Abstract
The present study is planned to examine the antioxidant activity (AA) and the protective effect of the essential oil of Citrus limon (EOC) against aspirin-induced histopathological changes in the brain, lung, and intestine of female rats. For this purpose, 28 albino rats were classified to control group (group C), aspirin group (group A), EOC group (group EOC), and pretreatment with EOC and treated with aspirin group (group EOC + A). The antioxidant activities of EOC were evaluated by three different assays including reducing power, β-carotene, and scavenging of hydrogen peroxide (H2O2). Our results found that EOC represents, respectively (0.064 ± 0.013 and 0.027 ± 00 mg Quer E/100 µL), of flavonoid and flavonol. Then, it exhibited a potential activity of reducing power (at 300 mg/mL, which was found to be 0.82 ± 0.07), β-carotene-linoleic acid (AA% = 69.28 ± 3.5%), and scavenging of H2O2 (IC50 = 0.23 ± 0.008 mg/mL). In vivo, aspirin given to rats at the dose of 600 mg/kg body weight induced histomorphological damage in brain, lung, and intestine. However, our data found that the pretreatment with EOC offered a significant protection against the injury induced by aspirin. It can be concluded that the protective effect of EOC can be due to its antioxidant activities.
Introduction
The environmental pollutants, radiation, chemicals, and drugs are the most cause of production of free radicals, which are responsible for chronic disorder in humans, including diabetes, cancer, atherosclerosis, arthritis, hepatic injury, and gastrite. 1 Therefore, to prevent or inhibit the disaster disease, many scientific studies resort to use the medicinal plants. The latter have been used since antiquity to relieve and cure the human diseases, because these plants often have many effects, such as to treat inflammation, fever, headache, cold, cough, diarrhea, dysentery, gonorrhea, hemorrhage, sore throat, typhoid, and urinary tract ailments. In fact, their therapeutic uses are due to the presence of many bioactive natural compounds called secondary metabolites. Among these metabolites, there are the essential oils; they are complex mixtures of volatile organic compounds that are characterized by a strong odor. 2 They can be extracted from different parts of the plant including flowers, barks, stem, leaves, roots, fruits, and other parts. 3 They have various medicinal and therapeutic properties, including the anti-infectious, antifungal, antiviral, anti-inflammatory, cardiovascular, endocrine-regulating, and digestive effects. 4
Citrus limon is a member of the Rutaceae family, having several pharmacological properties including antimicrobial, antiparasitic, and antioxidant activities. 5,6 It has been widely used in traditional medicine to treat various diseases. Indeed, infusion of leaves of this plant is used to treat many anomalies, such as obesity, diabetes, blood lipid lowering, cardiovascular diseases, brain disorders, and certain types of cancer. 7 Furthermore, the essential oils of fruit of citrus plants are used in the food industry to give flavor to drinks and foods. They are also a constituent for the pharmaceutical industry for the preparation of drugs, soaps, perfumes, and other cosmetics as well as for home cleaning products. 8 Additionally, our recent published studies 9,10 found that the essential oil of leaves of C. limon had a protective effect oil against a high dose of aspirin-induced acute liver and kidney damage in female Wistar albino rats. We found also, in vitro, that the essential oil of C. limon (EOC) has a potent cytoprotective effect against aspirin-induced toxicity in intestine epithelial cells (IEC)-6 cells.
Consistently, this present research work is aimed to explore the antioxidant activities and the protective effect of EOC against the injury of aspirin in rats in others organs such as brain, lung, and intestine through histological study.
Materials and methods
Drugs and chemicals used
Aspirin, dimethysulfoxide (DMSO), sodium nitrite (NaNO2), sodium hydroxide (NaOH), aluminum chloride (AlCl3), potassium acetate (CH3CO2K), potassium ferricyanide (K3Fe (CN)6), chloroform, β-carotene, ascorbic acid, trichloroacetic acid, linoleic acid, quercetin, Butylated hydroxytoluene (BHT), hydrogen peroxide (H2O2), and sodium chloride (NaCl) were purchased from Sigma-Aldrich Chemicals Co. (St Louis, Missouri, USA).
