Abstract
Lambda-cyhalothrin (LTC) is a synthetic pyrethroid with a broad spectrum of insecticidal and acaricidal activities used to control wide range of insect pests in a variety of applications. The aim of this study was to examine (i) the potency of LTC to induce oxidative stress response in rat erythrocytes in vitro and (ii) the role of caffeic acid (20 μM) and/or quercetin (10 μM) in preventing the cytotoxic effects. Erythrocytes were divided into four portions. The erythrocytes of the first portion were incubated for 4 h at 37°C with different concentrations (0, 50 and 100 μM) of LTC. The others portions were pretreated with caffeic acid and/or quercetin for 30 min prior to LTC incubation. Lipid peroxidation, protein oxidation, antioxidant enzyme activities and DNA damage were examined. LTC at different concentrations causes increased levels of lipid peroxidation, protein oxidation, DNA damage and decreased antioxidant enzyme activities. Combined caffeic acid and quercetin pretreatments significantly reduced the levels of lipid peroxidation markers, that is thiobarbituric acid reactive substance (TBARS), protein carbonyls (PCO) and decreased DNA damage in LTC portion. Further, combined caffeic acid and quercetin pretreatment maintain antioxidant enzyme activities and glutathione content near to normal values. These results suggest that LTC exerts its toxic effect by increasing lipid peroxidation, altering the antioxidant enzyme activities and DNA damage. Caffeic acid and quercetin pretreatments prevent the toxic effects of LTC, suggesting their role as a potential antioxidant.
Keywords
Introduction
Humans are potentially exposed to pesticides either directly, as workers in greenhouses and in agriculture, or indirectly, via food consumption. In addition, it is likely that a significant amount of these pesticides and their metabolites reach rivers and estuaries via run-off from farmland that are potentially toxic to wildlife. 1 Lambda-cyhalothrin (LTC), a synthetic pyrethroid insecticide, has been extensively used in the last two decades in many of the developing countries for combating agricultural pests and insects of veterinary as well as human concern. The synthetic pyrethroid insecticides are widely applied in view of the fact that they have shown to possess a high insecticidal activity as well as a broad spectrum of high initial toxic action on several types of pest. The toxicity of pyrethroid insecticides to mammals has received much attention in recent years because animals exposed to these insecticides showed changes in their physiological activities besides other pathological conditions.2–4 Recent studies indicate that oxidative stress is the major mechanism of synthetic pyrethroid-induced toxicity in various tissues (kidney, liver, brain, etc.).5–7
Erythrocytes are prone to oxidative stress because of their constant exposure to high oxygen tension, high content of polyunsaturated fatty acid in their membranes coupled with high amount of haemoglobin-bound iron. 8 ,9 However, the erythrocytes are armed with efficient antioxidant machinery in the form of antioxidant enzymes, such as superoxide dismutase (SOD), catalase (CAT), glutathione-S-transferase and antioxidant molecules such as vitamins C and E that scavenge reactive oxygen species to maintain cellular integrity. 10 A crucial function for the preservation of the integrity of red cells is maintenance of their low intracellular calcium concentration against a very steep electrochemical gradient. 11 This is mediated both by a low membrane permeability to Ca2+ in the inward direction and by an active efflux mechanism catalyzed by the membrane-bound enzyme Ca2+-ATPase. 12 Functional alterations such as inhibition of Na+/K+ ATPase and Ca2+ATPase property and elevated cellular and membrane lipid peroxidation contribute to various pathological conditions. 13 ,14
During the last decades, phenolic compounds, naturally occurring secondary plant metabolites, have received considerable attention due to their antioxidant properties. 15 ,16 Quercetin and caffeic acid exhibit a wide range of physiological and pharmacological activities relevant to human health, such as anticarcinogenic, anti-inflammatory and antiviral actions. 17 ,18 Many of these effects are supposed to be related to its antioxidant property. Quercetin can act by scavenging free radicals, chelation of divalent cations, inhibition of some enzymes and protecting against DNA damage. 19 ,20 Caffeic acid phenethyl ester (CAPE) has anti-inflammatory, 21 anticarcinogenic, antiviral, immunomodulatory, 22 and antioxidant activities. 23 In vitro studies showed that CAPE is protective against hepatotoxicity in mice, 24 reduced the levels of intracellular hydrogen peroxide (H2O2) and oxidized bases in DNA. 25 Although, several studies have shown that exposure to pesticides may induce genotoxic effects in occupationally exposed human populations. 26 ,27 No much attention has been paid to study the mechanism of synthetics pyrethroids especially, LTC on the genotoxicity effects, oxidative damage and integrity of mammalian erythrocytes. For this reason, the present work is designed to investigate the in vitro effects of the insecticide LTC in rat erythrocyte by evaluation of its capability to induce oxidative damage and genotoxicity. At the same time, the present work is essentially planned for evaluation of the possible protective effects of two natural products, viz. quercetin and caffeic acid against in vitro cytotoxicity effects of the insecticide LTC.
