Abstract
Trazodone (TRZ) is an antidepressant drug commonly used in the treatment of depression, anxiety, and insomnia. Although some studies demonstrated the adverse effects of TRZ related to cardiovascular system, the conflicting results were observed in these studies. Therefore, we aimed to investigate the cardiac adverse effects of TRZ in rats at repeated doses in our study. In accordance with this purpose, TRZ was administered orally to rats at 5, 10, and 20 mg/kg doses for 28 days. Electrocardiogram records, serum aspartate aminotransferase (AST), lactate dehydrogenase, creatine kinase-myoglobin band, cardiac troponin-T (cTn-T) levels, DNA damage in cardiomyocytes, and histologic view of heart tissues were evaluated. In addition, glutathione (GSH) and malondialdehyde (MDA) levels were measured to determine the oxidative status of cardiac tissue after TRZ administration. Heart rate was decreased, PR interval was prolonged, and QRS and T amplitudes were decreased in 20 mg/kg TRZ-administered group compared to the control group. Serum AST and cTn-T levels were significantly increased in 10 and 20 mg/kg TRZ-administered rats with respect to control rats. DNA damage was significantly increased in these groups. Additionally, degenerative histopathologic findings were observed in TRZ-administered groups. Although there was no difference in MDA levels between groups, GSH levels were significantly decreased in 10 and 20 mg/kg TRZ-administered groups compared to the control group. Our results have shown that TRZ induced cardiotoxicity in rats dose-dependently. It is assumed that oxidative stress related to GSH depletion may be accompanied by these adverse effects.
Introduction
Trazodone (TRZ) is an antidepressant drug of the serotonin receptor antagonists and serotonin reuptake inhibitors class used for the treatment of depression, anxiety, and insomnia. 1,2 The most frequently reported adverse effects related to TRZ treatment are orthostatic hypotension, syncope, headaches, dizziness, balance disorders, somnolence, and dry mouth. 3 Although it is widely used due to its lower anticholinergic properties and cardiac adverse effects compared to tricyclic antidepressants, and cardiac adverse effects than tricyclic antidepressants, the results of limited clinical studies investigating cardiovascular adverse effects of TRZ are conflicting. 4 It has been noted that TRZ may cause possible cardiovascular adverse effects because of the increased number of cardiotoxicity reports associated with TRZ treatment over the past years. 5,6 Studies have shown that postural hypotension, electrocardiogram (ECG) anomalies (such as prolonged PR interval, long QT syndrome), and cardiac arrhythmias were induced at therapeutic and subtherapeutic doses of TRZ. 7 –12
Many drugs may cause unexpected cardiotoxic side effects in the clinical stage, even though they pass animal testing stage. 13 Insomnia is a condition widespread in the general population, and TRZ is widely used as an alternative to benzodiazepines due to its hypnotic, anxiolytic, and sedative actions and the lack of the addiction risk in TRZ treatment of this indication. 14 It was determined that TRZ was used in 26.6% of insomnia cases. 15 At this point, we emphasized that it was important to determine the possible cardiovascular adverse effects of TRZ which was frequently used for this treatment. Therefore, we aimed to investigate the cardiotoxic effects of TRZ administration at pharmacological repeated doses in rats by determining serum cardiac biomarkers (including aspartate aminotransferase (AST), creatine kinase-myoglobin band (CK-MB), lactate dehydrogenase (LDH), and cardiac troponin-T (cTn-T) levels, ECG parameters, DNA damage in cardiomyocytes, and histopathological investigation of cardiac tissue. Additionally, we also aimed to evaluate the impact of oxidative status in a possible pathology by determining the levels of glutathione (GSH) and malondialdehyde (MDA), which are the most important markers used in the assessment of oxidative status.
Materials
The chemicals used during the experiment were obtained from the following sources: ketamine (Ketalar, Pfizer, Turkey), xylazine (Rompun, Bayer, Turkey), and TRZ (Desyrel® 50 mg, Angelini Pharma). AST, CK-MB, LDH, cTn-T, GSH, and MDA levels were measured with ELISA kits from CusabioBiotech, China.