Collection and extraction of EOC
At maturity, the leaves of C. limon were collected and identified by botanist in the Faculty of Science of Gafsa (Dr Lefi El Kadri). After dreading the aerial parts of this plant at 25°C for 15 days in a shaded and ventilated atmosphere, the essential oil sample was extracted from 50 g with 500 mL of distilled water by hydrodistillation. The latter was carried with the same protocol and under the same conditions those described in our previous study. 9
Total flavonoid content determination
The total flavonoid content of the EOC was carried out with the method of Kavoosi and Rowshan. 11 In this procedure, 100 µL of essential oil (0.1 mg/mL in DMSO) were mixed with 300 µL of NaNO2 solution (5%). After 5 min, 300 µL of AlCl3 solution was added. Then, 2 mL of 1-M NaOH was added after 6 min. The volume of the solution was made up to 10 mL with distilled water. The mixture was incubated at 37°C for 15 min. The absorbance was measured at 510 nm, and quercetin was used as standard. The results were expressed as milligrams of quercetin equivalent (QuerE) per 100 µL of essential oil. Triplicate determinations were performed.
Total flavonols content determination
Total flavonol content was determined as described by Kosalec et al. 12 Briefly, 500 μL of EOC (0.1 mg/L) was mixed to 100 μL of AlCl3; 100 μL of CH3CO2K (1 M) and 2.8 μL of distilled water were added to the mixture. The absorbance was determined at 415 nm after allowing the mixture to stand for 30 min in the dark at room temperature using quercetin as a standard. Results were expressed as milligrams of QuerE per 100 µL of essential oil.
In vitro antioxidant activities of EOC
Reducing power
The reducing power assay was determined according to the method of Chu et al.
13
In brief, 1 mL of various concentrations of EOC (50–300 mg/µL) was mixed with 2.5 mL of 1% K3Fe (CN)6 and 2.5 mL of potassium phosphate buffer (0.1 M, pH 6.6); 2.5 mL of 10% trichloroacetic acid was added after incubation at 50°C for 25 min and the mixture was centrifuged at 300 g for 10 min. At the end of the assay, 2.5 mL of the upper layer was mixed with 0.5 mL of 0.1% ferric chloride (FeCl3) and 2.5 mL of distilled water. After incubation for 30 min at 28°C, the absorbance was measured at 700 nm. Ascorbic acid as standard synthetic used as positive control. The EC
β-Carotene bleaching assay
The method of β-carotene bleaching assay was carried out according to the method of Koleva et al. 14 In the assay; the linoleic acid formed free radical upon the abstraction of a hydrogen atom from one of its diallylic methylene groups which attacks the unsaturated, β-carotene molecules. Then, β-carotene was oxidized and broken down in parts; subsequently, the system loses its chromophore and characteristic orange color, which can be monitored spectrophotometrically.
About 0.5 mg of β-carotene was dissolved in 1 mL of chloroform and then mixed with 25 µL of linoleic acid and 200 mg of Tween 40. The chloroform in the mixture was removed by rotary evaporator followed by the addition of 100 mL of distilled water and the reaction mixture was shaken vigorously to prepare the β-carotene/linoleic acid emulsion; 100 µL of EOC or standard solution was added to 25 µL of the reagent mixture. Then, the reaction mixture was incubated in a water bath at 50°C for 2 h. BHT was used as a positive control. The antioxidant activity (AA%) was measured using the following formula
where A sample 0 is the absorbance of the sample at t = 0 min, A sample 120 is the absorbance of the sample at t = 120 min, A control 0 is the absorbance of the control at t = 0 min, and A control 120 is the absorbance of the control at t = 120 min. All determinations were performed in triplicate.
Scavenging of H2O2
The scavenging activity of EOC toward H2O2 radicals was performed by the method of Kavoosi and Rowshan. 11 In brief, 1 mL of H2O2 (50 mM in 100 mL phosphate buffer pH 7.4) was mixed to 100 µL of essential oils (0–0.3 mg/mL in DMSO). Then, the mixture was shaken vigorously and incubated for 60 min at 37°C. After that, the absorbance of the reaction mixture was evaluated at 230 nm against a blank solution containing phosphate buffer without H2O2.
The experiment was repeated in triplicate. The percentage of H2O2 scavenging was obtained by the equation as follows: H2O2 scavenging activity (%): [Absorbance control – Absorbance sample/Absorbance control] × 100. IC50 was calculated from the graph plotting percentage inhibition against different essential oil concentrations.
