Abstract
Parkinson’s disease (PD) is the second most common age-related neurodegenerative disease after Alzheimer’s disease, characterized by loss of dopaminergic neurons in substantia nigra pars compacta, accompanied by motor and nonmotor symptoms. The neuropathological hallmarks of PD are well reported, but the etiology of the disease is still undefined; several studies assume that oxidative stress, mitochondrial defects, and neuroinflammation play vital roles in the progress of the disease. The current study was established to investigate the neuroprotective effect of agmatine on a rotenone (ROT)-induced experimental model of PD. Adult male Sprague Dawley rats were subcutaneously injected with ROT at a dose of 2 mg/kg body weight for 35 days. Agmatine was injected intraperitoneally at 50 and 100 mg/kg body weight, 1 h prior to ROT administration. ROT-treated rats that received agmatine showed better performance on beam walking and an elevated number of rears within the cylinder test. In addition, agmatine reduced midbrain malondialdehyde as an indication of lipid peroxidation, pro-inflammatory cytokines including tumor necrosis factor alpha and interleukin-1β, and glial fibrillary acidic protein. Moreover, agmatine was responsible for preventing loss of tyrosine hydroxylase-positive neurons. In conclusion, our study showed that agmatine possesses a dose-dependent neuroprotective effect through its antioxidant and anti-inflammatory activities. These findings need further clinical investigations of agmatine as a promising neuroprotective agent for the future treatment of PD.
Introduction
Parkinson’s disease (PD) is a neurodegenerative disease that is characterized by a gradual loss of dopaminergic neurons in the midbrain substantia nigra pars compacta (SNc) and consequently degeneration of striatal dopaminergic nerve terminal fibers, 1 which can eventually deplete striatal dopamine levels. 2 PD is characterized by special clinical signs including rigidity, slow movement, resting tremor, postural instability, and gait weakness, attributable to loss of dopaminergic neurons. 3,4 These symptoms are progressive and become notable probably because of the spread of pathology in the basal ganglia, 5 with the involvement of oxidative stress resulting from the elevated reactive oxygen species (ROS) produced by many sources, including mitochondrial dysfunction, dopamine metabolism, neuroinflammation of dopaminergic neurons, and aging. 6,7 Among these, ROS and subsequent neuroinflammation appear to play a necessary role in the initiation as well as the progression of PD. 7
Rotenone (ROT) is a mitochondrial complex I inhibitor; it is classified as an insecticide and has been broadly used as a pesticide in agriculture. 8 ROT-treated animals represent an ideal experimental animal model of PD with construct validity. 9 Most of the pathological features seen in human PD pathogenesis, such as the loss of dopaminergic neurons in SNc and neuroinflammation in addition to augmented oxidative stress in the nigrostriatal dopaminergic pathway, have been proved in the ROT model. 10 Therefore, the ROT-treated rats is an appropriate model for the purpose of investigating new therapeutic agents which target oxidative stress and neuroinflammation in PD.
Regarding the oxidative stress process, several studies reported the neuroprotective role of antioxidants in the management of PD. Many natural compounds such as S-allylcysteine and ginkgo biloba extract protect against 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine-induced PD through their free radical scavenging activity. 11 The antioxidant activity of demethoxycurcumin, a natural derivative of curcumin, was investigated in the ROT model of PD and showed an improvement in motor deficits and striatal dopamine level. 12 Treatment with spermidine, a polyamine compound, ameliorated oxidative stress, neuroinflammation, and restored striatal dopamine level in ROT-induced PD. The neuroprotective effect of spermidine is mediated through its antioxidant and anti-inflammatory properties. 13
Agmatine is a polyamine, which is synthesized after decarboxylation
Materials and methods
Chemicals and kits
Agmatine sulfate and ROT were purchased from Sigma-Aldrich (St Louis, Missouri, USA). The antibody used in the current study such as polyclonal rabbit anti-glial fibrillary acidic protein (GFAP) was purchased from Life Technologies, Thermo Fisher Scientific (Waltham, MA, USA). Anti-tyrosine hydroxylase (TH) was obtained from Cell Marque (Rocklin, CA, USA). Histostain-Bulk-SP IHC Kit broad spectrum was procured from Life Technologies, Thermo Fisher Scientific. Enzyme-linked immunosorbent assay (ELISA) kits for tumor necrosis factor alpha (TNF-α) and interleukin-1 beta (IL-1β) were obtained from Cloud-Clone Corp. (Houston, TX, USA).
