Abstract
The present study investigates the potential ability of selenium, considered as an antioxidant with pharmacological property to alleviate oxidative stress and hematological parameter disorders induced by methimazole, an antithyroid drug. Pregnant Wistar rats were randomly divided into four groups of six each: group I served as negative control and received a standard diet; group II received 250 mg/L of methimazole in drinking water and a standard diet; group III received both methimazole (250 mg/L, orally) and selenium (0.5 mg/kg of diet) supplemented to the standard diet; group IV served as positive control and received a supplement of selenium in the diet (0.5 mg/kg of diet) as sodium selenite (Na2SeO3). Treatment was started from the 14th day of pregnancy until day 14 after delivery. Methimazole reduced the number of red blood cells, hemoglobin concentration and hematocrit in mothers and their pups. Besides, plasma iron, vitamins B9, B12, C and E levels were reduced. Lipid peroxidation increased, objectified by high malondialdehyde levels and lactate dehydrogenase activity in plasma, while glutathione, glutathione peroxidase, superoxide dismutase and catalase activities showed a significant decline. Co-administration of selenium through diet improved all the parameters cited above. It can be concluded that the administration of selenium alleviates methimazole-induced toxicity, thus demonstrating its antioxidant efficacy.
Introduction
Methimazole (MMI), an antithyroid drug, has been used for the management of hyperthyroidism. 1 In fact, MMI reduces the risk of teratogenic effect in pregnancy, when transmitted through placenta or milk. 2 Epidemiologic findings suggest that MMI causes anemia and agranulocytosis. 1,3 Moreover, there is the impairment of the cognitive, behavioral and physical growth of children, the immune status and morbidity due to infections. 4
Recent lines of evidence indicate that several pathological conditions, like hypothyroidism induced by MMI, are associated with oxidative stress. 5–7 The mechanism of MMI toxicity may be due, in part, to the disruption of the prooxidant/antioxidant balance, leading to tissue injury via oxidative damage to critical biomolecules such as lipids, proteins, and DNA. 5,8 The hematological system is the major target of oxidative stress. 9
Living beings have a complex of antioxidant (enzymatic and non-enzymatic) system to be protected against the deleterious effects of free radicals. Selenium, considered as a non-enzymatic antioxidant, has a large number of biological functions in humans. It is part of the active center of the glutathione peroxidase enzyme. 10,11 So, plasma glutathione peroxidase activity reflects selenium consumption. 12–15 Specifically, plasma would be a short-term indicator of selenium status as well as antioxidant defence, but erythrocytes longer-term indicators. 16 Indeed, according to Alicigüzel et al., 17 plasma antioxidant status such as glutathione peroxidase, catalase, superoxide dismutase influences the components of the erythrocytes antioxidant system. Moreover, selenium interacts with other nutrients such as vitamins E and C, overlapping its action and affecting the antioxidant/prooxidant balance of the cell. 18
Inorganic selenium compounds, used throughout our experiments as well as in other findings, 19 are generally provided as a nutritional source due to their easy absorbance by duodenum. 20 They are also transferred during gestation and lactation from mothers to fetuses/neonates. 5,21
Selenium supplementation has been proved to have protective effects against several pathological states such as MMI-induced cerebrum, cerebellum and kidney impairment, 5,8 cancer 22 and diabetes. 23 Based on these facts, we hypothesize that selenium supplementation could be a useful method to protect against hematological impairment, thus allowing a further exploration of its protective potential. This could therefore be a step towards the protection of human fetuses/neonates against hypothyroidism and its adverse effects, including hematological disorders. Consequently, the present study may open new prospects for selenium to act as a nutritional supplement for the pregnant and breast feeding MMI-treated patients, improving health and contributing to the advancement of human nutrition.
To the best of our knowledge, no investigation has so far reported the protective role of selenium against the disturbance of hematological parameters in pregnant and lactating rats submitted to MMI. The present study assesses the antioxidant and hematological protective potential of selenium against MMI-induced oxidative damage in pregnant and lactating rats and their suckling pups.
Materials and methods
Chemicals
Sodium selenite (Na2SeO3), MMI (C4H6N2S), glutathione (oxidized and reduced), nicotinamide adenine dinucleotide phosphate reduced form (NADPH), 5-5′-dithio-bis-2-nitrobenzoic acid (DTNB) and thiobarbituric acid (TBA) were purchased from Sigma (St. Louis; Missouri, USA). All other chemicals were of analytical grade and were purchased from standard commercial suppliers.
Animals
Adult Wistar rats, weighing about 140 g and aged 8–9 weeks, were purchased from the Central Pharmacy (SIPHAT, Tunisia). They were housed at ambient temperature 22 ± 3°C with a 12-hour light/dark cycle and a minimum relative humidity of 40%. Commercial diet (SICO, Sfax, Tunisia) and tap water were given ad libitum. The concentration of selenium in standard diet (0.17 mg/kg of diet) was determined, after mineralization, by the Electrothermic Atomic Absorption Spectrometry technique (ET-AAS). Measurements were performed on a Perkin-Elmer 5100/Zeeman Atomic Absorption Spectrometer with a 196-nm wavelength.
The general guidelines for the use and care of living animals in scientific investigations were followed. 24 The handling of the animals was approved by the Tunisian Ethical Committee for the care and use of laboratory animals.
Experimental design
After a 1-week acclimatization in the laboratory conditions, pairs of male and virgin female rats were kept overnight in each cage. Pregnant female rats were inspected daily by the presence of the vaginal plug, which indicated day zero of pregnancy.
