Abstract
Alzheimer’s disease (AD) is characterized by a robust inflammatory response elicited by the accumulation and deposition of amyloid-β (Aβ) within the brain. Aβ induces detrimental inflammatory responses through toll-like receptors (TLRs) signaling pathway. Thymoquinone (TQ), the main active constituent of Nigella sativa oil, has been reported by several previous studies for its potent anti-inflammatory effect. The aim of this study is to elucidate the effect of TQ in improving learning and memory, using a rat model of AD induced by a combination of aluminum chloride (AlCl3) and
Introduction
Alzheimer’s disease (AD) is a progressive neurodegenerative disorder and the most common cause of dementia. 1 Besides the main pathological hallmark of AD, amyloid-β (Aβ) deposits, neuroinflammation was more recently described to play a major role in AD pathogenesis. Toll-like receptors (TLRs) are a family of 11 member (TLR-1-11) receptors which were originally discovered based on their response to invading microorganisms. 2 TLRs are expressed in a variety of immune-related cells such as macrophages, monocytes, astrocytes, and microglia as well as nonimmune cells such as epithelial and endothelial cells within the brain parenchyma. 3,4
TLRs are transmembrane proteins with an extracellular leucine-rich repeat domain and an intracellular toll–interleukin-1 receptor (TIR) domain. 5 TLRs activation triggers two branches of downstream signaling pathways: Myeloid differential factor 88 (MyD88)-dependent and TIR domain-containing adapter-inducing interferon-β (TRIF)-dependent (or MyD88-independent) pathways. 5 Agonism of all TLRs is dependent upon the dimerization of TLR signaling complex that includes recruitment of MyD88 through their TIR domain. Following this recruitment, MyD88-dependent signaling pathway is activated leading to the activation of nuclear factor-κB (NF-κB) with subsequent production of a number of pro-inflammatory cytokines such as TNF-α and IL-1β. 6,7 Among TLRs family, TLR-3 and TLR-4 can utilize the adaptors TRIF independently of MyD88 to trigger TRIF-dependent signaling pathway. This leads to the activation of interferon regulatory factor 3 (IRF-3) with subsequent production of several Type I interferon inducible gene as well as initiating the late phase of NF-κB activation. 5,8 Since TLR-4 ligand-induced inflammatory cytokines production was impaired in TRIF-deficient mice as well as MyD88-deficient mice, 5,9 signals for both MyD88 and TRIF may be required for the maximum expression of cytokines. 5 TLR-2 and TLR-4 play an important role in initiating the inflammatory responses following Aβ deposition. 10 –12 Previous studies have shown that TLR-2 and TLR-4 are molecular targets for Aβ isoforms to trigger neuroinflammatory activation. 13,14 Moreover, TLR-2 knockout mice were protected from cognitive impairment upon immunization with Aβ. 15 Also, chronic administration of a detoxified TLR-4 ligand potently ameliorated the cognitive functions in a mouse model of AD. 16
Thymoquinone (TQ), the bioactive constituent of the volatile oil of black cumin seeds (Nigella sativa), has been traditionally used in Middle East and Southeast Asian countries for its multiple health beneficial activities. It has been extensively studied for its wide range of biological activities, including anti-inflammatory, antioxidant, antihistaminic, and antitumor properties. 17 –20 In vivo, TQ possesses a number of therapeutic effects with respect to central nervous system neurodegeneration as shown by its antioxidant and its anti-inflammatory potency. 21 –23 While the anti-neuroinflammatory effect of TQ is mediated through its ability to diminish NF-κB activation and its subsequent cytokines including TNF-α and IL-1β, 21,24 the molecular mechanisms underlying TQ effect remain unclear.
There is no clear understanding of the order of cellular changes that account for AD progression and pathogenesis. 25 To understand cellular changes that are involved in AD pathogenesis, several studies recently measured gene expressions in AD brains and age-matched control subjects. 26 –30 In recent years, the development of real-time polymerase chain reaction (RT-PCR) techniques has revolutionized gene expression studies. 31 –33 Therefore, the aim of this study is to elucidate the therapeutic effects of TQ on inflammation which is associated with learning and memory impairment in AD. We characterized the learning and memory ability, the gene expression profiles of TLR-2 and TLR-4 and their signaling pathway molecules and the pro-inflammatory cytokine profiles of brain TNF-α and IL-1β as well as the histopathological changes in AD rat model, in order to underline the molecular mechanisms and the possible signaling molecules in relation to three different doses of TQ.
Material and methods
Tested compounds
TQ (2-Isopropyl-5-methyl-1,4-benzoquinone) and all chemicals were obtained from (Sigma-Aldrich Co., USA).