Animals and experimental procedure
Adult male Sprague Dawley rats (250–300 g; n = 60) were purchased from the breeding unit of the Egyptian Organization of Biological Products and Vaccines (Helwan, Egypt). Animals were randomized and housed (four rats per cage) under controlled environmental conditions of light and dark cycle at a constant temperature (22°C ± 2°C). The animals were allowed free access to standard pellet diet for 1-week acclimatization period prior to the experimental procedures and given tap water ad libitum. The experimental protocol for animal experimentation was approved by the animal care and use committee at the Faculty of Pharmacy, Helwan University.
The rats were divided into four experimental groups (n = 15 rats); the groups were named as follows: Group I: Control group, rats were injected with the vehicle. Group II: ROT group, ROT (2 mg/kg body weight) was injected subcutaneously (s.c.) for 35 days to induce the PD in rats. The regimen used to induce parkinsonism in rats in our study was adopted as previously reported.
29
Group III: Agmatine 50 mg/kg + ROT group, rats received agmatine intraperitoneally (i.p.) at a dose of 50 mg/kg/day,
30
1 h before ROT administration. Group IV: Agmatine 100 mg/kg + ROT group, rats received agmatine i.p. at a dose of 100 mg/kg/day,
31
1 h before ROT administration.
Dimethyl sulfoxide was used to dissolve ROT to get a 50× stock solution and stored at −80°C for further use. Before injection, the ROT stock solution was dissolved and further diluted in sunflower oil to prepare a final concentration of 2 mg/ml, where agmatine was dissolved in cold normal saline. Agmatine was administered 1 week prior to ROT and continued for 35 days. Motor function was evaluated by narrow beam test beginning from day 30 until day 33, and cylinder test at the day of 34. The animals were euthanized 24 h after the last injection of ROT or in combination of ROT with agmatine.
Tissue collection
At the end of the experiment at day 35, the rats had been decapitated and the skulls were opened to rapidly separate the brain. The brain was put on an ice-plate and cut along the midline to isolate the two cerebral hemispheres and separate the midbrain. The midbrain regions dissected from six rats were used for biochemical studies. For immunohistochemistry analysis, other six midbrains were postfixed in 4% paraformaldehyde solution for 48 h and subsequently rinsed with 10% sucrose solutions three times a day for three consecutive days at 4°C.
The midbrains from each group were homogenized in potassium chloride (KCl) buffer containing 10 mM Tris–hydrochloride, 1 mM ethylenediaminetetraacetic acid, 140 mM sodium chloride, 300 mM KCl, and 0.5% Triton X-100 at pH 8.0 supplemented with phosphatase inhibitors and protease. The tissue homogenates were centrifuged at 14,000 ×g for 20 min at 4°C to obtain the post-mitochondrial supernatant for determination of markers of oxidative stress and pro-inflammatory cytokines using spectrophotometric measurements and ELISAs.
Behavioral evaluation
All the behavioral experiments had been performed by a blind observer and carried out in late afternoon in quiet environment and under dim light.
Elevated narrow beam test
The narrow beam test was used to evaluate bradykinesia in the rats according to a previously settled method. 32 The rats had been allowed to explore the beam for three trials 3 days earlier than the experimental day and during this exploration rats had been encouraged and rewarded with cocoa food pellets in the goal box. The time taken to cross the beam had been recorded. The maximum time allowed for the task was 120 s. If the rat was unable to start to cross the beam within 2 min, it was removed from the beam and excluded from the analysis.
Rearing behavior in the cylinder test
In order to evaluate the motor performance of rats' forelimbs, rats were positioned in a clear cylinder and were evaluated for their rearing behavior. During rearing behavior, the forelimbs touched the internal wall of the cylinder. For this test, each rat was placed in a clear plexiglass cylinder (diameter = 20 cm, height = 30 cm) for 5 min. The test was performed in late afternoon under dim light in order to induce rats to explore. The numbers of rears were counted. To be counted as a rear, the rats have to raise the forelimbs above the shoulder level and make contact with the cylinder wall by either one or both forelimbs. Removal of both forelimbs from the cylinder wall and touch with the table floor was considered as a requisite before another rear. 33
Determination of oxidative stress biomarkers
Malondialdehyde (MDA) assay was carried out according to the method of Mihara and Uchiyama, 34 reduced glutathione (GSH) was measured according to the method of Ellman, 35 and BioVision (Milpitas, CA, USA) superoxide dismutase (SOD) assay kit was used to determine the activity of SOD in midbrain homogenate according to the previous method of McCord and Fridovich . 36
Estimation of pro-inflammatory cytokines by ELISAs
The levels of pro-inflammatory cytokines such as IL-1β and TNF-α were measured using commercially available ELISA kits obtained from Cloud-Clone Corp. (USA). The levels of IL-1β and TNF-α were estimated according to the manufacturer’s instructions.
