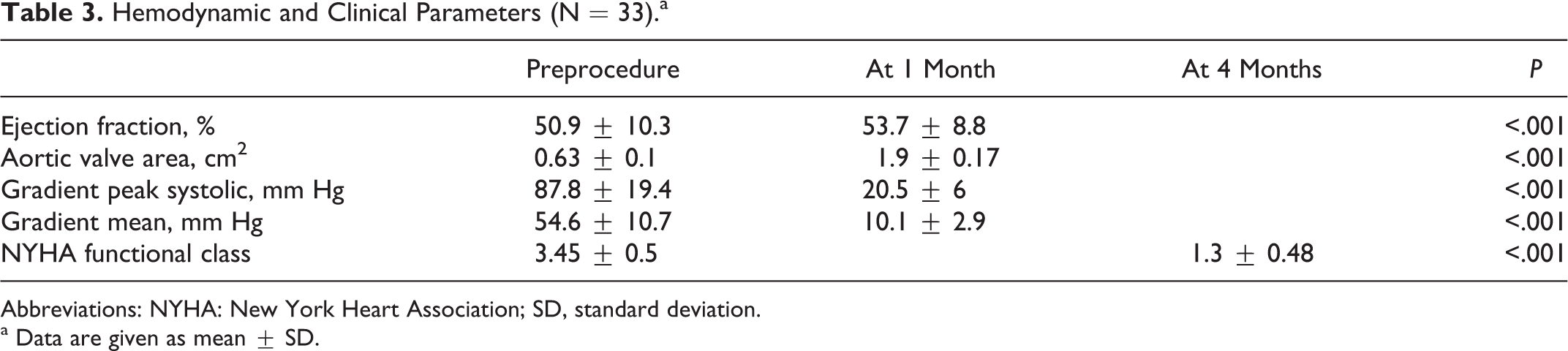
Abbreviations: NYHA: New York Heart Association; SD, standard deviation.
a Data are given as mean ± SD.
Biochemical parameters are shown in Table 4. Levels of troponin T, creatinine kinase-MB (CK-MB),
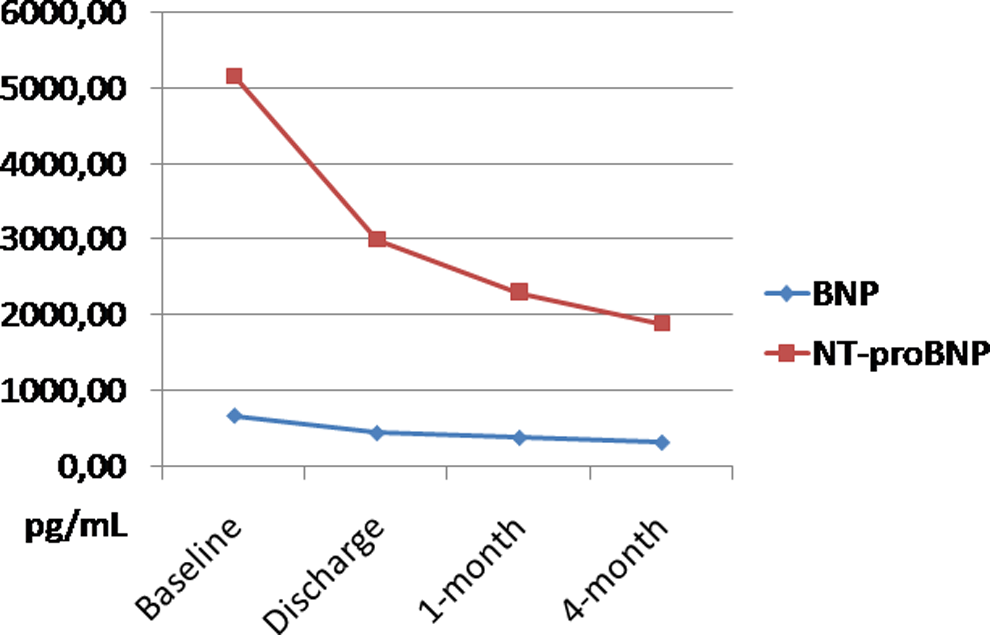
The follow-up of B-type natriuretic peptide (BNP) and N-terminal proBNP (NT-proBNP) levels after transcatheter aortic valve implantation (TAVI).