Animals
Ninety six Sprague-Dawley male albino rats were purchased from the animal house of National Research Center, Cairo, Egypt. All animals were housed in plastic cages, kept in a conditioned atmosphere at 25°C, and fed standard laboratory pellets with tap water ad libitum. Rats were allowed to acclimatize for 2 weeks prior to the experiment. Animal handling and experimental procedures were conducted in accordance with the Guide for the Care and Use of Laboratory Animals (Eighth Edition, 2011, published by The National Academies Press, 2101 Constitution Ave. NW, Washington, DC 20055, USA) and approved by the ethics committee at the Faculty of Pharmaceutical Sciences and Pharmaceutical Industries, Future University in Egypt (registered REC-FPSI-9/54).
Animal model establishment
In this study, we established a model for AD using long-term
Animals and their treatment outlines
After 2 weeks of acclimatization, rats were randomly divided into eight equal groups (n = 12) as follows. Control group: rats received normal saline and corn oil to serve as negative control group. Control+TQ (CTQ) groups: Three groups of rats received TQ dissolved in corn oil orally at 10, 20, and 40 mg/kg/dose levels daily for 14 consecutive days to serve as positive control groups. AD-model group: Rats received intraperitoneally AlCl3 (10 mg/kg) and
Step through passive avoidance test
On the 11th day following the treatment, the passive avoidance test was performed according to the methods described previously.
41
A step through passive avoidance apparatus was used. It consisted of Plexiglas box divided into two compartments (dark/light) with identical dimensions (25 × 25 × 27 cm3), which were connected by an opaque guillotine door (8 × 8 cm2). The light compartment was illuminated with 220-V 40-W bulb placed 15 cm above the center. The dark compartment is constructed of black Perspex panels and had 40 parallel bars of stainless steel grid floor (0.3 cm in diameter, set 1.2 cm apart) available for foot electric shock (50 Hz, 1.5 mA, and 3 s). Each compartment has a wall, which can be opened or closed for placement and removal of the rats.

Schematic representation of step through passive avoidance experiments. Rats underwent three exploration session with 180 s habituation in 1 day, the following day rats were subjected to one training session with 60 s habituation, presentation of aversive stimulus, a foot shock, (50 Hz, 1.5 mA, and 3 s) at 3 s intervals. After 24 h, rats were subjected to memory retention test with no electrical shock, the cutoff time was 180 s, and the retention latency of rats to enter the dark compartment was record.
Brain tissue sampling and preparation
Rats were anaesthetized using thiopental and killed by cervical dislocation. The whole brain of each rat was rapidly placed on ice then dissected from olfactory bulb to the cerebellum. It was then washed with isotonic saline and dried on filter paper. Each brain was divided sagittal into two portions. The first portion (right hemibrain) was stored at −80°C for biochemical analyses. A 20–30 mm-thick piece of tissue was sagittal cut from the second portion of the brain (left hemibrain) containing hippocampus using cryostat (SLEE, Germany) for further RT-PCR investigation of TLR-2, TLR-4, MyD88, TRIF, IRF-3, and NF-κB. The rest of the second portion of the brain was fixed in 10% formalin for histological investigation.
Quantitative RT-PCR
RNA extraction
Total RNA was isolated from 200 μL of brain tissue using the mRNeasy Mini Kit (Qiagen, Germany) according to the manufacturer’s instructions. For normalization of sample-to-sample variation, housekeeping gene (β-actin) was assayed in each sample with the same steps as TLR-2, TLR-4, MyD88, TRIF, IRF-3, and NF-κB genes. RNA was dissolved in 50 μL of RNase-free water and then stored at −80°C until analysis.
Reverse transcription
Total RNA was reverse transcribed using QuantiTect® Reverse Transcription Kit (Qiagen, Germany) in 4 µL from Quantiscript RT buffer, 1µL from Quantiscript Reverse Transcriptase, 1 µL from the RT primer mix, and 14 µL of the template RNA (Qiagen, Germany). For synthesis of cDNA, the RT reaction was incubated at 42°C for 15 min, then at 95°C for 3 min. The cDNA product was stored at −20°C until analysis.
RT-PCR detection
For quantitative real-time PCR (qRT-PCR; QuantiTect SYBR Green, 2 μL of the cDNA product was used as template in 25-μL total reaction volume containing the following: 12.5 μL of Quantitect SYBER Green PCR Master Mix, 8 μL of RNase-free water, and 2.5 μL of Quantitect Primer Assay. The primers used for amplification were readily made by Qiagen. qRT-PCR was performed with Qiagen rotor gene Q6 Plex RT-PCR system (Qiagen, Germany) at PCR initial activation at 95°C for15 min, followed by 40 cycles of 94°C for 15 s and 55°C for 30 s and 72°C for 30 s. Data were analyzed with Rotorgene Q software (Qiagen, Germany), with the automatic Ct setting for assigning baseline and threshold for Ct determination. The relative expression level of each individual gene after normalization to (β-actin) was calculated using the 2−ΔΔCt method.