TH and GFAP immunohistochemistry
Immunohistochemistry was used for quantitative determination of TH and GFAP protein expression using suitable kits. The technique used for immunostaining was the labeled streptavidin–biotin (LSAB) method according to Elias et al. 37 Paraffin-embedded brain blocks were cut into 5-µm-thick coronal sections and mounted onto positively charged adhesive slides. The cut sections were air dried for 30 min and then fixed in a 65°C oven for 1 h. The three pretreatment steps (deparaffinization, rehydration, and unmasking) were performed using the Trilogy working solution (Cell Marque, USA). Slides were placed in a Coplin jar filled with 200 ml of Trilogy working solution. The jar was securely positioned in the autoclave. The autoclave was adjusted so that the temperature reached 120°C and remained stable for 15 min. After this, the Coplin jar was removed to allow the slides to cool for 30 min. Sections were rinsed in phosphate-buffered saline (PBS, pH 7.4). Quenching of endogenous peroxidase activity was performed by immersing slides in 3% hydrogen peroxide in methanol for 10 min and then in distilled water for 5 min. The background staining was blocked by putting two to three drops of 10% goat nonimmune serum blocker on each slide and incubating them in a humidity chamber for 10 min. After rinsing the slides three times with cold PBS, Histostain-Bulk-SP IHC Kit, broad spectrum was used as follows: slides were incubated with biotinylated goat anti-polyvalent for 10 min, washed four times with PBS, followed by 10 min incubation with streptavidin–horseradish peroxidase complex. The slides were washed with PBS, and tissue sections were visualized by adding two to three drops of peroxidase substrate 3, 3′-diaminobenzidine (DAB) as a chromogen. DAB was rinsed. Afterward, counterstaining with Mayer’s hematoxylin for 5 min and cover slipping were performed as the final steps before slides were examined under light microscope.
Immunostaining was evaluated by examination of slides under a bright field microscope (CX21, Olympus, Japan), and images were captured through a digital camera for measurement of intensity. Intensities of immunostained cells were estimated by densitometry using image analysis software (Leica Qwin 500, Cambridge, England).
Statistical analyses
The results were expressed as the mean value ± SEM. Statistical analysis and graphical representations were performed using GraphPad Prism, version 3 (GraphPad Software Inc., San Diego, California, USA), using a one-way analysis of variance, followed by a Tukey’s test to determine the statistical significance between various groups. The value p < 0.05 and onward was considered as significant.
Results
Effect of agmatine on behavioral parameters in ROT-induced PD in rats
Narrow beam test
Narrow beam test was performed to evaluate the motor activity in rats. The time taken to cross the beam was significantly increased in ROT group by approximately 164.6% (p < 0.001) compared with the control group. Agmatine 50 and 100 mg/kg prior to ROT injection significantly decreased the time of beam walking, showing marked improvement in locomotor performance by approximately 46.5% (p < 0.05) and 53.4% (p < 0. 01), respectively, compared to the ROT group (Figure 1(a)).