Biochemical Parameters (N = 33).a
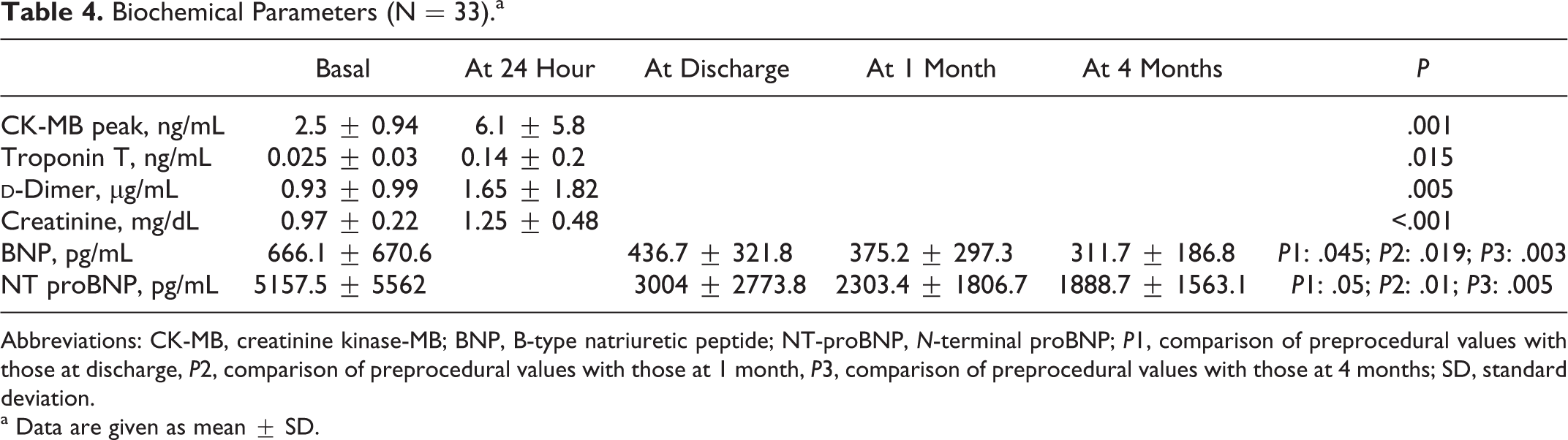
Abbreviations: CK-MB, creatinine kinase-MB; BNP, B-type natriuretic peptide; NT-proBNP, N-terminal proBNP; P1, comparison of preprocedural values with those at discharge, P2, comparison of preprocedural values with those at 1 month, P3, comparison of preprocedural values with those at 4 months; SD, standard deviation.
a Data are given as mean ± SD.
Discussion
We observed improvements in the clinical and echocardiographic parameters of our patients after bioprosthetic valve placement by means of TAVI and noted that hematologic parameters, in particular MPV, which is an indicator of platelet activation, decreased progressively.
Platelets have been found to have an association with the development of atherosclerosis and acute coronary syndromes. Larger and hyperreactive platelets accelerate intracoronary thrombus formation, leading to a cascade of clinical events, such as acute coronary syndrome. 16 Mean platelet volume is one of the important platelet production indices that may relate to platelet function. It has been shown that platelet size, measured as MPV, correlates with their reactivity. 17 Pereg et al found that MPV admission correlates with impaired response to thrombolysis in patients with ST-elevation myocardial infarction. 18 Several studies indicate that high MPV and high platelet reactivity are associated with overall vascular mortality, including myocardial infarction. Slavka et al 19 demonstrated that patients with an increased MPV were at higher risk for death due to ischemic heart disease. Bilen et al 20 found that bicuspid aortic valve was associated with elevated MPV values. In addition, they showed a significant correlation between MPV and aortic stiffness and aortic elastic modulus. Korkmaz et al 21 demonstrated a significant and independent association between coronary artery calcification and MPV. Huczek et al 22 found that MPV was a strong, independent predictor of impaired angiographic reperfusion and 6-month mortality in patients treated with primary percutaneous coronary intervention for ST-elevated myocardial infarction. Additionally, Butterworth et al 23 reported increased MPV in patients with acute ischemic stroke compared to the control group.
