Abstract
Given that particulate matter (PM) has an established role in inducing oxidative stress, inflammation, and skin aging, it is plausible that PM could exacerbate inflammatory skin conditions such as xerosis. Xerosis represents a significant dermatological concern among older adults within aging populations. We conducted an investigation into the efficacy of antioxidants, such as dieckol, punicalagin, epigallocatechin gallate (EGCG), and resveratrol, against PM10 in a skin barrier–disrupted mouse model. A skin barrier–disrupted mouse model was induced by tape stripping. This study investigated the antioxidative and anti-inflammatory properties of antioxidants on PM-induced changes using the skin barrier–disrupted mouse model. Tape strips were attached to the back of 7-week-old nude mice and removed quickly. To investigate variations in skin hydration, levels of inflammatory cytokines, and indicators of keratinocyte differentiation, mice underwent treatment with several compounds: a control vehicle (100 μL), PM10 100 μL (100 μg/mL), PM10 100 μL (100 μg/mL) with antioxidants 100 μL (Punicalagin 5 μM, Dieckol 5 μM, EGCG 1 μM, resveratol 1 μM) for 1 week. To assess their effects, different analysis were conducted using measurements of skin moisture, real-time polymerase chain reaction, enzyme-linked immunosorbent assay for detecting inflammatory cytokines, and immunofluorescence staining to identify markers of keratinocyte differentiation. While PM10 decreased water content in disrupted skin, all antioxidants preserved skin hydration in the skin barrier–disrupted mice, regardless of the presence of PM10. All antioxidants also inhibited the upregulation of inflammatory cytokines, such as interleukin (IL)-1β, IL-4, IL-6, IL-8, and tumor necrosis factor-alpha and normalized the downregulation of keratinocyte differentiation markers against PM10 in skin barrier–disrupted mice. This study elucidated the protective effects of antioxidants-namely, punicalagin, dieckol, EGCG, and resveratrol-in mitigating the impact of PM10 on skin barrier integrity and inflammation in a disrupted skin barrier mouse model, highlighting their potential utility in dermatological treatments.
Introduction
Since the 2000s, levels of environmental particulate matter (PM) have risen markedly, attributable to emissions from industrial factories, power generation facilities, vehicle exhaust, construction activities, and the dispersion of natural dust by wind.1,2 This increase has positioned PM as a significant public health concern. PM is classified by size into coarse (PM10: <10 μm), fine (PM2.5: <2.5 μm), and ultrafine (PM0.1: <0.1 μm) particles. While PM is known to damage the cardiovascular and respiratory systems, 3 it is also associated with various skin problems, such as atopic dermatitis, acne, and psoriasis.4,5 Jin et al. demonstrated that PM can infiltrate the intercellular space by traversing the stratum spinosum in mice with compromised skin barriers. 6 Furthermore, according to Kim et al., exposure to PM2.5 amplifies trans-epidermal water loss and augments the penetration of fluorescein isothiocyanate dextran in both organotypic skin and mouse models, thereby impairing skin barrier function. 4
The precise mechanism underlying inflammatory skin diseases induced by PM remains incompletely elucidated, with research ongoing to understand the roles of skin barrier disruption, inflammation, and oxidative stress as pivotal factors in the pathogenesis and etiology of diverse skin disorders.7,8 Xerosis, characterized by dryness of the skin or mucous membranes, commonly affects areas like the chins or conjunctiva. It manifests prominently in older individuals with compromised or impaired skin, heightened protease activity, and reduced natural moisturizing factor production, attributed to filaggrin proteolysis.9–11 With advancements in healthcare contributing to a rapidly expanding older demographics, the incidence of severe xerosis among older adults is on the rise, often considered a consequence of aging rather than a distinct pathological condition. Notably, xerosis is closely associated with pruritus, significantly impacting the overall quality of life for older individuals. 12 Consequently, proactive measures are essential to address the aging demographic and escalating levels of PM, with the use of antioxidant-rich moisturizers being paramount in effectively alleviating xerosis and associated pruritus.
