Abstract
Aging is a multifactorial process associated with functional deficits, and the brain is more prone to developing chronic degenerative diseases such as Parkinson’s disease. Several groups have tried to correlate the age-related ultrastructural alterations to the neurodegeneration process using in vivo pharmacological models, but due to the limitations of the animal models, particularly in aged animals, the results are difficult to interpret. In this work, we investigated neurodegeneration induced by rotenone, as a pharmacological model of Parkinson’s disease, in both young and aged Wistar rats. We assessed animal mobility, tyrosine hydroxylase staining in the substantia nigra pars compacta (SNpc), and TdT-mediated dUTP-biotin nick end labeling-positive nuclei and reactive oxygen species production in the striatum. Interestingly, the mobility impairment, dopaminergic neuron loss, and elevated number of apoptotic nuclei in the striatum of aged control rats were similar to young rotenone-treated animals. Moreover, we observed many ultrastructural alterations, such as swollen mitochondria in the striatum, and massive lipofuscin deposits in the SNpc of the aged rotenone-treated animals. We conclude that the rotenone model can be employed to explore age-related alterations in the ontogeny that can increase vulnerability in the striatum and SNpc, which may contribute to Parkinson’s disease pathogenesis.
The aging process is accompanied by biochemical and subcellular alterations, which eventually result in cognitive decline or somatosensory impairment. This can be considered a major risk factor for the development of neurodegenerative diseases such as Parkinson’s disease. A remarkable neuropathological feature of Parkinson’s disease is the loss of dopaminergic neurons in the substantia nigra, thus reducing the release of dopamine in the striatum (Ehringer and Hornykiewicz 1960), which results in bradykinesia, muscle rigidity, and shaking. These features are based on environmental and genetic factors (Foltynie et al. 2002) and are largely associated with the aging process (called late onset Parkinson’s disease), although there are also many familiar cases of early onset Parkinson’s disease (Gershanik 2003).
With regard to the movement control pathways, Parkinson’s disease is an affection of the extrapyramidal system, which coordinates and processes motor commands, and differs from the pyramidal system, which involves the pyramidal cells from the primary motor cortex to generate movement. The nigrostriatal pathway, which involves the striatum and substantia nigra, among other structures, is the part of the extrapyramidal system that controls the motor actions and is affected during the normal aging process (Emborg 2004). Dopamine neurotransmission from the substantia nigra pars compacta (SNpc) functions by inhibiting the release of acetylcholine in the striatum (Lehmann and Langer 1983) and modulating the release of glutamate. Thus, the abrogation of dopamine neurotransmission in the striatum can cause hyperactivity of the cortical glutamatergic synapses (Lindefors and Ungerstedt 1990), and the elevation of glutamate levels in the synaptic cleft can cause excitotoxicity (Cepeda, Li, and Levine 1996). It has been previously reported, by our group, that striata, from aged animals, display markers of enhanced vulnerability when compared to younger animals (Ureshino et al. 2010). Furthermore, it was also observed that the aged striata produce a greater amount of reactive oxygen species (ROS), reduce the mitochondrial membrane potential and mitochondrial Ca2+ overload, followed by a reduction in the antiapoptotic protein, Bcl-2, suggesting that it could be related to the increased cell death observed in this structure (Ureshino et al. 2010).
Several types of Parkinson’s disease models have been described (Emborg et al. 1998; Tieu 2011), but none of them can entirely reproduce the symptomatology of the disease in humans. Some transgenic models have been generated to study the defective proteins that are found in familiar cases of Parkinson’s disease such as PINK1, Parkin, LRRK2, and α-synuclein (Dawson, Ko, and Dawson 2010). The compounds commonly used to generate pharmacological models of Parkinson’s disease are as follows: (1) electron transport chain inhibitors such as 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (Vila et al. 2000) and rotenone (Gerlach et al. 1991; Langston and Ballard, 1983), (2) compounds that elevate dopamine levels in the axon terminals of the SNpc, which can be oxidized and promote toxicity such as 6-hydroxydopamine (Saner and Thoenen 1971). Since Parkinson’s disease is mainly an old-age pathology, the use of aged animal models is suitable in order to combine the age-related loss of function to the pathophysiological features of the disease. A large majority of pharmacological models use young animals to promote nigral degeneration and try to associate those models to the “idiopathic” form of Parkinson’s disease. Here, we evaluated the effects of aging in a pharmacological model of Parkinson’s disease, with respect to the ultrastructural neuronal morphology and cell death in the striatum and SNpc, which may potentially be related to the manifestation of the disease in rat models.
Material and Method
Animals and Treatments
In the present study, 4- to 5-months-old (young) and 24- to 25-months-old (aged) female Wistar rats 2BAW were used. They were maintained at 25°C, under a 12-hr dark–light cycle, and fed with standard chow ad libitum in the animal housing facility at the Institute of Pharmacology and Molecular Biology located on the campus of the Federal University of São Paulo. All of the procedures performed in the present study, including behavior tests and histopathological experiments, were previously submitted, approved, and are in accordance with the ethical standards of the institution where the studies were conducted (CEP 0308/12).
