Abstract
Petroleum coke (PC) is a coal-like product that is produced during the refinement of crude oil and bituminous sand. Fugitive dust from open storage of PC in urban areas is a potential human health concern. Animal inhalation studies suggest that PC leads to an adverse pulmonary histopathology, including areas of fibrosis and chronic inflammation; however, little is known about its impact on human health. In order to identify biomarkers and cellular pathways that are associated with exposure, we performed two-dimensional liquid chromatography–mass spectrometric analyses on secreted proteins from two human lung culture models. A total of 2795 proteins were identified and relatively quantified from an immortalized cell line and 2406 proteins from primary cultures that were either mock treated or exposed to particulate matter with a diameter of 2.5–10 μm PC or filtered urban air particulates for 16 h. Pathway analysis on secretomes from primary lung cultures indicated that PC exposure suppressed the secretion of proteins involved in the organization of the extracellular matrix and epithelial differentiation. Because these cellular processes could facilitate fibrosis, we performed chronic 12-day exposure studies on three-dimensional human lung cultures consisting of epithelia and stromal fibroblasts. Relative to mock-treated cells, matrix metallopeptidase 9 levels in the conditioned media were lower by 4 days postexposure and remained suppressed for the duration of the experiment. Immunocytochemical staining of collagen III, a marker associated with fibrosis, showed increased accumulation in the epithelial layer and at the air–liquid interface.
Introduction
Refinement of oil sands has increased dramatically over the past 20 years as higher prices for crude oil and new technologies have enabled profitable extraction, transport, and processing. Bituminous sand deposits are found in many countries, but the largest reserves are located in Western Canada. Recent upgrades to refineries in Chicago and Detroit have allowed processing of Canadian oil sand bitumen into fuel products. A by-product of this refinement is petroleum coke (PC), a black carbonaceous material that resembles coal but with a higher silt content. Storage of PC in open piles and uncontrolled release of dust have aroused health concerns from residents of the surrounding communities. It is currently unknown the extent to which PC, as an airborne fine particulate matter (PM), initiates or exacerbates lung disease either alone or in conjunction with other contaminants present in the urban environment.
Animal studies have shown that PC is not carcinogenic via inhalation or dermal exposures, and there is low risk of developmental toxicity or reproductive effects, 1 which was reviewed by Caruso et al. 2 Adverse effects were observed in the trachea and lungs of rats and/or monkeys during a 2-year inhalation study. These included inflammatory changes and a net increase in tissue weights due to accumulation of macrophages, neutrophils and leukocytes, as well as pulmonary interstitial inflammation, and sclerosis and focal fibrosis. The severity of these outcomes correlated with the dose and duration of PC exposure and were described as non-reversible. 1
Epidemiological and clinical research have provided compelling evidence that exposure to fine PM air pollution, and black carbon in particular, 3 contributes to the development of a wide range of diseases including asthma, 4 chronic obstructive pulmonary disease, 5 lung cancer, 3 cardiovascular disease, 6 Alzheimer’s disease, 7 diabetes, 8 and an increased risk of birth defects. 9 Size and molecular composition of the particulates are key determinants for the health effects associated with exposure. Inhaled particles in the 1–10 μm diameter range have the capacity to reach the distal lung, where they deposit on the airway epithelium. Inhalable coarse PM with a diameter of 2.5–10 μm (PM10) and fine particles with diameter <2.5 μm (PM2.5) are most frequently cited in the literature, with the latter having the strongest impact on human health. Environmental PM is a mixture of organic compounds, such as black carbon, polycyclic aromatic hydrocarbons, and cellular material, and inorganic material, such as silica and heavy metals. PC is roughly 90% carbon with the remainder primarily consisting of sulfur and residual hydrocarbons, and enrichment of vanadium and nickel is observed. Dissecting the contribution of individual components and combinatorial effects are ongoing objectives of PM-related health research.
Animals traditionally have had a central role in toxicology, but future research will rely more heavily on human models. 10 –12 Problems inherent in animal research include tremendous cost and time requirements, low sample throughput, difficulties in extrapolating high-dose animal studies to low-dose human chronic exposures, and lack of data on human intraspecies variability. Advances in stem cell research and three-dimensional (3-D) organ cultures will likely make it possible in the near future to test the potential toxicants in human organotypic systems that incorporate primary cells to reconstruct liver, heart, lung, brain, and other tissues. 13 For the lung, we can now move beyond two-dimensional (2-D) immortalized bronchial and alveolar cell lines and assay more physiological in vitro models that present a pseudostratified and polarized phenotype and include ciliated epithelial cells and mucin-producing goblet cells, which together develop high transepithelial electrical resistance. 14 “Omics” technologies can then be applied to these human models to thoroughly mine changes in RNA transcript levels, protein abundance and modification, and/or metabolite profiles. Bioinformatics and holistic systems biology approaches are used to interpret the large data sets that these technologies produce and decipher the cellular and molecular mechanisms associated with a toxic response. The expectation is that this type of mechanistic toxicology will develop into an important tool for assessment of human health risk. 15
The goal of the present work was to identify biomarkers and pathways associated with PC exposure in human lung epithelial cell cultures. PC was ground to a fine PM level, and exposures were compared against a standard PM collected from an urban environment. Quantitative secretome analysis was performed in 2-D immortalized and 3-D primary human lung cell models. Results from these studies suggest that proteins associated with maintenance of the extracellular matrix (ECM) are secreted to a lesser extent after PC exposure and that long-term exposure may lead to an increased accumulation of collagen, a hallmark of fibrosis.
Methods
Cell cultures
A549 cells were purchased from the ATCC. This adherent, human adenocarcinoma lung cell line has an undifferentiated alveolar phenotype and was maintained in F-12K culture medium supplemented with 10% fetal bovine serum and 100 U/ml Pen/Strep. EpiAirway™ and EpiAirwayFT™ primary human lung cultures were purchased from MatTek Corporation (Ashland, Massachusetts, USA). Both of these 3-D cultures consisted of ciliated epithelial cells, mucus-producing goblet cells, and basal cells, and the EpiAirwayFT cultures contained an additional sublayer of primary stromal fibroblasts. Primary cultures were delivered on cold packs and immediately prepared for incubation at 37°C as directed by the manufacturer. Cultures were fed with a proprietary serum-free medium supplied by the company through a porous support membrane and were treated the day after delivery without freezing. Cell cultures were maintained in incubators at 37°C, 5% carbon dioxide.