Antioxidants are thought to exhibit preventive properties and alleviate oxidative stress associated with inflammatory skin conditions triggered by PM exposure.13–15 Dieckol is a type of natural polyphenol compound derived from marine algae. Dieckol exhibits a range of biological activities, including antioxidant, anti-inflammatory, anticancer, antiviral, and antidiabetic effects.16–18 Punicalagin, a natural polyphenol present in pomegranates (Punica granatum), is a type of ellagitannin extracted from pomegranates and is potent antioxidant and anti-inflammatory properties. It is believed to help reduce oxidative stress, combat free radicals, and potentially protect against certain chronic disease.19–21 Epigallocatechin gallate (EGCG) is a type of catechin, which is a natural phenol and antioxidant found primarily in green tea. EGCG is a potent bioactive compound with multiple health benefits, particularly due to its antioxidant, anti-inflammatory, anticancer, and cardiovascular protective properties.15,21 Resveratrol, naturally derived from polyphenolic compounds synthesized by plants in response to stressors like infection, also possesses antioxidant properties and inhibits the formation of reactive oxygen species (ROS) and the release of pro-inflammatory cytokines.22–24 Recent studies have affirmed the preventive effects of these antioxidants against PM-induced elevations in serum immunoglobulin E (IgE) levels, mast cell proliferation, inflammatory cytokine release, and alterations in keratinocyte differentiation markers in mouse models of atopic dermatitis induced by 2,4-dinitrochlorobenzene (DNCB). 25 Furthermore, investigations have been extended to the effects on cultured human sebocytes and/or outer root sheath (ORS) cells. 26 In this context, the authors examined the efficacy of various antioxidants against PM10 in mouse models with disrupted skin barriers.
Methods
Treatment with PM10, dieckol, punicalagin, EGCG, or resveratrol in skin barrier–disrupted mouse models
Six-week-old nude were procured from Orient Bio Inc. (Sungnam, Republic of Korea) and acclimated for 1 week prior to experimentation. All animal care and treatment procedures strictly adhered to established laboratory animal guidelines. This study complied with the ethical standards outlined in the ARRIVE guidelines and animal care and treatment protocols were in accordance with the guidelines of the use of Laboratory Animals. Animal experiments were approved by the Institutional Animal Care and Use Committee of the Kyungpook National University (IRB No. 2021-0068). To induce skin barrier disruption, the tape-stripping method was employed, following established protocols, using black D-squame tape (Biobridge, Seoul, Korea).27,28 The tape was cut into 1.5 cm 2 strips, applied to the mice’s back and swiftly removed. Following tape stripping, the dorsal skin of each experimental mouse was topically treated with one of the following: a control vehicle, 1X phosphate-buffered saline (PBS) (100 μL), PM10 (100 μL at a concentration of 100 μg/mL), or a combination of PM10 (100 μL at a concentration of 100 μg/mL) and antioxidants 100 μL (5 μM dieckol, 5 μM punicalagin, 1 μM EGCG, 1 μM resveratrol) for a duration of 1 week.
We applied PM10 and antioxidants to the 1.5 cm² area where tape was attached, and only the treated area was excised. The tissue was divided into three parts, each measuring 0.5 × 1.5 cm, for different experimental purposes, including ELISA, RNA extraction, and immunofluorescence (IF). The areas without tape attachment were excluded from the experiment. Five mice were used for each group.
PM10 (resembling the European reference material ERM-CZ120) was procured from Sigma-Aldrich (St. Louis, MO, USA). PM10 acts as a carrier for harmful substances such as transitional metals, endotoxins, organic chemicals, and ultrafine particles. PM10 was diluted in PBS and applied to the xerosis mouse model. CO2 inhalation was employed for mouse euthanasia, and dorsal skin samples were subsequently collected.
Measurement of water content
The water content of the skin was measured using a device (JASKEY, Skin Analyzer, Korea) placed on the dorsal mouse skin after 1 week of treatment with either the control vehicle or the specified PM10 and antioxidant formulations.
Quantitative real-time polymerase chain reaction (qRT-PCR)
Total RNA was isolated using TRIzol (Invitrogen, MA, USA) reagent after lysing mouse dorsal skin samples in a TissueLyser II (Qiagen, Germany). ImProm-II™ Reverse Transcriptase and oligo (dT) primers (Promega, Madison, WI, USA) were used for synthesizing cDNA at 72°C for 10 min and 42°C for 90 min. Sequences for PCR primers are summarized in Table 1. Real-time PCR was performed on a StepOnePlus™ Real-Time PCR System (Applied Biosystems, Foster City, CA, USA) using 50 ng of cDNA, 10 pM primers, and SYBR premix (Applied Biosystems). The cycling conditions for amplification were as follows: 95°C for 10 min, followed by 40 cycles at 95°C for 15 s and 60°C for 60 s. The StepOnePlus™ Real-Time PCR analysis software (Applied Biosystems) was used for calculating and quantifying the PCR products.