Initially, the optimal dose was determined by treating the young and aged animals with 1 to 3 mg/kg of rotenone (R8875 Sigma-Aldrich, St. Louis, MO, USA) for 12 days (Betarbet et al. 2000).
The most effective dose was found to be 1.5 mg/kg/day for up to 12 days by subcutaneous injection, which was administered at 3 p.m. everyday, in the dorsal region of the animal, with a 1 ml/kg rotenone solution (1.5 mg/ml). The rotenone was solubilized in DMSO and diluted in corn oil at a ratio of 1:10. To avoid animal death, rotenone was withdrawn if the animals presented akinesia before 12 days of treatment. Animals were grouped and defined as the following: YCT (young control, 6 animals), YR (young treated with rotenone, 6 animals), ACT (aged control, 6 animals), and AR (aged treated with rotenone, 9 animals were initially treated, 3 survived until the end of the study). In all of the procedures, the animals were euthanized 15 days after the first injection.
Behavior Test and Semiquantification of tyrosine hydroxylase (TH)-positive Cells in SNpc
After 12 days of rotenone treatment, young and aged animals were subjected to an open-field test (Insight, model EP 154) to monitor behavior and mobility. On the 13th day, the animals were allowed to adapt to the new environment. On the 14th day, an experimental test was performed. Briefly, three animals at a time were moved from the animal maintenance room into the experimental room, located in the same animal housing facility. The animals were allowed to adapt to the new room for at least 10 min. After the adaptation period, each animal was gently moved from the box to the equipment, and the crossing and rearing rates were counted for 3 min. At the conclusion of the test, the open-field testing equipment was cleaned with 10% ethanol (v/v), and the next animal was tested.
The dopaminergic neuron distribution was evaluated in the SNpc by using an anti-TH immunofluorescence assay. For this experiment, animals used in the behavior experiments were euthanized by decapitation, the skull was gently opened, and the brain was rapidly removed. Brains were immersed in an artificial cerebral–spinal fluid (aCSF) solution (2 mM KCl, 1 mM CaCl2, 26 mM NaHCO3, 1.25 mM NaH2PO4, 1 mM MgCl2, 2 mM MgSO4, 10 mM glucose, 248 mM sucrose, and pH 7.4;Ureshino et al. 2014) and then sliced in a vibratome, into sagittal sections (∼2.5 mm thick) containing the striatum and midbrain. Slices were fixed in 4% paraformaldehyde (w/v) in 0.1 M phosphate buffered saline (PBS), followed by cryoprotection with a 20% sucrose solution (w/v) in PBS at room temperature, overnight. Slices were frozen in isopentane and dry ice (∼−30° C) and cryosectioned into slices (∼25 μm thick). The frozen sections of the brains (∼25 µm thick) were washed in PBS and blocked with 10% horse serum + 1.5% Triton X-100 in PBS. Then the sections were incubated with an anti-TH primary antibody (1:1,000; EMD Millipore, Burlington, MA, USA) overnight at 4° C. The sections were then washed and incubated with a fluorescent secondary antibody (1:500 anti-mouse conjugated to Alexa fluor 594, Invitrogen, Carlsbad, CA, USA) for 2 hr at room temperature. The slides were mounted with Fluormont, and the TH fluorescence (Ex/Em: 590/617 nm), of the SNpc, was observed with a high-resolution fluorescence microscope (Nikon TE 300, Nikon, Japan). The images were acquired (1 slide of SNpc per animal, with 10× and 20× objectives), the fluorescence was collected using the 20× objective, and the data were analyzed using the BioIP software (v. 9999.0.0.5; Anderson Engineering, NJ, USA). Fluorescence intensity was presented in arbitrary units (AU).
Apoptotic Nuclei and Neurons Quantification
Frozen brains were fixed in 4% buffered formaldehyde and cryosectioned, as described above. The TdT-mediated dUTP-biotin nick end labeling (TUNEL) assays (Apop Tag Plus, Millipore, Burlington, MA, USA) were performed with 10-µm thick sections placed onto silane-treated glass slides. The striatum sections were rinsed in 0.1 M PBS, and the samples were permeabilized with 2:1 ethanol—acetic acid mixture for 5 min at −20° C. The endogenous peroxidase activity was blocked with 1% H2O2 for 20 min. The samples were treated with equilibration buffer and incubated with the TdT enzyme at 37° C for 1 hr. After the addition of the stop buffer, the samples were incubated with anti-digoxigenin–peroxidase for 30 min and then the reaction was developed with 0.5% 3.3’-diaminobenzidine. To further identify the neurons in the striatum, the samples were submitted to immunofluorescence labeling (as described in the subsection Behavior Test and Semiquantification of tyrosine hydroxylase (TH)-positive Cells in SNpc), with an anti-NeuN antibody (1:1000; EMD Millipore, Burlington, MA, USA). The secondary antibody used was an anti-mouse conjugated to Alexa fluor 594 (1:500, Invitrogen, Carlsbad, CA, USA), and the nuclei were stained with Hoescht 33342 (Molecular Probes, Eugene, OR, USA). The slides were mounted with Fluoromont and observed with a high-resolution fluorescence microscope (Leica SP5, Germany). Images were acquired from 1 slide of striatum per animal, and 7 fields from each slide were analyzed. TUNEL-positive nuclei were counted within an area of approximately 2.2 × 106 µm2 and presented in absolute values, and the neurons and nuclei were counted in alternate random fields with an area of 4,000 µm2 (approximately 280 fields/slide), within an area of approximately 1.1 × 106 µm2.