Twenty-four pregnant female rats of Wistar strain were randomly divided into four groups of six each. Group I served as a negative control and received standard diet containing 0.17 Na2SeO4 mg/kg; group II received orally 250 mg/L of MMI C4H6N2S and a standard diet; animals of group III were treated orally with MMI (250 mg/L of drinking water) and 0.5 mg/kg of selenium added to the standard diet as Na2 SeO3; group IV served as positive control and received a standard diet supplemented with Na2SeO3 (0.5 mg/kg of diet). Treatments were started from the 14th day of pregnancy until day 14 after delivery. The dose of MMI and the start of treatment were chosen according to Schwartz et al., 25 since MMI induced the classical picture of hypothyroidism without lethal effects. The selenium dose (0.5 mg/kg of diet) used in our experiments and in other findings would give high protection against hypothyroidism 26 and stress conditions. 5,20
Pregnant female rats were allowed to deliver spontaneously 3 weeks after coitus. No delay and no death were observed in the delivery of the treated groups. Within 24 hours after birth, the litters were reduced to 8 pups for each mother (four males and four females if possible) to ensure standardized nutrition and maternal care. 27 So, 192 pups were sacrificed on postnatal day 14.
Biochemical assays
On day 14 after delivery, the animals were anesthetized with chloral hydrate by intraperitoneal way. Forty-eight (48) pups of each group were weighed. Blood samples were collected from the brachial artery of pups and aortic puncture of dams in 5-mL test tubes containing EDTA for determination of hematological parameters. Other blood samples were pooled into heparinized tubes and centrifuged at 2200 × g for 15 minutes. Plasma samples were withdrawn and kept at −20°C until biochemical determinations. The enzyme activities were determined within the same day of collecting in plasma diluted in phosphate buffer.
Determination of hematological parameters
White blood cells, red blood cells, hematocrit, hemoglobin, mean corpuscular volume, mean corpuscular hemoglobin and mean corpuscular hemoglobin concentration were analyzed by an electronic automate Coulter MAXM (Beckman Coulter, Inc, Fullerton, California, USA).
Assays of iron, vitamins B9 and B12
Plasma levels of iron were measured by a colorimetric method using a commercial kit (ref 61075, Biomerieux, France). Those of vitamins B9 and B12 were determined by immuno-electro-chemiluminescence analysis (Elecsys B9 and B12 immunoassays for Elecsys 2010 system, ROCHE diagnostics, USA).
Hydrophilic antioxidants
Ascorbic acid (vitamin C) determination was performed as described by Jacques-Silva et al. 28 Protein in plasma was precipitated in 10 volumes of a cold 4% trichloroacetic acid solution. An aliquot of sample (300 µL) was adjusted with H2O to a final volume of 1 mL and incubated at 38°C for 3 hours, then 1 mL of H2SO4 65% (v/v) was added to the medium. The reaction product was determined using color reagent containing 4.5 mg/mL dinitrophenyl hydrazine and CuSO4 (0.075 mg/mL). The data were expressed as µg ascorbic acid/mL.
Lipophilic antioxidants
Vitamine E (α-tocopherol) level was assayed by the extraction method of Katsanidis and Addis 29 using high-performance liquid chromatography (HPLC). Roughly, plasma was deproteinized with alcohol (methanol) containing α-tocopherol acetate as an internal standard and extraction was performed using hexane. Twenty microliters were injected into a reverse-phase analytical column (5 µm C18; 4.6 × 150 mm, Hypersil™, Supelco, Bellefonte, PA, USA). The mobile phase was methanol–water (95:5). The wavelength was programmed at 292 nm. Flow rate was 1.5 mL/min (α-tocopherol was eluted within 10 minutes, and α-tocopherol acetate was eluted at 14 minutes). The calibration curve was linear over the concentration range of 0.2–20 μg/mL. The regression equation was y = 0.0877 × x (r2 = 0.999) where x was a concentration of α-tocopherol in μg/mL. The detection limits (LOD) calculated as signal-to-noise ratio equal to 3 was 40 ng/mL.
Malondialdehyde measurement
The plasma malondialdehyde levels, a lipid peroxidation index, were determined spectrophotometrically according to Draper and Hadley. 30 Briefly, from the plasma sample, 0.5 mL was mixed with 1 mL of a solution of 5% trichloroacetic acid and centrifuged at 2500×g for 10 minutes. An amount of 1 mL of TBA reagent (0.67%) was added to 500 µL of supernatant and heated at 90°C for 15 minutes. The mixture was then cooled and measured for absorbance at 532 nm using a spectrophotometer (Jenway UV-6305, Essex, England). The malondialdehyde values were calculated using 1,1,3,3-tetraethoxypropane as standard and expressed as nmoles of malondialdehyde/mL.
Lactate dehydrogenase activity
Plasma lactate dehydrogenase activity was measured according to the method of Bergmeyer et al., 31 using commercial reagent kits purchased from Biomaghreb (ref. 20012, Ariana, Tunis, Tunisia). Lactate dehydrogenase activity was expressed as IU/L.
Glutathione peroxidase activity
The assessment of glutathione peroxidase activity was determined using a commercial kit (catalog No. RS 505; Randox, Ltd, Uk). Glutathione peroxidase catalyzes the oxidation of reduced glutathione by cumene hydroperoxide. In the presence of reduced glutathione reductase and nicotinamide adenine dinucleotide phosphate reduced form (NADPH), the oxidized reduced glutathione is immediately converted to the reduced form with a concomitant oxidation of NADPH–NADP+. The decrease in absorbance at 340 nm was measured. 32 One unit of glutathione peroxidase activity was defined as the amount of enzyme that catalyzed the oxidation by cumene hydroperoxide of 1.0 nmol of reduced glutathione to oxidized glutathione per minute at pH 7.0 and 37°C.
Total superoxide dismutase activity
Superoxide dismutase activity was measured according to the method of Beutler. 33 One hundred microliters of the plasma were mixed with 1.5 mL of a Tris–HCl buffer (pH 8.5) and 1000 µL of 15 mM pyrogallol and then incubated at 25°C for 10 minutes. The reaction was determined by adding 50 µL of 1 N HCl and the absorbance was measured at 440 nm. One unit was determined as the amount of enzyme that inhibited the oxidation of pyrogallol by 50%. The activity was expressed as U/mL.