ELISA determination
The levels of IL-1β and TNF-α were measured in homogenates prepared from the right hemisphere using ELISA Kit according to the manufacturer’s protocol (R&D systems, Minneapolis, MN, USA).
Histological examination
The brain tissue was fixed in 10% formalin for 1 week, washed in running tap water and dehydrated in ascending series of ethanol (50–90%), followed by absolute alcohol. The samples were cleared in xylene and immersed in a mixture of xylene and paraffin at 60°C. The tissue was then transferred to pure paraffin wax of the melting point 58°C and then mounted in blocks and left at 4°C. The paraffin blocks were sectioned on a microtome at thickness of 5 µm and mounted on clean glass slides and left in the oven at 40°C to dryness. The slides were deparafinized in xylene and then immersed in descending series of ethanol (90–50%). The ordinary haematoxylin and eosin (H&E) stain was used to stain the slides.
Statistical analysis
Data are expressed as mean ± standard error of mean. Statistical analysis was performed using one-way analysis of variance followed by Tukey’s post hoc for multiple comparisons test using GraphPad Prism software (version 5, GraphPad Prism Software Inc., La Jolla, CA, USA). For all analyses, the level of statistical significance was set at p < 0.05.
Results
TQ improves learning and memory ability in AD rat model
Performance of the rats in the step-through passive avoidance testing is shown in Figure 1(a) and (b): The step-through latency decreased markedly in the AD model group compared to the control group (p < 0.05). The step-through latency significantly increased by TQ treatment at all levels of doses compared to AD group (p < 0.05). TQ middle dose (20 mg/kg) caused an increase in the step-through latency significantly compared to TQ low dose (10 mg/kg) and TQ high dose (40 mg/kg) by 161.2% or 150.2% after 24 h and by 152.2% or 99.5% after 48 h, while TQ high dose (40 mg/kg, orally) did not alter the step-through latency compared to TQ low dose (10 mg/kg) and TQ middle dose (20 mg/kg). Administration of TQ to CTQ groups did not cause any significant change in the step-through latency compared to the control group.
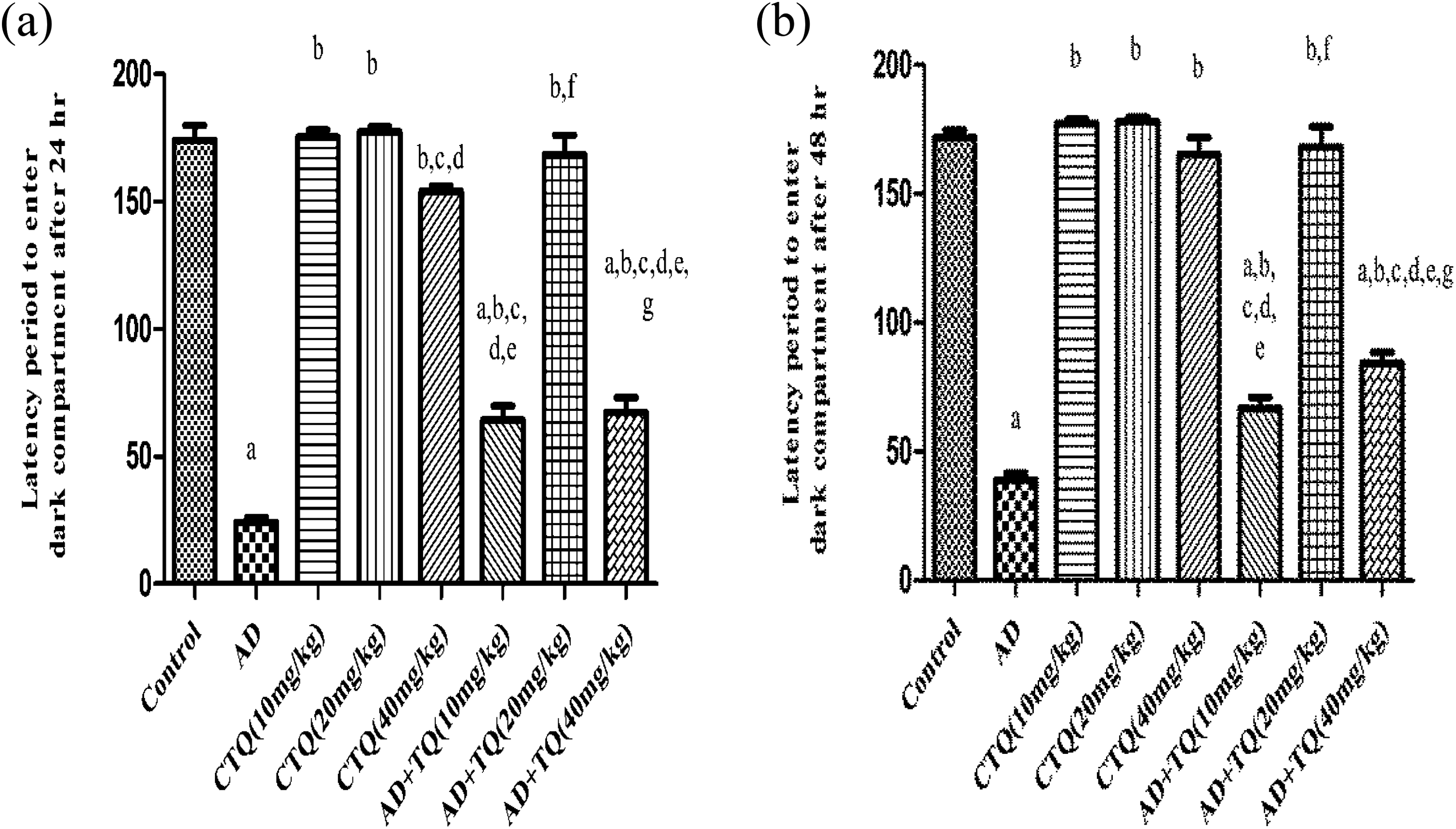
Effect of TQ on latency of step-through avoidance test in AD rats. (a) The effect of three different doses of TQ (10, 20, and 40 mg/kg, orally) on the step through latency after 24 h of AD experimental model. (b) The effect of three different doses of TQ (10, 20, and 40 mg/kg, orally) on the step through latency after 48 h of AD experimental model. Data are expressed as mean ± SEM, n = 12. a to g: significant difference compared to control group, AD group, CTQ(10 mg/kg), CTQ(20 mg/kg), CTQ(40 mg/kg), AD+TQ(10 mg/kg), AD+TQ(20 mg/kg), and AD+TQ(40 mg/kg) at p < 0.05 using one-way ANOVA followed by Tukey’s post hoc test. TQ: thymoquinone; AD: Alzheimer’s disease; CTQ: control+TQ; ANOVA: analysis of variance; SEM: standard error of mean.