Electron Microscopy
Two animals from each group were selected for the ultrastructural analysis. Animals were anesthetized with halothane, followed by an intracardiac perfusion-fixed with 2.5% glutaraldehyde and 2% formaldehyde in 0.1 M PBS. After the perfusion, the skull was gently opened and the brain was rapidly removed and sliced into ∼1-mm fragments. Coronal 1,000-µm thick sections from the dorsolateral region of the striatum and SNpc were performed at 1.8 mm and −4.80 mm from the bregma, respectively (Paxinos). Samples were rinsed with 0.1 M cacodylate buffer, postfixed in 1% osmium tetroxide/1.5% potassium ferrocyanide for 30 min, rinsed with water, contrasted in saturated aqueous uranyl solution, rinsed again with water, dehydrated in absolute alcohol, cleared in propylene oxide, and embedded in Epon resin. Semithin sections (500 nm thick) were obtained using an ultramicrotome (Ultracut R, Leica, Germany), stained with alcoholic toluidine blue, and examined under a Olympus BX41 light microscope for selecting the area to be observed under the electron microscope. Blocks were then trimmed to obtain ultrathin sections (100 nm thick). Sections were subsequently contrasted with uranyl acetate and lead citrate for the examination under the electron microscope (1200 EXII, JEOL, Japan). Electron micrographs were obtained using a digital camera (Multiscan 791, GATAN, USA). Lysosome area was quantified by measuring the organelle and cytoplasm and was presented as a lysosome/cytoplasm ratio. The area measurements were performed using the ImageJ software (v. 1.43u; National Institutes of Health, Bethesda, MD, USA).
ROS Measurements
ROS measurements were performed immediately after the preparation of brain slices. Animals were euthanized, and the brains were rapidly and gently removed. Afterward, the brains were sliced in a vibratome (approximately 400 µM thick), immersed into a sucrose-supplemented aCSF solution (2 mM KCl, 1 mM CaCl2, 26 mM NaHCO3, 1.25 mM NaH2PO4, 1 MgCl2, 2 mM MgSO4, 10 mM glucose, 248 mM sucrose, and pH 7.4) and then incubated in normal aCSF solution (2 mM KCl, 2 mM CaCl2, 26 mM NaHCO3, 1.25 mM NaH2PO4, 2 mM MgSO4, 10 mM glucose, 124 mM NaCl, and pH 7.4; Ureshino et al. 2014). The solution was perfused and constantly aerated in a vibratome and maintained at room temperature.
Slices were incubated with the ROS indicator CM-H2DCFDA (‘5- (and-6) -chloromethyl-2’, 7’ dichlorodihydrofluorescein diacetate [DCFDA], acetyl ester, Molecular Probes; 5 µM for 30 min in the dark). The measurements were carried out in a fluorimeter (FlexStation III—Molecular Devices, MDS Analytical Technologies Inc., San Jose, CA, USA), and dichlorodihydrofluorescein (DCF) fluorescence was extracted using the SoftMax Pro software (v. 5.4) and presented in AU. The wavelengths for excitation and emission used in the DCFDA fluorescence assay were Ex/Em: 488/505 nm.
Statistical Analysis
The results were expressed as mean ± standard error of the mean and analyzed by two-way analysis of variance with a Bonferroni posttest. A p value of < .05 was considered to be statistically significant.
Results
Characterization of the Pharmacological Model of Parkinson’s Disease
In order to establish a pharmacological model of Parkinson’s disease in young and aged rats, it was first necessary to determine the optimal dosage of rotenone to be administered. Based on the initial behavioral and immunofluorescence experiments, it was determined that the most effective treatment was 1.5 mg/kg/day of rotenone for up to 12 days. Some of the animals displayed Parkinson’s disease symptoms, such as fragility and motor impairment, before the 12th day of treatment, and in those cases, the rotenone injections were withdrawn until the end of the treatment period. In at least 50% of the young animals, the injections were withdrawn by the 7th day of treatment, but all of the animals survived until the 12th day (6 animals treated). On the other hand, only 30% of the aged animals survived the rotenone injections for 12 days, although the remaining animals survived until the end of the injection protocol (initially 9 animals were treated, see Method section). Animals treated with rotenone showed motor activity impairment, lethargy, and walked more slowly but did not have a staggering gait. However, those animals continued to gain weight and did not display any loss of muscle strength in the limbs. Figure 1a shows that the YR group displayed a decrease in TH labeling in the SNpc, when compared to the YCT group, which is indicative of degeneration in this structure. In the aged animals, there was a marked reduction in the TH labeling in the ACT group, which was more pronounced in the AR group. Accordingly, the fluorescence of TH-labeled cells was reduced in both rotenone-treated groups when compared to the YCT group, suggesting a degenerative progress in the dopaminergic cells of the SNpc (Figure 1b), which was further enhanced during aging.