Materials and methods
Chemicals
LTC is a synthetic pyrethroid insecticide (C23H19 ClF3NO3). CAS chemical name (a-cyano-3-phenoxybenzyl-3-[2-chloro-3,3,3-trifluoro-1-propenyl]-2,2-dimethylcyclo-propanecarboxylate) with a CAS registry number 91465-08-6. A commercial formulation of LTC, named ‘KARATEs 5EC’ (Syngenta Agrochemicals, Greensboro, USA), was used in the experiments. All other chemical products used in this study were purchased from Sigma Chemical Co. (St Louis, France).
Animals and care
Adult Wistar rats (aged 8–9 weeks; weighing 140–150 g) were obtained from the Central Pharmacy (SIPHAT, Tunis, Tunisia).The animals were housed at the following conditions: 22 ± 3°C temperature, 45 ± 5% humidity and 12 h light–dark cycle, and the animals were left to acclimatize for 1 week before the experiments. They were fed standard laboratory diet and provided with water ad libitum.
All animal procedures were in strict conformation with the Institute Ethical Committee guidelines for the care and use of laboratory animals.
Preparation of erythrocytes
Rats were killed under mild diethyl ether anaesthesia. Blood was drawn by cardiac puncture into tubes containing 3.8% trisodium citrate solution. Blood was centrifuged at 1000×g for 10 min at 4°C, and the plasma and buffy coat were removed. The erythrocytes were washed three times with ice-cold phosphate-buffered saline ([PBS] 145 mM NaCl, 1.9 mM NaH2PO4 and 8.1 mM Na2HPO4) and centrifuged. The packed erythrocytes after the final wash were resuspended in phosphate buffer (0.1 M, pH 7.4) at 1:9 dilution and used for incubations.
Treatment of erythrocytes
Erythrocytes were divided into four portions. The first portion was incubated for 4 h at 37°C with different concentrations of lambda-cyhalothrin (50 and 100 μM). The above concentrations of another pesticide had been applied in earlier in vitro investigation on mammalian erythrocytes. 29 ,30 The second portion was preincubated with caffeic acid (20 μM) for 30 min followed by incubation with 100 μM of LTC for 4 h. The third portion was preincubated with quercetin (10 μM) for 30 min followed by incubation with 100 µM of LTC for 4 h. These concentrations of caffeic acid and quercetin were chosen because earlier studies showed that these concentrations afforded significant amelioration in lowering cytotoxicity. 31 ,32 One negative control contained erythrocyte alone and two others served as positive controls, containing CAPE or quercetin (QE). The same incubations were used on blood samples collected by cardiac puncture into tubes containing 3.8% solution of trisodium citrate as an anticoagulant to perform the comet assay.
Determination of oxidative biomarkers
Lipid peroxidation in erythrocyte was estimated colorimetrically by measuring thiobarbituric acid reactive substances (TBARS) which were expressed in terms of malondialdehyde (MDA) content according to Draper and Hadley method. 33 The MDA values were calculated using 1,1,3,3-tetraethoxypropane as the standard and expressed as nmoles/mg protein.
Protein oxidation was determined based on the reaction of the carbonyl groups with 2,4-dinitrophenylhydrazine (DNPH) to form 2,4-dinitrophenylhydrazone. 34 Samples were read at 370 nm and carbonyl content was calculated using the molar absorption coefficient for aliphatic hydrazones (22,000 M−1 cm−1) and expressed as nmol carbonyl/mg protein.