Effects of TQ on TLRs cascade elements
Figure 2(a) shows that treatment of rats with TQ at all levels of doses has significantly reduced TLR-2 expression compared to AD group reaching 8.062-, 1.391-, and 6.308-fold versus10.43-fold, respectively, (p < 0.05). Treatment of rats with TQ at all levels of doses significantly reduced TLR-4 expression compared to AD group reaching 8.374-, 1.310-, and 5.503-fold versus 11.44-fold, respectively, (p < 0.05) as presented in Figure 2(b).

Effect of TQ on TLR-2/TLR-4 on the receptors levels. (a) The effect of three different doses of TQ (10, 20, and 40mg/kg, orally) on TLR-2 gene expression in brain tissue of AD experimental model. (b) The effect of three different doses of TQ (10, 20, and 40 mg/kg, orally) on TLR-4 gene expression in brain tissue of AD experimental model. Data are expressed as mean ± SEM, n = 12. a to g: significant difference compared to control group, AD group, CTQ(10 mg/kg), CTQ(20 mg/kg), CTQ(40 mg/kg), AD+TQ(10 mg/kg), AD+TQ(20 mg/kg), AD+TQ(40 mg/kg) at p < 0.05 using one-way ANOVA followed by Tukey’s post hoc test. TQ: thymoquinone; TLR: toll-like receptor. AD: Alzheimer’s disease; CTQ: control+TQ; ANOVA: analysis of variance; SEM: standard error of mean.
In Figure 3, treatment of rats with TQ at all levels of doses has significantly reduced MyD88 expression compared to AD group reaching 6.300-, 1.282-, and 4.052-fold versus 8.477-fold, respectively, (p < 0.05). Treatment of rats with TQ at all levels of doses significantly reduced TRIF expression compared to AD group reaching 7.003-, 1.302-, and 5.150-fold versus 9.774-fold, respectively, (p < 0.05) as seen in Figure 4.

Effect of TQ on MyD88: The effect of three different doses of TQ (10, 20, and 40 mg/kg, orally) on MyD88 gene expression in brain tissue of AD experimental model. Data are expressed as mean ± SEM, n = 12. a to g: Significant difference compared to control group, AD group, CTQ(10 mg/kg), CTQ(20 mg/kg), CTQ(40 mg/kg), AD+TQ(10 mg/kg), AD+TQ(20 mg/kg), AD+TQ(40 mg/kg) at p < 0.05 using one-way ANOVA followed by Tukey’s post hoc test. TQ: thymoquinone; AD: Alzheimer’s disease; MyD88: myeloid differential factor 88; CTQ: control+TQ; ANOVA: analysis of variance; SEM: standard error of mean.