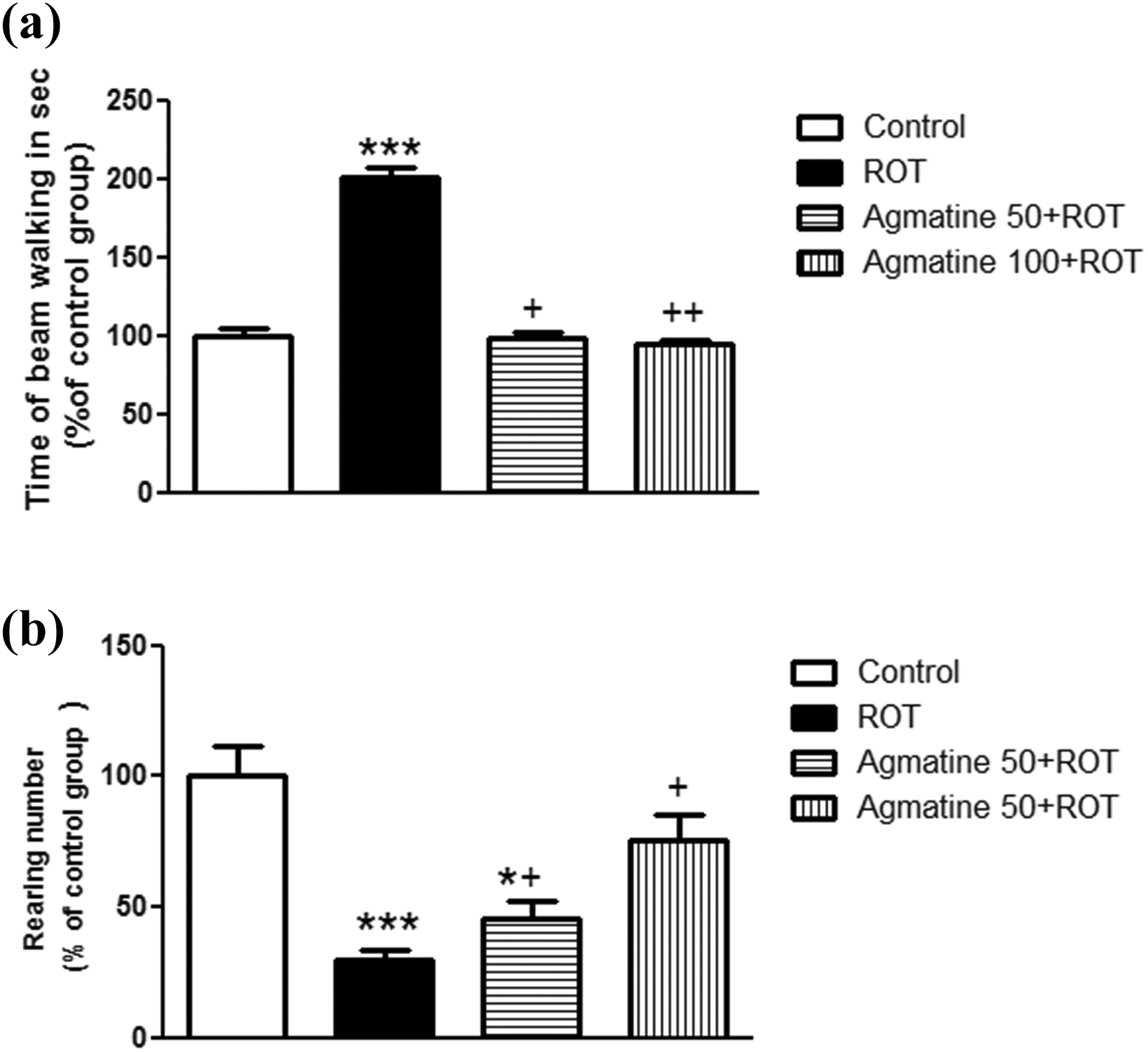
Effect of agmatine on behavioral parameters in ROT-induced PD in rats (n = 15, mean ± SE). Agmatine was given i.p. daily at doses of 50 and 100 mg/kg 1 h before s.c. ROT injection at a dose of 2 mg/kg for 33 days. (a) The time of beam walking. (b) The rearing numbers. ***p < 0.001: significantly different from control group; *p < 0.05: significantly different from control group; ++ p < 0.01: significantly different from ROT group; + p < 0.05: significantly different from ROT group. i.p: intraperitoneally; s.c: subcutaneous; ROT: rotenone; PD: Parkinson’s disease.
Rearing numbers
The number of rearing behavior in the cylinder test was significantly decreased in ROT group by approximately 70.16% (p < 0.001) compared to the control group. Administration of agmatine 50 and 100 mg/kg before ROT treatment significantly increased the rearing number by 60% and 152.72% (p < 0.05), respectively, compared to the ROT group (Figure 1(b)). However, a dose of 50 mg/kg agmatine was significantly different from the control group.
Antioxidant effect of agmatine in ROT-induced PD in rats
MDA level in midbrain
MDA is a lipid peroxidation marker, and the MDA level was significantly increased (215.14%; p < 0.001) in the ROT group compared to the control group. The increase in MDA was significantly restored (20.79% and 29%; p < 0.001), respectively, in treatment groups with agmatine 50 and 100 mg/kg compared to the ROT group (Figure 2(a)). Moreover, these results were found to be still higher than the control group.