In vivo
Animals
Twenty-eight female albino Wistar rats with a body weight of 150–200 g obtained from the Animal Ecophysiology Laboratory (Sfax, Tunisia) were used in this study. They maintained for a 2-week adaptation period under the same conditions of temperature (22 ± 2°C), relative humidity (50 ± 4%), and a constant photoperiod (12-h light/12-h dark cycle) and had free access to feed and water. The animals were housed seven rats per each cage.
The experimental protocol was approved by the Faculty Ethics Committee in our institution with ethics approval number 1204. Animals were maintained in accordance with international guidelines for care and use of living animals in scientific investigations (Council of European Communities 1986).
Experimental design
The experiment was carried out with the same protocol as described in our previous study. 9 Briefly, the rats were randomly divided into four groups: group 1: control rats were administrated orally by gavage with 0.9% NaCl; group 2: EOC was given orally for 56 days and then given orally NaCl for 4 days, respectively (about 1.0% w/w with a dose of 1 mL/kg); 15 group 3: rats orally received normal saline and treated with aspirin in acute dose of 600 mg/kg which was suspended in normal saline and administered orally for 4 days; 9 and group 4: rats received EOC for 56 days followed by oral administration of aspirin for 4 days. The body weight of the rats was monitored each 3 days. After the end of experiment, the animals from each group were sacrificed rapidly by decapitation to avoid the effect of stress.
Histopathological study
After weighing the organs (lung, brain, and intestine), they were immediately fixed in fixative solution (4% formaldehyde in phosphate buffer (pH = 7.6)). The tissues were then cut and dehydrated by passing through different grades of ethanol. Then, they were washed with toluene to remove the ethanol. They embedded in paraffin and cut into 5-µm section using microtone and they stained with hematoxylin and eosin. The tissues were then observed and examined under a light microscope.
Statistical analysis
All analyses were performed with Graph Pad Prism (GraphPad Software, San Diego, California, USA). Significant differences between treatments were carried out using one-way analysis of variance, followed by Tukey’s post hoc test for multiple comparisons with acceptable statistical level of significance set to 0.05. All values were expressed as the means ± standard deviation.
Results
Essential oil yield
Our result showed that the essential oil obtained from the leaves of C. limon was a light yellow liquid with a strong odor with and the yield of hydrodistillation was 1.10 ± 04%.
Total flavonoid and flavonols contents
The total flavonoid and flavonol contents in the EOC were examined using Folin–Ciocalteu and AlCl3colorimetric assays. The result reported in Table 1 showed that the amount of total flavonoid and flavonol in the EOC were found to be 0.064 ± 0.013 and 0.027 ± 00 mg QuerE/100 µL essential oil, respectively.
Total flavonoids and flavonols contents of EOC.

EOC: Essential oil of Citrus limon; QuerE: quercetin equivalents; SD: standard deviation.
a Values are mean ± SD, n = 3.
Antioxidant activity
Ferric reducing antioxidant power assay
The results of the ferric reducing antioxidant power (FRAP) assay expressed the antioxidant capacity of the extract based on its reducing properties. It is found that the reducing power increased with increasing concentration. As shown in Figure 1, the reducing power of EOC at 300 mg/mL was found to be 0.82 ± 0.07 and thus significantly lower than that of ascorbic acid, which was used as positive control (1.78 ± 0.16), at the same concentration.

Reducing power of EOC using ascorbic acid as control. The values are expressed by means ± SEM (n = 3). EOC: essential oil of Citrus limon; SEM: Standard error of the mean.
Antioxidant capacity of EOC by β-carotene assay
The AA in the assay using β-carotene as model system, as shown Figure 2, at the absorbance, decreased toward a lower value (control), whereas this decrease becomes stable for oil and standards compared to that of control. Result indicated that the AA through the β-carotene/linoleate mode system of EOC was found to be 69.28 ± 3.5%. This activity appeared lower than the BHT as positive control (81.3 ± 0.54%) (Table 2)

Antioxidant activity of EOC and BHT by β-carotene-bleaching method. EOC: essential oil of Citrus limon; BHT: Butylated hydroxytoluene.