Animals
Adult male Sprague-Dawley rats weighing approximately 300–350 g from the Research Center for Animal Experiments at Anadolu University were used in the experiments. The rats were housed in well-ventilated rooms at 24 ± 1°C in a 12-h dark 12-h light cycle, in the laboratory 1 week prior to the beginning of the experiment to ensure compliance with the working environment, provided with standard animal food and tap water. All experiments were carried out after the approval of Local Ethical Committee on Animal Experimentation of Anadolu University, Eskisehir, Turkey (File Registration Number: 2015-08). The rats were assigned randomly into the following administered groups: Control group: Animals received distilled water orally for 28 days (n = 8). TRZ-5 group: Animals received 5 mg/kg dose TRZ orally for 28 days (n = 8). TRZ-10 group: Animals received 10 mg/kg dose TRZ orally for 28 days (n = 8). TRZ-20 group: Animals received 20 mg/kg dose TRZ orally for 28 days (n = 8).
Pharmacological doses, which were determined in previous studies, were chosen as the study doses of TRZ. 16 –21 Furthermore, the clinical dose of TRZ for the treatment of depression is 250–600 mg per day and the proper dose for the treatment of insomnia is 25–100 mg before sleep. 22 The doses studied were also in accordance with the guidelines extrapolating human doses to animal doses. 23 All drugs dissolving in distilled water were administered at a volume of 1 mL/100 g body weight. The concentrations were adjusted to deliver the intended dose levels of the base compound. The treatment period was in accordance with the guideline OECD 407—repeated dose oral toxicity study in rodents. 24
Methods
ECG recordings
Animals were anaesthetized by intraperitoneal injection of 1.5 mg/kg urethane for ECG recording. 25 Then, needle electrodes were inserted in right arm, left arm, and chest position II using Biopac MP36 data acquisition system (Biopac Systems, Santa Barbara, California, USA). Records included measurements of heart rate (speed of the heartbeat), PR interval (time from the onset of the P wave to the start of the QRS complex), QT interval (time from the start of the Q wave to the end of the T wave), QRS (a combination of the Q wave, R wave, and S wave), and T wave (positive deflection after each QRS complex). 26
Determination of serum cardiac biomarker levels
Under anesthesia, blood samples for biochemical analyses were collected from the right ventricular tissue of the rat via syringe. The heart tissues were removed after the euthanazation of rats by withdrawing large amounts of blood from their hearts.
Serum specimens were prepared by centrifuging the blood samples collected at the gel-bottomed tubes for 15 min at 1000 × g at 4°C after 30 min of drawing the blood to allow clotting. Serum AST, CK-MB, LDH and cTn-T levels were measured using the commercially available kits according to manufacturer’s instructions.
Determination of DNA damage in cardiomyocytes
The heart tissue was homogenized in a Potter-Elvehjem PTFE pestle and glass tube (Sigma-Aldrich Chemie GmbH, Taufkirchen, Germany) in 1.5 ml of ice-cold Merchant medium (pH 7.4). Portions of the cell suspensions were placed in agarose. The comet experiment was basically carried out according to the procedure described by Singh et al. 27 Cardiac cells (10 μl) containing 1 × 105 cells/ml were suspended in 75 μl 1% (w/v) low melting point agarose while iced microscope slides were covered with 1% normal melting point agarose in phosphate buffered saline; 85 μl of this resulting suspension was allowed to adjust for 5 min at 4°C by applying a microscope slide (precoated with 1% normal melting point agarose) to form a microgel. The slides were immersed in a freshly prepared lysis solution (2.5 mM NaCl, 100 mM Na2EDTA, 10 mM Tris, 1% Triton X-100, and 10% DMSO, pH 10) for 1 h at this temperature followed by 20 min of electrophoretic buffer (1 mM Na2EDTA, 300 mM NaOH, pH 12.5) to expose DNA loosening and alkaline unstable damage. Following this procedure, the slides placed next to each other in a horizontal gel electrophoresis bath (24V, 300mA, 20 min) filled with fresh electrophoretic buffer were washed with 0.4 M Tris buffer for 5 min to neutralize excess alkaline and incubated for 1 h with SYBR Green I and covered with cover slips. Images were taken from slides using an Olympus BX (40×, Olympus, Tokyo, Japan) 50 fluorescence microscope. All measurement data were analyzed using software image analysis using BS 200 ProP (BAB Imaging System, Ankara, Turkey). One hundred cells were counted on each slide.