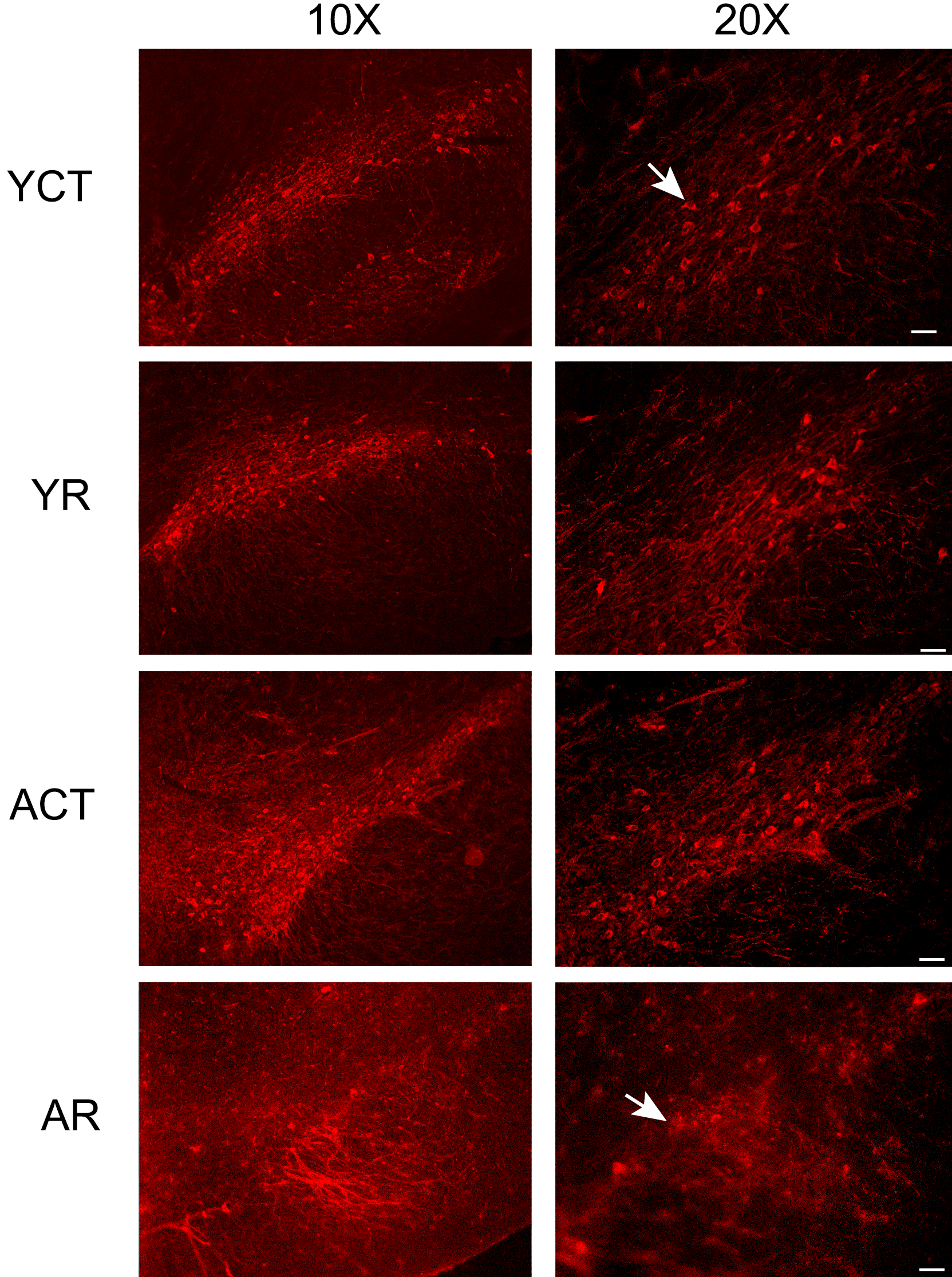
Rotenone reduces animal mobility and dopaminergic neuron labeling in the substantia nigra pars compacta (SNpc) of young and aged rats. Photomicrographs (a) and fluorescence quantification (b) of immunofluorescence labeling of tyrosine hydroxylase (TH) in the cryosections of the SNpc, from YR and AR animals. The representative photomicrographs (taken with an original objective 10× to show the whole SNpc, and an original objective 20× was employed to observe the details of the TH-positive cells) show that TH-positive cells are sparser in the SNpc of the rotenone-treated tissues compared to the control groups. Note that in the ACT group, the TH labeling is less intense, when compared to the YCT group. Arrows indicate a well-defined cell body and spines in the YCT group, but in the AR group, the labeling is sparser and more diffuse, indicating a degenerative process. (c) Open-field tests were performed and the number of crossings and rearings of each animal were counted, for 3 min. * indicates a significant difference compared to YCT; @ indicates a significant difference between ACT and AR (in Figure 1c) with p ≤ .05. ACT = aged control, AR = aged treated with rotenone, AU = arbitrary units, YCT = young control, YR = young treated with rotenone.