Antioxidant activity (%) of essential oil of Citrus limon and BHT using β-carotene bleaching assay.

a The values are expressed by means ± SEM, (n = 3).
H2O2 scavenging assay
H2O2 scavenging activity shows the ability of EOC and standard BHT to inhibit the H2O2. The IC50 of EOC had higher value (0.23 ± 0.008 mg/mL) as compared to BHT (0.08 ± 0.002 mg/mL) indicating that EOC had least ability in hydroxyl radical scavenging (Figure 3).

Antioxidant capacity of the EOC by the H2O2 method. EOC: essential oil of Citrus limon; H2O2: hydrogen peroxide.
In vivo
Effect on body weight
As shown in Figure 4 and Table 3, the body weight of the rats in the control group (C), EOC, and EOC + A before 56 days is normal. However, in the aspirin treated group (A), the body weight of the rats significantly (p < 0.001) decreased for 4 days as compared to control (C). And we noted, in the group pretreated with EOC (EOC + A), an increase in the body weight as compared to the rats treated with aspirin alone (A).

Effects of aspirin and EOC on body weight of rats. Each value represents the mean ± SEM of seven rats per group. EOC: essential oil of Citrus limon.
Effects of treatments on animal body weight (g).a

A: aspirin; EOC: essential oil of Citrus limon.
a All values are expressed as mean ± standard deviation, n = 7 for each treatment group.
b p < 0.001 significantly different from A group.
Relative organs weight analysis
Table 4 reveals that administration of aspirin induced an increase in the relative weight of the brain, stomach, kidney, and liver (0.82 ± 0.14, 3.31 ± 0.31, 1.00 ± 0.09, and 4.56 ± 0.83), respectively. But, the weight of the lung and heart decreased (0.73 ± 0.25 and 0.29 ± 0.08), respectively. Compared with the group (A), a significant (p ≤ 0.05) decrease in the relative weight of the brain, stomach, kidney, and liver (0.68 ± 0.15; 1.44 ± 1.02, 0.88 ± 0.09, and 3.67 ± 0.27), respectively, and an increase in the weight of heart and lung (0.37 ± 0.04, 1.02 ± 0.09), respectively, were resulted.
Relative organs weight to body weight in rats for all treated groups.a

EOC: essential oil of Citrus limon.
a The results are expressed as mean ± SEM, n = 7 rats/group.
b p ≤ 0.05 compared to control group (C).
c p ≤ 0.05 compared to control group (A).
Light microscopic examination
Histopathological observations of brain tissue
In the control group (C), the histological section of the brain showed a normal structure appearance. But, in the group of rats treated with aspirin (A), a degenerative morphological change was observed such as changes of neurons which appear to have pyknotic nuclei and has large spaces (vacuoles). However, the pretreatment with EOC prevented these changes and maintained a normal architecture of nerve cells with a reduced number of pycnotic nuclei and the cytoplasmic vacuole (Figure 5).

Histological examination of the different groups of rat brain tissues (hematoxylin and eosin staining at ×40): C: group 1 (normal rats); EOC: group 2 (rats treated with EOC); A: group 3 (rats treated with aspirin); EOC + A: group 4: EOC + aspirin-treated rats. Normal villus, degeneration and desquamation of surface epithelium, and shortening and fusion of villus. EOC: essential oil of Citrus limon.
Histopathological observations of lung tissues
As shown in Figure 6, the lungs section of control group showed the normal alveolar and bronchioles spaces, whereas in the group of rats treated with aspirin, our observation showed that aspirin induced damage in lung tissue, showing degeneration of the alveolar septa, fibrosis, disruption of the connective tissues and elastic fibers, and the congestion of the blood capillaries, which were also blocked with large aggregation of the blood cells. The pretreatment of EOC reduced the toxic effects of aspirin and reduced injuries which were observed in the lung tissues of the group (EOC + A).