Histological analysis of heart tissue
Heart samples were fixed in paraformaldehyde (4%) in phosphate buffer with pH 7.2 at 20–22°C for 2 h and were dehydrated with the graded alcohol series. The samples were treated with the LR White mixture for 1 h at the same temperature to improve the permeability, followed by sectioning at 700 nm (0.7 μm) using samples embedded in LR White (Electron Microscopy Sciences, Fort Washington Pennsylvania, USA) and a Leica EM UC7 ultra-microtome (Leica Microsystems, Wetzlar, Germany). The resulting semi-thin sections were stained with 1% toluidine blue/borax (pH 8.4) for 2 min and observed with a Leica DM 750 light microscope (Leica Microsystems, Wetzlar, Germany). 28
Determination of GSH and MDA levels in heart tissue
Heart tissues were cleaned of blood in phosphate buffer solution (NaCl: 8 g, KCl: 0.2 g, KH2PO4: 0.2 g, Na2HPO4: 1.14 g, weighed in 1 l distilled water, pH 7.4). After cleaning blood and other contaminants, equal parts were separated and frozen in liquid nitrogen and stored at −20°C until they were used in biochemical analysis. The GSH and MDA levels in heart tissue were determined using commercially available kits according to the instructions of the manufacturer.
Statistical analysis
All data were expressed as mean ± standard error. Statistical analyses of the groups were performed using the SigmaPlot v.10 package program (Systat Software, Chicago, USA). In all experiments, Kruskal–Wallis analysis of variance following Tukey test as a post hoc test was performed. p < 0.05 was considered statistically significant.
Results
Effects of TRZ administration on ECG parameters
According to our ECG recordings, heart rate was significantly decreased in TRZ-20 group compared to the control group. In the TRZ-20 group, PR interval was significantly prolonged compared to the control group. When the groups were compared in terms of QRS amplitude, significant decrease in the amplitude was observed in TRZ-20 group compared to the control and TRZ-5 groups. Furthermore, T-wave amplitudes of TRZ-adminitered groups were significantly decreased compared to the control group (Table 1).
Effects of TRZ administration on ECG parameters.a
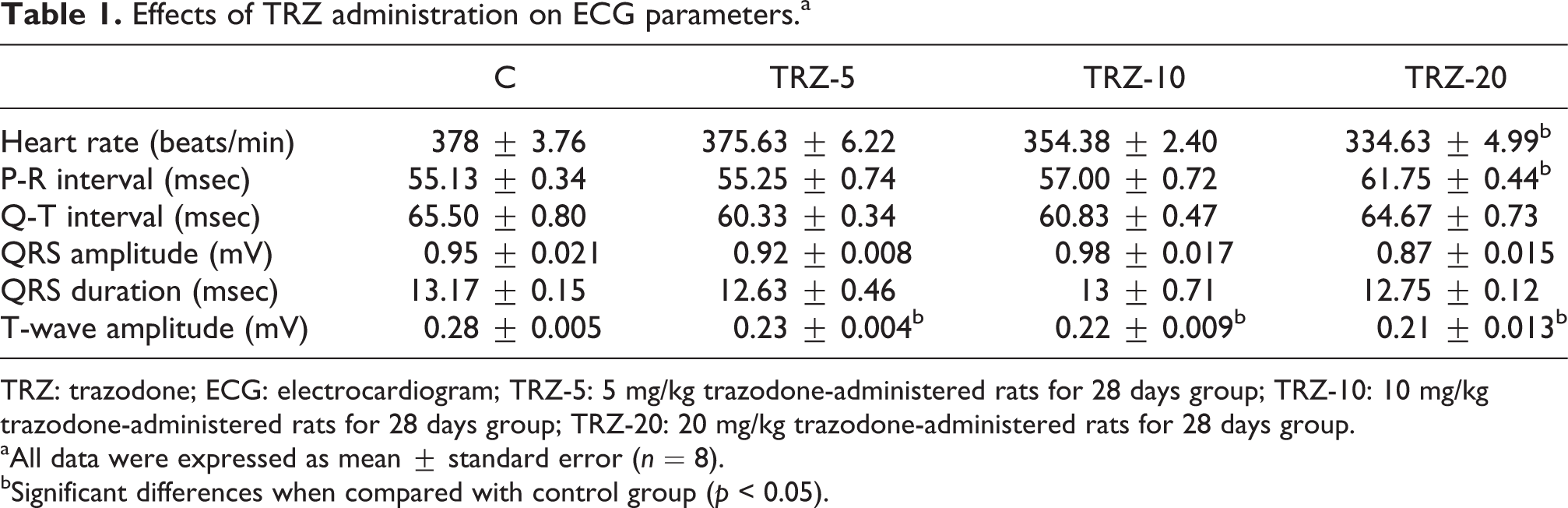
TRZ: trazodone; ECG: electrocardiogram; TRZ-5: 5 mg/kg trazodone-administered rats for 28 days group; TRZ-10: 10 mg/kg trazodone-administered rats for 28 days group; TRZ-20: 20 mg/kg trazodone-administered rats for 28 days group.
aAll data were expressed as mean ± standard error (n = 8).
bSignificant differences when compared with control group (p < 0.05).