Effect of TQ on TRIF: The effect of three different doses of TQ (10, 20, and 40 mg/kg, orally) on TRIF gene expression in brain tissue of AD experimental model. Data are expressed as mean ± SEM, n = 12. a to g” Significant difference compared to control group, AD group, CTQ(10 mg/kg), CTQ(20 mg/kg), CTQ(40 mg/kg), AD+TQ(10 mg/kg), AD+TQ(20 mg/kg), AD+TQ(40 mg/kg) at p < 0.05 using one-way ANOVA followed by Tukey’s post hoc test. TQ: thymoquinone; TRIF: toll–interleukin-1 receptor domain-containing adapter-inducing interferon-β; AD: Alzheimer’s disease; CTQ: control+TQ; ANOVA: analysis of variance; SEM: standard error of mean.
Treatment of rats with TQ at all levels of doses significantly diminished IRF-3 expression compared to AD group reaching 8.474-, 1.159-, and 5.847-fold versus 10.06-fold, respectively, (p < 0.05) as demonstrated in Figure 5.

Effect of TQ on IRF-3: The effect of three different doses of TQ (10, 20, and 40 mg/kg, orally) on IRF-3 gene expression in brain tissue of AD experimental model. Data are expressed as mean ± SEM, n = 12. a to g: Significant difference compared to control group, AD group, CTQ(10 mg/kg), CTQ(20 mg/kg), CTQ(40 mg/kg), AD+TQ(10 mg/kg), AD+TQ(20 mg/kg), AD+TQ(40 mg/kg) at p < 0.05 using one-way ANOVA followed by Tukey’s post hoc test. TQ: thymoquinone; IRF-3: interferon regulatory factor 3; AD: Alzheimer’s disease; CTQ: control+TQ; ANOVA: analysis of variance; SEM: standard error of mean.
Treatment of rats with TQ at all levels of doses for 14 consecutive days caused a significant decrease in NF-κB expression compared to AD group reaching 6.134-, 1.180-, and 5.325-fold versus 8.415-fold, respectively, (p < 0.05) as shown in Figure 6.
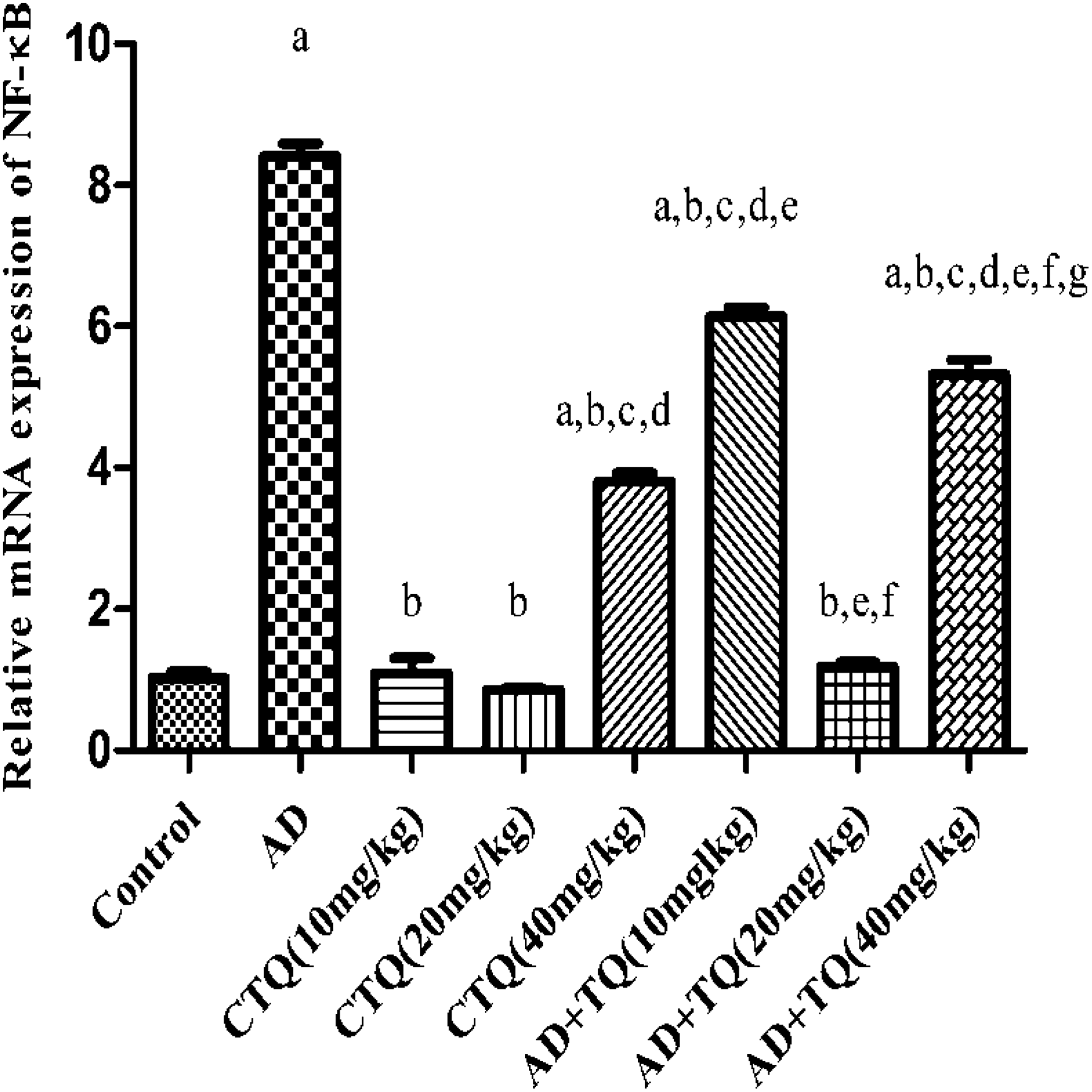
