Abstract
This study investigated whether hyperthermia within the first 24 h after presentation was associated with long-term neurological outcomes after acute carbon monoxide (CO) poisoning. This retrospective study included 200 patients with acute severe CO poisoning. Hyperthermia (≥ 37.5°C) developed during the first 24 h after presentation in 55 (27.5%) patients, and poor long-term neurological sequelae assessed at 23 months after acute CO poisoning developed in 19.5% of the patients. The incidence of poor long-term neurological outcomes was significantly higher in the hyperthermia group than in the normothermia group. Patients with poor long-term neurological outcomes had higher maximum temperatures than patients with good outcomes. No significant difference was found in the time of hyperthermia onset within the first day according to the neurological outcomes. Hyperthermia (adjusted odds ratio (aOR) 5.009 (95% confidence interval (CI) 1.556–16.126)) and maximum temperature (aOR 2.581 (95% CI 1.098–6.063)) within the first 24 h after presentation to the emergency department were independently associated with poor long-term neurological outcomes. Body temperature measurements, which are easily and noninvasively recorded at the bedside in any facility, help to predict the risk for poor long-term neurological outcomes. This study carefully emphasizes fastidious control of pyrexia, particularly during the early period after acute CO poisoning.
Introduction
Although the mechanism by which hyperthermia induces brain damage has not been elucidated, increased release of neurotransmitters, such as glutamate, and proinflammatory cytokines, increased free radical production, and changes in adenosine triphosphate metabolism have been suggested. 1 Previous laboratory and clinical studies have reported the deleterious effect of elevation of the body or central nervous system temperature on histopathological and functional outcomes in various brain injury models, including cardiac arrest, cerebral ischemia, traumatic brain injury, and intracranial hemorrhage. 1 –3 Thus, monitoring body temperature and active control of hyperthermia are recommended as treatment strategies for these brain injuries. 4 Furthermore, targeted temperature management (TTM) is widely used to limit the extent of initial brain lesions in patients with these injuries.
Acute carbon monoxide (CO) poisoning accounts for more than 50,000 annual emergency department (ED) visits. 5 The major problem associated with CO poisoning is the risk of neurological sequelae faced by survivors, even after acute treatment. Neurological sequelae after CO poisoning are divided into acute persistent neurological sequelae (develop immediately after acute poisoning) and delayed neurological sequelae (DNS; develop after an initial complete clinical recovery from acute poisoning). From 10.1% to 14% of patients with severe acute CO poisoning exhibit persistent severe neurological sequelae, rendering them dependent on others throughout their lifetimes. 6,7 The neurological sequelae that develop after accidental CO poisoning cost the United States approximately US$1 billion annually in terms of loss of lifetime earnings. 8 Therefore, prediction and prevention of neurological sequelae are important for individuals with CO poisoning. However, in clinical practice, our only therapeutic option for CO poisoning is hyperbaric oxygen (HBO) therapy, and the effectiveness of HBO therapy for the prevention of neurological sequelae is controversial. 9 Recently, the need to treat patients with CO poisoning using a series of potential treatments has been emphasized. 9
Elevated body temperature, which often occurs in response to cellular injury, activation of inflammatory cascades, changes in the thermoregulatory mechanism due to hypothalamic lesions, or infection 10 is observed in 26.1% of patients with acute CO poisoning. 11 Brain temperatures measured by magnetic resonance spectroscopy are abnormally high in the acute phase (approximately 1 week) and remain abnormal in the subacute phase (approximately 2 weeks) of CO poisoning. 12,13 Small increases in brain temperature substantially exacerbate histopathological damage in cerebral neurons independent of the initial injury severity. 14 Although these findings imply an effect of hyperthermia on neurological outcomes in patients with acute CO poisoning, whether this effect occurs in patients with acute CO poisoning has not been investigated. Furthermore, monitoring body temperature and controlling hyperthermia in the acute stage after CO poisoning have not been considered as potential treatment strategies despite their implementation in cases of cardiac arrest or other brain injuries. 1 –3
Therefore, we hypothesized that hyperthermia after acute severe CO poisoning would be associated with poor neurological outcomes. We also hypothesized that patients with earlier onset of hyperthermia would have poorer outcomes than those with later onset.