Catalase activity
Catalase activity was assayed by the method of Aebi. 34 Enzymatic reaction was initiated by adding an aliquot of 20 µL of the homogenized tissue and the substrate (H2O2) to a concentration of 0.5 M in a medium containing 100 mM phosphate buffer, pH 7.4. Changes in absorbance were recorded at 240 nm. Catalase activity was calculated in terms of μmol H2O2 consumed/min/mL.
Glutathione levels
Plasma glutathione levels were determined by Ellman’s method, 35 modified by Jollow et al., 36 based on the development of a yellow colour when 5,5-dithiobis-2 nitro benzoic acid was added to compounds containing sulfhydryl groups. Briefly, 3 mL of sulfosalicylic acid (4%) was added to 500 µL of plasma in phosphate buffer for deproteinization and the mixture was centrifuged at 2500×g for 15 minutes; Ellman’s reagent was then added to 500 µL of supernatant. The absorbance was measured at 412 nm after 10 minutes. Total glutathione levels were expressed as µg/mL.
Statistical analysis
The data were analyzed using the statistical package program Stat view 5 Soft Ware for Windows (SAS Institute, Berkley, California, USA). Statistical analysis was performed using one-way analysis of variance (ANOVA) followed by Fisher’s Protected Least Significant Difference (PLSD) test as a post hoc test for comparison between groups (treated groups [MMI, methimazole + selenium, selenium] vs [negative controls]) and (methimazole + selenium) vs (methimazole, selenium). All values were expressed as mean ± SD. Differences were considered significant if p < 0.05.
Results
The effects of MMI on general health of rats
Death was not observed during the experimental period (22 days). However, few clinical signs such as reduced activity, increasing weakness and slight diarrhea were observed in MMI-treated group.
Hematological parameters
Compared with the control group, red blood cells' number, hemoglobin concentration and hematocrit were reduced by 52%, 23% and 25% in pups and by 31%, 30% and 31% in their mothers exposed to MMI treatment, respectively. Other erythrocyte parameters such as mean corpuscular volume, mean corpuscular hemoglobin and mean corpuscular hemoglobin concentration were unchanged in mothers. Whereas, in their progeny, mean corpuscular volume and mean corpuscular hemoglobin increased by 35% and 37%, respectively. White blood cells increased in mothers, indicating an activation of their defense mechanism and were unchanged in their pups compared with those of the control group (Table 1 ). Co-treatment with selenium improved all the parameters cited above to near normal values.
Hematological parameters of mothers and their offspring controls or treated with methimazole (MMI), methimazole + selenium (MMI + Se) or selenium (Se) from the 14th day of pregnancy until day 14 after delivery
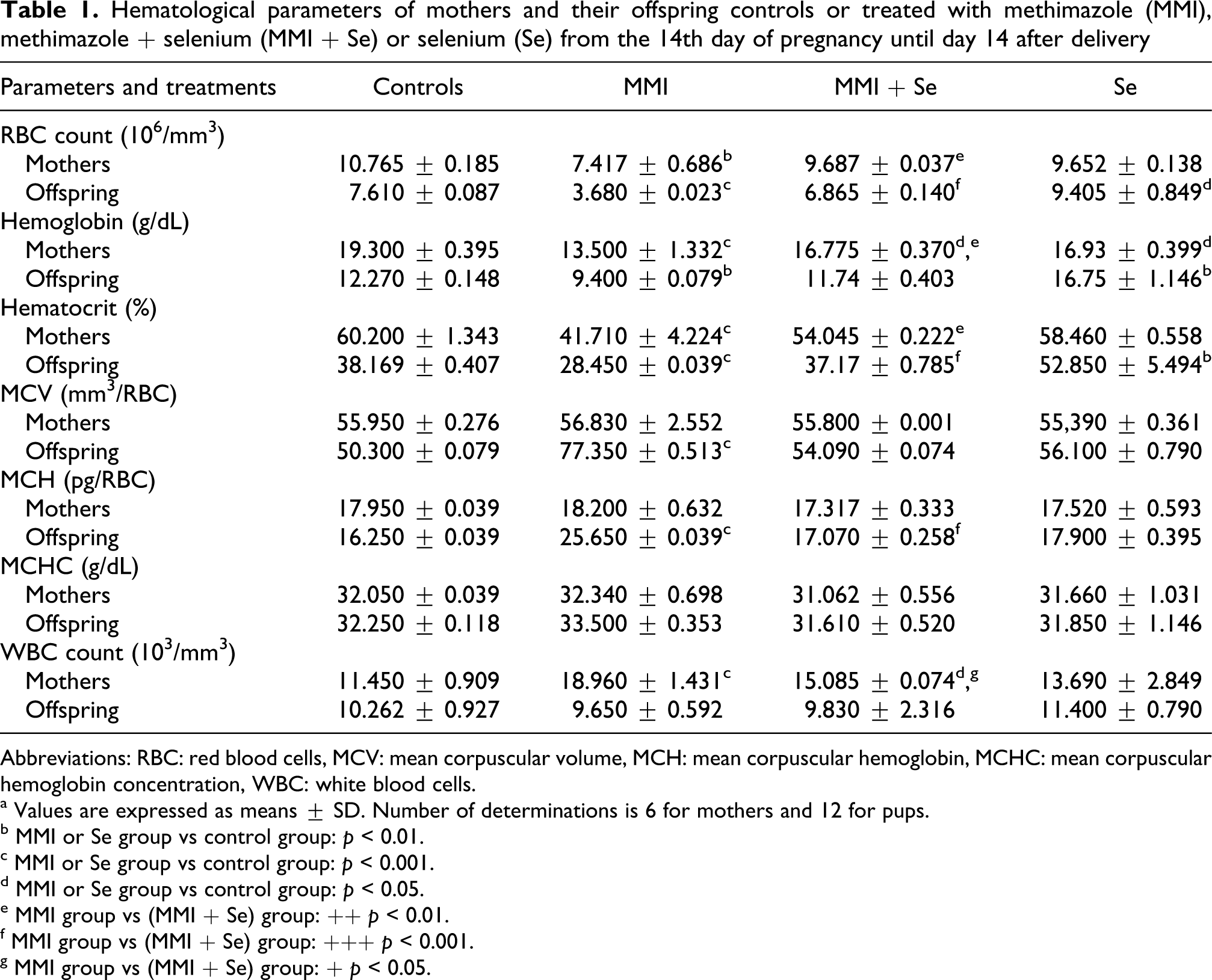
Abbreviations: RBC: red blood cells, MCV: mean corpuscular volume, MCH: mean corpuscular hemoglobin, MCHC: mean corpuscular hemoglobin concentration, WBC: white blood cells.