Treatment protocol
PC was collected from the intersection of W Fort Street and Rosa Parks Boulevard in Southwest Detroit, Michigan, USA, adjacent to a storage facility along the Detroit River. The sample was crushed using a mortar and pestle, and 160 g was milled to a mean size of 158.7 μm (SD 100.9 μm) by the Jet Pulverizer Co. (Moorestown, New Jersey, USA). The final milled PC, measured by a HORIBA LA950 Laser (Kyoto, Japan), particle size analyzer, had a mean size of 2.85 µm and a median size of 2.58 µm with an SD of 1.39 µm (99% <7.20 µm). Control urban particulate matter (UP), collected by air filtration over a period of 12 months, was purchased from the National Institutes of Standards and Technology (NIST; part number 1648a). PC and UP stock suspensions were prepared in phosphate-buffered saline (PBS) at a concentration of 5 mg/ml, aliquoted and frozen at −80°C until further use. To fully resuspend particles, stock suspensions were thawed, vortexed for 1 min, and subjected to at least 15 min of bath sonication. Prior to treatment, A549 cells (log-phase growth in 96-well or 100 mm plates) were washed twice with PBS and then returned to the incubator for 30 min in serum-free medium. EpiAirway (35 mm inserts transferred to 100 mm plates) and EpiAirwayFT (12 mm inserts transferred to 6-well plates) cultures were prepared as recommended by the company and treated 24 h after delivery at the upper mucociliary liquid–air interface. At 3 h posttreatment, free liquid was removed from the top surface, and cultures were returned to the incubator. For long-term treatments, culture media was replenished every 2 days. In some studies, EpiAirwayFT cultures were maintained in 10 ng/ml of human recombinant transforming growth factor β1 (TGF-β1; Life Technologies) as a control for fibrosis.
Cell viability assays
A549 cells were plated at 10,000 cells/well in 96-well plates. The following day, cells were mock treated (with PBS) or treated in quadruplicate with various concentrations of either PC or UP as described above. After 16 h of exposure, the media were replaced with fresh serum-free media containing 10% Alamar Blue™ reagent (Thermo Scientific) and the plates returned to the incubator. Every 30 min thereafter, until the 3-h time point, plates were scanned at 570 and 600 nm absorbance. Blank readings were made with wells containing medium only, and negative control wells contained medium plus Alamar Blue (no cells). Percentage reduction of Alamar Blue reagent was calculated as described by the manufacturer for each time point, and these values were plotted over time to calculate the rate of reduction and then normalized to the rate of reduction found in untreated cells. This experiment was repeated at least three times.
Toxicity in EpiAirwayFT cells was assayed by lactate dehydrogenase (LDH) release into the culture medium (LDH-Cytotoxicity Assay Kit II, RayBiotech, Inc.). LDH oxidizes lactate to generate NADH (Norcross, Georgia, USA), which then reacts with WST reagent to generate a color change that is read at an optical density (OD) of 450 nm on an absorbance plate reader. Cultures were treated in triplicate samples, and each sample was analyzed in triplicate. Cytotoxicity (%) was calculated as (test conditioned media − background)/(lysate in media − background) × 100 and were averaged for the samples.
Secretome preparation
After 16 h of mock treatment or exposure to 100 μg/ml of either PC or UP (each in triplicate), media were carefully removed from cells and pelleted at low speed to remove floating cells (10 min at 800 × g, 4°C). Supernatants were transferred to prewashed Amicon Ultra-15 filter (MilliporeSigma, Burlington, Massachusetts, USA) units with a 10,000 nominal molecular weight limit cutoff membrane and centrifuged for 40 min at 5000 × g, 4°C. Concentrated samples were washed 3× with 10 ml of 5 mM HEPES, pH 8.5, under the same centrifugation parameters. Samples (<1 ml) were transferred to 1.5 ml tubes, and the protein concentrations were estimated using the bicinchoninic acid assay assay.
Protein digestion and isobaric tag labeling
Twenty microgram of each sample was denatured by boiling for 5 min in 0.1% RapiGest (Waters, Milford, Massachusetts, USA). Samples were cooled and reduced with 5 mM DTT (30 min, 60°C) and alkylated with 20 mM iodoacetamide (30 min, room temperature (RT)). Proteins were digested overnight at 37°C with trypsin (Promega, Madison, Wisconsin, USA) in a final concentration of 33 mM HEPES, pH 8.5, and 10% acetonitrile. RapiGest was precipitated by acidification to 0.5% trifluoroacetic acid, removed by centrifugation, and then peptides were purified using Pierce 100 μl C18 tips. Dried samples were resuspended in buffer containing 100 mM HEPES, pH 8.5, 50% acetonitrile, and 0.5 mg of a unique TMT10plex™ isobaric tag (Thermo, Thermo Fisher Scientific, Waltham, Massachusetts, USA) for 1 h at RT. Reactions were quenched with 0.4% hydroxylamine, acidified to 2.5% trifluoroacetic acid, and the nine samples were combined. The final sample was purified by C18 chromatography and then dried by vacuum centrifugation.
Two-dimensional liquid chromatography–mass spectrometry
Peptides were resuspended in 5% acetonitrile/0.1% formic acid/0.005% trifluoroacetic acid and separated over a reverse phase PLRP-S column (Agilent, Agilent Technologies, Santa Clara, California, USA) using mobile phases A (2% acetonitrile) and B (98% acetonitrile), with both buffers adjusted to pH 10.0 using ammonium hydroxide. First dimension fractions were collected using a TriVersa NanoMate (Advion, Ithica, New York, USA) system paired to a LTQ-XL mass spectrometer (Thermo). Second dimension peptide separation on 20 fractions was performed using reverse-phase chromatography under acidic conditions (0.1% formic acid) and a 75-min gradient with an EASY nLC-1000 ultra high pressure liquid chromatography (UHPLC) system (Thermo). Mass spectrometry was performed with an Orbitrap Fusion (Thermo). Mass spectrometry 1 (MS1) scans were acquired at 120,000 resolution in the range of 350–1600 m/z. MS2 scans were performed on the top 10 most abundant multiply charged ions, with dynamic exclusion set for 30 s, using collision-induced dissociation (CID) collision energy set at 35% and the ion trap for detection. TMT tags were analyzed in MS3 scans using synchronous precursor selection, higher-energy C-trap dissociation (HCD) collision energy set at 65%, and orbitrap detection (60,000 resolution).