Antioxidant effect of agmatine in ROT-induced PD in rats (n = 6, mean ± SE). Agmatine was given i.p. daily at doses of 50 and 100 mg/kg 1 h before s.c. ROT injection at a dose of 2 mg/kg for 35 days. (a) Midbrain MDA level. (b) Midbrain GSH level. (c) Midbrain SOD activity. ***p < 0.001: significantly different from control group; +++ p < 0.001: significantly different from ROT group; ++ p < 0.01: significantly different from ROT group; + p < 0.05: significantly different from ROT group; & p < 0.05: significantly different from agmatine 50 + ROT group. ROT: rotenone; i.p: intraperitoneally; s.c: subcutaneous; MDA: malondialdehyde; GSH: glutathione; SOD: superoxide dismutase; PD: Parkinson’s disease.
GSH level in midbrain
GSH is an essential antioxidant in cells defense system. GSH level was significantly decreased (60.12%; p < 0.001) in the ROT group compared to the control group. This level was significantly restored by oral administration of agmatine 50 and 100 mg/kg prior to ROT injection by approximately 39.31% (p < 0.01) and 68.27% (p < 0.001), respectively, compared to the ROT group (Figure 2(b)). However, these results were found to be significant from the control group. In addition, pre-administration of agmatine 100 mg/kg significantly increased the GSH level by approximately 20.79% (p < 0.05) compared to agmatine 50 mg/kg.
SOD activity in midbrain
SOD is the major antioxidant enzyme against superoxide radical. ROT administration significantly decreased (46.3%; p < 0.001) SOD activity when compared with the control group. Pre-administration of agmatine 50 and 100 mg/kg significantly increased SOD activity (24.2%; p < 0.05 and 46.2%; p < 0.001), respectively, compared to the ROT group (Figure 2(c)). However, these levels were still less than that of the control group.
Anti-inflammatory effect of agmatine in ROT-induced PD in rats
TNF-α level in midbrain
TNF-α is a major pro-inflammatory cytokine in the early stage of the inflammation process. ROT administration caused a marked increase in the level of midbrain TNF-α (82.9%; p < 0.001) compared to the control group. Treatment with agmatine 50 and 100 mg/kg before ROT administration showed a significant decrease in the level of TNF-α by approximately 11.9% (p < 0. 01) and 22.8% (p < 0.001), respectively, compared to the ROT group; also these results were found to be elevated in comparison with the control group. Moreover, administration of agmatine 100 mg/kg significantly decreased the TNF-α level by approximately 12.5% (p < 0. 01) compared to agmatine 50 mg/kg (Figure 3(a)).

Anti-inflammatory effect of agmatine in ROT-induced PD in rats (n = 6, mean ± SE). Agmatine was given i.p. daily at doses of 50 and 100 mg/kg 1 h before s.c. ROT injection at a dose of 2 mg/kg for 35 days. (a) Midbrain TNF-α level. (b) Midbrain IL-1β level. ***p < 0.001: significantly different from control group; +++ p < 0.001: significantly different from ROT group; ++ p < 0.01: significantly different from ROT group; && p < 0.01: significantly different from agmatine 50 + ROT group. ROT: rotenone; i.p: intraperitoneally; s.c: subcutaneous; PD: Parkinson’s disease; TNF-α: tumor necrosis factor-alpha; IL-1β: interleukin-1 beta.
IL-1β level in midbrain
IL-1β is a potent pro-inflammatory cytokine; IL-1β was found to be elevated (134.74%; p < 0.001) significantly in the ROT group compared to the control group. Pretreatment with agmatine 50 and 100 mg/kg showed a significant decrease in the level of IL-1β by approximately 13.5% (p < 0. 01) and 29.4% (p < 0.001), respectively, compared to the ROT group (Figure 3(b)); also these results were found to be remarkable from the control group.
Effect of agmatine on striatal GFAP-positive cells in ROT-induced PD rats
GFAP is a biomarker for glial cell activation; immunohistochemical analysis showed a significant increase in the optical density of GFAP-positive cells in ROT-injected rats by 30% (p < 0.001) compared to control rats (Figure 4(a) and (b)). Interestingly, agmatine 100 mg/kg treatment prior to ROT injection significantly decreased the density of GFAP-positive cells by 9% (p < 0.05) compared to the ROT-injected rats. However, these data were found to be significantly different from the control group.