Effects of TRZ administration on cardiac biomarkers
Serum AST levels of 10 and 20 mg/kg TRZ-administered groups were increased significantly compared to the control group. No significant differences were observed among the TRZ-administered groups (Table 2).
Effects of TRZ administration on cardiac biomarkers.a

TRZ: trazodone; AST: aspartate aminotransferase; CK-MB: creatine kinase-myocardial band; LDH: lactate dehydrogenase; cTn-T: cardiac troponin T; TRZ-5: 5 mg/kg trazodone-administered rats for 28 days group; TRZ-10: 10 mg/kg trazodone-administered rats for 28 days group; TRZ-20: 20 mg/kg trazodone-administered rats for 28 days group.
aAll data were expressed as mean ± standard error (n = 8).
bSignificant differences when compared with control group (p < 0.05).
When serum CK-MB and LDH levels were compared among groups, serum CK-MB and LDH levels obtained from the TRZ-administered groups were indistinguishable from the control group. No significant differences were observed among the TRZ-administered groups in terms of serum CK-MB and LDH levels (Table 2).
A significant increase in the serum troponin-T level was observed in the 10 and 20 mg/kg TRZ-administered groups compared to the control. Among the TRZ-administered groups, the serum cTn-T level did not show any significant differences (Table 2).
Effects of TRZ administration on cardiomyocyte DNA
DNA damage was expressed as the tail moment. The tail moment is, by definition, the width of the tail, a product of the tail DNA (tail moment = tail length × tail DNA% / 100). 14 Tail moments in TRZ-adminitered groups were significantly higher than the control group. Furthermore, the exposure to 10 and 20 mg/kg TRZ significantly increased DNA damage when compared to TRZ-5 group. At the same time, tail moment was significantly increased in TRZ-20 group compared to TRZ-10 group (Figure 1).

Effects of TRZ administration on cardiomyocyte DNA: (a) tissue comet assay photo of control group; (b) tissue comet assay photo of TRZ-5; (c) tissue comet assay photo of TRZ-10; (d) tissue comet assay photo of TRZ-20. All data were expressed as mean ± standard error (scale bars: 10 µm; n = 8). *p < 0.05: significant differences when compared with control group; #p < 0.05: significant differences when compared with TRZ-5; and +p < 0.05: significant differences when compared with TRZ-10. TRZ: trazadone; TRZ-5: 5 mg/kg trazodone-administered rats for 28 days group; TRZ-10: 10 mg/kg trazodone-administered rats for 28 days group; TRZ-20: 20 mg/kg trazodone-administered rats for 28 days group.
Effects of TRZ administration on the cardiac histology
Light microscopic longitudinal and cross examination of heart tissues of control group showed normal structure without any lesions. Prominent small vacuoles in the sarcoplasm accompanying normal histological architecture and cardiac muscle structure were observed in TRZ-5 group. In 10 mg/kg TRZ-administered group, larger vacuoles were determined in the sarcoplasm accompanying irregular myofibrils, hypertrophy, and fibrotic cardiac muscle in some regions. Degenerative changes including myocardial degeneration with fibrosis, asymmetrical hypertrophy of cardiac myocytes, irregular myofibrils, myofibrillar loss, intracellular edema, and inflammation were observed in TRZ-20 group (Figure 2).

Effects of TRZ administration on the cardiac histology—Longitudinal (a, c, e, g) and cross (b, d, f, h) sections of heart tissues obtained from groups. (a, b): Control group showing normal structure without any lesions. (c, d): TRZ-5 group with prominent small vacuoles in the sarcoplasm (arrowhead). (e, f): TRZ-10 group with larger vacuoles in the sarcoplasm (arrowhead) accompanying irregular myofibrils and hypertrophy (*). Fibrotic cardiac muscle can be seen in some regions (arrow). (g, h): TRZ-20 group showing signs of hypertrophic cardiomyopathy. Intracellular edema (arrowhead) and inflammation, myocardial degeneration with fibrozis (arrow) and asymmetrical hypertrophy (*) of cardiac myocytes. TRZ: trazodone; TRZ-5: 5 mg/kg trazodone-administered rats for 28 days group; TRZ-10: 10 mg/kg trazodone-administered rats for 28 days group; TRZ-20: 20 mg/kg trazodone-administered rats for 28 days group (scale bars: 30 µm; n = 8).