Mass spectrometry data analysis
RAW files for each sample were grouped and analyzed by MaxQuant (version 1.5.2.8). 16 MS2 spectra were searched against the UniProt complete human database (20,159 sequences; downloaded on June 23, 2015; http://www.uniprot.org) using the Andromeda search engine. Search parameters included up to two missed tryptic cleavages; 20 ppm match tolerance for the orbitrap and 0.5 match tolerance for the ion trap; and variable modifications for deamidation (N and Q residues), oxidation (M residues), and TMT (K residues and peptide N-termini). A false discovery rate cutoff of 1% was used at the peptide and protein levels for positive identification. Quantitation information was extracted from MS3 scans with a reporter mass tolerance of 0.01 Da. Peptide quantitation data were used to calculate the overall protein signal intensity. Three groups were analyzed: Control, PC, and UP, with each group represented by biological triplicates. Data were accepted only if there were reporter ion intensity values for each sample, except in the case where there were three nulls in one group compared to three measurements in the comparative group. Reporter ion intensity data were averaged across the three samples for each group, and then ratios were formulated for PC/Control and UP/Control. Proteins were ranked based on these ratios from lowest to highest, corresponding to decreased or increased secretion following treatment, respectively. The lowest 10 percentile list of proteins for each cell culture type and each treatment group were analyzed for inclusion in protein: protein interaction networks using the Reactome FI plugin 4 for Cytoscape (version 3.3) and biological function enrichment using Gene Ontology (GO) mapping (http://geneontology.org).
Enzume-linked immunosorbent assays
Enzume-linked immunosorbent assay (ELISA) kits for matrix metallopeptidase 7 (MMP-7), MMP-9, and TGF-β 1 were purchased from RayBiotech. Conditioned cultured media were removed every 2 days from EpiAirwayFT cultures, aliquoted, and frozen at −80°C. Then, 50 μl of MMP-9, 200 μl of MMP-7, or acidified 200 μl of TGF-β1 of thawed conditioned media were incubated on 96-well ELISA plates in duplicate. Methodology was performed as described by the manufacturer, and readings were made with an absorbance plate reader at OD 450 nm. Concentration of protein in the conditioned media was estimated by comparing test OD 450 nm against a standard curve, and averaged for triplicate biological samples.
Immunocytochemistry
At the treatment end point, EpiAirwayFT cultures were washed with PBS and separated from their plastic tissue culture support inserts with a scalpel. Samples were fixed with 4% paraformaldehyde in PBS, blocked with bovine serum albumin (BSA), and stained with anti-collagen III antibody (Abcam; ab7778), followed by Alexa Fluor 488 goat anti-rabbit immunoglobulin G (Invitrogen, a brand of Thermo Fisher Scientific, Waltham, Massachusetts, USA); or costained with anti-E cadherin antibody-Alexa Fluor 568 (ab206879) + anti-fibronectin antibody-Alexa Fluor 647 (ab198934) + anti-vimentin antibody-Alexa Fluor 647 (ab195877) conjugates (Abcam). Samples were mounted with medium containing DAPI and visualized with a Zeiss LSM 780 (Oberkochen, Germany) confocal microscope and a Plan-Apochromat 63×/1.40 oil objective. Cultures were imaged in 3-D with Z-stack layering, and data were analyzed with ZEN software (version 2.3; Carl Zeiss Microscopy). Laser strength, detection wavelengths, exposure time, and digital gains were kept constant across all experiments for Alexa Fluors 488, 568 and 647.
Results
Toxicity of PC sized PM10 or less
In order to ascertain the potential effects of airborne PC on human lung cultures, a sample of green PC was ground to a size of 10 μm or less. An urban PM10 standard from NIST was also included in these experiments. The size distributions were similar but broader for filtered urban particulates, with a median particle size of 2.58 μm for PC and 5.85 μm for the NIST standard (Figure 1(a)). Cell viability was tested in A549 cell cultures in exponential growth phase using the Alamar Blue assay up to PM concentrations of 200 μg/ml. PC did not significantly alter cell growth or death over a 16-h time period at any concentration tested (Figure 1(b)). Exposure to UP up to 100 μg/ml did not affect the viability of cells, but 200 μg/ml treatment reduced viability by about a third compared to mock-treated cells. Therefore, 100 μg/ml was chosen as a nontoxic treatment for evaluating cellular responses to PC and UP exposure.

Assessment of PC-induced toxicity in A549 cells. (a) A sample of PC was ground and sieved to a particle size of 10 µm or less. Size characteristics of a control urban PM10 (UP) from NIST is also shown. (b) Cell viability was measured using the Alamar Blue™ assay. Cells in logarithmic growth were treated in quadruplicate for 16 h with the indicated concentrations of PC or UP. The rate of Alamar Blue reduction was then assessed over the next 3 h. Error bars represent standard error for 3. separate experiments PC: petroleum coke; PM: particulate matter; UP: urban particulate matter; NIST: National Institutes of Standards and Technology.
Toxicity in primary cultures was determined by an enzymatic assay that measures the release of LDH into the culture medium. EpiAirwayFT cultures were exposed to 100 μg/ml of PC or UP at the air–liquid interface (Figure 2(a)) for 3 h and then assessed over 12 days posttreatment. LDH release varied in treated cultures between 0.58% and 2.86% of maximum levels and was not statistically different compared to mock-treated cultures (Online supplemental Table S1) indicating low toxicity. Although PC samples were ground to a slightly finer size compared to UP particles (Figure 1(a)), PC aggregated to a greater extent in solution (compare the size of black particles in Figure 2(b) with (c)).