Determination of antioxidant enzyme activities
CAT activity was determined according to Aebi method. 35 One unit of enzyme activity was defined as the activity required to degrade 1 μmol of H2O2 in 60 s. Enzyme activities was expressed nmol/min/mg protein.
SOD activity was determined according to the method of Marklund and Marklund 36 by measuring the inhibition of pyrogallol autoxidation. One unit of SOD is defined as the amount of enzyme that inhibits the rate of pyrogallol autoxidation in 50%.
Glutathione peroxidase (GPx) activity was measured according to Flohe and Gunzler method. 37 The enzyme activity was expressed as nmoles of GSH oxidized/min/mg protein.
Determination of nonenzymatic antioxidant
The total glutathione (tGSH) content was measured by Ellman method. 38 This method was based on the development of a yellow colour when 5,5-dithiobis-2-nitrobenzoic acid was added to the compounds containing sulphydryl groups. The tGSH levels were expressed as nmol/g haemoglobin (Hb).
Assay of membrane-bound enzymes
Na+/K+-ATPase was assessed in erythrocyte membrane preparation according to the Quigley and Gotterer method. 39 The Na+/K+-ATPase activity was measured as the difference between total ATPase activities and ouabain-insensitive ATPase activities.
The Ca2+-ATPase activity was measured according to the Desaiah et al.’s method. 40 The inorganic phosphate released by the action of ATPases was estimated by the Fiske and Subbarrow method. 41
Protein content
The protein content in the erythrocyte membrane was estimated according to the method of Lowry et al., 42 using bovine serum albumin (BSA) as a standard.
Single-cell gel electrophoresis (comet assay)
The comet assay (alkali method) was performed according to the method developed by Singh et al. 43 with a few modifications. Briefly, 20 μl total blood of each sample was mixed with 120 μl of 0.5% low melting point agarose in PBS at 37°C and pipetted onto 8 microscope slides precoated with a layer of 1.5% normal melting point agarose prepared in PBS. Slides were then immersed in a lysis solution (2.5 M NaCl, 100 mM Na2EDTA, 10 mM Tris, pH 10.0–10.5, 1% lauroyl sarcosine, with 1% Triton X-100 and 10% dimethyl sulphoxide added fresh) for 1 h at 4°C. After lysis, the slides were placed in an alkaline electrophoresis buffer (300 mM NaOH, 1 mM Na2EDTA, pH 13.0). Once completed, the slides were washed three times with neutralizing solution (0.4 M Tris, pH 7.5) and scored using a fluorescent microscope equipped with an excitation filter 546 nm and a barrier filter of 590 nm at ×400 magnification. The tail intensities (%) of 100 comets on each slide were determined using specialized Image Analysis System (Comet Assay IV, Perceptive Instruments Ltd, UK).
Statistical analysis
Data were expressed as mean ± SD. One-way analysis of variance and Student–Newman–Keuls post hoc tests were performed on the data for intergroup comparisons. Database management and statistical analysis were performed using SPSS (SPSS 11, Chicago, IL, USA) statistical software package. Differences were considered significant if p < 0.05.
Results
Effect of caffeic acid and quercetin pretreatments on LTC toxicity
The levels of TBARS and PCO were significantly increased in rat erythrocytes incubated with LTC at 50 μM (44.8% and 41.7%, respectively) and at 100 μM (180.6% and 120.1%, respectively) compared with control (Table 1). Combined CAPE and QE significantly reduced the levels of lipid peroxidation (62.24%; p < 0.05) and protein oxidation (61.5%; p < 0.05) compared with LTC-treated erythrocytes.
Effects of caffeic acid (20 μM) and quercetin (10 μM) on the levels of TBARS, PCO and total glutathione in normal and LTC-treated rat erythrocytes a

CAPE: caffeic acid phenethyl ester, LTC: lambda-cyhalothrin, PCO: protein carbonyl, QE: quercetin, TBARS: thiobarbituric acid reactive substance, tGSH: total glutathione.
a Values are given as mean ± SD of six experiments in each group.
b Significantly different from control group (p < 0.05).
c Significantly different from LTC-treated group (100 μM; p < 0.05).