To evaluate motor behavior, an open-field test was performed with the YR and AR animals (Figure 1c). The mobility of the YR and AR animals was reduced, when compared with the respective control groups. It is worth mentioning that the ACT group presented a significant impairment in mobility when compared to the YCT animals, which might indicate that aging can cause motor activity impairment, which was further compromised by the rotenone treatment (Figure 1c). The rearing capacity was also impaired in the ACT and AR animals. The reduction in crossing and rearing rates is indicative of mobility and balance impairments. The AR animals displayed a prominent reduction in the crossing and rearing rates, which could be due to the observed neuronal loss detected in the striatum and SNpc.
Apoptotic Nuclei and Neuron Quantification
The effect of rotenone treatment was also evaluated by the presence of fragmented apoptotic nuclei, as detected by the TUNEL assay and NeuN labeling. As shown in Figure 2, the sections of the striatum from the YCT animals displayed only a few apoptotic nuclei, while the sections from the YR group contained an elevated number of fragmented nuclei. The rate of cell death in the striata of aged rats was greater than that observed in the young animals, and the treatment with rotenone exacerbated this degeneration in the aged group. In the SNpc of all the groups, only a few apoptotic nuclei were detected (data not shown).
The number of neurons was found to be reduced in the striata of the AR animals, as shown in Figure 2. Interestingly, the ACT group also had a reduced number of neurons compared to the YR animals, suggesting enhanced age-related neuron vulnerability in this structure.
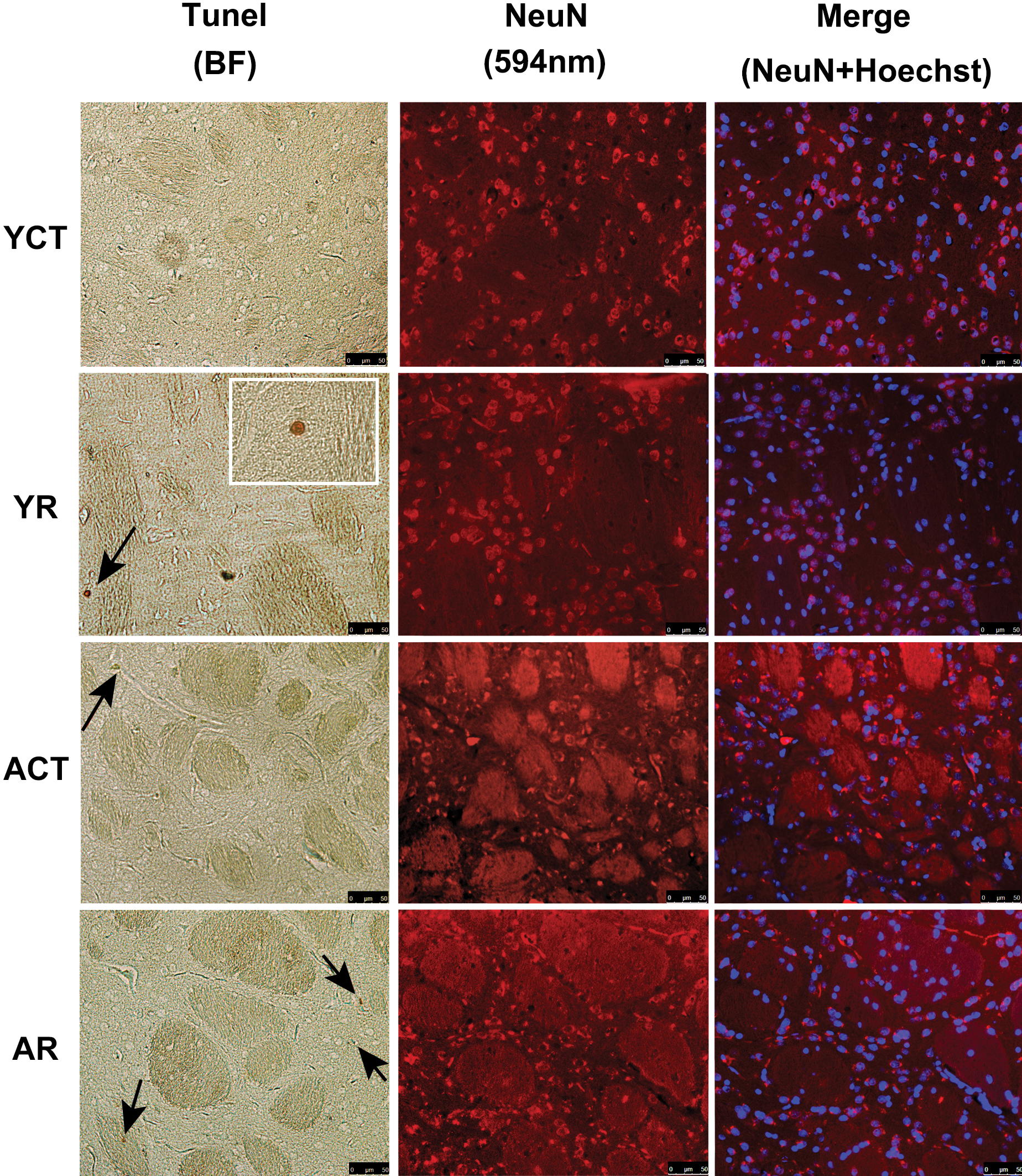
Rotenone and age-related increase in cell death, with a reduction in the number of neurons in the striatum. (a) TUNEL staining of fragmented nuclei in the striata sections (10 µm) from YR and AR animals. Representative photomicrographs (using an original objective 20×) are shown and the arrows indicate TUNEL-positive nuclei (brown), which is detailed in zoom box (using an original objective 63×) of the YR group. In addition to the TUNEL assay, immunofluorescence staining for anti-NeuN in the striatum was performed, and the nuclei were counterstained with Hoescht 33342. (b) The histograms (mean ± SE) show the morphometric analysis of the total number of TUNEL-positive cells in the striatum. (c) The number of quantified striata neurons is shown as a percentage