Effects of TRZ administration on GSH and MDA levels in cardiac tissue
Significant decreases in the GSH levels of cardiac tissues were observed in TRZ-10 and TRZ-20 groups when compared to the control group. Among the TRZ-administered groups, the GSH levels of cardiac tissues did not show any significant differences (Table 3).
Effects of TRZ administration on GSH and MDA levels in cardiac tissue.a

TRZ: trazodone; GSH: glutathione; MDA: malondialdehyde; TRZ-5: 5 mg/kg trazodone-administered rats for 28 days group; TRZ-10: 10 mg/kg trazodone-administered rats for 28 days group; TRZ-20: 20 mg/kg trazodone-administered rats for 28 days group.
aAll data were expressed as mean ± standard error (n = 8).
bSignificant differences when compared with control group (p < 0.05).
When MDA levels of cardiac tissues were compared among groups, the MDA levels of cardiac tissues obtained from the TRZ-administered groups were indistinguishable from the control group (Table 3).
Discussion
The cardiovascular adverse effects of TRZ were investigated in repeated pharmacological doses in our study which we performed independently of other risk factors related to cardiotoxicity. Pathological cardiovascular symptoms attracted attention in our 10 and 20 mg/kg TRZ-administered groups, especially in 20 mg/kg TRZ-administered group. In our study, it was determined that serum AST and troponin-T levels were increased, heart rate and amplitudes of QRS and T wave were decreased, PR interval was prolonged, DNA damage was induced with TRZ administration in rats. Intracellular edema and inflammation, myocardial degeneration with fibrosis, and asymmetrical hypertrophy of cardiac myocytes were observed in the histological sections of the heart tissue in 20 mg/kg TRZ-administered group, while larger vacuoles in the sarcoplasm, irregular myofibrils, fibrotic cardiac muscle in some regions, and hypertrophy were observed in the histological sections of the heart tissue in 10 mg/kg TRZ-administered group. On the other hand, when the oxidative status in the heart tissue after TRZ administration was evaluated, it was found that GSH levels were decreased at high dose groups. Consequently, it could be concluded that detected cardiac toxicity findings were accompanied by oxidative stress in the cardiac tissue.
As an important marker, ECG that measure electrical potential changes in the myocardium allows determining of possible myocardial pathologies by monitoring cardiac functions. 10,29 –32 The normal ECG consists of P wave, QRS complex, and T wave. The P wave consists of the electrical potentials that occur when the atrium is contracted before depolarization occurs. QRS complex refers to the depolarization before ventricular contraction. T wave indicates the repolarization state of the ventricles. 33 –35 Many drugs can cause ECG abnormalities even in patients without any cardiovascular disease history. 30 The PR interval represents the required time for an electrical impulse to be conveyed from the sinoatrial node via the atrioventricular node to the Purkinje fibers. 36 The PR interval was significantly increased in 20 mg/kg TRZ-admnistered group. The prolongation of PR interval indicated that atrioventricular conduction was weakened and delayed. 37,38 As different from studies that showed the ineffectiveness of TRZ on the PR interval, some studies indicated disturbances of atrioventricular conduction, atrioventricular block, and prolongation of PR interval after TRZ administration. 39 –44 PR interval is used to assess β-adrenergic receptor and channel functions including calcium, potassium, and sodium. 14,45 Prolonged PR interval can be accepted as a sign of β-adrenergic receptor and channel antagonism. 10,46 Some studies have shown that chronic treatment of TRZ leads to desensitization of β-adrenergic receptors. 47 –49 Additionally, TRZ has been found to block cardiac calcium channel, 50 cardiac potassium channel, and cardiac sodium channels. 8,12,51,52 Furthermore, some studies indicate that prolonged PR interval is based on hypertrophy and other structural changes. 53,54 Hypertrophic cardiomyopathy, which was observed in the rats that were administered high dose TRZ, might have caused the PR interval prolongation. When the T amplitude was compared between groups, it was found that there was a significant decrease in all TRZ-administered groups. Clinical studies indicated decreasing of T-wave amplitude after TRZ administration. 39,55,56 Ion channel blocking with xenobiotics may cause the decrease in T-wave amplitude. 57 Additionally, decrease of T-wave amplitude may be associated with ischemia and hypertrophic processes. 58,59 Bradycardia may be resulted from the direct effect of the drug on ion channels that determine the potential duration of action of cardiomyocytes and/or the inhibition of the cardiac conduction system. 52 In our study, heart rate was decreased after 20 mg/kg TRZ administartion. Also, other studies have shown that bradycardia was developed within 2 h after TRZ administration, at around the time of the peak plasma concentrations. 41,52