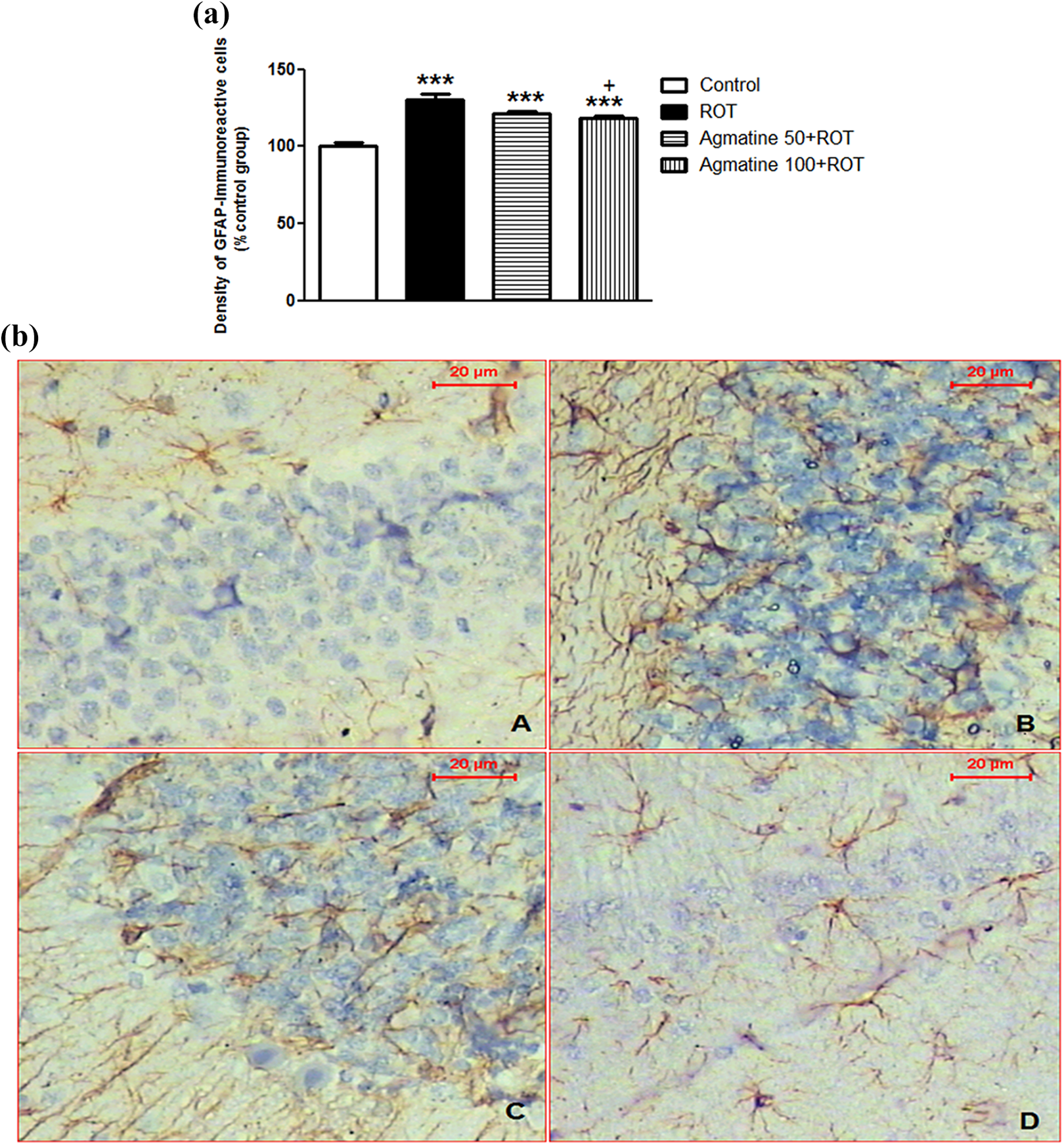
(a) Effect of agmatine on striatal GFAP in ROT-induced PD in rats. (n = 6, mean ± SE). Agmatine was given i.p. daily at doses of 50 and 100 mg/kg 1 h before s.c. ROT injection at a dose of 2 mg/kg for 35 days. ***p < 0.001: significantly different from control group; + p < 0.05: significantly different from ROT group. (b) Micrograph of sections from striatum region of the brain showed the density of GFAP-immunoreactive cells in different groups: (A) control group, (B) ROT group, (C) agmatine (50 mg/kg) + ROT, and (D) agmatine (100 mg/kg) + ROT. Agmatine was given i.p. daily at doses of 50 and 100 mg/kg 1 h before s.c. ROT injection at a dose of 2 mg/kg for 35 days. ROT: rotenone; i.p: intraperitoneally; s.c: subcutaneous; PD: Parkinson’s disease; GFAP: glial fibrillary acidic protein.