The levels of nonenzymatic antioxidant tGSH was found to be significantly decreased in LTC-treated erythrocytes at 100 μM (47.8%; p < 0.05) compared with control. Combined CAPE and QE restored the tGSH levels to normal levels compared with control and significantly increased the tGSH levels (89.6%; p < 0.05) compared with LTC-treated erythrocytes.
Alterations in ATPases activities
Table 2 shows the effect of in vitro incubation with LTC on the activity of Na+/K+-ATPase and Ca2+-ATPase activities. Na+/K+-ATPase and Ca2+-ATPase activities were significantly decreased in LTC-treated group at 100 μM (43.32% and 34.25%, respectively) compared to control group. CAPE and QE pretreatments significantly increased the activity of Na+/K+-ATPase (101.24%; p < 0.05) and Ca2+-ATPase (32.48%; p < 0.05) compared with LTC-treated erythrocytes.
Effects of caffeic acid and quercetin pretreatment on ATPases activities in LTC-treated erythrocytes a

CAPE: caffeic acid phenethyl ester, LTC: lambda-cyhalothrin, QE: quercetin.
aValues are given as mean ± SD of six experiments in each group.
b Significantly different from control group (p < 0.05).
c Significantly different from LTC-treated group (100 μM; p < 0.05).
Enzymatic antioxidant activity
Table 3 shows the activities of SOD, CAT and GPx in normal, LTC-treated erythrocytes and CAPE or/and QE pretreatment erythrocytes. The activities of CAT, SOD and GPx were significantly decreased in LTC-treated erythrocytes at 100 μM (49.21%, 38.93% and 58.46%, respectively) compared to control group. Pretreatment with QE alone significantly increased the CAT (83.23%; p < 0.05) and GPx (59.26%; p < 0.05) activities compared to LTC-treated group at 100 μM. Further, CAPE and QE pretreatments significantly increased the CAT (88.66%; p < 0.05), SOD (44.97%; p < 0.05) and GPx (100%; p < 0.05) activity compared to LTC-treated group at 100 μM.
Effects of caffeic acid and quercetin pretreatment on catalase, superoxide dismutase and glutathione peroxidase activities in LTC-treated erythrocytes a

CAPE: caffeic acid phenethyl ester, CAT: catalase, GPx: glutathione peroxidase, LTC: lambda-cyhalothrin, QE: quercetin, SOD: superoxide dismutase.
a Values are given as mean ± SD of six experiments in each group.
b Significantly different from control group (p < 0.05).
c Significantly different from LTC-treated group (100 μM; p < 0.05).
Effect of caffeic acid and quercetin pretreatments on LTC-induced DNA damage
Table 4 and Figure 1 represent the DNA damage in LTC, CAPE and/or QE pretreated rat blood. The extent of DNA damage was calculated by means of percentage tail DNA and tail length. LTC treatment at 100 μM for 4 h significantly increased the percentage tail DNA (227.23%; p < 0.05) and tail length (33.52%; p < 0.05) compared to the control group (Figure 1). Moreover, CAPE and QE pretreatments significantly decreased the percentage tail DNA (63.02%; p < 0.05) and tail length (30.93%; p < 0.05) compared to LTC-treated group at 100 μM.