of the total number of nuclei. Note that the aged striatal neurons (NeuN-positive labeling) are reduced compared to the young controls (YCT) and are reduced further upon rotenone treatment. * indicates a significant difference compared to YCT; @ indicates significant difference between AR and all groups (in Figure 2b). p ≤ .05. AR = aged treated with rotenone, BF = Bright-field, SE = standard error, YR = young treated with rotenone.
Electron Microscopy and ROS Measurements
As shown in Figure 3a (upper panel), the YCT animals exhibited typical ultrastructural features of neurons in the striatum and SNpc (Figure 3a in A and B). Additionally, the ACT group displayed lipofuscin granules (residual bodies), typically found during aging (Figure 3b in C). Lipofuscin was recognized as lysosome-containing vesicles, with marked vacuolization with lipid-like vesicles inside, surrounded by an electron-dense material. Ultrastructural changes were observed in both the YR and the AR groups (Figures 3a and 3b). Neuronal death in the SNpc was massive at the beginning of treatment but was not detected after 12 days of treatment (data not shown). Consequently, the ultrastructural evaluation of residual neurons in the SNpc may not reveal all of the ultrastructural changes of the rotenone-treated animals. While the young animals did not exhibit major ultrastructural changes in the surviving neurons of the SNpc, the presence of some apoptotic cells (not shown) and lipofuscin granules was observed. The AR animals displayed large amounts of lipofuscin granules when compared to the ACT animals (Figure 3b in C). Analysis on the amount of lipofuscin granules present in the AR animals revealed a statistical difference when compared to the ACT animals (Figure 3d). Striatal neurons from the YCT animals presented fewer swollen mitochondria (Figure 3b in D) compared to the ACT group and contained numerous multivesicular bodies (Figure 3a in E). Additionally, the AR animals presented copious amounts of mitochondrial swelling and cristae disarrangement in the striatum (Figure 3b in D).
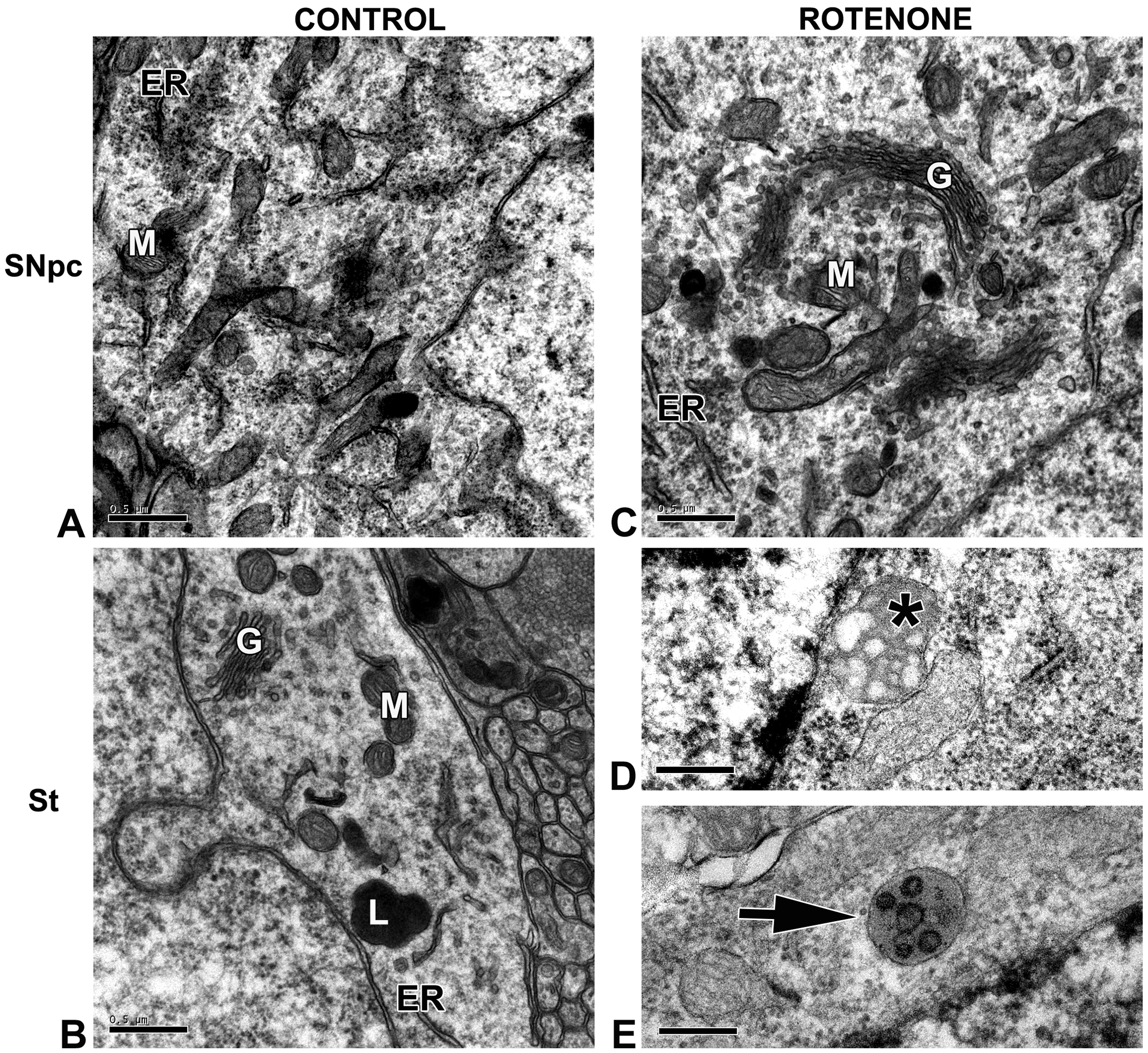
Animal model of Parkinson’s disease has increased levels of ROS in the striatum along with extensive mitochondrial swelling and in the aged group and massive deposits of lipofuscin granules in the substantia nigra pars compacta (SNpc) of AR animals. Representative electron micrographs of the dorsolateral striatal (St) or SNpc neurons from YCT and ACT animals (A and B) or YR and AR animals (C, D, and