a Values are expressed as means ± SD. Number of determinations is 6 for mothers and 12 for pups.
b MMI or Se group vs control group: p < 0.01.
c MMI or Se group vs control group: p < 0.001.
d MMI or Se group vs control group: p < 0.05.
e MMI group vs (MMI + Se) group: ++ p < 0.01.
f MMI group vs (MMI + Se) group: +++ p < 0.001.
g MMI group vs (MMI + Se) group: + p < 0.05.
Hematopoietic factors (iron, vitamin B9 and vitamin B12 levels)
Our results showed that plasma levels of iron, vitamin B9 and vitamin B12 were lower in the MMI-treated mothers (−38%, −40% and −21%) and their suckling pups (−27%,−47% and −21%) than in the negative controls. When selenium was supplemented to the diet of MMI-treated rats, these parameters reached normal values (Table 2 ).
Plasma levels of iron, vitamins B12 and B9 of mothers and their offspring controls or treated with methimazole (MMI), methimazole + selenium (MMI + Se) or selenium (Se) from the 14th day of pregnancy until day 14 after delivery
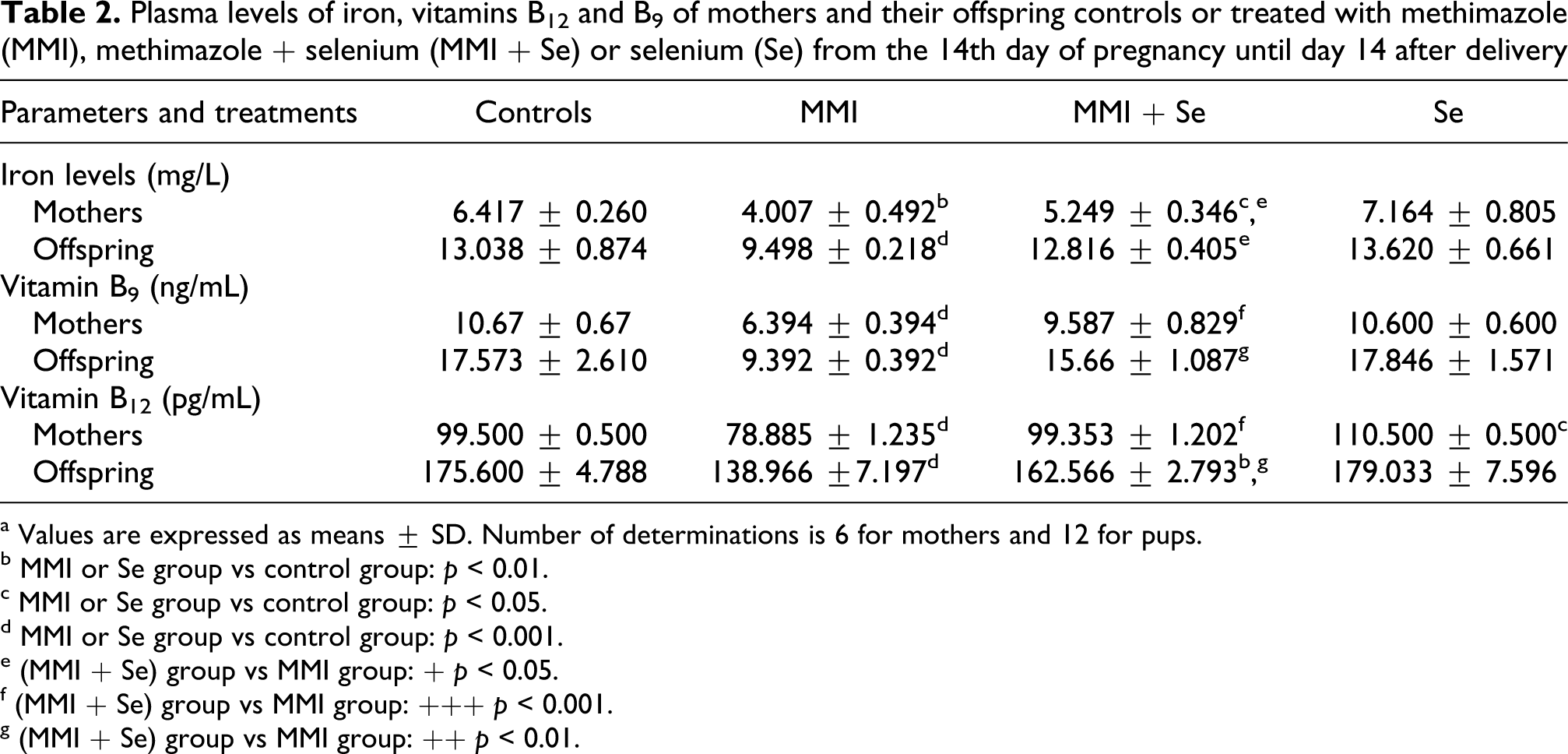
a Values are expressed as means ± SD. Number of determinations is 6 for mothers and 12 for pups.
b MMI or Se group vs control group: p < 0.01.
c MMI or Se group vs control group: p < 0.05.
d MMI or Se group vs control group: p < 0.001.
e (MMI + Se) group vs MMI group: + p < 0.05.
f (MMI + Se) group vs MMI group: +++ p < 0.001.
g (MMI + Se) group vs MMI group: ++ p < 0.01.