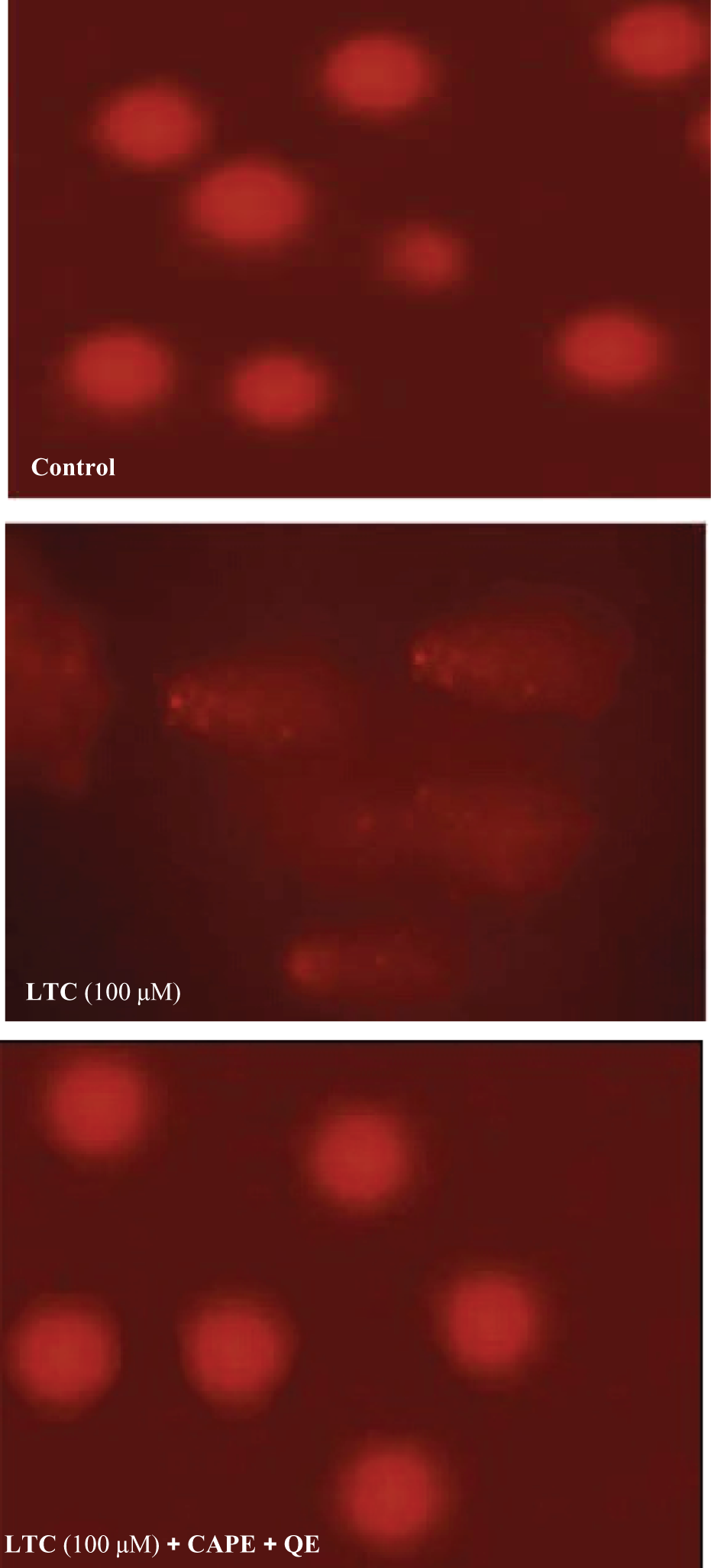
Effects of lambda-cyhalothrin (LTC), caffeic acid and quercetin pretreatment in rat blood for comet assay.
Effects of LTC, caffeic acid and quercetin on DNA damage (% tail DNA, tail length) a
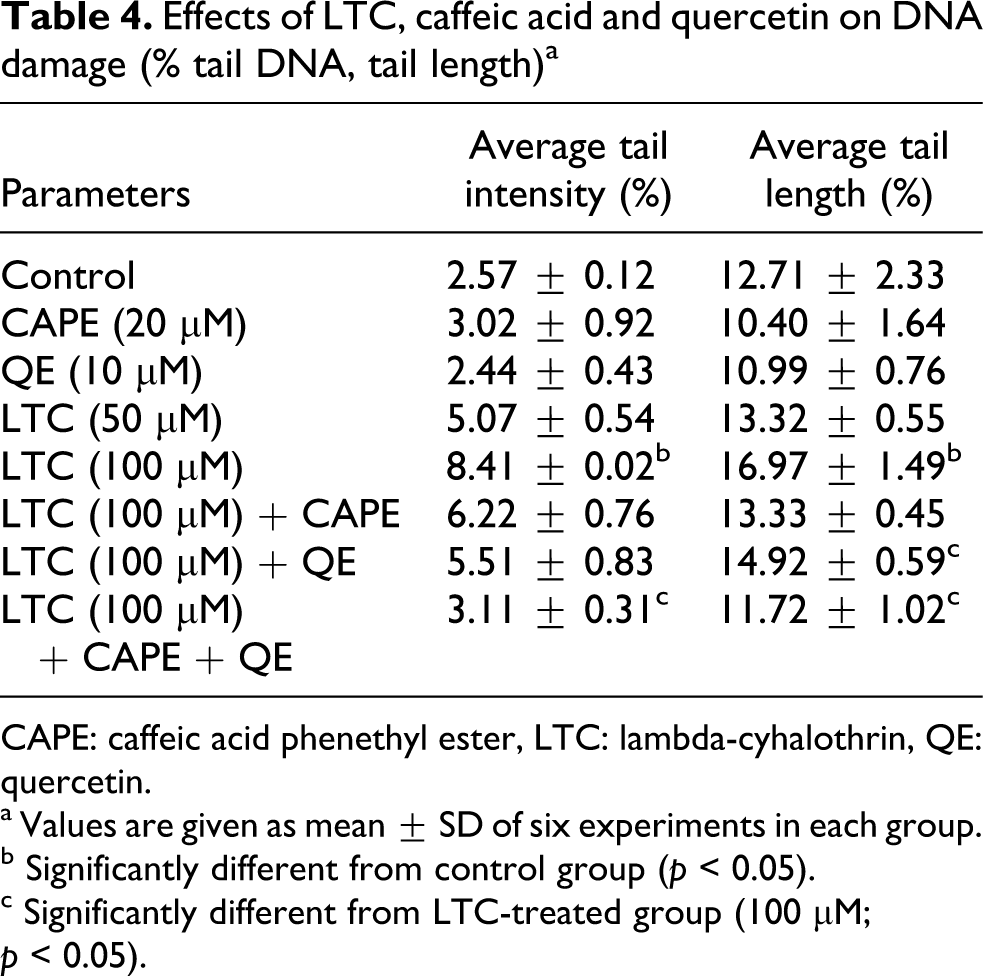
CAPE: caffeic acid phenethyl ester, LTC: lambda-cyhalothrin, QE: quercetin.
a Values are given as mean ± SD of six experiments in each group.
b Significantly different from control group (p < 0.05).
c Significantly different from LTC-treated group (100 μM; p < 0.05).
Discussion
Erythrocytes represent a convenient model to investigate not only the degree of lipid peroxidation owing to the presence of polyunsaturated fatty acids but offer themselves as good models to study the oxidative damage to proteins as well. 44 Oxidants are known to produce alterations in erythrocyte membranes as manifested by a decreased cytoskeletal protein content, and production of high-molecular weight proteins 45 ,46 which can lead to abnormalities in erythrocyte shape and disturbances in the microcirculation. 47 Oxidative damage of erythrocyte membrane (lipid/protein) have been implicated in haemolysis associated with some haemoglobinopathies, oxidative drugs, transition metal excess, radiation and deficiencies in some erythrocyte antioxidant systems. 48 Accordingly, it is pertinent to investigate the susceptibility of erythrocytes to synthetic pyrethroid induced oxidative damage.
In present study, incubation of rat erythrocytes with high concentration of LTC (100 μM) significantly increases lipid peroxidation (TBARS), protein oxidation (PCO) and significantly decreased membrane Na+/K+ and Ca2+-ATPase activities. These results indicate its efficacy to alter membrane integrity and fluidity in mammalian cells. In agreement with previous findings, Michelangeli