Abstract
Background:
Acrylamide is a very common compound even reaching up to our daily foods. It has been studied in a wealth of cell lines on which it proved to have various toxic effects. Among these cell lines, human lung adenocarcinoma cell line (A549) is one of that on which acrylamide’s toxicity has not been studied well yet.
Aim:
We intended to determine the half maximal inhibitory concentration (IC50) dose of acrylamide and to investigate its cytotoxic, anti-proliferative and apoptotic effects on A549 cells.
Methods:
We determined the IC50 dose by 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyl-2H-tetrazolium bromide (MTT) assay. Then, the mode of cell death was evaluated by flow cytometry using Annexin-V fluorescein isothiocyanate (FITC)/propidium iodide (PI) staining. Next, we performed transmission electron microscopy (TEM) and confocal microscopy analyses for morphological alterations and apoptotic indices.
Results:
According to the MTT assay results, A549 cell viability decreases proportionally with increasing acrylamide concentrations and IC50 for A549 was 4.6 mM for 24 h. Annexin-V FITC/PI assay results indicated that acrylamide induces apoptosis in 64% of the A549 cells. TEM and confocal microscopy analyses showed nuclear condensations, fragmentations, cytoskeleton laceration, and membrane blebbing, which are morphological characteristics of apoptosis.
Conclusion:
Our research suggests that acrylamide causes cytotoxic, anti-proliferative, and apoptotic effects on A549 cells at 4.6 mM IC50 dose in 24 h.
Introduction
Acrylamide is a highly reactive and water-soluble chemical having a formula of C3H5NO. It is utilized in a large spectrum of areas such as laboratories, constructions, polymer industry, ore processing, cosmetics, and wastewater treatment. This high prevalence of acrylamide poses a significant risk to human health due to its various documented clastogenic, genotoxic, and mutagenic effects in experimental animals. 1,2 Beyond that, acrylamide is also found in food commodities, especially those processed at higher temperatures (>120°C) during roasting, frying, grilling, or baking. 3 Actually, rather than being naturally in these commodities, acrylamide has been discovered to form by Michael addition reaction between amino acids (mainly asparagine) and reducing sugars, which is highly catalyzed by increasing temperature. This discovery further extended acrylamide toxicity to our daily foods along with its already existing effects, which prompted the studies regarding the acrylamide to increase abruptly.
Besides the negative effects aforementioned, acrylamide is reported to have inhibitory effects on both proliferation and differentiation of various carcinogenic cell lines in a time- and dose-dependent manner including human neuroblastoma and glioblastoma cell lines. 4 In addition, acrylamide has an inhibitory effect on neural cell adhesion molecule expression in human neuroblastoma cells, which is important for cell proliferation, survival, motility, and axon guidance. 5 Furthermore, low-dose acrylamide is suggested to increase HepG2 cancer cell line proliferation, but to decrease human lung adenocarcinoma cell line (A549) viability while not changing MDA-231, HeLa, and PC-3 cell line viabilities. 6 Moreover, acrylamide was demonstrated to have apoptotic effects on human astrocytoma-derived cell line U-1240 in a time- and dose-dependent manner. 7,8 Lung carcinoma is one of most widespread cancers with high mortality rate in the world. A549 cell line is a widely used model cell line for the toxicity studies regarding lung metabolism and carcinogenesis. It is reported that A549 cell line might be one of the highly sensitive to acrylamide-induced toxic effects 6 ; however, no the half maximal inhibitory concentration (IC50) value for acrylamide on A549 cells has been reported clearly yet. There are few studies investigating the effect of acrylamide on A549 cells that are not elaborate and specific. In this study, we aimed to determine an IC50 value for acrylamide on A549 cells and to ascertain acrylamide’s effects on cell proliferation, viability, and apoptosis by using flow cytometry, transmission electron microscopy (TEM), and confocal microscopy.
Materials and methods
Chemicals
Acrylamide, Dulbecco’s modified Eagle’s medium (DMEM), fetal bovine serum (FBS), penicillin–streptomycin, dimethyl sulfoxide (DMSO), phalloidin, and 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyl-2H-tetrazolium bromide (MTT) were obtained from Sigma-Aldrich (St Louis, Missouri, USA). Trypsin/EDTA[A: Please provide expansion for “EDTA.”] solution was purchased from Biochrom (Berlin, Germany). Annexin-V fluorescein isothiocyanate (FITC) apoptosis kit was purchased from BD Pharmingen (San Diego, California, USA). A549 cell line was obtained from American Type Culture Connection (Rockville, Maryland, USA).