In our study, observed ischemic findings such as prolonged PR interval, decreased T-wave amplitude, and hypertrophy after high doses of TRZ administration were accompanied by the increased levels of biomarkers which indicated cardiotoxicity. Cardiovascular adverse effects can occur after acute or chronic drug treatment and can affect function (e.g. alteration of the mechanical function of the myocardium) and/or structure (e.g. morphological damage or loss of cellular/subcellular components of the heart) or vasculature. 60 There are also specific circulating biomarkers that may be used to signal potential drug-induced cardiotoxicity. The release of biomarkers to blood from myocytes occurs depending on the structural damage after drug exposure. 61 –63 High levels of AST in blood reflect myocardial damage, although it does not have complete specificity to the myocardium. In terms of serum AST levels, significant increases were observed in the groups after high doses of TRZ administration. The short half-lifes of CK-MB and LDH, which indicate myocardiyal injury, make them important for determining the acute myocardial damage. 64,65 At this point, it could be concluded that serum CK-MB and LDH levels were not altered after TRZ administration because of their short half-lifes. However, troponins, considered to be the most important biomarkers of myocardial ischemic damage and have the highest cardiac specificity in both clinical and animal model studies, are kept at circulation around 7–14 days. 66,67 In our study, cTn-T levels were increased after TRZ-administration dose-dependently. At this point, we emphasized that TRZ administration induced myocardial injury in rats related to the increased levels of AST and troponin-T levels. In other studies, ischemic findings were also reported associated with TRZ treatment. 41,68,69
Drugs cause myocardial injury due to cell death, imbalance of calcium homeostasis, inducing oxidative stress, disrupting membrane integrity, and cellular components. 66,67 The relationship between cardiovascular diseases and oxidative stress has been determined by numerous animal studies and clinical trials. It is known that oxidative stress cause disorders such as myocardial hypertrophy, contractile dysfunction, development of interstitial cardiac fibrosis, and endothelial dysfunction. 70 –73 In the heart, reactive oxygen species are produced during normal cellular functions of mitochondria during oxidative phosphorylation as well as enzymatic reactions catalyzed by xanthine oxidase, NAD(P)H oxidases, and cytochrome P450. 74 Additionally, oxidative stress is also induced with reactive metabolites which are formed via phase I biotransformation reactions after drug exposure in the heart. GSH depletion is considered as an indicator of oxidative stress in the heart tissue. 72,75 –77 Similar to GSH depletion, increasing MDA levels, which is an end product of lipid peroxidation, in the heart tissue also indicates oxidative stress. After myocardial ischemia, increase of MDA levels in the heart was established. 71 In our study, although there were no differences in terms of MDA levels, GSH levels were significantly decreased after TRZ administration. According to these results, it seems that TRZ administration may induce oxidative stress in the heart. Also, in vitro studies showed that TRZ and its reactive metabolite caused oxidative stress by GSH depletion and increasing MDA level. 76,78,79 Besides, xenobiotic-induced oxidative stress may cause damage to cellular components including DNA (e.g. DNA base modification), consequently the loss of cellular function. 80 –83 Comet, micronucleus, chromosomal aberration, and sister chromatid exchange assays are commonly used to determine the mutagenic and genotoxic effects induced by xenobiotics. 84 –87 Although studies are insufficient to investigate the genotoxic effects of TRZ, it has been shown that trazodone have clastogenic and mutagenic effects on human lymphocytes in vitro. 85 According to the results of our study, there was a significant increase in tail moment indicating DNA damage in TRZ-administered rats. Besides, increasing oxidative stress in cardiac tissue may induce structural changes such as cardiomyopathy, cardiac hypertrophy, and dysfunction of contractile protein. 83,88 –90 At this point, it is concluded that the histopathological findings in cardiac tissue might be associated with oxidative stress which is generated after TRZ administration in our study.
In conclusion, our study, which investigated the effects of repeated TRZ administration on cardiac parameters, showed that TRZ administration at high doses might have the potential to cause cardiotoxic effects. The presence of cardiovascular risk factors such as sex, age, cardiovascular disease history, and electrolyte anomalies may be more critical for patients undergoing TRZ therapy. Therefore, it may be important to adjust TRZ doses in the presence of these risk factors.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