Materials and methods
Study design and setting
This investigation was a retrospective, observational study performed via a chart review at a single academic tertiary care center with an annual ED census of 40,000 patients. The study design was approved by the Institutional Review Board of Chonnam National University Hospital (Gwangju, South Korea, IRB number: CNUH-2018-046).
Participant selection
The inclusion criteria for this study were as follows: patients 18 years or older who presented to our ED with CO poisoning within 24 h between January 2014 and February 2017 and who had an indication for HBO therapy at ED presentation. Patients who presented to the ED at least 1 year prior to the study were included, because most DNS resolve over a period of 3–12 months 15 and we were interested in examining long-term neurological outcomes (persistent neurologic sequelae lasting for at least 12 months regardless of the onset of neurologic sequelae (acute persistent neurologic sequelae or DNS)).
Two trained physicians who were blinded to the patient outcomes determined whether the patients met the definition of CO poisoning, the indications for HBO therapy for CO poisoning, and the presence of any exclusion criteria. CO poisoning was defined as a history of exposure to CO and an initial carboxyhemoglobin (COHb) level exceeding 5% (10% in smokers) at our ED or the primary hospital. The indications for HBO for nonpregnant patients were defined as the presence of any neurological deficit, including loss of consciousness and seizure, and a COHb level greater than 25% regardless of symptoms. The exclusion criteria were pregnancy, a history of an altered mental state before the CO poisoning event, a history of taking anti-inflammatory medications (corticoids), death upon arrival or at the scene, mixed poisoning with other drugs, discharge against the doctor’s orders, transfer before the final outcome was determined, no recorded body temperature during the first 24 h, and treatment using a protocol that did not adhere to our hospital’s guidelines for CO poisoning (Figure 1).

Algorithm for subject selection. Indication for hyperbaric oxygen therapy* was defined as having any neurological deficits, including loss of consciousness and seizure, and a COHb level above 25% regardless of symptoms. COHb: carboxyhemoglobin.
Our hospital’s HBO therapy consists of 100% oxygen with increasing absolute pressures (from 1.0 ATA to 2.8 ATA) for 20 min, 2.8 ATA for 40 min, and decreasing absolute pressures (from 2.8 ATA to 1.0 ATA) for 20 min. If the indication for HBO therapy does not resolve after one HBO session, an additional session is provided. The maximum number of HBO sessions within 24 h after presentation is three. When a patient with indications for HBO requires mechanical ventilation (MV) or a vasopressor infusion due to refractory hypotension, the patient is treated with MV at a Fraction of inspired oxygen level of 1.0 instead of HBO.
Methods and measurements
The following data were directly collected from electronic medical records: age, sex, laboratory results at presentation, and hospital duration.
Two trained physicians who were blinded to the patient prognosis collected the following information by reviewing patient medical records and entering the data into a standardized Excel spreadsheet: duration of exposure to CO; source of CO; time interval from the last exposure to CO to arrival at our ED; intentionality of exposure to CO; Glasgow Coma Scale (GCS) score and vital signs at presentation; body temperature during the first 24 h after presentation; non-administration of HBO therapy during the first 24 h; the time interval between arrival at the ED and the initiation of the first HBO treatment; the number of HBO sessions during the first 24 h of admission; coinfection within the first 48 h; development of complications during hospitalization; and GCS score at discharge. Non-administration of HBO therapy during the first 24 h was defined as a case in which HBO therapy could not be performed at all or after one or two sessions of HBO therapy within the first 24 h due to the patient’s general conditions, such as respiratory failure, despite the need for HBO therapy. A coexistent infection was considered if a positive urine culture or leukocyturia (≥ 20 cells in a visual field), a positive blood culture, a positive sputum culture, or radiological evidence of pneumonia was observed. 16
The investigated complications during hospitalization included respiratory failure (need for MV support regardless of the reason), hypotension (need for intravenous vasopressor infusion to maintain blood pressure), and myocardial injury (diagnostic electrocardiography changes or troponin I levels > 0.7 ng mL−1).