PC-induced toxicity in a 3-D human lung model. (a) Cross-sectional diagram of the EpiAirwayFT tissue model. Cultures are fed through the lower microporous membrane, and particle exposure occurred at the upper air–liquid interface. (b, c) Phase contrast images of EpiAirwayFT cultures after exposure to either PC (b) or UP (c). The plane of the image was captured at the air–liquid interface after 12 days exposure and represent areas of greater particulate matter density. Red bars at the bottom left corners represent 10 μm distance. PC: petroleum coke; 3-D: three-dimensional; UP: urban particulate matter.
Secretome analysis after exposure of immortalized or primary human lung cultures with petroleum coke or standard urban PM10
In order to identify candidate biomarkers for exposure to airborne PC, A549 cells were exposed to either vehicle, PC, or UP. After 16-h treatment, the medium was carefully removed and analyzed for relative abundance of the secreted proteins. Proteins were digested with trypsin and labeled with a unique TMT isobaric tag. Samples were then combined and simultaneously analyzed by liquid chromatography–mass spectrometry (LC/MS). The advantage of this approach compared to label-free quantitative approaches is that it negates the variability that occurs between sequential chromatographic runs. Furthermore, peptide identification was made with MS2 scans, whereas quantitation was performed in MS3 which greatly enhances accuracy. 17 Results showed that a total of 3373 proteins were identified in the medium, 2795 of which contained quantitative information for every label (Table 1). Only 367 proteins showed ≥50% increase in abundance after PM treatment, whereas 1585 proteins had ≤50% in relative levels. In order to compare an immortalized 2-D model of human lung epithelial cells with a 3-D model of primary human lung cells, we replicated this experiment with EpiAirway cultures. In contrast to A549 cells, the primary cultures were quiescent and produced a layer of mucus on the apical surface. Results from these experiments showed a similar trend as A549 cells but in decreased levels. That is, there were 850 less proteins identified in the secretomes of EpiAirway cultures with quantitative information for every label, and the number of proteins with greater or less than 50% average signal intensity compared with the control cells were 9 and 76, respectively (Table 1). Full protein identification lists can be found in the supplementary data (Online supplemental Tables S2 and S3).
Statistics for protein mass spectroscopy identification and relative quantitation by isobaric labels.

Gene ontology term pathway enrichment analysis for proteins with decreased secretion after PM exposure
As it was clear that inflammatory or other secretory biomarkers were not induced to a greater extent after PM exposure in our model systems, we then utilized systems biology approaches to determine whether there were global changes in secretory proteins that could be compared to a physiological outcome. The goal of this approach is not to identify a handful of proteins that change in response to toxicant with absolute precision but rather to begin to decipher how the emergent properties of systemic changes in the secretome could affect health and disease. To this end, we chose an arbitrary cutoff of the lowest 10 percentile of proteins when the relative secretion of PM treatment versus control is compared, that is, proteins with a net decrease in secretion after exposure. Characteristics of the set of proteins generated by this filtering strategy are presented in Table 2. Note that reductions in protein secretion were more pronounced in A549 cell cultures, which possibly reflects the greater growth rate of these cells.
Characteristics for protein sets used for pathway enrichment analyses.

PC: petroleum coke; UP: urban particulate matter.
aEach set represents the lowest 10 percentile of proteins secreted in treatment versus control (i.e. decreased secretion).
bRatio represents the (average of treated protein signal intensities/average control protein signal intensities).
Next, the protein sets were analyzed for gene ontology (GO) enrichment. The GO database groups proteins according to their cellular component, biological process, or molecular function. We analyzed for pathway enrichment using the biological process classification, which is distinguished from the molecular function classification as having more than one distinct step.
Results from this analysis showed a clear distinction between A549 and EpiAirway cultures (Figure 3). Notably, significant association for “response to chemical,” “regulation of cell migration,” and “epithelial cell differentiation” GO terms were found only in EpiAirway secretomes in response to either PC or UP, whereas processes related to gene expression were significantly associated exclusively for A549 secretomes. The only biological process that was significant only for PC in both A549 and EpiAirway exposed cultures was “extracellular matrix organization.”

GO enrichment analysis of secretory proteins in PC and urban PM10 (UP) exposed cell cultures. Protein group lists comprising the lowest 10 percentile of secreted proteins were compared against the Biological Process classification of GO terms. p-Values were calculated as a hypergeometric distribution, which estimates the probability of finding x number of proteins within a user list out of a total of y number of proteins in a particular GO term annotation list. GO term groups are not mutually exclusive. Values in brackets represent the term accession number. *p ≤ 0.05. GO: Gene Ontology; PC: petroleum coke; PM: particulate matter; UP: urban particulate matter
Protein–protein interaction modeling of proteins with decreased secretion after PM exposure
Another way to determine pathway enrichment is by visualizing protein–protein interaction networks. Figure 4 shows this type of modeling with the protein sets generated from cell cultures exposed to PC as described above. Proteins are depicted as nodes, and known interactions between proteins as edges. Nodes are color coded according to known and predominant biological functions. PC elicited different responses in A549 and EpiAirway cultures. Proteins associated with RNA processing (yellow nodes) were highly represented with A549 cultures (Figure 4(a)). Also of significance were proteins involved in transcriptional regulation (with a key node centered on RELA/p65) and extracellular remodeling (key node around FN1/fibronectin). In contrast, extracellular remodeling was the most prominent biological function when the reactome analysis was applied to EpiAirway (Figure 4(b)), and proteins associated with immune response were highly represented as well (red and green nodes, respectively). Secretion of proteins associated with gene expression were not affected by PC treatment to a greater extent in the primary cultures.