et al. 49 reported that synthetic pyrethroid compounds are more hydrophobic than other classes of insecticides, and they could penetrate to the cell membrane easily. Further, according to World Health Organisation, 50 LTC toxicity might be due to the release of cyanohydrins, which were unstable under physiological conditions and further decomposed to cyanides and aldehydes which in turn could act as a source of free radicals. Our results corroborate a previous study which demonstrated that by-products of lipid peroxidation have been shown to cause profound alterations in the structural organization and functions of the cell membrane including decreased membrane fluidity, increased membrane permeability, inactivation of membrane-bound enzymes like ATPases and loss of essential fatty acids. 51 Pretreatment with CAPE (20 μM) and QE (10 μM), 30 min priori to LTC incubation prevents membrane cells against LTC toxicity by reducing the levels of TBARS, PCO levels and maintains ATPase activities near to normal values. We suggest that the wide mixture of phenolic compounds such as CAPE and QE may interact to produce synergistic protection against LTC toxicity in rat erythrocytes. The antioxidant properties of these compounds might lie in their ability to insert into cell membranes and modify the lipid packing order and fluidity. 52 ,53 Oteiza et al. 54 reported that in biological membranes and in membrane models, the interaction between phenolics compounds and the lipid bilayer results in either binding at the lipid water interface or inclusion in the bilayer interior, with the different location or distribution of these molecules determined by their chemical properties. In addition, it has been suggested that flavonoids may be concentrated near the membrane surface in order to scavenge aqueous oxygen radicals.