Hyperthermia and maximum temperature
Body temperature was measured at presentation at the ED, every 2–3 h during hospitalization, and upon complaint of feeling febrile or signs of upcoming fever, such as chills. A digital thermometer (MT 200®; Microlife, Clearwater, Florida, USA) with a measurement range of 32–42.9°C at a dry axillary site was used to measure the body temperature. The accuracy of the thermometer is ±0.1°C for temperatures ranging from 34°C to 42°C according to the manufacturer.
Hyperthermia was defined as an axillary temperature ≥ 37.5°C within the first 24 h after arrival at the ED after CO poisoning. 3,17 The maximum temperature was defined as the highest recorded temperature in the first 24 h after presentation. Three classification approaches were used to account for possible correlations between an elevated body temperature and outcomes: as a continuous variable, as an ordinary variable (by 0.5°C), and as a dichotomous variable (≥ 37.5°C) in separate models.
Timing of the onset of hyperthermia
The timing of the onset of hyperthermia was defined as the time between presentation to the ED and the first recorded hyperthermia. The timing of the onset of hyperthermia was assessed using the following criteria: (1) time interval ≤ 8 h (early) versus 8 h < time interval ≤ 16 h (middle) versus 16 h < time interval ≤ 24 h (late) and (2) continuously (hours from presentation to first temperature ≥ 37.5°C).
Outcomes
The long-term outcomes were assessed by structured telephone interview using the Glasgow Outcome Scale (GOS) and divided into good (GOS score of 4–5) and poor (GOS score of 1–3) outcomes. The five outcome categories of the GOS are as follows: death (score of 1), vegetative state (unable to interact with the environment, score of 2), severe disability (able to follow commands but unable to live independently, score of 3), moderate disability (able to live independently but unable to return to work or school, score of 4), and good recovery (able to return to work or school, although the patient might experience some minor neuropsychological deficits, score of 5). Patients whose physicians were not able to contact for follow-up were excluded from further statistical analyses.
Data analysis
The patients were divided into two groups based on the development of hyperthermia within 24 h after arrival at the ED (hyperthermia group vs. normothermia group) to review baseline patient characteristics. Descriptive statistics used proportions, means and standard deviations, and medians and interquartile ranges. For continuous variables, Student’s t-test or the Mann–Whitney U test was used for comparisons between two groups according to the normality of the data, which was tested using the Shapiro–Wilk test. Differences among more than two groups were examined using the Kruskal–Wallis test and the Mann–Whitney U test with Bonferroni’s adjustment as a post hoc comparison. For categorical variables, the χ 2 test or Fisher’s exact test was used.
For each analysis of the temperature variable, the potential confounders that were suggested as initial prognostic factors for CO poisoning (age, initial GCS score, arterial HCO3 −, white blood cell (WBC) count, and C-reactive protein (CRP), blood urea nitrogen (BUN), and creatinine (Cr) levels) 18 and the variables related to HBO therapy (non-administration of HBO therapy within the first 24 h and the time interval between arrival at the ED and initiation of the first HBO session) were tested in the univariate analysis. Although the beneficial effect of HBO therapy on preventing neurological sequelae remains controversial, the variables related to HBO therapy were entered into the analysis to exclude the effect of HBO therapy on neurological outcomes. 19 A multivariate logistic regression model using the entering technique was applied to significant factors obtained in the univariate analysis, coexistent infection within the first 48 h, and the temperature variable. The coexistence of infection within the first 48 h was entered into the multivariate regression model to exclude the confounding effect of coexistent infections, which might be a cause of hyperthermia and might account for or even contribute to the outcome. Body temperature was entered in the form of a continuous variable, an ordinary variable (by 0.5°C) or a dichotomous variable (≥ 37.5°C) in each separate model. Before modeling, if two or more univariate variables retained in the multivariate analysis were highly correlated in the linear regression analysis, one variable was removed to avoid collinearity. Estimated odds ratios (ORs) and 95% confidence intervals were calculated for all significant variables.
A p-value < 0.05 was considered significant. All statistical analyses were performed using SPSS 21.0 (SPSS, Chicago, Illinois, USA) and Stata version 13.1 (StataCorp, College Station, Texas, USA).
Results
Table 1 presents the baseline clinical characteristics of the 200 patients who presented to our ED at a mean of 2 h after acute exposure to CO and who required HBO therapy.