Plasma lipid peroxidation
Our results revealed an increase of plasma lipid peroxidation in MMI-treated group as evidenced by the enhanced malondialdehyde levels in mothers (+28%) and their offspring (+26%) when compared to negative controls (Figure 1 ). The supplementation of selenium alleviated lipid peroxidation induced by MMI treatment and significantly modulated the plasma malondialdehyde levels in both mothers and pups.
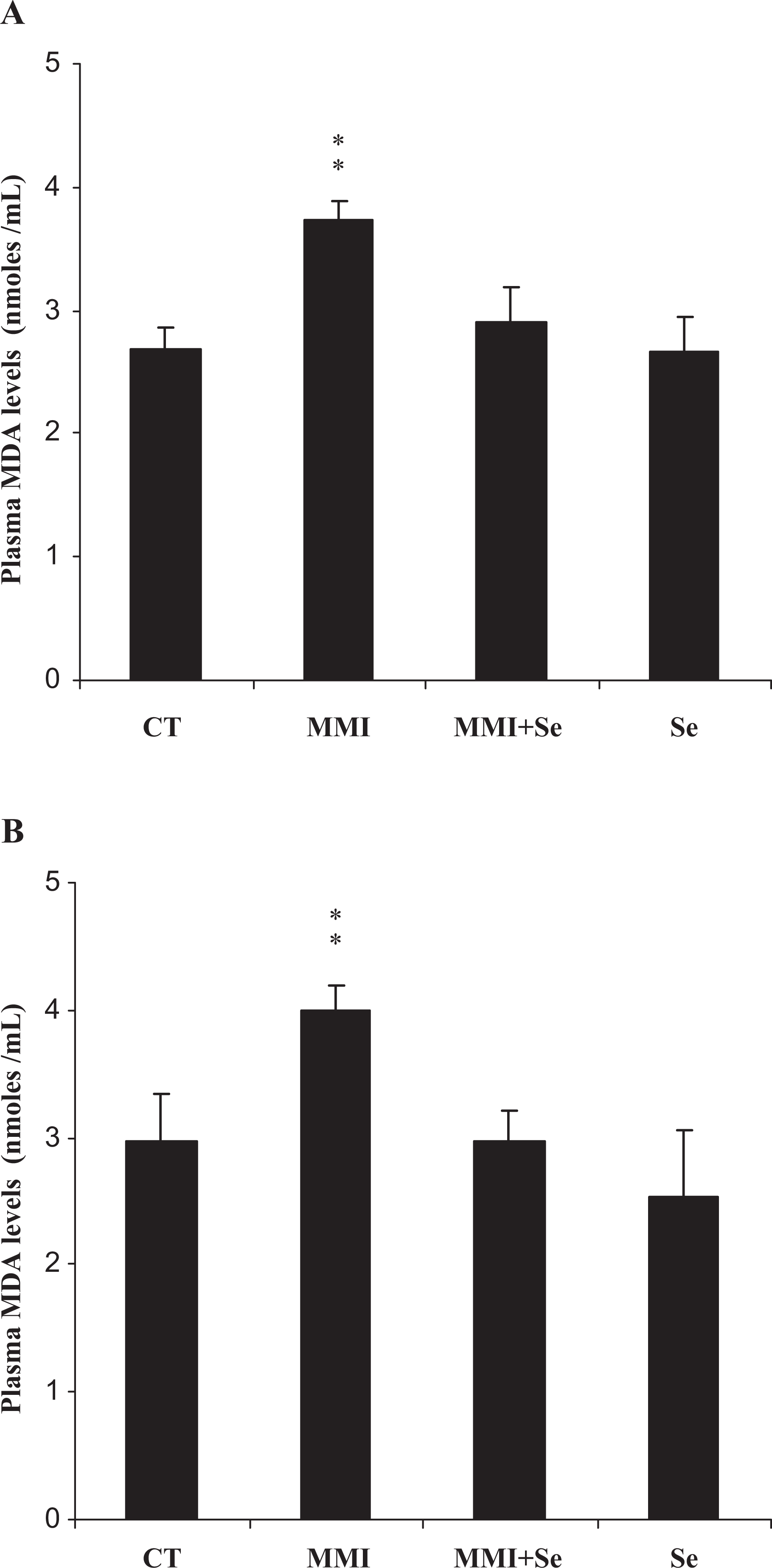
Plasma malondialdehyde (MDA) levels of mothers (A) and their offspring (B), controls (CT) or treated with methimazole (MMI), methimazole + selenium (MMI + Se) or selenium (Se) from the 14th day of pregnancy until day 14 after delivery. Values are expressed as means ± SD. Number of determinations is 6 for mothers and 12 for pups, MMI or Se group vs control group: **p < 0.01.
Plasma lactate dehydrogenase activity
In MMI group, lactate dehydrogenase activity, a potential marker of chemical products toxicity, significantly increased in mothers (+57%) and their offspring (+41%) when compared to negative controls. The administration of selenium to (methimazole + selenium) group reduced lactate dehydrogenase activity (Figure 2 ).