Effect of TQ on NF-κB: The effect of three different doses of TQ (10, 20, and 40 mg/kg, orally) on NF-κB gene expression in brain tissue of AD experimental model. Data are expressed as mean ± SEM, n = 12. a to g: Significant difference compared to control group, AD group, CTQ(10 mg/kg), CTQ(20 mg/kg), CTQ(40 mg/kg), AD+TQ(10 mg/kg), AD+TQ(20 mg/kg), AD+TQ(40 mg/kg) at p < 0.05 using one-way ANOVA followed by Tukey’s post hoc test. TQ: thymoquinone; NF-κB: nuclear factor-κB; AD: Alzheimer’s disease; CTQ: control+TQ; ANOVA: analysis of variance; SEM: standard error of mean.
Interestingly, TQ middle dose (20 mg/kg) significantly downregulated the expression of TLR-2 by 82.74% and 77.94% and the expression of TLR-4 by 84.35% and 63.30%, the expression of MyD88 by 79.65% and 68.36%, the expression of TRIF by 25.90% and 76.75%, the expression of IRF-3 by 18.19% and 77.96%, and the expression of NF-κB by 80.76% and 77.84%, compared to TQ low dose (10 mg/kg) and TQ high dose (40 mg/kg) (p < 0.05). Regarding the effect of TQ on CTQ groups, administration of TQ low (10 mg/kg) and middle (20 mg/kg) doses to CTQ groups caused no significant changes in the expression of TLR cascade elements compared to the control group (p < 0.05). On the other hand, administration of TQ high dose (40 mg/kg) to CTQ group significantly upregulated the expression of TLR cascade elements compared to control group (p < 0.05), which is the opposite effect seen in the AD group.
Effects of TQ on the pro-inflammatory cytokines
Fourteen consecutive days of TQ treatment at all levels of doses caused a significant decrease in the rats brain content of TNF-α compared to AD group reaching 39.85, 18.22, and 30.37 versus 65.30, respectively, (p < 0.05) as shown in Figure 7(a).
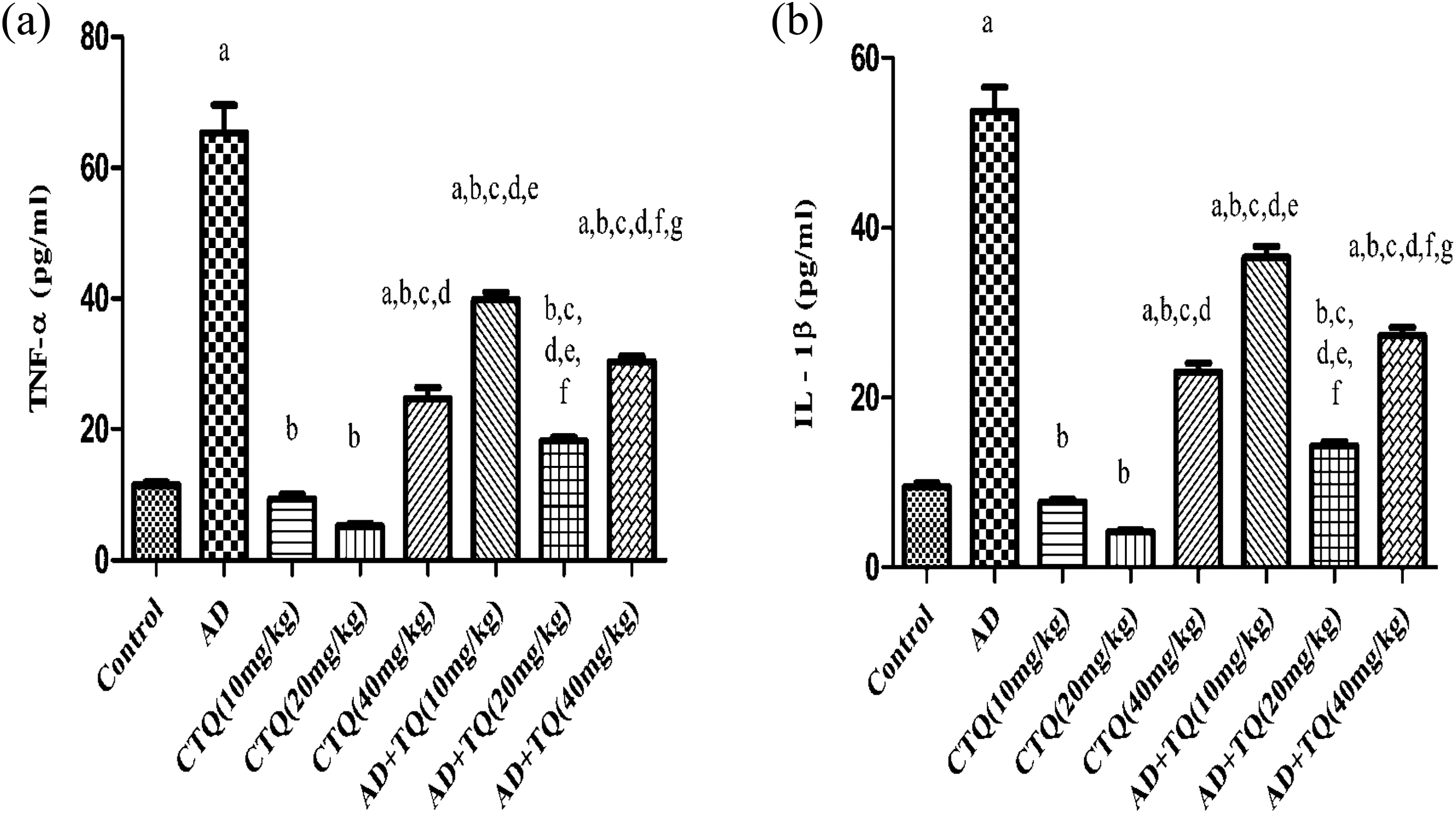