Cell culture
A549 cells were cultured in DMEM supplemented with 10% (v/v) FBS and 1% (v/v) penicillin–streptomycin (100 U/mL–100 µg/mL) at 37°C in a humidified atmosphere of 95% air and 5% carbon dioxide. Confluent (80–90%) A549 cells were harvested with 0.25% trypsin-1 mM EDTA solution and plated at a density of 3 × 103 cells per well into 96-well microtiter tissue culture plates and incubated for 24 h before acrylamide administration.
MTT assay
To determine cytotoxicity of acrylamide on A549 cells, MTT colorimetric assay was used. Firstly, 100 mM stock solution of acrylamide was freshly prepared with distilled water and filtered through 0.22 µm sterile filter before use. Further dilutions were performed with culture medium. A549 cells at a density of 3 × 103 cells per well were seeded in a 96-well microtiter plate and treated with different acrylamide concentrations ranging from 0.5 mM to 20 mM for 24 h at 37°C. After the incubation period, 20 μL of MTT solution (5 mg/mL in distilled water) per well was added and the plates were incubated for 2 h at 37°C. Afterwards, the medium in each well was removed, 200 µL of DMSO was added per well at room temperature to dissolve formazan blue crystals 9 and then plates were read on an ELISA reader (ELx808, Winooski, Vermont, USA) at 540 nm wavelength. Each group was repeated in four times. The percentage of cell viability was calculated by the following formula 10 :

IC50 was determined with the corresponding graph according to the percentage of cell proliferation.
Flow cytometry
The apoptotic effects of acrylamide on A549 cells were evaluated by Annexin-V FITC/propidium iodide (PI) staining. For Annexin-V staining test, cells were incubated with the IC50 of acrylamide for 24 h. After the incubation period, 100 µL of untreated and acrylamide-treated cells were transferred to separate tubes and 100 µL of Annexin-V reagent was added to each tube. Tubes were incubated for 20 min at room temperature at dark, then analyzed with Muse™ Cell Analyser (Merck, Millipore, Hayward, California, USA). Samples were prepared in four times and according to the user manual of Muse® Annexin-V and Dead Cell Assay Kit.
TEM analysis
Ultrastructural changes in acrylamide-treated A549 cells were analysed by TEM (FEI Technai BioTWIN, Limnen, The Netherlands). The IC50 of acrylamide was applied to A549 cells. After 24 h, untreated and acrylamide-treated cells were harvested and centrifuged at 1200 r/min for 5 min. Pellets were fixed in 2.5% glutaraldehyde overnight at 4°C, embedded in agar, and re-fixed in 2% osmium tetroxide. After dehydration in ascending alcohol series (70%, 90%, 96% and 100%), samples were embedded in epoxy resin. About 100 nm-thick sections were cut and stained with lead citrate and uranyl acetate respectively before TEM examination. 10
Confocal microscopy
A549 cells were plated onto sterilized coverslips in a petri dish and exposed to the IC50 of acrylamide for 24 h at 37°C. After 24 h, untreated and acrylamide-treated cells were fixed in glutaraldehyde/paraformaldehyde (2%) at room temperature and stained with phalloidin and acridine orange. The preparations were observed under a confocal microscope (Leica TCS-SP5 II, Wetzlar, Germany) based on changes in nuclei and other cell structures using Leica Confocal Software version 2.00. 11
Statistical analysis