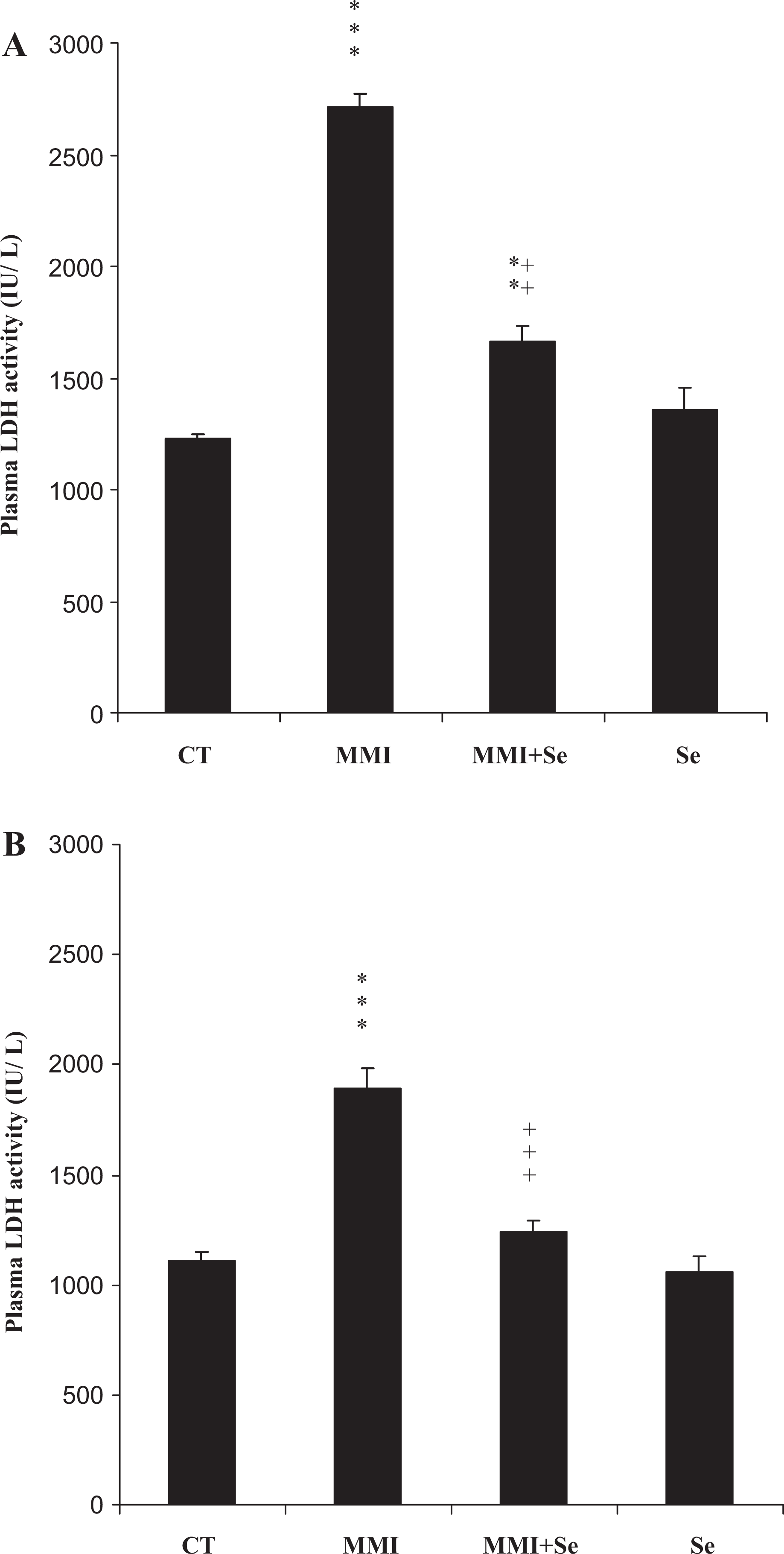
Plasma lactate dehydrogenase (LDH) activities of mothers (A) and their offspring (B), controls (CT) or treated with methimazole (MMI), methimazole + selenium (MMI + Se) or selenium (Se) from the 14th day of pregnancy until day 14 after delivery. Values are expressed as means ± SD. Number of determinations is 6 for mothers and 12 for pups. MMI or Se group vs control group: **p < 0.01; ***p < 0.001, MMI group vs (MMI + Se) group: ++p < 0.01; +++p < 0.001.
Plasma enzymatic antioxidants
In the MMI-treated rats, plasma superoxide dismutase, catalase and glutathione peroxidase activities decreased significantly by 41%, 44% and 54% in mothers and by 27%, 31% and 22% in their pups, respectively, when compared to negative controls (Table 3 ). The administration of selenium ameliorated the enzyme activities in (methimazole + selenium) group when compared to MMI group. Treatment with selenium alone increased the selenoenzyme activity (p < 0.05) in mothers, when compared to negative controls.
Plasma enzymic antioxidants activities (glutathione peroxidase, catalase and superoxide dismutase), non-enzymic antioxidants contents (glutathione) of mothers and their offspring controls or treated with methimazole (MMI), methimazole + selenium (MMI + Se) or selenium (Se) from the 14th day of pregnancy until day 14 after delivery

a Values are expressed as means ± SD. Number of determinations is 6 for mothers and 12 for pups.
b Superoxide dismutase: U/mL.
c Catalase: µmoles H2O2 degraded/min/mL.
d Glutathione peroxidase: U/mL.
e Glutathione µg/mL.
f MMI or Se group vs control group: p < 0.001.
g MMI or Se group vs control group: p < 0.05.
h MMI or Se group vs control group: p < 0.01.
i (MMI + Se) group vs MMI group: p< 0.01.
j (MMI + Se) group vs MMI group: p < 0.05.
k (MMI + Se) group vs MMI group: p < 0.001.
Plasma non-enzymatic antioxidants
Glutathione levels
The levels of non-enzymatic antioxidants such as glutathione levels significantly decreased in mothers (−46%) and pups (−51%) in MMI-exposed group when compared to controls. However, supplementation of selenium to the standard diet maintained the levels of this antioxidant at those of control (Table 3).
Hydrophilic antioxidant (vitamin C)
The data presented in Figure 3 show significant changes in the activity of vitamin C, potent hydrophilic antioxidant, during the treatment of rats with MMI, selenium and their combination. The exposure of rats to MMI caused a significant decrease in plasma vitamin C levels in mothers (−48%) and pups (−18%). In animals exposed to selenium and MMI, the plasma vitamin C levels reached the normal values, while no significant changes were obtained after treatment with selenium alone.