E). (a) In A and B, representative neurons of young animals displaying organelles with a normal appearance: mitochondria (M), Golgi apparatus (G), endoplasmic reticulum (ER) and lysosome (L). In C, a YR animal presenting normal ultrastructural appearance of all organelles: mitochondria (M) and Golgi apparatus (G) in the SNpc, whereas swollen mitochondria with disarranged cristae were found (asterisk in D), and multivesicular bodies (arrow in E) were seen in the striatal neurons. (b) In A and B, representative micrographs of neurons in the SNpc and striatum, respectively, of aged animals displaying normal ultrastructure: Golgi apparatus (G), endoplasmic reticulum (ER), mitochondria (M), and the typical findings of aging neurons—lipofuscin granules (L). In C, an AR animal showing massive deposits of lipofuscin granules, characterized by vacuolized lipid-like vesicles inside, surrounded by electron dense material. (L). In D, swollen mitochondria (arrow) are observed within a striatal neuron (arrow). (c) For ROS measurements, brain slices (400 µm) were loaded with CMH2DCFDA, and the fluorescence was measured by fluorimetry. Note that the steady-state ROS level is elevated in the striata from ACT animals, compared to the YCT group. (d) Lipofuscin granule quantitation, expressed as the percentage of area per µm2 of cytoplasm. * indicates a significant difference compared to YCT with p ≤ .05. ACT = aged control, AR = aged treated with rotenone, AU = arbitrary units, ROS = reactive oxygen species, YCT = young control, YR = young treated with rotenone.
ROS generation was assessed by measuring DCF fluorescence (Figure 3c). In the YCT group, a smaller amount of ROS production was observed, compared to the other groups. As expected, ACT tissues had a higher basal fluorescence level, which is commonly observed during the aging process, and surprisingly the AR group did not display any enhancement in ROS production when compared to the ACT group.
Discussion
In this work, we proposed to develop a pharmacological model of Parkinson’s disease in aged animals by rotenone treatment in order to compare the age-related effects in the striatum and SNpc. The results showed a remarkable age-related neurodegeneration in both structures, accompanied by an elevation in ROS generation, lipofuscin overload in the lysosomes, compromised mitochondrial morphology, and elevated cell death along with a reduced number of neurons, in the rotenone-treated animals, which could possibly be associated to animal akinesia.