Real-time PCR primers used in this study.

Enzyme-linked immunosorbent assay (ELISA)
To evaluate the protein expression levels of interleukin (IL)-1β, IL-4, IL-6, IL-8, and tumor necrosis factor-alpha (TNF-α) in mouse tissues, ELISA kits for IL-1β, IL-4, IL-6, IL-8, and TNF-α (R&D Systems, Minneapolis, MN, USA) were used. To measure the protein levels, tissues from the dorsal mouse skin treated with PM10 and antioxidants were placed in a tube containing lysis buffer (100 mM Tris, 150 mM NaCl, 1 mM EGTA, 1 mM EDTA, 1% Triton X-100, 0.5% sodium deoxycholate, pH 7.4) containing phosphatase/protease inhibitor cocktail (Roche, Mannheim, Germany). TissueLyser II (Qiagen, Hiden, Germany) and Stainless Steel Beads (Qiagen) were used to homogenize these tissues. For protein quantification, 10 µL of the sample lysates or BSA standards at various concentrations were added to 200 µL of 1X Bradford solution (Bio-Rad, Hercules, CA). The protein concentration was measured at 595 nm. After protein quantification with the lysates, following the manufacturer’s instructions. Protein concentrations of IL-1β, IL-4, IL-6, IL-8, and TNF-α were measured using specific ELISA kits according to manufacturers’ protocols. In brief, equal amounts of lysate were added to each well of a plate coated with a specific antibody. Next, an HRP-conjugated antibody that binds to the target protein was added followed by a substrate treatment, and a stop solution addition to halt the reaction when color development occurs. The absorbance was measured at 450 nm to confirm the expression of the protein with a spectrophotometer (VersaMax; Molecular Devices, CA, USA).
Immunofluorescence staining
The excised mouse tissues from the dorsal skin treated with PM10 and antioxidants were embedded in a frozen section compound, OCT compound (Tissue-Tek; Sakura Finetek, Torrance, CA, USA), in a freezer at −80°C. Tissues were cut to a uniform thickness of 8 μm using a cryostat (Leica CM3050 S; Leica, Heidelberg, Germany) and fixed in 4% paraformaldehyde (Sigma-Aldrich) for 10 min. Following the blocking step with 5% normal donkey serum (Abcam, Cambridge, UK) for 1 h, the primary antibodies (loricrin, filaggrin, and involucrin, all from Abcam) were incubated overnight at 4°C, each at a dilution of 1:100. Subsequently, after rinsing three times with phosphate-buffered saline, visualization of expression was achieved using Alexa Fluor 488–labeled donkey anti-rabbit secondary antibody (Molecular Probes, Eugene, OR, USA), followed by counterstaining with DAPI (4′,6-diamidino-2-phenylindole) for 10 min.
Statistical analysis
Data were evaluated as mean ± standard deviation. Triplicate experiments were conducted. ANOVA (One-way ANOVA) was used for statistical analysis, and *P < 0.05, **P < 0.001, ***P < 0.005, ****P < 0.0001 were considered statistically significant.
Results
Antioxidants reversed PM10-induced reduction of skin hydration in skin barrier-disrupted mice
In skin barrier–disrupted mice, the depletion of corneocytes, which serve to inhibit water loss, led to a decrease in skin water content. Our study sought to determine the efficacy of various antioxidants—namely, dieckol, punicalagin, EGCG, and resveratrol—in addressing xerosis by augmenting skin water content. After tape stripping, the water content in the stratum corneum of mice was measured following treatment with PM10 or antioxidants. PM10 exposure resulted in a reduction of skin water content, which was ameliorated by the application of antioxidants (Figure 1). The effectiveness of the antioxidants in enhancing skin water content was ranked as follows: resveratrol > EGCG > dieckol > punicalagin.