Effect of agmatine on striatal TH-positive cells in ROT-induced PD in rats
TH is a rate-limiting enzyme of dopamine neurotransmitter biosynthesis; TH immunohistochemical analysis was performed to estimate the expression of healthy TH-positive dopamine neurons. The results showed that ROT injection caused a significant loss of TH-positive neurons by 25% (p < 0.05) when compared to vehicle-injected control rats (Figure 5(a) and (b)). In contrast, agmatine 100 mg/kg prior to ROT injection provided significant protection to dopamine neurons (23.2%; p < 0.001) when compared to ROT injection of rats; however, agmatine 50 mg/kg was still significantly different from the control group.

(a) Effect of agmatine on striatal TH in ROT-induced PD in rats. (n = 6, mean ± SE). Agmatine was given i.p. daily at doses of 50 and 100 mg/kg 1 h before s.c. ROT injection at a dose of 2 mg/kg for 35 days. *p < 0.05: significantly different from control group; ***p < 0.001: significantly different from control group; +++ p < 0.001: significantly different from ROT group. (b) Micrograph of sections from striatum region of the brain showed the density of TH-immunoreactive neurons in different groups: (A) control group, (B) ROT group, (C) agmatine (50 mg/kg) + ROT, and (D) agmatine (100 mg/kg) + ROT. Agmatine was given i.p. daily at doses of 50 and 100 mg/kg 1 h before s.c. ROT injection at a dose of 2 mg/kg for 35 days. ROT: rotenone; i.p: intraperitoneally; s.c: subcutaneous; PD: Parkinson’s disease; TH: tyrosine hydroxylase.
Discussion
Agmatine is the endogenous metabolite of
ROT is a potent neurotoxic, which selectively inhibits complex I of the mitochondrial electron transport chain (NADH dehydrogenase), reduces adenosine triphosphate (ATP) production consequently, and leads to selective degeneration of nigrostriatal dopaminergic neurons. 40 ROT administration in rats induced behavioral and motor changes that are considered to be probably found in people suffering from PD as evidenced by hypokinesia, bradykinesia, and rigidity. 9,40,41 In our study, ROT-injected rats showed an increase in the time of beam walk, as well as a decrease in the rear numbers indicating defects in motor functions; this can be explained by striatal dopamine depletion evidenced by low density of TH-positive cells. Agmatine treatment was beneficial in attenuating the observed motor dysfunction in a dose-dependent manner, indicating its potential effect in the protection of dopaminergic neurons and to keep the density of TH-immunopositive neuronal cells as shown in our results.
One of the major factors associated with the progression and development of PD is the oxidative stress. 42 Several studies propose that free radical oxidation damage especially of neuronal lipids, proteins, and nucleic acids is tremendous in the brains of PD patients due to the formation of free radicals and ROS. 43,44 Unlike other organs, the brain has been discovered to be more susceptible to oxidative stress due to its excessive lipid content and its noticeably elevated oxygen metabolism and the low level of antioxidant defenses. 45,46 A massive generation of free radical leading to cell death can be obtained with any defect in the mitochondrial electron transport chain (complex I). 47 Systemic administration of ROT, which is considered as a complex I inhibitor, causes reduction in ATP production and ROS generation. 48
ROS stimulate polyunsaturated fatty acid oxidation in a process referred to as lipid peroxidation. MDA is an essential product of lipid peroxidation which construct adducts with proteins and DNA bases, consequently leading to cellular damage. Significantly elevated levels of MDA in the ROT-treated rats were observed as compared to the control rats. Interestingly, agmatine treatment significantly reduced MDA levels as compared to the ROT-injected rats. Bhutada et al. reported that agmatine ameliorates oxidative stress due to lipid peroxidation in the rat brain. 49 The suppression of lipid peroxidation by agmatine in the present study may have been mediated by the detoxification of peroxy radicals and ROS, which further supports the antioxidant role of agmatine.
Reduced GSH is an essential antioxidant and has a quintessential role in hydrogen peroxide scavenging. Elevated MDA production due to oxidative stress is correlated with the decrease in the bioavailability of GSH tripeptide in midbrain. Therefore, the low levels of GSH in the brain might point out a state of oxidative stress. Pearce et al. reported low levels of GSH in the SNc of PD patients. 50 In the current study, the GSH level was significantly decreased in ROT-injected rats as compared to the control rats, while administration of agmatine significantly elevated GSH level indicating the antioxidant potential effect of agmatine which was previously reported by many studies. 26,28,38 The effect of agmatine on GSH may involve its direct antioxidant effects of agmatine or the prevention of ROT-induced GSH oxidation.