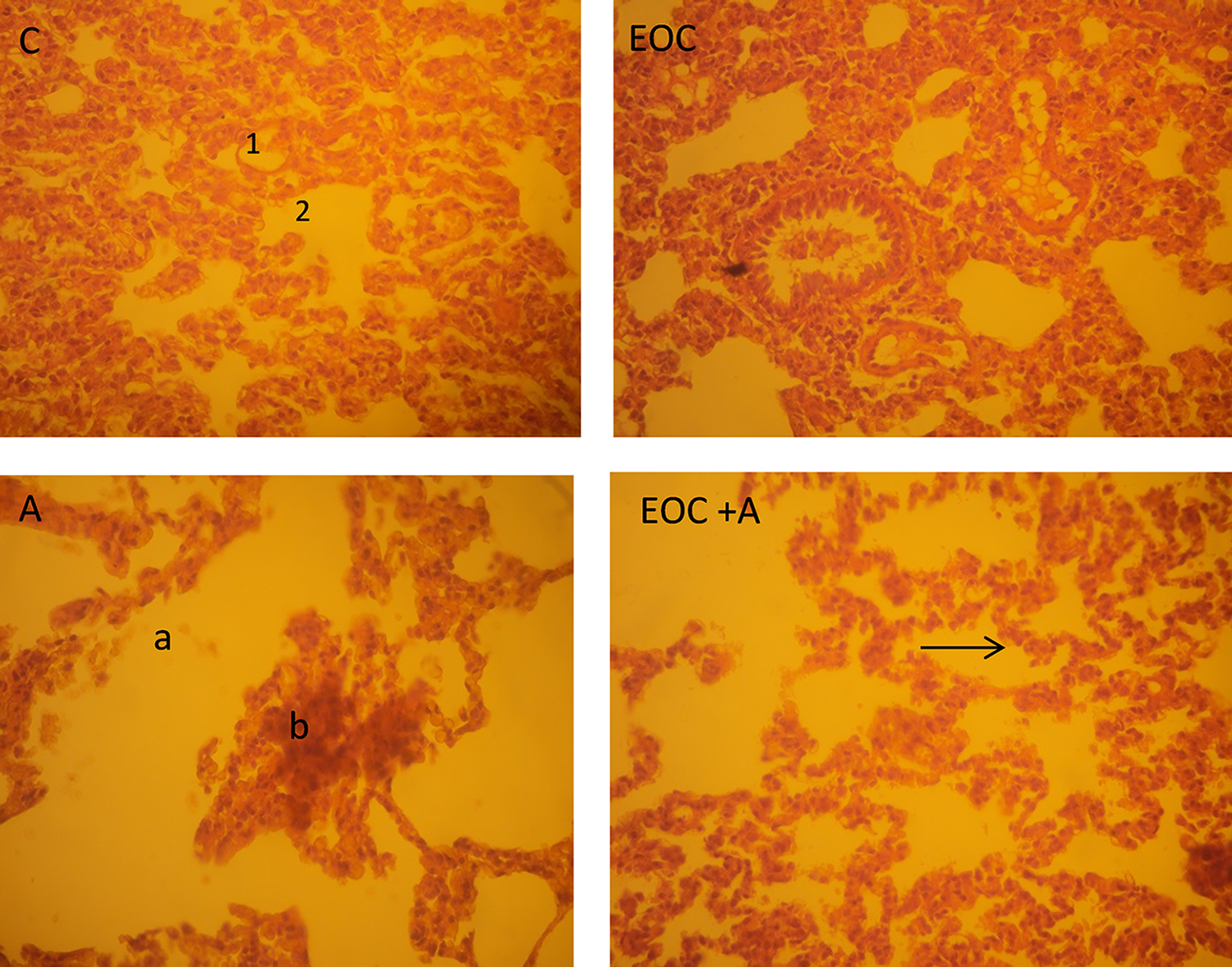
Photomicrographs of section of lung of control (C), a exposed and treated rats (EOC + A), H&E ×40. 1: Alveoli; 2: alveolar sac; 3: bronchiole; a: air space enlargement; b: edema interalveolar septum (arrow). EOC: essential oil of Citrus limon.
Histopathological observations of intestine tissues
The tissue section of the intestine of the rats in the control group showed normal intestinal structures. However, in the aspirin-treated group, considerable changes including atrophy of surface epithelium, cystic dilation in crypts, and crypt loss were observed. In the section of animals of EOC + A, a remarkable improvement of the histological analysis was noted compared to the aspirin group. However, no alteration in the section of the intestine was observed in animals treated only with EOC (Figure 7).