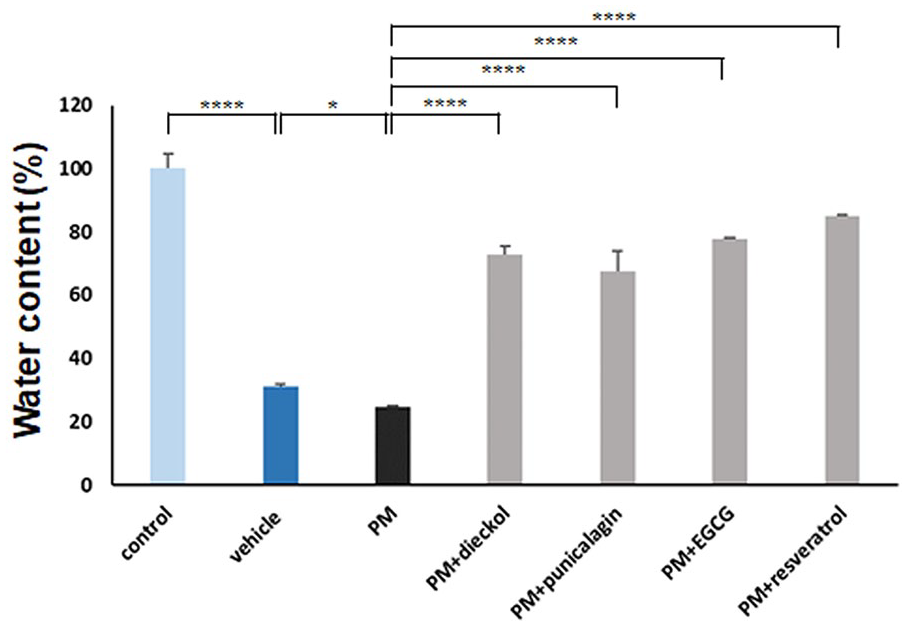
The preventive effects of antioxidants against the reduction in skin water content in skin barrier–disrupted mice treated with or without PM10. An increase in skin water content was observed after treatment with dieckol, punicalagin, epigallocatechin gallate (EGCG), and resveratrol, while PM10 treatment decreased skin water content in tape stripping–induced skin barrier–disrupted mouse models. Data in the bar graphs represent the mean ± standard deviation from three independent experiments (*P < 0.05, **P < 0.001, ***P < 0.005, ****P < 0.0001).
Preventive effects of antioxidants against the upregulation of inflammatory cytokines in PM10-treated, tape stripping–induced skin barrier–disrupted mice
Skin barrier disruption in mice was induced through tape stripping technique. The expression levels of inflammatory cytokines, such as IL-1β, IL-4, IL-6, IL-8, and TNF-α, were notably elevated relative to the control group, as depicted in Figures 2 and 3. Treatment with PM10 resulted in an upregulation of both gene and protein expression, consistent with the inflammatory response observed in other models of skin inflammation induced by PM10. Thus, PM10 exacerbates xerosis by inducing the overexpression of these genes and proteins.

The preventive effect of antioxidants on the amplification of inflammatory cytokines in PM10-exposed, tape stripping–induced skin barrier–disrupted mice was examined. Initially, the expression levels of genes associated with IL-1β, IL-4, IL-6, IL-8, and TNF-α were found to be elevated in the tape stripping–induced skin barrier–disrupted mice relative to those in the control group. Upon treatment with PM10, the skin barrier–disrupted mice exhibited greater increases in gene expression levels than the group treated solely with vehicle. These findings suggested that PM10 has the potential to exacerbate inflammation and disrupt the skin barrier by further stimulating gene expression. Additionally, subsequent administration of antioxidants resulted in a significant reduction in the expression levels of IL-1β, IL-4, IL-6, IL-8, and TNF-α relative to those observed following exclusive PM10 exposure. The data depicted in the bar graphs represent the mean ± standard deviation derived from three independent experiments (*P < 0.05, **P < 0.001, ***P < 0.005, ****P < 0.0001).

The protein levels of IL-1β, IL-4, IL-6, IL-8, and TNF-α exhibited an elevation in tape stripping–induced skin barrier–disrupted mice relative to those observed in the control group. Upon treatment with PM10, these skin barrier–disrupted mice demonstrated a more pronounced increase in protein expression levels compared with those subjected solely to vehicle treatment. These findings demonstrated the exacerbating effects of PM10 on inflammation and barrier disruption through further augmentation of protein expression. Additionally, following antioxidant treatment, a significant decrease in the protein expression levels of IL-1β, IL-4, IL-6, IL-8, and TNF-α was observed compared with those after exclusive PM10 exposure. The data presented in the bar graphs represent the mean ± standard deviation derived from three independent experiments (*P < 0.05, **P < 0.001, ***P < 0.005, ****P < 0.0001).