Baseline characteristics and clinical course of patients with acute severe CO poisoning stratified according to the development of hyperthermia within the first 24 h of admission.

CO: carbon monoxide; HBO: hyperbaric oxygen; ED: emergency department; MV: mechanical ventilation; GCS: Glasgow Coma Scale.
a The duration of exposure to CO was available for 110 patients (85 patients in the normothermia group and 25 patents in the hyperthermia group).
b The time interval from the end of exposure to CO to presentation at our ED.
c Initial lactate levels were available for 193 patients (138 patients in the normothermia group and 55 patients in the hyperthermia group).
d HBO therapy.
Fifty-five (27.5%) patients showed hyperthermia (37.9°C (37.6–38.3°C)) during the first 24 h after presentation. The hyperthermia group had a lower initial GCS score and arterial HCO3 −; higher WBC counts and CRP, BUN, Cr, and lactate levels; and a higher frequency of elevated troponin I levels at presentation than the normothermia group (Table 1). The frequencies of both non-administration of HBO therapy due to a critical condition and administration of more than one HBO session were higher in the hyperthermia group than in the normothermia group.
During hospitalization, the hyperthermia group exhibited a higher prevalence of respiratory failure, hypotension, and myocardial injury than the normothermia group. At discharge, the hyperthermia group more frequently exhibited a neurological deficit (GCS < 13) than the normothermia group (2.8% in the normothermia group vs. 29.1% in the hyperthermia group; p < 0.001).
During long-term follow-up, the incidence of poor long-term neurological outcomes was 19.5% at a median follow-up of 23 months. A significantly higher incidence of poor long-term neurological outcomes was observed in the hyperthermia group (47.3% in the hyperthermia group vs. 9% in the normothermia group; p < 0.001; Table 2). Patients with a poor long-term neurological outcome had a higher maximum temperature than patients with a good outcome (37.5°C (36.9–38.2°C) in the poor long-term neurological outcome group vs. 36.9°C (36.7–37.3°C) in the good long-term neurological outcome group; p < 0.001; Figure 2).