MTT assay data were analysed using SPSS software (Statistics for Windows, Version 21.0, IBM Corp. Armonk, New York, USA) with one-way ANOVA test, followed by the Tukey’s post hoc test. The mean ± standard deviation (SD) values were shown as a supplement (Online Supplement 1). Flow cytometry data were analyzed using Minitab® 17.3.1 software (trial version) with two proportions
Results
MTT assay
As seen in Figure 1 and Online Supplement 1, ascending acrylamide doses are inversely proportional to A549 cell viability. Increasing acrylamide concentrations decreased A549 cell viability in a dose-dependent manner. The IC50 dose of acrylamide for A549 cells in 24 h was found to be 4.6 mM. When we compared different acrylamide doses-treated cells with untreated cells, we determined significant differences between them (all

Cell viability percentages according to acrylamide doses determined by MTT assay after 24 h of acrylamide treatment (IC50: 4.6 mM). Dots show means and bars show standard deviations of four separate experiments. (a)
Flow cytometry
The results of flow cytometry assay are shown in Figure 2 and Table 1. The viable cell percentage for untreated and acrylamide-treated cells (IC50 = 4.6 mM) were found as 76.51% and 34.96%, respectively, and a significant difference (
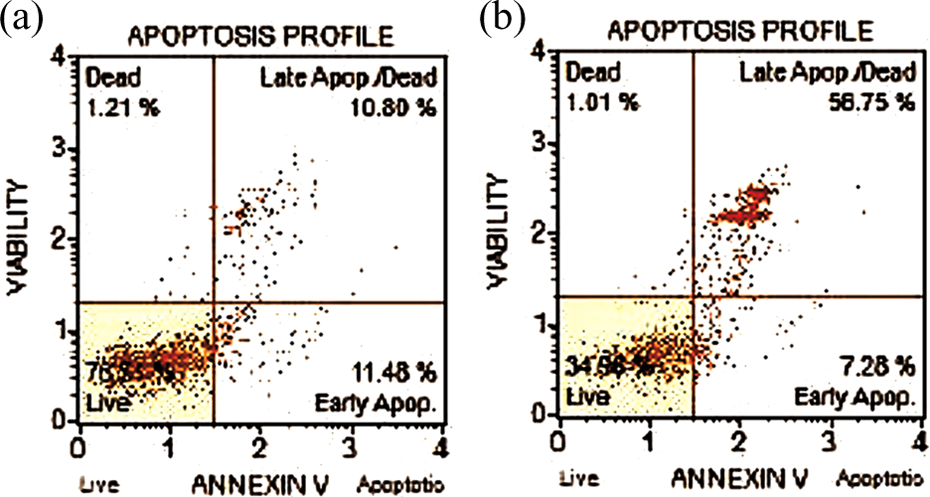
Viability and apoptosis percentages of A549 cells after 24 h with Annexin-V FITC/PI staining. (a) Untreated cells and (b) acrylamide-treated cells. A549: human lung adenocarcinoma cell line; FITC: fluorescein isothiocyanate; PI: propidium iodide.
Viability and apoptosis percentages of untreated and acrylamide-treated A549 cells after 24 h (IC50: 4.6 mM).

Q1
a
TEM analysis
In TEM analysis, A549 cells exposed to acrylamide revealed several ultrastructural changes such as blebbing on the cell membrane, vacuolization, formation of apoptotic bodies and holes as well as nuclear condensation, reduction and fragmentation. The untreated cells showed no conspicuous alterations and exhibited nearly normal cell ultrastructure (Figure 3(a) to (c)).

TEM micrographs of A549 cells. (a) A normal cell with homogeneous cytoplasm and smooth-bordered nucleus, (b) an acrylamide-treated cell with an irregularly shaped and indented nucleus containing chromatin clumping (
Confocal microscopy
In confocal microscope analysis, it is evident that acrylamide caused condensation and fragmentations of cell nuclei and DNA, membrane blebbing, vacuolizations, apoptotic bodies, and cytoskeleton lacerations on A549 cell line (Figure 3(d) to (f)).
Discussion
To our knowledge, this is the first study to focus on A549 cells using different methods for evaluation of acrylamide’s effects.
In the current study, we suggested for the first time IC50 for acrylamide-treated A459 cells, which was found as 4.6 mM. In the literature, IC50 value for acrylamide-treated PC-12 cells and HepG2 cells were reported as 5 and 7.18 mM, 12,13 which are greater than our IC50 value. This indicates that A549 cells are more sensitive to acrylamide than PC-12 and HepG2 cells. There are other studies which do not specify IC50 value after acrylamide treatment, but interval of IC50 value can be inferred from their data. In this manner, IC50 of acrylamide-treated rat cortical neurons 14 and V79 cells 15 are extrapolated to be 10 and 4–5 mM, respectively. Rat cortical neurons are more resistant to acrylamide, while V79 cells are as sensitive as A549 cells, IC50 of both are close. Considering that V79 cells are lung cells derived from hamster, this similarity is not surprising.