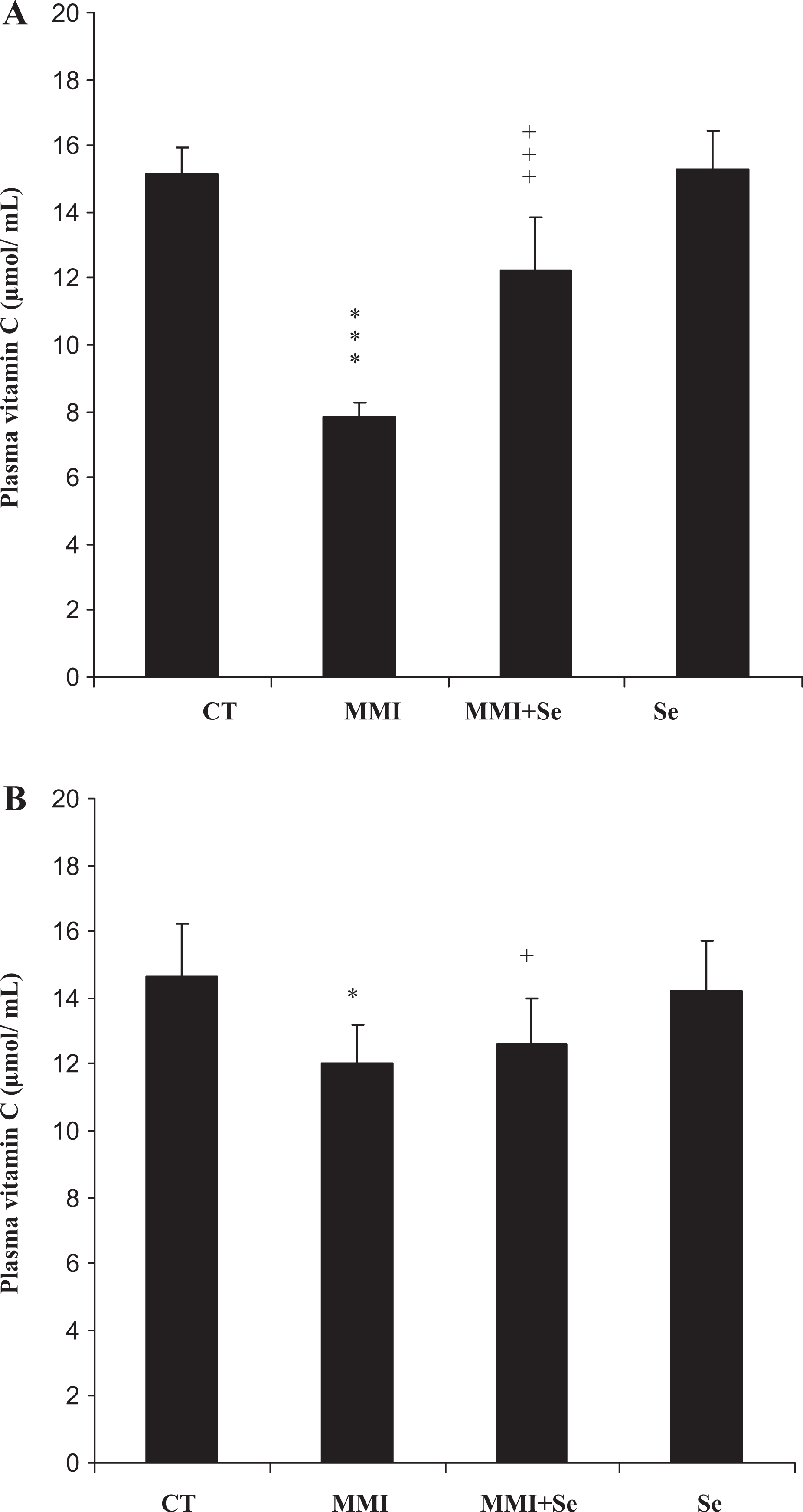
Plasma vitamin C levels of mothers (A) and their offspring (B), controls (CT) or treated with methimazole (MMI), methimazole + selenium (MMI + Se) or selenium (Se) from the 14th day of pregnancy until day 14 after delivery. Values are expressed as means ± SD. Number of determinations is 6 for mothers and 12 for pups. MMI or Se group vs control group: *p < 0.05; ***p < 0.001, MMI group vs (MMI + Se) group: + p < 0.05; +++p < 0.001.
Lipophilic antioxidant (vitamin E)
α-Tocopherol is the major lipophilic antioxidant in lipoproteins. Plasma levels of non-enzymatic antioxidants like vitamin E markedly decreased in mothers (−45%) and pups (−37%) in the MMI-treated group, when compared to controls. Selenium supplemented in the diet of MMI-treated group improved this parameter, when compared to MMI group (Figure 4 ).

Plasma vitamin E levels of mothers (A) and their offspring (B), controls (CT) or treated with methimazole (MMI), methimazole + selenium (MMI + Se) or selenium (Se) from the 14th day of pregnancy until day 14 after delivery. Values are expressed as means ± SD. Number of determinations is 6 for mothers and 12 for pups. MMI or Se group vs control group: *p < 0.05; **p < 0.01, MMI group vs (MMI + Se) group: + p < 0.05.