Morphopathological changes in intestine tissues of control (C), treated with aspirin (A), pretreated with EOC, and treated with aspirin (EOC + A) showing preservation of normal (H&E ×400). Degenerate and vacuolated neurocytes (dark arrow), normal neurons; normal Glial cell. EOC: essential oil of Citrus limon.
Discussion
An essential oil is a liquid concentrated with vegetable substances, obtained by extraction or distillation of volatile molecules from the plant. They constituted with terpenes and aromatic molecules. Essential oils from different plants have different properties, depending on the original composition. Therefore, based on their therapeutic uses and their important biological activities, the use of essential oils is interesting.
In the present study, the yield of the EOC was determined and then estimated at 1.1 ± 0.041%. This value is different from others varieties of C. limon determined in Algeria (Eureka, Lisbon, and Meyer varieties) which signaled, respectively (0.64, 0.90, and 0.46%). 16 Consistently, these differences can attribute to several factors, including geographical origin, time collection of the plants, maturity, seasonal climate, extraction process, and storage condition. 17
In addition, this study is aimed to investigate the antioxidant protective effect of EOC in rats as well as its phytochemical and bioactive compounds. Indeed, it is known that the essential oil contains aromatic and aliphatic acid, ester, and phenolic compounds. 5 Thus, in our present study, moderate flavonoids amount was found in C. limon oil (0.064 ± 0.013 mg QuerE/100 µl essential oil). These compounds are well known for their strong antioxidant activities by scavenging the free radical and by the prevention against various diseases. In addition, a least amount of flavonols (0.027 ± 00 mg QuerE/100 µL of essential oil) was determined in this work. These results confirmed previous study 9 showing that EOC had a potential total phenol of about 0.131-mg gallic acid equivalent (GAE)·per 100 µL of essential oil. In fact, polyphenols are used as natural antioxidants and are important agents because of their health benefits for humans, that is, decreasing the risk of liver disease, inflammation, and cardiovascular and degenerative diseases by reduction of oxidative stress and counteraction of macromolecular oxidation. 18 In the context, we determined the antioxidant activities of EOC in our study. Indeed, the free radical scavenging activity of EOC was tested through three assays. First, the FRAP assay, in the reducing power, the presence of reductants (antioxidants) in the samples is able to reduce the complex Fe3+ to Fe2+ by donating an electron. In our study, EOC exhibited a moderate reducing power at 300 mg/mL and was found to be 0.82 ± 0.07 as compared to ascorbic acid (1.78 ± 0.16), at the same concentration. Second, the second activity was β-carotene-linoleate system; data obtained in our study agree with those reported by Song et al. 19 showing that essential oil has antioxidative activities against linoleic acid oxidation.
Furthermore, the effect of EOC on the inhibition of free radical was assessed by H2O2 scavenging. It is known that H2O2 was considered as a toxicant to the cell by converting itself into hydroxyl radical in the presence of metal ions and superoxide anion and produced singlet oxygen through reaction with superoxide anion or with hypochlorous acid in living systems. It may degrade certain heme proteins as hemoglobin. 20 In the study, the result found that EOC exhibited dose-dependent (H2O2) scavenging ability. The IC50 value at which H2O2 was scavenged by 50% was found to be 0.23 ± 0.008 mg/mL as compared to BHT (0.08 ± 0.002 mg/mL).
Interestingly, these antioxidant activities of EOC can be attributed to their redox properties which can play an important role in neutralizing free radicals, quenching singlet and triplet oxygen, or decomposing peroxids. 11 Furthermore, it can be due to their composition especially due to the presence of citral compound. Our previous study 10 found that citral had AA. Additionally, the antioxidant properties can be attributed to the phenolic and flavonoid contents of the essential oil. In fact, it is known that the antioxidative effects of essential oil depend on the content of phenolic compounds. 21 Previous studies 22 found that there were a significant linear correlation between total phenol content and antioxidant capacity of some plant medicinal.
In addition, in the present study, we found that aspirin administered at 600 mg/kg dose in rats affected the body weight which induced an increase in the relative weights of organs such as brain, stomach, kidney, and liver and a decrease in the relative weights of organs such as lung and heart. We found that the administration of aspirin for the 4 days reduced the body growth. This reduction in growth may be due to the loss of appetite and the reduced food consumption. These results were correlated with the study of Olaibi et al., 23 which showed that the administration of aspirin with dose 150 and 300 mg/kg during 2 weeks induced a decrease in body weight.