Rotenone is a powerful pharmacological tool used to induce behavioral and biochemical alterations that resemble some features of Parkinson’s disease (Betarbet et al. 2000). Sherer et al. (2003) has described a pharmacological model of Parkinson’s disease with rotenone administration and observed that rotenone causes high toxicity and mortality, and some animals had motor symptoms before the end of the 28-day treatment period. Similarly, in our study, we noted that aged animals presented a high mortality rate before the 12th day of treatment, which suggests that this group is more sensitive to rotenone toxicity. With regard to the use of aging models, some studies have used mature (14- to 15-month-old) or aged (18-month-old) rats to evaluate rotenone toxicity in the nigrostriatal pathway. Wang et al. (2015) reported no changes in striatal dopamine content in young animals treated with rotenone, whereas aged rats presented reduced dopamine content in the striatum after the administration of the toxin. They also reported a reduction in glutathione and increased levels of malondialdehyde in the SNpc of aged animals. In the present study, we show that ACT animals presented higher levels oxidative stress in the striatum, which was also observed in the Parkinson's disease model (Figure 3c). Furthermore, Phinney et al. (2006) only reported a loss of dopaminergic neurons in mature rats treated with rotenone, and no alterations in α-synuclein staining patterns or protein aggregation were observed in the experimental groups. Here, we show that the YR and AR animals displayed decreased TH labeling in the SNpc (Figure 1b), and in agreement with the findings of Phinney et al. (2006), there were no observed differences in α-synuclein labeling in the animals treated with rotenone (data not shown). On the other hand, Cannon et al. (2009) showed that rotenone causes behavioral alterations along with a reduction in the number of dopaminergic neurons in the SNpc and the loss of striatal dopamine nerve terminals in young and middle-aged rats, treated with several doses of rotenone. It is plausible that the use of different protocols of rotenone treatment might degenerate the aged nigrostriatal pathway differently. Perhaps, our most notable contribution is the ultrastructural evidence for an impaired nervous system response to toxin exposure in old animals (24- to 25-months-old). This was demonstrated by the presence of altered mitochondria in the striatum and enlarged lipofuscin overloaded lysosomes in the remaining neurons of the SNpc, particularly in the AR animals (Figure 3).
With regard to striatum toxicity, Ferrante et al. (1997) previously reported that rotenone can cause striatal damage, without an alteration in the number of SNpc neurons, but only in young rats. In the present study, we report a tissue-specific degeneration in the nigrostriatal pathway, which could be associated with the mobility impairments, as evidenced by the observed akinesia and loss of equilibrium (Figure 1). Since the striatum of the rotenone-treated animals has numerous TUNEL-positive cells (Figure 2), which strongly suggests a degenerative process, we could speculate that the observed motor impairment might occur secondarily to the loss of dopaminergic neurons, particularly during the aging process. In fact, it has been previously reported that there is an elevated vulnerability in the striatum of aged rats (Ureshino et al. 2010), which could be potentiated by rotenone toxicity. Furthermore, it is plausible that dopaminergic neuron cell death in the SNpc occurs during the initial phase of the acute rotenone treatment, since sporadic TUNEL-positive nuclei were only detected in this structure after 12 days of rotenone treatment (data not shown).
In the present work, we observed scattered mitochondrial alterations in the striatum of YR rats and massive mitochondrial swelling and altered cristae in the striatum of AR rats, which can affect cell viability and lead to cell death. Accordingly, multivesicular bodies were also observed in the striatum of rotenone-treated animals, which might indicate an attempt to recycle damaged organelles. Surprisingly, the mitochondria of the few surviving dopaminergic neurons in the SNpc were not altered. It is worth mentioning that rotenone was not able to elevate ROS production in the striata of aged animals over that observed in age-matched controls and is probably due to the saturation of ROS in the system (Figure 3b). It is possible that the mitochondrial disruption can cause the release of proapoptotic factors such as cytochrome c or Smac/DIABLO and lead to an elevated rate of cell death (Du et al. 2000; Smaili et al. 2003), which is more pronounced in the rotenone-treated animals.
Therefore, this study suggests that the dysfunction in the nigrostriatal pathway in the AR animals could be related to subcellular alterations in the striatal neurons due to mitochondrial demise, which can ultimately lead to cell death. Our findings are in accordance with other reports on the mitochondrial–lysosomal axis theory of aging, since mitochondria and lysosomes are the most vulnerable organelles during the aging process (Brunk and Terman 2002; Terman, Gustafsson, and Brunk 2006). We can speculate that lipofuscin overload in the remaining nigral neurons possibly occurred due to the impairment of organelle turnover, presumably leading to the accumulation of damaged mitochondria, thus enhancing oxidative stress in rotenone-exposed tissues. The animal model presented in the present study can be utilized to investigate and explore new neuroprotective strategies toward the goal of preventing degeneration in the striatum during the aging process.
Footnotes
Acknowledgments
The authors would like to thank Andre Aguillera, Marcia Araguth, and Patricia Milanez for kind assistance in electron microscopy sample processing and figures format.
Authors’ Contribution
All authors (RU, AC, GP, RS, SS, AE) contributed to conception or design; data acquisition, analysis, or interpretation; drafting the manuscript; and critically revising the manuscript. All authors gave final approval and agreed to be accountable for all aspects of work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Fundação de Amparo à Pesquisa do Estado de São Paulo (FAPESP—2012/08273-3; 2013/20073-2), Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq—PVE 401236/2014-5), and Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES).