Long-term neurological outcomes in patients with acute severe CO poisoning stratified according to the development of hyperthermia within the first 24 h of admission.

CO: carbon monoxide; GOS: Glasgow Outcome Scale.
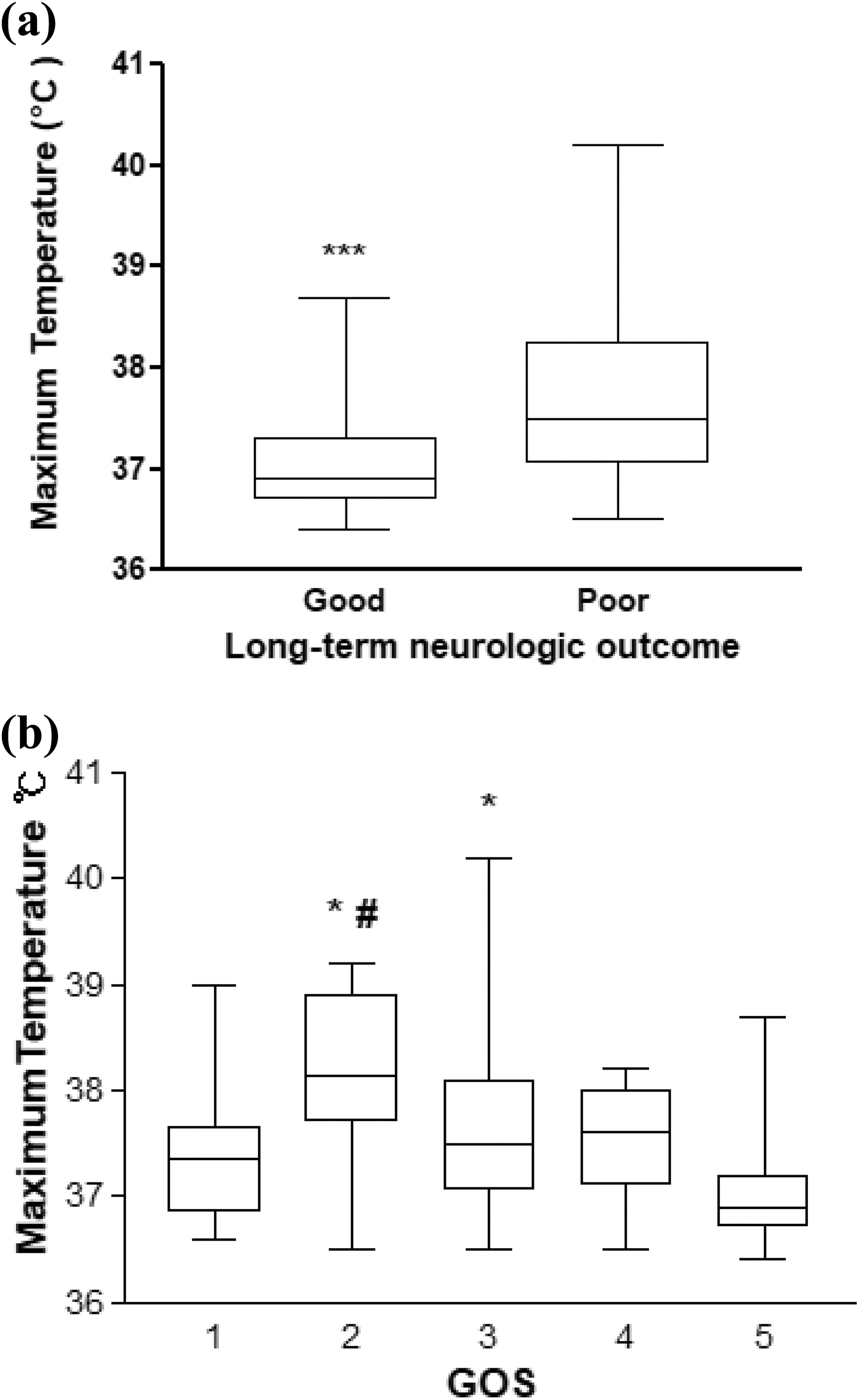
Maximum temperature recorded within the first 24 h after presentation in patients stratified according to long-term neurological outcomes and GOS scores. (a) Patients with poor long-term neurological outcomes had a higher maximum temperature than patients with good long-term neurological outcomes (37.5°C (36.9–38.2°C) in the poor long-term neurological outcome group vs. 36.9°C (36.7–37.3°C) in the good long-term neurological outcome group). ***p < 0.001 compared to poor long-term neurological outcomes. (b) The maximum temperature was 37.4°C (36.8–37.7°C) for patients with a GOS score of 1, 38.2°C (37.9–38. 8°C) for patients with a GOS score of 2, 37.5°C (37.1–38.1°C) for patients with a GOS score of 3, 37.6°C (37.1–38°C) for patients with a GOS score of 4, and 36.9°C (36.7–37.2°C) for patients with a GOS score of 5. A significantly lower maximum temperature was recorded in patients with a GOS score of 1 than in patients with a GOS score of 2 (p = 0.037). The maximum temperature recorded in patients with a GOS score of 5 was significantly lower than in patients with GOS scores of 2 (p < 0.001) and 3 (p < 0.001). #p < 0.05, compared to a GOS score of 1; *p < 0.001, compared to a GOS score of 5. GOS: Glasgow Outcome Scale.
Hyperthermia developed 6.7 h (2.0–9.7 h) after presentation, and no difference was observed in the time interval from arrival at the ED to onset of hyperthermia according to long-term neurological outcomes (7.8 h (2.3–11.8 h) in the good long-term neurological outcomes group vs. 4.8 h (0.1–9.0 h) in the poor long-term neurological outcomes group, p = 0.088; see Online Supplemental Data 1).
In the multivariate logistic regression analysis adjusted for age; initial GCS; arterial HCO3 −; Cr, CRP, and troponin I levels at presentation; variables related to HBO therapy; and coexistent infection within the first 48 h after presentation, the body temperature (as a dichotomous variable (body temperature ≥ 37.5°C), as a continuous variable, and as an ordinal variable (by 0.5°C) in separate models), was associated with long-term neurological outcomes (Table 3 and Figure 3).