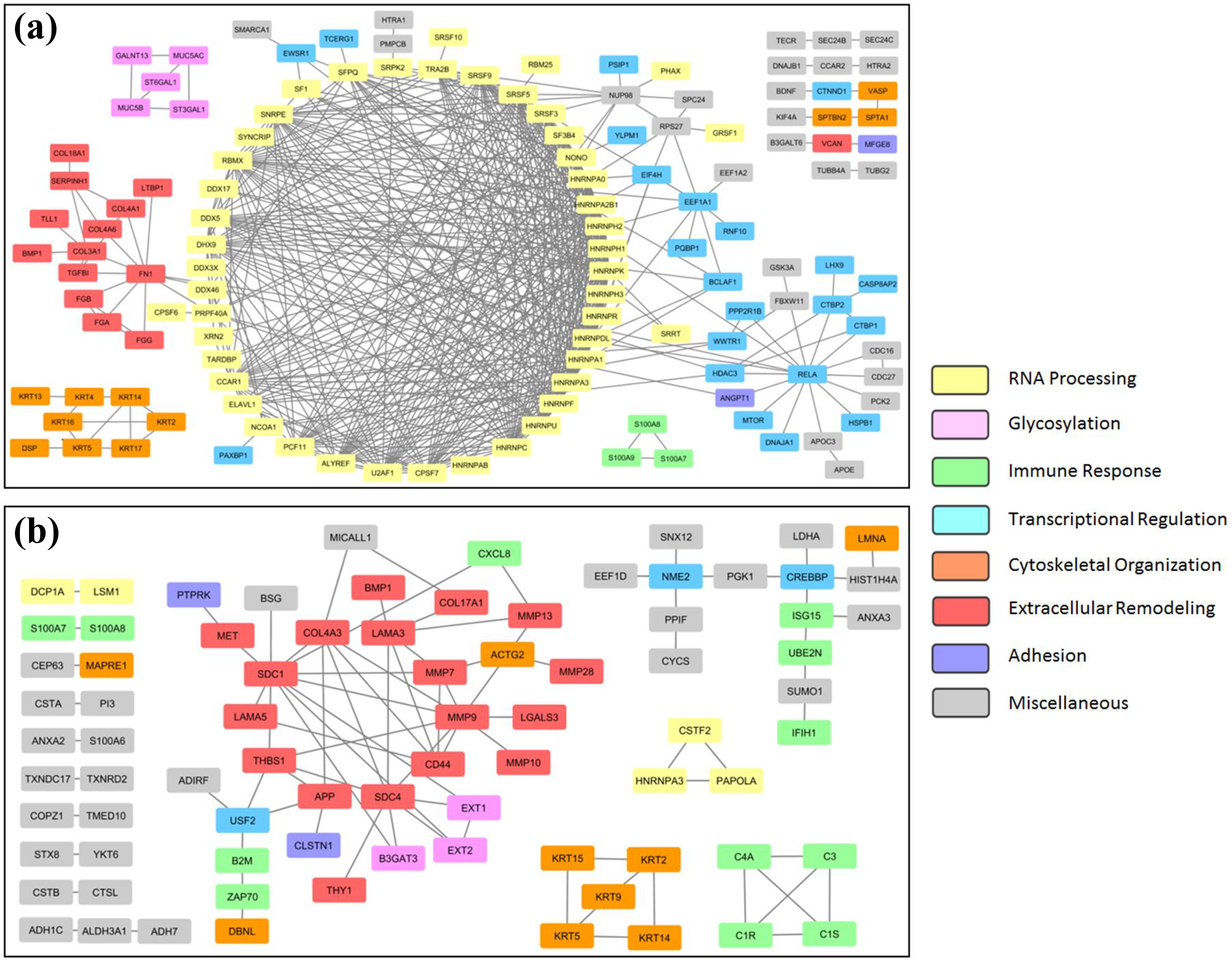
Secretory protein interaction networks of cell cultures exposed to PC. Proteins with decreased representation in the secretomes of PC-exposed A549 (a) and EpiAirway (b) cell cultures are visualized as nodes and connections with their interacting partners as edges. Nodes are color coded according to their known biological functions. PC: petroleum coke.
Although extracellular remodeling was a significant feature of both A549 and EpiAirway protein–protein interaction analyses, breaking this classification down further by mapping the protein sets to reactome pathways reveals differences between these cell models (Table 3). Proteins involved in degrading the ECM, including CD44, matrix MMPs, and laminins, were highly enriched in the data set of EpiAirway cultures treated with PC, and to a lesser extent UP, whereas the A549 data set was significantly enriched in proteins that synthesize or modify collagen after either PC or UP exposure. For A549 cells, messenger RNA (mRNA) splicing was the most significant subtype of RNA processing. In addition, after PC exposure, chemokines and their receptors were significantly associated with EpiAirway and O-linked glycosylation of mucins with A549 (Table 3). Proteins with at least 25% increase in secretion following PC or UP exposure in A549 or EpiAirway cultures did not show significant correlation with GO terms containing the phrase “extracellular matrix” (data not shown). Therefore, these data suggest that PC decreases the secretion of proteins associated with ECM organization and remodeling in human lung cultures, but there are cell model-dependent differences in the types of ECM proteins which are affected.
Protein–protein interaction mapping for proteins with lowered secretion in A549 and EpiAirway cell cultures.a

PC: petroleum coke; UP: urban particulate matter; ECM: extracellular matrix.
aNB: color-coding in first column corresponds to that of Figure 4.
bNumber of proteins in the reactome pathway.
cNumber of hits within the PC or UP groups of secreted proteins that fall within the lowest 10 percentile range of treated versus control.
d p < 0.005.
e p < 0.05.
The ECM is a complex environment involved in structural support and regulation of intercellular communication. To determine how PC might alter the ECM of primary human lung cultures, proteins with decreased secretion after treatment were mapped to reactome pathways (Figure 5). Proteins associated with “extracellular matrix organization” had the highest probability of any pathway. Visualization of the “degradation of the extracellular matrix” network shows potential impairment of most of the subpathways (hits are highlighted in purple). This includes degradation of aggrecan, brevican, decorin, E-cadherin, elastin, fibrillin, fibronectin, laminin-332, laminin-511, NID1, osteopontin and perlecan and binding of MMP to CD44, CD147, and α2-macroglobulin.

Pathway enrichment analysis of primary human lung cell secretory proteins with decreased abundance in response to PC. “Extracellular Matrix Organization” was the most highly significant pathway in this reactome analysis (FDR < 5 × 10−4) of EpiAirway cells, with a majority of the protein hits within the “Degradation of the Extracellular Matrix” subgroup (depicted here). Positive hits are highlighted in purple, and non-hits include proteins (green boxes) and protein complexes (blue boxes). PC: petroleum coke; FDR: false discovery rate.