Effect of TQ on the pro-inflammatory cytokines. (a) The effect of three different doses of TQ (10, 20, and 40 mg/kg, orally) on Tumor necrosis factor-alpha (TNF-α) level in brain tissue of AD experimental model. (b) The effect of three different doses of TQ (10, 20, and 40 mg/kg, orally) on Interleukin-1beta (IL-1β) level in brain tissue of AD experimental model. Data are expressed as mean ± SEM, n = 12. a to g: Significant difference compared to control group, AD group, CTQ(10 mg/kg), CTQ(20 mg/kg), CTQ(40 mg/kg), AD+TQ(10 mg/kg), AD+TQ(20 mg/kg), AD+TQ(40 mg/kg) at p < 0.05 using one-way ANOVA followed by Tukey’s post hoc test. AD: Alzheimer’s disease; TQ: thymoquinone; CTQ: control+TQ; ANOVA: analysis of variance; SEM: standard error of mean.
Figure 7(b) reveals that treatment of rats with TQ at all levels of doses significantly reduced the brain content of IL-1β compared to AD group reaching 36.55, 14.32, and 27.27 versus 53.65, respectively, (p < 0.05). Remarkably, TQ middle dose (20 mg/kg) significantly diminished the brain content of TNF-α by 54.27% and 40% and IL-1β by 99.60% and 47.48% compared to the TQ low dose (10 mg/kg) and TQ high dose (40 mg/kg; p < 0.05) as presented in Figure 7(a) and (b).
Administration of TQ low dose (10 mg/kg) and TQ middle dose (20 mg/kg) to CTQ groups did not cause any significant changes compared to the control group (p < 0.05). While administration of TQ high dose (40 mg/kg) to CTQ group significantly increased the brain content of TNF-α and IL-1β compared to the control group as shown in Figure 7(a) and (b).
Effects of TQ on brain morphological structure
Microscopic examination of H&E-stained brain sections of the control group rats showed normal morphological structure of the striatum as shown in Figure 8(a).