MTT assay results of the present study clearly showed that acrylamide exerts anti-proliferative and cytotoxic effect on A549 cells in a dose-dependent manner. Previous studies reported that acrylamide is an anti-proliferative and cytotoxic agent for many cell lines including keratinocytes, 16 nerve cells, 17 and fibroblast cells 18 as normal cell lines and hepatocellular carcinoma, 13 pheochromocytoma, 12 and human astrocytoma cells 19 as carcinogenic cell lines. The anti-proliferative effect of acrylamide is suggested to be due to its DNA damaging effect. After DNA damage, p21, p27, and p53 (damage associated proteins) are activated and cell proliferation is stopped in the G0/G1 phase to repair DNA. Ataxia telangiectasia mutated/Rad3-related kinases, which are responsible for regulation of DNA repair, were reported to be implicated in this G0/G1 arrest. 19 Accordingly, the amounts of DNA damage associated proteins and checkpoint proteins (Cdk2 and cyclin D1) after acrylamide treatment were found to be increased. 19
Apoptosis is known as regulated cell death and composed of series of consecutive changes, which are characterized by some apparent cell alterations including cell shrinkage and fragmentation into membrane-bound apoptotic bodies, genomic DNA cleavage, cytoplasmic, and nuclear condensation. 20 –22 We have also determined such alterations in the acrylamide-treated A549 cells using different methods. Firstly, our flow cytometry results showed that acrylamide increased the percentage of the late apoptotic cells almost 5.5 times compared to the untreated cells. In addition, the viability of acrylamide-treated cells was less than the half of that of untreated cells. These findings support that acrylamide induces cell death mainly by apoptosis. Mehri et al. 12 found that approximately 43% of acrylamide-treated PC-12 cells underwent to apoptosis. In our study, this percentage was 64%. As mentioned above, this further indicates that A549 cells are more sensitive to acrylamide toxicity. Secondly, TEM reveals to detect the alterations of cell organelles and is a crucial method for toxicity researches. However, there are a few cell culture studies using TEM analysis for acrylamide toxicity. In a previous study, Chen et al. 8 observed acrylamide-treated astrocytoma cells with TEM and detected mitochondrial abnormalities, cytoplasmic vacuole formation, pyknosis, and vesicular matrix compartments. In our study, we also detected similar alterations with TEM, which indicated acrylamide induced toxicity on A549 cells. To demonstrate the morphological alterations, as a third method, we used confocal microscopy, which clearly shows the cytoplasmic and nuclear degenerations. Unfortunately, we could not detect any previous reports in the literature about the use of confocal microscopy in acrylamide-treated cell lines. In this regard, we think that our present study is unique. Under confocal microscope, we detected apoptotic bodies and nuclear fragmentations, which clearly indicated that acrylamide leads to apoptosis in A549 cells.
In the literature, apoptotic effect of acrylamide has been documented. 7,8,23 Previous reports explain how acrylamide acts on cells. According to these reports, acrylamide is an electrophile, that is, electron-deficient, and exerts its toxicity with interacting covalent bonds of nucleophilic residues of macromolecules, especially cysteine residues including DNA and cellular proteins. 24 Besides, acrylamide interferes with kinesin proteins, which are responsible for the spindle fibers formation during cell division, thereby inhibiting proliferation. 25
As for the apoptotic mechanism, the main reason for acrylamide toxicity was reported to be stemmed from reactive oxygen species (ROS), that is, oxidative stress. 26,27 Acrylamide treatment causes the increase in mitochondrial reactive oxygen radicals. Then, Nrf2, which is a transcription factor responsible for activation of antioxidant-related genes, is activated to some extent and thus the synthesis of glutathione (GSH) and other antioxidants are upregulated. 28,29 High amount of acrylamide leads to GSH depletion via conjugation and excessive ROS formation. 30 In addition, heat shock proteins, the regulators of protein folding in extreme conditions, are activated during oxidative stress. 31 When the balance between antioxidant production and ROS formation are disrupted, as reported above, acrylamide inflicts DNA damage and G0/G1 cell cycle arrest occurs and the levels of pro-apoptotic indices such as Bax/Bcl, caspase 3 and 9, and cytoplasmic cytochrome c release are elevated. 8,27,32 Eventually, the cells undergo apoptosis. Also, the studies showing antioxidants preventing acrylamide toxicity 8,12,13 also proves that acrylamide mainly exerts its damaging effect by oxidative stress.
In conclusion, the present study revealed an IC50 dose for acrylamide-treated A549 cells. Moreover, cytotoxic, anti-proliferative, and apoptotic effects of acrylamide on A549 cells have been clearly demonstrated. Our results make a contribution to the elucidation of the relation between acrylamide and A549 cells. We believe that additional researches are needed to bring to light the exact mechanism of acrylamide on these cells.
Supplemental material
Supplement_1 - Acrylamide-derived cytotoxic, anti-proliferative, and apoptotic effects on A549 cells
Supplement_1 for Acrylamide-derived cytotoxic, anti-proliferative, and apoptotic effects on A549 cells by S Kacar, D Vejselova, HM Kutlu and V Sahinturk in Human & Experimental Toxicology
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplementary material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