SOD is a one of the most important antioxidant enzymes implicated in free radical detoxification. Deficiency of SOD activity further contributes to an increase in oxidative stress in ROT-injected rats, which is indicated by higher levels of MDA and lower GSH levels. Inhibition of the electron transport chain leads to the generation of a huge amount of superoxide radicals, 51 and the lowered SOD activity would be deleterious in the scenario when the production of superoxide radical is elevated. Our present results showed decreased SOD activity in ROT-injected rats compared to the control group. Furthermore, agmatine was able to improve SOD activity in ROT-injected group, this way enhancing the antioxidant defense system. This result corroborated a previous report showing an increase in the activity of SOD following agmatine administration. 52
Furthermore, the neuroinflammation process is triggered and sustained via different mechanisms in ROT-treated rats. The mitochondrial respiratory chain complex I dysfunction induced by ROT injection leads to a massive release of ROS, which can activate glial cells. 53 Many studies reported the participation of neuroinflammation in PD, which is generally characterized by glial cell activation. 54,55 Glial cells activation was noted in ROT-injected rats as proved by elevated expression of GFAP, which is considered to be an indicator of astrocyte glial cells activation, and subsequent neuroinflammation which is in agreement with previous studies. 5,56,57 However, the activation of astrocytes was attenuated by agmatine treatment as evidenced by the decreasing expression of GFAP; therefore, our results show that agmatine suppressed astrocyte activation, which is considered as a dopaminergic neurodegeneration initiator and, consequently, rescued dopaminergic neurons.
A large number of studies have reported that glial cells activation encourages the release of pro-inflammatory cytokines such as TNF-α and IL-1β that subsequently leads to neurodegeneration of dopaminergic neurons. 58,59 The neuroinflammatory process, which includes the secretion of pro-inflammatory cytokines, and glial cell activation are key factors in the PD neurodegenerative process. 56
To investigate the possible beneficial effects of agmatine on the inflammatory pathway, the levels of TNF-α and IL-1β as pro-inflammatory cytokines were measured. The midbrain levels of TNF-α and IL-1β were elevated following ROT injections similar to previous reports. 5,60 Interestingly, agmatine treatment to ROT-injected animals caused a decrease in the levels of TNF-α and IL-1β concomitant with the low level of GFAP and glial cell activation, thereby suggesting that agmatine plays a significant role in controlling TNF-α and IL-1β levels by counteracting the activation of glial cells. The effects of agmatine in our study are similar to those found in previous studies where it was observed that agmatine can reduce pro-inflammatory cytokines and glial cell activation. 61,62,63
TH is the rate-limiting enzyme that catalyzes the initial step of dopamine biosynthesis, which is synthesized in the dopaminergic neurons of the SNc area. DA is stored inside the synaptic vesicles and, in response to stimuli, liberated in the striatum region to produce its physiological function. 64 In PD patients, more than 70% of the dopaminergic neurons in the SNc are lifeless, leading to the retraction of dopaminergic nerve terminals in the striatum and depletion of dopamine in the striatum. 65 The dopaminergic neurons degeneration in the SNc and decreased density of striatal nerve terminals are regarded to be one of the pathological hallmarks of PD. 1 In the current study, immunohistochemical examination of TH showed that ROT injection causes a significant loss of striatal TH-positive dopaminergic neurons which agree with previous studies. 5,33 Meanwhile, agmatine treatment to ROT-injected rats significantly protected against the ROT-induced loss of dopaminergic neurons. Taken together, our results suggest that agmatine has a neuroprotective effect that prevents the loss of dopaminergic neuron induced by ROT in rats.
Conclusions
In conclusion, our study reports that agmatine treatment in a dose-dependent manner ameliorates the ROT-induced dopaminergic neurodegeneration in vivo through its antioxidant and anti-inflammatory activities. Agmatine protected ROT-induced oxidative stress by reducing MDA level and increasing both GSH level and SOD activity. Agmatine suppressed the overactivation of glial cells by reducing GFAP-positive cells and also inhibited neuroinflammation via abrogating the cytokines levels, IL-1β and TNF-α. In addition, it also maintained TH-positive cells. Overall, our study highlights the potential clinical efficacy of agmatine in neurodegenerative diseases including PD.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