Univariate and multivariate analyses for predictors of long-term neurological outcomes.

OR: odds ratio; CI: confidence interval; HBO: hyperbaric oxygen; BT: body temperature.

Association between the maximum temperature within the first 24 h after presentation and long-term neurological outcomes in patients with acute severe CO poisoning. The data are presented as the marginal probability with 95% CIs of a good long-term neurological outcome after acute CO poisoning given the maximum temperature after controlling for age, initial GCS score, arterial HCO3 −, the serum troponin I, Cr, and CRP levels at presentation, variables for HBO therapy, and the coexistence of an infection within 48 h. CO: carbon monoxide; CI: confidence interval; GCS: Glasgow Coma Scale; Cr: creatinine; CRP: C-reactive protein; HBO: hyperbaric oxygen.
Discussion
Based on the findings of the present study, hyperthermia within the first 24 h of admission was independently associated with long-term neurological outcomes in patients with acute severe CO poisoning. These results are consistent with the well-established deleterious effect of hyperthermia on patients with other brain injuries. 1 –3 Measurements of body temperature, which can be easily and noninvasively recorded at the bedside in any facility, help to predict the risk for poor long-term neurological outcomes.
In contrast with our findings, body temperature measured at least 12 days after acute CO poisoning was not correlated with the development of cerebral white matter damage. 13 Late measurements of temperature may partially account for the lack of a relationship between the temperature and outcomes in a previous study.
However, we noted that the maximum temperature recorded in patients with a GOS score of 1 was significantly lower than that of patients with a GOS score of 2; this result differed from the positive association observed between body temperature and the risk of poor long-term neurological outcomes in the multivariate analysis (Figure 2). Patients with unfavorable functional neurological recovery (a prolonged coma and brain death) after cardiac arrest had more frequent initial hypothermia (body temperature < 35°C). 20 The usual temperature course in patients with unfavorable neurological outcomes after cardiac arrest is an initial decrease followed by a fever. 1,21 A failure or delay in temperature elevation due to vasodilation related to the cardiovascular toxicity of CO or loss of shivering related to severe brain injury may produce the significantly lower temperatures observed in patients with GOS scores of 1.
We speculated that patients with an earlier onset of hyperthermia had a higher risk of poor outcomes based on a previous study showing earlier onset of hyperthermia (≥ 37.58°C) without a focus of infection (a fever of central origin) than for hyperthermia with infection during the poststroke period. 22,23 However, we failed to observe a relationship between the time of onset of hyperthermia and the outcomes of CO poisoning. The temporal profiles of the associations between body temperature and outcomes are inconsistent in patients with other brain injuries. Similar to our findings, the onset time of hyperthermia (≥ 38°C) that developed within 72 h after completion of TTM in patients with cardiac arrest was not related to the neurological outcomes. 10 One study did find an association between infarct size and body temperature on admission, 24 but other studies did not show an association with body temperature on admission 3,25 or within 6 h. 26 Hyperthermia (>37.5°C) that initiates within the first day of ischemic stroke onset but not afterward is associated with poor neurological outcomes, 27 whereas the body temperatures recorded on days 2 and 3 but not within the first day are correlated with functional outcomes after ischemic stroke. 25
In addition to being caused by infections, hyperthermia can develop due to changes in the thermoregulatory mechanism due to hypothalamic lesions or activation of inflammatory cascades in patients with CO poisoning. 4,28 Interestingly, increased pressure in the chamber during HBO therapy using the ideal gas law has been suggested to cause hyperthermia. 29 However, the cause of hyperthermia is not always evident in clinical practice. In this study, coexistence of an infection within the first 48 h was not associated with long-term neurological sequelae. Consistently, previous clinical studies showed that neither infections nor inflammation, which produce a fever, exerted a significant effect on death or functional outcomes after stroke in contrast to hyperthermia. 24,27 Hyperthermia was associated with unfavorable functional outcomes in patients with stroke regardless of the cause of hyperthermia. 27,30
The results of this study obviously lead to the question of whether control of hyperthermia, particularly within the first 24 h, yields improved outcomes. This study only reported an association between hyperthermia and poor outcomes but not a causal relationship between these variables. However, we carefully recommend that clinicians should direct standard treatment toward the immediate control of hyperthermia in patients within the first 24 h after acute severe CO poisoning based on the causal relationship between hyperthermia and poor neurological outcomes in patients with other brain injuries. 1 –3 Furthermore, the current situation, for which a proven therapy for CO-associated brain injury is not available, and the lack of ethical problems related to the control of hyperthermia in clinical practice support our suggestion. We are awaiting evidence of a positive effect of therapeutic hypothermia on prevention of neurological sequelae. 31 Until then, control of hyperthermia should be stressed as the goal in the management of patients with acute CO poisoning.