Validation of changes in abundance of secreted MMP-9
Advantages of proteomic quantitation include non-biased protein selection, accuracy, and a large number of proteins that can be analyzed in a single experiment. Other methodologies with greater sensitivity and precision can then be utilized on individual proteins-of-interest to confirm important findings. To this end, we have determined changes in protein secretion in EpiAirwayFT cultures after PC or UP exposure using ELISA. EpiAirwayFT are different from EpiAirway cultures in that they contain an additional sublayer of stromal fibroblasts (Figure 2(a)). Because our initial findings suggested that the ECM is affected by particulate exposure, and that fibrosis is associated with an accumulation of ECM proteins, we surmised that utilizing a tissue model that includes fibroblasts would be best suited for further study. Fibroblasts are a central component of fibrosis and have important roles in wound healing and tissue repair in organs with prominent epithelial derivation such as lung, skin, liver, and kidney. 18 Following epithelial insult, fibroblasts migrate to the provisional wound matrix and deposit additional ECM components such as collagen, resulting in fibroblast-enriched granulation tissue.
MMP-9 is associated with degradation of the ECM including type I and III collagen proteins 19 and LC/MS-based secretome analysis indicated its decreased abundance following PC and UP treatment in EpiAirway cultures (Online supplemental Table S3). Monitoring of secretion by ELISA showed decrease of MMP-9 following PC and UP treatment in EpiAirwayFT cultures (Figure 6). Relative to mock-treated cells, MMP-9 levels in the conditioned media were lower by 4 days postexposure and remained suppressed for the duration of the experiment. In addition, MMP-7 (Matrilysin) abundance was analyzed, but a component of the proprietary media preparation for these cultures was found to inhibit the assay; and TGF-β1 protein was undetected in the conditioned media by this methodology (JC, unpublished data).

Detection of MMP9 secretion by ELISA. EpiAirwayFT primary human lung cultures were exposed on the apical surface to 100 μg/ml of PC or UP for 3 h, then secretion of MMP9 into the conditioned media was monitored every 2 days for a total of 12 days. Data represent the average of 3 samples, and abundance of test samples are shown relative to mock-treated samples (PBS only). MMP: matrix metalloproteinases; PC: petroleum coke; UP: urban particulate matter; PBS: phosphate-buffered saline; ELISA: enzyme-linked immunosorbent assay.
Collagen III deposition following particulate exposure
Following a toxic insult, fibroblasts are thought to deposit large quantities of matrix proteins on the affected site, predominantly type I and III collagens, which increase the tensile strength of the wound. Inability to clear excessive collagen deposition may lead to irreversible pulmonary fibrotic scarring. 20 –22 Collagen III accumulation was visualized by immunocytochemistry as a marker for changes associated with fibrosis. Compared to mock-treated cultures, collagen III abundance was greatly enhance in both PC- and UP-treated cultures, suggesting that this outcome is not unique to PC but PM10 particulates in general (Figure 7). Accumulation was limited to the epithelial layer, and in some instances, more heavily concentrated at the air–liquid interface. As a control for fibrosis-like morphology, cultures were exposed to TGF-β1. These cultures also demonstrated enhanced collagen III deposition in the epithelial layer (Figure 7).

3-D scanning of collagen III expression. EpiAirwayFT cultures were not treated (NT, PBS only) or exposed to 100 μg/ml of PC or UP for 3 h, or 10 ng/ml of TGF-β1 for 12 days. Cultures were harvested after 12 days, fixed, and stained with reagents to detect collagen III (green) or nuclei (blue). Two different samples are depicted for each treatment group: a and c from the first culture, b and d from the second culture. a and b represent virtual reconstructed cross-sections of the 3-D cultures. Arrows indicate locations of the confocal slices at the upper air-liquid interface that are shown for c and d, with images captured just below the coverslip (135 μm ×x 135 μm). Yellow bars represent a distance of 10 μm. 3-D: three-dimensional; PC: petroleum coke; UP: urban particulate matter; PBS: phosphate-buffered saline; TGF: transforming growth factor.
Particulate exposure effect on epithelial–mesenchymal transition markers
As noted earlier, GO analysis of secretory proteins with decreased abundance showed that the “epithelial cell differentiation” classification was indicated for EpiAirway cultures exposed to either PC or UP. This leaves open the possibility that epithelia could become de-differentiated in the presence of particulate exposure. Although migration and proliferation of stromal fibroblast and recruitment of bone marrow-derived fibrogenic cells account for a large portion of ECM-producing fibroblasts/myofibroblasts found within wound sites, there is some evidence in the literature that epithelial cells may have a role in this process. 23,24 In this model, epithelial cells would transition to a mesenchymal phenotype, lose intercellular adhesion, migrate into the interstitium, and contribute to the synthesis of the fibrotic matrix. 20,25
Hallmarks of epithelial–mesenchymal transition (EMT) include repression of the epithelial adhesion molecule E-cadherin and increase in expression of mesenchymal markers such as vimentin and fibronectin. 26,27 EMT transitioning was assessed by a qualitative and semiquantitative approach using confocal microscopy (Online supplemental Figure S4 and Figure 8). EpiAirwayFT cultures were mock treated or exposed to PC, UP, or TGF-β1. At 12 days postexposure, cultures were fixed and stained with reagents to visualize E-cadherin (red), vimentin (green), fibronectin (pink), and nuclei (blue). All samples were stained simultaneously, and imaging parameters were kept constant for the EMT markers. Epithelial cadherin, or E-cadherin, is a transmembrane protein involved in cell–cell adhesion. E-cadherin expression was only observed in the epithelial layer and treatment led to increased expression, primarily in UP and TG cultures (UP = TG > PC > NT). In contrast, vimentin and fibronectin staining clearly showed a fibrillar pattern (Online supplemental Figure S4). All 3 treatments led to loss of vimentin and fibronectin expression in the epithelial layer (Figure 8). Vimentin expression was not evident in untreated fibroblasts, but treatment led to increased expression just above the membrane support (UP > PC > TG). Fibronectin expression was present throughout the fibroblast layer in untreated cultures, and its expression increased with UP treatment and decreased after exposure to PC or TG.