Previous studies found increased platelet activation in patients with AS. Stenotic valve-associated turbulent flow causes shear stresses, inducing platelet activation. 4 –9 An association between stenosis and platelet activation has been observed in other groups of valvular heart disease. Varol et al 24 showed that MPV was significantly elevated in patients with mitral stenosis and sinus rhythm compared to the controls. Mitral valve stenosis represents a relatively low-gradient stenosis, whereas AS is a high-gradient valvular defect. This indicates that AS can generate even higher blood markers of platelet activation. Varol et al 3 also found increased MPV in AS. In this study, we report for the first time that MPV values decrease as a result of improvements in severe AS following TAVI.
To our best knowledge there is no study in the literature evaluating MPV after any cardiac procedure. Platelet activation and aggregation increases after invasive procedures. Our results at discharge (avarege hospital stay duration was about 1 week after procedure) revealed decreased MPV values that is likely due to decreased turbulance and shear stress secondary to increased aortic valve area. Patients were given heparin during the procedure and because of the reasons mentioned above MPV values were not obtained immediately after the procedure.
A variety of mechanisms have been suggested to explain the association between platelet activation and adverse events. Platelets represent an important linkage between inflammation, thrombosis, and atherogenesis; in addition, they promote recruition of leukocytes and progenitor cells to sites of vascular injury and inflammation and release of proinflammatory, anti-inflammatory, angiogenic factors, and microparticles into the circulation. 25 Various types of matrix metalloproteinases have been reported to be present in platelets. 26 Platelets secrete chemokines and cytokines that mediate vascular inflammation, which in turn are activated by substances released from cells of the vascular wall. 27 Platelets are known to have a major effect on the formation of atherosclerotic plaques; therefore, they play an essential role in the pathogenesis of atherothrombosis. In a recent study, patients with aortic valve sclerosis were found to have increased platelet hyperaggregability and platelet aggregation in response to adenosine diphosphate (ADP), as well as reduced antiaggregant response to sodium nitroprusside, which is a nitric oxide donor. 28 There are also studies demonstrating an association between increased matrix metalloproteinase and calcific aortic valves. 29
In response to myocardial wall stress, BNP and NT-proBNP are released, which correlate well with pulmonary capillary pressure. 30 Sherif et al 31 showed that TAVI was associated with a greater valve area and smaller valvular gradient compared with surgical aortic valve replacement as well as with lower natriuretic peptide levels. Following TAVI, we also found increased aortic valve area and decreased gradients on echocardiography and progressively decreasing BNP and NT-proBNP levels.
Cardiac markers, including troponin T, CK-MB, and
Study Limitations
Our study has some limitations. It was a single-center study and the number of patients was small mostly due to the high number of exclusion criteria. Data on the effects of various drugs on platelet size are insufficient. Previous in vitro studies found no effect of aspirin on platelet size. 32 However, it has been shown that clopidogrel significantly inhibits ADP-induced increases in MPV in vitro. 33 The patients not receiving clopidogrel and aspirin were started on these drugs at least 10 days before TAVI, and we continued dual antiplatalet therapy for up to 6 months. Thus, there were no changes in giving platelet inhibitors during the follow-up. Some studies reported a time-dependent increase in MPV with the use of EDTA as an anticoagulant agent. 34 However, more recent studies have shown that, if measurements are performed within 2 hours after blood collection, increase in MPV will be less than 0.5 fL. 35 To minimize the effect of EDTA on platelet size, we used standardized sample tubes, and all samples were processed early (at 30 minutes) after blood collection.
Conclusion
Transcatheter aortic valve implantation has become the treatment of choice for severe AS in patients having a high risk of surgery. The clinical and echocardiographic parameters improved significantly after TAVI. In addition, we observed a progressive decrease in MPV values, suggestive of decreased platelet activation. Further studies are needed to assess the relationship of MPV with mortality. Considering its known association with AS, this study is the first to show decreased MPV following TAVI, suggesting that MPV may be used as a simple and inexpensive test for clinical and hematologic changes in TAVI.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