These findings were confirmed by histopathological observation of the brain, intestine, and lung. This alteration can be due to the oxidative stress induced by aspirin. It is known that the brain is an important organ whose function is to maintain the normal function of the other organs. However, any damage or malfunction resulting from a stress condition can have serious consequences for the total body. Indeed, the scientific research reveals that the nervous tissue is more sensitive to oxidative stress than other tissues because of its low AA. 24 Besides, the nervous tissue has a high concentration of polyunsaturated fatty acids (after adipose tissue), which are easy targets for oxidative damage by free radicals due to unsaturated bonds. 25,26 In the present study, the administration of aspirin induced severe degenerative changes in neurons of brain. It can be explained that the oxidative stress result by the aspirin can lead to peroxidation of membrane lipids which can disrupt membrane fluidity and cell compartmentation and can result in cell lysis. It is known that the central nervous system is particularly susceptible to the damaging effects of reactive oxygen species (ROS).
In addition, in our study, aspirin induced damage in lung tissue which can be highlighted by degeneration of the alveolar septa, fibrosis, disruption of the connective tissues and elastic fibers, and the congestion of the blood capillaries.
These findings are in agreement with those reported by Taylan et al., 27 which demonstrated that aspirin at 100 mg/kg induced damage in lung tissue. This can be explained by the induction of oxidative stress that plays a role in Aspirin (ASA)-lung. Generally, the oxidative stress induced tissue damage by oxygen-free radicals, which was considered as the hallmark of several chronic disorders and cell death. The ROS is important mediator of cellular injury and can contribute to a variety of diseases.
Our data also show that aspirin induced injury in intestine histological study atrophy of surface epithelium, cystic dilation in crypts, and crypt loss. The obtained results were similar to those reported by Lichtenberger et al. 28 showing that aspirin administered at 100 mg/kg induced injury to the surface of intestinal mucosa in rats. This finding shows that aspirin induced damage in the intestinal epithelial cells IEC-6. 10 A number of other studies have shown that aspirin causes ulcer in small intestine, 29 –31 which explains that aspirin can induce acute surface injury to the tissue by Cyclooxygenase (COX)-independent mechanisms. Aspirin induced COX-inhibition and inhibits the synthase of prostaglandins which promote the barrier properties of the tissue. Therefore, aspirin induced the depletion of cytoprotecteur of prostaglandins, which induced injury to the tissue.
Nowadays, scientific research reveals that the antioxidant property of the plant extracts gives beneficial effect to human health to protect many physiological functions. 32 In the context, our data showed that the pretreatment with EOC implicated a strong protective effect against aspirin-induced injury in tissues as revealed by the increase of the body weight in the EOC + A group rats. And it is also revealed by the ameliorative weights of some organs. Histopathological brain, lung, and intestine section of the rats treated with the EOC reports normal appearance of the structure as compared to the control group.
Based on these findings, it is clear that the observed protective activity of EOC can be attributed to the presence of phenolic, flavonoid, flavonol, and their composition of terpenes. EOC can block the harmful action of the free radicals caused by aspirin; it scavenges and detoxifies the organism. The study of Saumya and Bacha 20 proved that the antioxidants may protect the body against ROS toxicity either by preventing the formation of ROS, by bringing interruption in ROS attack, by scavenging the reactive metabolites, or by converting them to less reactive molecules. It is noted that the AA of essential oils is of great interest because they may preserve foods from the toxic effects of oxidants. 33 Essential oils that are able to scavenge free radicals may play an important role in some disease preventions such as brain dysfunction, cancer, heart disease, and immune system decline. 34
Conclusion
In the present work, the AA from EOC was evaluated. It exhibited potential AA by ferric reducing antioxidant power (FARP), β-carotene, and assays, and it possessed moderate flavonoid and flavonol content. These antioxidative properties could be useful for the prevention of aspirin-induced injury to brain, lung, and intestine. As our investigations stand at present, complementary studies of oxidative stress of each organ will be necessary to confirm the protective effect of EOC.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.