We noted that administration of HBO therapy was independently associated with the neurological outcomes only in the regression model including body temperature as a continuous variable but not in the other models. The inconsistent result may be due to the small number of patients with non-administration of HBO therapy. Whether HBO therapy is effective for improving long-term neurological outcomes is unclear. 32 However, because the aim of our study is to correlate hyperthermia with neurological outcomes and not to focus on the preventive effectiveness of HBO therapy, we did not further analyze the effect of HBO therapy in this study.
Our study has several limitations. The first important limitation is that the preferred peripheral sites for measuring temperature (i.e. oral, axillary, and tympanic) can differ among EDs. Axillary temperature measurements are a common, simple, noninvasive, and accessible method in any ED and display acceptable accuracy and precision as representative of the core body temperature. 33 –35 The precision of the axillary temperature measurements is comparable to the precision of other noninvasive temperature measurements (oral or temporal artery method). 36 However, because the range of normal body temperatures differs according to the site of temperature measurement, the cutoff values used to classify body temperatures as hyperthermic in this study should be changed. 37 Second, long-term neurological outcome assessments were not performed at the same fixed time point for all patients. However, the time interval from arrival at the ED after CO poisoning to the assessment of long-term neurological outcomes did not differ according to the outcome (27.5 months (18–30 months) for the poor outcome vs. 22 months (17–27.5 months) for the good outcome, p = 0.085). This time interval might be sufficiently long to observe improvements in reversible DNS regardless of the outcome. Additionally, no difference in the time interval was observed between the two groups stratified by the development of hyperthermia (Table 2). A third limitation was that the results obtained in this study had limited generalizability because of the underlying heterogeneity in clinical studies on CO poisoning regarding the severity of CO poisoning, treatment protocols (maximum HBO pressure, number of HBO sessions within 24 h, and duration of the compression or decompression period), and the source of CO. Therefore, these factors should be considered when interpreting the results of the present study. Fourth, because of the retrospective study design, there were missing data for some variables, such as the duration of exposure to CO and serum lactate. However, when the duration of exposure to CO was entered into the multivariate regression model, hyperthermia remained significant as an independent predictor of long-term neurological outcome (12.919 (1.609–103.696)). Lactate was highly correlated with arterial HCO3 − (r = –0.744, p < 0.001), which had already been entered into the multivariate regression model. Because of the collinearity between lactate and arterial HCO3 −, lactate could not be entered into the regression model. Additionally, we could not completely exclude the possible existence of undiagnosed subclinical infections due to the lack of a protocol to determine the cause of hyperthermia. However, infection was not associated with neurological outcome after stroke. 24,27 Further prospective research is needed to overcome these limitations originating from our retrospective study design.
Conclusion
In patients with acute severe CO poisoning, development of hyperthermia within the first 24 h after ED presentation is correlated with a risk of poor long-term neurological outcomes. Measurements of body temperature, which can be easily and noninvasively recorded at the bedside in any facility, help to predict the risk that patients will develop long-term neurological outcomes. Additionally, this study places careful emphasis on the fastidious control of pyrexia, particularly during the early period after acute CO poisoning. We await further clinical trials on the possible beneficial effects of therapeutic hypothermia. Until then, normothermia must remain the goal in the management of acute CO poisoning.
Supplemental material
Supplement - The impact of hyperthermia after acute carbon monoxide poisoning on neurological sequelae
Supplement for The impact of hyperthermia after acute carbon monoxide poisoning on neurological sequelae by JM Moon, BJ Chun, SD Lee, and MH Shin in Human & Experimental Toxicology
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