Furthermore, when PM10 was administered in conjunction with antioxidants, such as dieckol, punicalagin, EGCG, and resveratrol, a significant downregulation in the gene expression of IL-1β, IL-4, IL-6, IL-8, and TNF-α was observed relative to PM10 treatment alone. The efficacy ranking of various genes was conducted as follows: IL-1β—resveratrol > punicalagin > dieckol = EGCG; IL-4—resveratrol = punicalagin > dieckol = EGCG; IL-6—punicalagin > resveratrol = dieckol > EGCG; IL-8—resveratrol > dieckol > punicalagin > EGCG; TNF-α—dieckol > resveratrol > punicalagin > EGCG.
Likewise, protein expression also decreased upon treatment with a combination of PM10 and antioxidants. The efficacy ranking of the various proteins was as follows: IL-1β—punicalagin > resveratrol > dieckol > EGCG; IL-4—resveratrol > EGCG > punicalagin = dieckol; IL-6—EGCG > punicalagin > dieckol > resveratrol; IL-8—EGCG > dieckol = resveratrol > punicalagin; TNF-α—dieckol > punicalagin > resveratrol > EGCG.
Preventive effects of antioxidants on the upregulation of keratinocyte differentiation markers in PM10-treated, skin barrier–disrupted mice
Finally, immunofluorescence staining was conducted on dorsal skin specimens obtained from skin barrier–disrupted mice treated with PM10, with or without antioxidants. Loricrin, involucrin, and filaggrin were chosen as markers of keratinocyte differentiation. The reduction in expression levels of these keratinocyte differentiation markers, induced by PM10 treatment, was reversed in skin barrier–disrupted mice when co-treated with antioxidants (Figure 4).

Preventive effects of antioxidants on the downregulation of keratinocyte differentiation markers in tape stripping–induced skin barrier–disrupted mice after PM10 treatment. Antioxidants increased the levels of loricrin, involucrin, and filaggrin in the PM10-treated, skin barrier–disrupted mice.
Regarding filaggrin, the order of efficacy among the antioxidants was observed as follows: EGCG > resveratrol = punicalagin > dieckol. For involucrin, the order of efficacy was EGCG > dieckol = punicalagin > resveratrol. Similarly, for loricrin, the order of efficacy was resveratrol = EGCG > dieckol = punicalagin.
Discussion
In the International Classification of Diseases, xerosis cutis (synonyms: dry skin, xerosis, and xeroderma) is defined as a deficiency of epidermal hydrolipids (ICD-10: L85.3) in the skin. Both exogenous (e.g. environmental factors) and endogenous causes (e.g. inflammatory skin disease, aging) can lead to xerosis. 27 Since the skin barrier function is impaired and moisturizing factors are deficient, patients with xerosis exhibit decreased skin water content. Controversy persists over whether xerosis in older people should be considered a disease or a part of the aging process. Moreover, some authors even describe xerosis as low-grade atopic eczema. 28
The tape-stripping technique is recognized for its ability to generate mouse models with artificially disrupted skin barriers.29,30 According to Yang et al., the disruption of the barrier resulting from tape stripping can be attributed to the removal of corneocytes. 29 Our study corroborated an increase in the levels of inflammatory cytokines, such as IL-1β, IL-4, IL-6, IL-8, and TNF-α, in tape stripping–induced skin barrier–disrupted mice, as well as a downregulation of keratinocyte differentiation markers, such as loricrin, involucrin, and filaggrin. These findings were consistent with previous reports.31–33 However, due to the lack of the understanding of its mechanism of action, further studies should be conducted. Additionally, our findings were based on animal models, which may not fully replicate human skin conditions. Limited treatment duration may also not reflect the effect of the chronic exposure to PM10, that requires the conduction of further studies and their clinical validation. An intriguing observation relates to the increased inflammatory responses in mice with compromised skin following exposure to PM. However, the impact of PM treatment on healthy skin remains inadequately explored. Kim et al. recently highlighted the pivotal role of PM2.5 induced TNF-α in inhibiting filaggrin mediated by PM2.5. 4 While our study did not specifically address the effect of PM2.5 in skin barrier–disrupted mice, investigating the effects of PM treatment alone would be worthwhile. The size of the PM used in our study may bear significance, warranting further inquiry into this parameter.