Primary human lung cultures stained with EMT biomarkers. EpiAirwayFT cultures were not treated (NT, PBS only) or exposed to 100 μg/ml of PC or UP for 3 h or 10 ng/ml of TGF-β1 for 12 days. Cultures were harvested after 12 days, fixed, and stained with reagents to detect E-cadherin (red), fibronectin (pink), vimentin (green), and nuclei (blue). Layered confocal images were incorporated into a 3-D rendering, and virtual cross-sections from two samples are depicted for each treatment group. Vertical red and blue bars represent the approximate locations of the epithelial and stromal fibroblast layers, respectively. Yellow bars represent 10 μm. EMT: epithelial-mesenchymal transition; PC: petroleum coke; UP: urban particulate matter; 3-D: three-dimensional; PBS: phosphate-buffered saline.
Discussion
The pulmonary alveolar epithelium is the final barrier for inhaled particles. This interface, together with sentinel macrophages, controls pulmonary homeostasis by mounting a response to environmental insults, which can lead to cell death, impair normal re-epithelialization, or alter EMT leading to fibroproliferation. 18 Proteomic and pathway analyses were utilized here to investigate the secretory response of human lung epithelial cell cultures to petroleum coke PM, with the goal of identifying systemic cellular responses in an unbiased fashion. Two cell culture models were included in these studies: A549, an adenocarcinomic human alveolar basal epithelial cell line, and EpiAirway, primary cultures of human tracheal/bronchial epithelial cells. In addition to ground PC, a standard PM collected from urban air (UP) was included as a reference. We observed a consistent decrease in secretory response when either cell model was exposed to PC or UP relative to mock-treated cultures. In comparing the two models, secretomes of PM-exposed EpiAirway cultures showed a decreased number of protein changes than was observed with A549 cells. These primary cultures have also been reported to be toxicologically more resistant to ozone and a photochemically aged Synthetic Urban Mixture (SynUrb54) relative to A549 cells. 28 This is likely due to the different growth characteristics of these cultures because A549 are exposed while in exponential growth phase, whereas EpiAirway cultures are highly differentiated and largely quiescent.
A549 is a common model to study exposure effects of various types of PM. 29 The advantage of using a cell line is low cost and ease of use for an unlimited supply of isogenic cells, which facilitate the ability to perform replicative experiments. Pathway analyses for A549 secretomes showed greatest changes for proteins involved in RNA processing, including mRNA splicing, as well as those involved in gene expression. We have previously found these pathways to be altered during tumorigenic progression by quantitative phosphoproteomic analysis (JC, unpublished results), but this was an unexpected finding for a secretome study. Proteins involved in extracellular organization were also significantly enriched within the set of proteins showing decreased secretion, as well as enzymes which catalyze O-linked glycosylation of mucins.
EpiAirway secretome analysis showed marked differences relative to A549 cultures after PC PM exposure. Abundance of proteins involved in RNA processing and regulation of gene expression were not altered to a significant extent in EpiAirway cultures. Significant associations were found for proteins involved in immune response, including pro-inflammatory cytokines such as interleukin 8 (CXCL8), which were not activated in response to PC but showed a decrease in levels. Other GO pathways significantly enriched only in EpiAirway cultures were “response to chemical,” “regulation of cell migration,” and “epithelial cell differentiation.” As observed with A549 cultures, proteins associated with ECM organization were decreased in response to PC. However, clear differences emerged when the ECM proteins were analyzed in more detail. For A549 cultures, ECM proteins included fibronectin and several fibrinogen and collagen family members, whereas EpiAirway ECM proteins included MMP enzymes and laminins. As a group, the proteins less abundant in A549 secretomes after PC exposure were associated with biosynthesis and modification of collagen, whereas for EpiAirway, the proteins were involved in degradation of the ECM.
UP particles were more toxic to A549 cells in cell viability assays and elicited a stronger response in proteins involved in RNA processing and gene regulation. A different trend was observed for EpiAirway cultures, however, as PC exposure led to greater changes in the number of proteins involved in immune response and ECM regulation relative to UP. Moreover, in our analysis we found that PC caused significant changes in the pool of proteins associated with ECM regulation in both A549 and EpiAirway cultures, suggesting the potential importance of this pathway to inhalation exposure of human lung epithelia to PC. These data indicate PC has specific effects on cell systems and that toxicant-specific fingerprints may emerge from proteomic profiling of secretory proteins.