Antioxidant enzyme activities such as SOD, CAT and GPx are the main enzymes which play a role in the development of a response to enzymatic oxidative stress caused by synthetic pyrethroid. 55 ,29 The activity of CAT was found to be significantly decreased at 100 μM LTC concentration in comparison with the control value. After incubation rat erythrocytes with the highest concentration of LTC (100 μM), the SOD activity was also significantly decreased over the control value. Two main mechanisms may have played a role in the decrease of the antioxidative enzyme activities. The first involves consumption during the breakdown of free radicals and the high level of H2O2 or the inhibition of the enzyme by these radicals, whereas the second involves the direct inhibition of SOD and CAT by LTC. 56 In agreement with our results, El-Demerdash 29 found that CAT and SOD activities were significantly reduced by different concentrations of LTC in rabbit erythrocytes treated in vitro. In a report by Prasanthi et al., 30 the fenvalerate induced oxidative damage seen by alterations in the enzymatic defence system in erythrocytes. Results obtained by Yousef et al. 57 also showed that CAT and SOD activities were significantly decreased due to deltamethrin administration in rats. Pretreated rat erythrocytes in vitro with CAPE and QE showed a protective action against the decrease of CAT, SOD and GPx activities induced by LTC. The mechanism by which the phenolic compounds such as CAPE and QE increased the levels of SOD, CAT and GPx activities in the LTC-treated groups may involve the dismutation of superoxide anions (O2 −) and the decomposition of H2O2 and may represent an aspect of the cellular response to increased levels of ROS induced by LTC toxicity. Facino et al. 58 demonstrated that phenolics effectively inhibit collagen (type III) fragmentation induced by superoxide anion and hydroxyl radicals. Hence, CAPE and QE may prevent the synthesis and utilization of antioxidant enzymes. This may be the reason for increased SOD, CAT and GPx activities observed in erythrocytes pretreated with combined CAPE and QE compared to LTC-treated group at 100 μM.
Further, pesticides are well-known inducers of oxidative stress. 59 Oxidative stress in turn induce genotoxicity in many cell types. Therefore, to find out whether LTC-mediated DNA damage is due to oxidative stress or not, we measured cellular GSH levels, since reduced GSH is the major intracellular antioxidant that has been implicated in the regulation of apoptosis or necrosis. Our results indicate that incubated rat erythrocyte in vitro with high concentration of LTC (100 μM) leads to GSH depletion and drastic reduction of GSH levels. The mechanism by which reduction in GSH levels induces apoptosis is once again not known. The involvement of oxidative stress may induce mitochondrial membrane damage leading to the release of cytochrome c which is possibly responsible for LTC-induced apoptosis. Earlier Kannan et al. 60 have demonstrated that mitochondrial disruption following GSH depletion is responsible for induction of apoptosis in a B-cell lymphoma cell line. Pretreatment with CAPE and QE can protect against LTC-induced DNA damage by decreasing the percentage tail DNA and tail length, suggesting that caffeic and quercetin exhibit a scavenging propriety against LTC damage.
In conclusion, LTC could modify antioxidant enzyme activity and enhance lipid peroxidation and DNA damage. Caffeic acid and quercetin reduced the extent of lipid peroxidation and DNA damage. Quercetin and caffeic acid showed a protective action against the inhibition of CAT, SOD and GPx activities induced by LTC in rat erythrocytes.
Footnotes
Fatma Ben Abdallah and Hamadi Fetoui contributed equally to this work.
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