In a previous study, exposure to PM10 was found to elevate serum IgE levels and induced an increase in spleen weight in an atopic dermatitis mouse model induced by DNCB. 25 Additionally, it was observed to augment the mast cell population, along with the expression of inflammatory cytokines, including IL-1β, IL-4, IL-6, IL-17α, IL-25, IL-31, and thymic stromal lymphopoietin (TSLP), as well as keratinocyte differentiation markers like loricrin and filaggrin in mice with DNCB-induced atopic dermatitis. 25
Shin et al. demonstrated that resveratrol effectively suppresses PM10-induced inflammatory reactions in human keratinocytes by diminishing the activation of aryl hydrocarbon receptors and the formation of reactive oxygen species. 24 Seok et al. further confirmed the restoration of the PM10-induced inflammatory response by punicalagin and EGCG in keratinocytes. 21 Additionally, Ha et al. observed that dieckol mitigates PM10-induced prostaglandin E2 production. 18 Our recent studies have shown that Siegesbeckia herba extract and dieckol inhibit the expression of inflammatory cytokines and matrix metalloproteinases induced by PM in sebocytes and keratinocytes.26,34 Siegesbeckia herba extract is used as herbal medicine in China, Korea, Japan, and Vietnam and has recently been examined for its antioxidant and anti-inflammatory propertie. 35 Another compound of interest, THSG (2,3,5,4′-tetrahydroxystilbene-2-O-β-D-glucoside) from Polygonum multiflorum, exhibits diverse biological activities, including anti-inflammatory and antioxidant effects. Hwang et al. examined the efficacy of THSG in modulating Th2 immune responses in an ovalbumin-induced asthma animal model, suggesting its potential in asthma treatment by regulating inflammatory processes. 36 Natural functional foods, such as antioxidative blueberries and black rice, represent alternative therapies for atopic dermatitis. Hong et al. investigated the impact of fermented blueberry and black rice extracts containing Lactobacillus plantarum MG4221 on PM-induced expression of proinflammatory cytokines both in vitro and in vivo. Administration of fermented blueberry and black rice extracts reduced IL-1β, IL-6, and IL-8 levels in PM-treated HaCaT cells. 37 Moreover, oral supplementation resulted in decreased transepidermal water loss, reduced serum IgE production, and enhanced protein expression of filaggrin and involucrin in skin tissue. Polyphenolic compounds such as dieckol, punicalagin, EGCG, and resveratrol exhibit antioxidative and anti-inflammatory properties. One potential limitation is that environmental factors, such as ambient humidity and temperature, could have influenced skin hydration measurements. Additionally, variations in antioxidant bioavailability might have impacted the observed outcomes. Further studies are needed to explore the long-term effects of antioxidant treatment on skin exposed to chronic PM10. Clinical trials in human subjects with compromised skin barriers will also be critical in translating these findings into potential therapeutic interventions.
Conclusions
PM10 exposure can exacerbate xerosis by reducing skin moisture levels and increasing the expression of inflammatory cytokines. In our study, mice with compromised skin barriers treated with PM10 showed reduced levels of key keratinocyte differentiation markers, including loricrin, involucrin, and filaggrin. However, antioxidants such as dieckol, punicalagin, EGCG, and resveratrol were associated with protective effects against PM10-induced alterations in skin hydration, inflammatory cytokine expression, and keratinocyte differentiation marker production. These findings underscore the therapeutic potential of antioxidants in mitigating skin damage caused by environmental pollutants. The use of antioxidant-rich formulations could be a promising strategy for managing skin conditions exacerbated by pollution, particularly in older adults who are more susceptible to xerosis and other inflammatory skin disorders.
Footnotes
Author contributions
All authors contributed to the study conception. JHK and MHK performed the experiments. WJL contributed new reagents and analyzed data. WJL supervised the research and JHK and MHK wrote the manuscript. All authors read and approved the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by a grant of the Korea Health Technology R&D Project through the Korea Health Industry Development Institute (KHIDI), funded by the Ministry of Health & Welfare, Republic of Korea (grant number : RS-2024-00437643).
Ethics approval
Ethical approval for this study was obtained the Institutional Animal Care and Use Committee of the Kyungpook National University (IRB No. 2021-0068).
Animal welfare
The present study followed the ARRIVE guidelines and Institutional Animal Care and Use Committee of the Kyungpook National University for humane animal treatment and complied with relevant legislation.
Data availability statement
The data sets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