The ECM is composed of multiple proteins that provide structure to tissues and regulate information transfer for cellular functions including proliferation, migration, EMT, and apoptosis. It facilitates the production and movement of various secreted proteins and acts as a reservoir for growth factors and cytokines that, during tissue injury and repair, help to localize and direct the wound healing response. 30 In the human lung, ECM structure derives mainly from collagens, elastins, and laminins. Other proteins are involved in maintaining cell polarity and survival (e.g. fibronectin and glycosaminoglycans), cell–ECM adhesion (e.g. integrins and CD44), and cell–cell adhesion (e.g. ICAMs and cadherins). The balance of secreted proteases and antiproteases in the ECM is tightly regulated and has an impact on tissue remodeling and lung disease, with excessive degradation leading to emphysema, and a relative paucity of degradation to accumulation of ECM and fibrosis. 31
Fibrosis results from chronic tissue injury and can be defined as excessive deposition of ECM components that results in irreversible destruction of the normal tissue architecture which can ultimately lead to organ failure. Mutsaers et al. suggested that there are three components that are intrinsic to the fibrotic response: (i) a chronic stimulus, (ii) excessive synthesis of collagen and other ECM proteins, and (iii) inability to remove the excess ECM components due to downregulation of degradative enzymes which could clear scar tissue. 22 Known stimuli includes airborne environmental agents such as asbestos, silica, and carbon. 32,33 Aberrant collagen deposition is thought to derive predominantly from activated fibroblasts and myofibroblasts. 34 There is some uncertainty as to the origin of these cells, but they might derive from postembryonic lung fibroblasts, bone marrow-derived fibrocytes, or might result from an EMT event. Hardie et al. 18 (and references therein) has suggested that the inflammatory response may have a limited role in the induction of fibrosis because overexpression of various chemokines led to an increase in bronchoalveolar lavage inflammatory cells and tissue pneumonitis, but this was not accompanied by lung fibrosis or tissue remodeling. Results from our model of PC and UP exposure are consistent with research showing that particulate exposure over an extended period of time leads to lung fibrosis in vivo. In accordance with fibrotic mechanisms described above, our data show that fine PC, a type a black carbon, causes a decrease in the release of degradative ECM enzymes (i.e. this could lead to ECM accumulation) and proteins associated with epithelial differentiation (i.e. this could facilitate EMT). Other studies have shown that a decrease in MMP enzymes is associated with a fibrotic phenotype. For example, lungs of MMP12−/− and MMP13−/− mice had increased fibrosis as measured by collagen deposition in models of idiopathic pneumonia syndrome 35 and idiopathic pulmonary fibrosis, 36 respectively, compared to wild-type mice. Simm et al. found that prolonged treatment of primary rat fibroblasts to fly ash particulates led to increased collagen I and decreased MMP-9 mRNA levels. 37
Based on these initial findings, we tested whether long-term PC exposure could lead to a fibrotic-like phenotype in EpiAirwayFT primary human lung cultures, which are similar to EpiAirway but have an additional sublayer of stromal fibroblasts. In this model, PC and UP treatment led to decreased MMP9 secretion relative to untreated cells by 4 days and remained suppressed for the duration of the study (12 days). Furthermore, collagen III accumulation was observed in the vicinity of the air–liquid interface, the primary contact of particulate exposure. TGF-β is an extensively studied regulator of the ECM and has been linked to fibrosis of the lung as well as other tissues. EpiAirwayFT cultures were exposed to TGFβ1 as a control for fibrosis, and this led to a greater depth of the fibroblast layer (Online supplemental Figure S4) and increased collagen III production within the epithelial layer (Figure 7). Secretory TGF-β1 was not detected by either LC/MS or ELISA assays after exposure to PC or UP. However, circulating monocytes and tissue macrophages are thought to be the principal sources of TGF-β1 production during a fibrotic response. 20,38
Acute proteomic profiling of PC exposure of EpiAirway cultures indicated that some proteins associated with epithelial differentiation are secreted to a lesser extent compared to untreated cultures. These data suggested the possibility that particulate exposure could lead to dedifferentiation of epithelial cells. To test for EMT, long-term exposed EpiAirwayFT cultures were stained for expression of E-cadherin (epithelial levels decrease with EMT) as well as fibronectin and vimentin (epithelial levels increase with EMT). PC and UP produced unique staining profiles within the epithelial and fibroblast layers, but they were not consistent with classical EMT. For instance, E-cadherin expression increased significantly in the epithelial layer, whereas vimentin expression was found to be decreased. Dedifferentiated epithelial cells have been identified as important precursors of fibroblasts in experimental kidney and liver models of fibrosis 39,40 ; however, the role of EMT in lung fibrosis is more controversial. 41,42 TGF-β1 did not elicit an EMT phenotype (Figure 8), but TGF-β signaling is not sufficient to initiate an EMT program in the absence of a wound response. 25
In summary, we have characterized and quantified secretomes of immortalized and primary human lung cell cultures with and without acute exposure to PC or urban PM to a depth of about 2500 proteins. Pathway analyses suggest possible cellular mechanisms that could underlie fibrotic pathology that often accompanies particulate exposures, including PC. Because pathway modeling after acute exposure predicted that regulators of the ECM might be impaired, primary human lung cocultures consisting of epithelia and stromal fibroblasts were chronically exposed to PC for up to 12 days. Results showed an increase in accumulation of collagen III within the epithelial layer as well as the air–liquid interface, which is consistent with fibrotic morphology, and a decrease in MMP-9 secretion. PM exposure led to changes in intensity and/or localization of EMT markers, but epithelial reprogramming was not found to be in effect in this model. These studies suggest that primary human tissue models paired with systems biology approaches can detect early cellular changes that could form a mechanistic basis for predictions of adverse outcomes.
Supplemental Material
Supplemental Material, S1_Table - Petroleum coke exposure leads to altered secretome profiles in human lung models
Supplemental Material, S1_Table for Petroleum coke exposure leads to altered secretome profiles in human lung models by JA Caruso and PM Stemmer in Human & Experimental Toxicology
Supplemental Material
Supplemental Material, S2_Table - Petroleum coke exposure leads to altered secretome profiles in human lung models
Supplemental Material, S2_Table for Petroleum coke exposure leads to altered secretome profiles in human lung models by JA Caruso and PM Stemmer in Human & Experimental Toxicology
Supplemental Material
Supplemental Material, S3_Table - Petroleum coke exposure leads to altered secretome profiles in human lung models
Supplemental Material, S3_Table for Petroleum coke exposure leads to altered secretome profiles in human lung models by JA Caruso and PM Stemmer in Human & Experimental Toxicology
Supplemental Material
Supplemental Material, S4_Figure - Petroleum coke exposure leads to altered secretome profiles in human lung models
Supplemental Material, S4_Figure for Petroleum coke exposure leads to altered secretome profiles in human lung models by JA Caruso and PM Stemmer in Human & Experimental Toxicology
Footnotes
Acknowledgments
The authors thank Nicholas Schroeck and Shawn McElmurry for obtaining and processing the petroleum coke sample used for these studies and also thank Mary Gargano for assistance with cell cultures.
Availability of data and materials
The mass spectrometry proteomics data have been deposited to the ProteomeXchange Consortium via the PRIDE partner repository with the dataset identifier PXD005194.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was supported by a pilot grant from the Center for Urban Responses to Environmental Stressors (CURES), which is supported by P30 ES020957. The Wayne State University Proteomics Core, and this work, are supported through the NIH Center Grant P30 ES 020957, the NIH Cancer Center Support Grant P30 CA 022453, and the NIH Shared Instrumentation Grant S10 OD 010700. The microscopy, imaging, and cytometry resources core were supported, in part, by NIH Center grant P30 CA022453 to the Karmanos Cancer Institute at Wayne State University, and the Perinatology Research Branch of the National Institutes of Child Health and Development at Wayne State University.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