Discussion
In the current study, the exposure of adult rats to MMI during late pregnancy and the early postnatal periods profoundly affected their hematological parameters as well as those of their pups. A reduction in red blood cells number, hematocrit values and hemoglobin concentration was observed in mothers and pups. Other hematological parameters, mean corpuscular volume, mean corpuscular hemoglobin and mean corpuscular hemoglobin concentration, remained unchanged in mothers. Similar findings were showed in hypothyroid dogs, 37 hyperthyroid cats treated with MMI 38 and rats subjected to dietary restriction. 9 In their suckling pups, mean corpuscular volume and mean corpuscular hemoglobin increased. Disturbances in blood parameters, induced by MMI, could be due to a decline in plasma iron, folic acid (vitamins B9) and vitamin B12. In fact, Hoogstratten et al. 39 and Sahashi et al. 40 reported that anemia could be due to some hematopoietic factors' deficiency. The major ones are iron, vitamins B9 and B12. These factors are necessary for normal hematopoiesis which is useful for DNA synthesis and cellular division. In pregnant women, a deficit of iron often coexists with vitamin B12 and/or vitamin B9 deficiencies, 41 leading to megaloblastic anemia. 42 Co-administration of selenium through diet improved the parameters cited above. According to Van Nhien et al., 43 and Semba et al., 44 a strong association of low serum selenium levels with anemia was observed in Vietnamese children and adults living in the United States. They considered selenium as an essential trace element which might be used as a dietary supplement against anemia.
We also found that MMI impaired the immune function of the treated mothers during pregnancy and lactation periods. In fact, white blood cells increased in mothers, indicating an activation of their defense mechanism, but they were unchanged in their pups. These results confirmed our previous studies, 8 showing that MMI caused renal inflammatory response by increasing the infiltration of lymphocyte and polynuclear cells in kidney. When selenium was added in the diet of MMI-treated rats, white blood cells number reached near normal values. Previous reports of Smith et al. 45 and Van Nhien et al. 43 suggested that selenium was a vital trace element for the effective functioning of the human immune system.
It is well known that vitamins C (ascorbic acid) and E (α-tocopherol) are among the most important antioxidants influencing the immune system. 46 Vitamin C is a potent antioxidant that offers protection against oxidative damage to various tissues. 47 Vitamin E has proven beneficial in protecting cellular membranes against oxidation, 48 increasing resistance to stress and also considered a natural agent with anti-inflammatory properties. 49 In the current study, plasma vitamins C and E levels were also alleviated after treatment with selenium in (methimazole + selenium) group. These results spoke in favor of an improved immune function in mothers.
The impairment of hematological parameters could be due to oxidative stress probably generated by metimazole. Different mechanisms have been postulated to explain MMI-induced oxidative stress, such as lipid peroxidation and interaction with membrane components. In fact, lipid peroxidation represents one of the most frequent reactions resulting from free radicals' attack on biological structures. 50 In our experiment, the plasma malondialdehyde level, indicator of lipid peroxidation, significantly increased in the MMI-treated rats. Our results were in agreement with previous reports of Fetoui et al. 9 who demonstrated that the high plasma malondialdehyde levels could be seen as an indicator of oxidative stress and lipid peroxidation in the circulating erythrocytes of suckling and adult rats subjected to dietary restriction. Blood parameters impairment was also estimated by lactate dehydrogenase activities in plasma. This enzyme was recognized as a potential marker to assess chemical products toxicity. 51 In our experimental study, plasma lactate dehydrogenase activities in MMI-treated mothers and their pups significantly increased.
In view of our results, we suggest that exposure to MMI probably causes oxidative damage in the blood tissue of adult and suckling rats and that selenium protects it against oxidative damage, thus reinforcing its antioxidant potential. In fact, the co-administration of selenium to MMI-treated group restored malondialdehyde levels and lactate dehydrogenase activities to near normal values. Thus, selenium might be useful as a nutritional supplement agent against oxidative stress.
The most important antioxidant aspect of selenium is its function in the active site of selenoenzyme glutathione peroxidase. 52 According to McPherson, 53 the latter antioxidant enzyme and others, such as superoxide dismutase and catalase, constitute mutually a supportive team of defense against reactive oxygen species. Our results showed that activities of these enzymes were significantly decreased in suckling rats as well as in their mothers treated with MMI when compared to those of negative controls. The decrease in the extracellular antioxidant potential may indicate that plasma antioxidants are the agents involved in the defense against reactive oxygen species that are used up most rapidly. 54 Thus, MMI-generated hematological disorders could probably be attributed to its pro-oxidant effect. Whereas, reduced catalase, total superoxide dismutase and glutathione peroxidase activities were improved by co-treatment with selenium. This is consistent with previous reports of Ahmad et al., 55 who suggested that the mechanistic approach of selenium supplementation against oxidative stress involved an increase in the antioxidant enzyme defenses. Thus, one possible action of selenium as a therapeutic agent through its antioxidant function may be the modification of enzymatic antioxidants in order to normalize the MMI-induced oxidative stress.
Likewise, glutathione, a crucial component of the antioxidant defense mechanism, functions as a direct reactive free-radical scavenger. The decreased glutathione level in MMI-treated rats may be due to its consumption in the scavenging free radicals probably generated by MMI. Our study showed that selenium improved glutathione levels, which participated in the protection against free radicals and reactive oxygen species. 56 Indeed, as a scavenger or a reducing agent, glutathione protects the living cells against hypoxia, toxicity, mutagenicity or transformation by radiation, carcinogens as well as a number of exogenous compounds such as drugs and pollutants.
Conclusion
This paper may constitute the first report which demonstrates that methimazole causes oxidative stress and anemia in adult rats and their suckling pups. Co-administration of selenium through diet to hypothyroid rats improved disorders in blood parameters, plasma enzymatic activities (superoxide dismutase, catalase, glutathione peroxidase) and non-enzymatic levels (glutathione and vitamins E and C). As a result, selenium could be a useful agent that provides protection against impairment in hematological parameters. The protective effect exhibited by selenium supplementation could involve the capability of selenium in activating and protecting various selenium-dependent enzymes
Footnotes
Acknowledgments
The authors are indebted to Mrs Serra Ben Saleh for her skillful technical assistance in vitamin E measurement. We also wish to extend our thanks to Mr Bejaoui Hafedh, teacher of English at Sfax Faculty of Science, who has helped proofread and edit this paper.
Authors Ahmed Hakim and Afef Troudi contributed equally to this work.
This work was supported by the DGRST grants (Appui à la Recherche Universitaire de Base ARUB 99/UR/08-73), Tunisia.
The authors declare that there are no conflicts of interest.