Micrographs of brain sections. Magnification 40×. (a) Control showing normal morphological structure of the striatum in cerebrum. (b) CTQ (10 mg/kg) rat showing normal morphological structure of the striatum in cerebrum. (c) CTQ (20 mg/kg) rat showing normal morphological structure of the striatum in cerebrum. (d) CTQ (40 mg/kg) rat showing normal morphological structure of the striatum in cerebrum. (e) AD rat injected with AlCl3/
Administration of TQ to CTQ groups at all levels of doses did not cause any significant changes in the brain normal morphological structure as presented in Figure 8(b) to (d). On the other hand, microscopic investigation of brain sections of AD-induced rats demonstrated the presence of amyloid plaques of various sizes in the striatum of the cerebrum as shown in Figure 8(e). Histological investigation of brain section of AD-induced rats treated with TQ at all levels of doses revealed more or less normal structure in the striatum of the cerebrum. Amyloid plaques were not observed after TQ middle (20mg/kg) and TQ high (40 mg/kg) dose administration in AlCl3-treated/
Discussion
TQ, 2-isopropyl-5-methylbenzo-1,4-quinone, bears a very simple molecular structure. It crosses the blood brain barrier and exerts diverse therapeutic effects with respect to neuroinflammation. 17,24 In this study, we observed that TQ treatment at all levels of doses succeed to diminish the Aβ plaques accumulation in brains of our AD model rat and improve their learning and memory abilities. Furthermore, TQ treatment at all levels of doses significantly suppressed the expression of TLR-2 and TLR-4, their adaptor proteins MyD88 and TRIF, and their downstream effectors IRF-3 and NF-κB in mRNA level as well as decreasing the levels of the consequent proinflammaory cytokines, IL-1β and TNF-α, release in protein level.
Functional TLR signal transduction is complex and relies on receptor dimerization as well as the presence of accessory proteins and co-receptors, which regulate the signaling pathways initiated by each receptor. 42 According to our results, TQ inhibits both MyD88- and TRIF-dependent pathways in TLR-2/TLR-4 signaling pathways. In MyD88-dependent pathway, the increased expression of MyD88 induced NF-κB increase. This was inhibited by TQ at all levels of doses. In TRIF-dependent pathway, all doses of TQ inhibited the gene expression of NF-κB which is induced by the increased expression of TRIF and IRF-3.
Importantly, TQ inhibits TLR-2 and TLR-4 and their downstream signaling molecule in a dose-independent manner. TQ middle dose (20 mg/kg) showed a significant effect compared to the other two doses, while TQ high dose (40 mg/kg) failed to record a better effect compared to the middle dose (20 mg/kg) and TQ low dose (10mg/kg) showed the lowest effect. On the other hand, the effects of TQ on CTQ groups were only detected at the high dose (40 mg/kg). While TQ low (10 mg/kg) and middle (20 mg/kg) doses didn’t cause any significant change in TLRs signaling pathway, TQ high dose (40 mg/kg) showed significant increase in TLR-2 and TLR-4 signaling pathways compared to the control group.
This could be explained by the affinity of a compound to a receptor which would directly influence the concentration of a compound on the binding site and duration of effect of the compound and consequently contribute to its biological action in vivo. It has been well-documented that molecules with the structural motif of α,β-unsaturated carbonyl group can react with biological nucleophiles such as sulfhydryl group by a Michael addition. 43,44 Therefore, we suggest that TQ at low (10 mg/kg) and middle (20 mg/kg) doses act as Michael reaction acceptor in which its α and β unsaturated group can react with –SH group in cysteine-rich region in the extracellular domain and TIR region (TIR homologous region) in the cytoplasmic domain of both TLR-2 and TLR-4 forming an irreversible disulfide bond leading to the inhibition of TLR-2 and TLR-4 gene expression 43 –45 , whereas TIR domains are required for downstream signal transduction. 42,46 Although some studies support the ability of TQ to attenuate inflammation and subsequent inflammatory injury by inhibiting TLR-4 expression, 47,48 conflicting reports showed the evidence that TQ facilitates MyD88/TLR-4 complex formation and subsequent NF-κB activation in live cells through induction of sialidase activity. 49,50
It has been proposed that the ligand-induced TLR activation is tightly controlled by sialidase activation which enables TLR desialyation via hydrolyzation of sialyl α-2,3-linked β-galactosyl residues distant from ligand binding to remove steric hindrance to TLR dimerization. 51 Importantly, TQ is a ligand which activates sialidase to promote a pro-inflammatory response. 49 –51 Therefore, we suggest that TQ administered to CTQ group at the high dose (40 mg/kg, orally) induced sialidase activity which would actually target TLR receptors, enabling desialylation of TLR receptors and facilitating TLR signaling pathway. This effect can account for the proinflammtory effect of TQ high dose when administered to CTQ group which is opposite to its effect in the AD group. This could be explained by the loss of sialidase enzyme activity in the brain of AD patients as proven in a study by Annunziata et al. 52 and Maguire. 53
Many cellular functions are regulated by changes in gene expression. Hence, quantification of transcription levels of genes plays a vital role in the understanding of gene function and of abnormal alterations in regulation that may result in a disease state. 54 The innovation of the RT-PCR technique plays a paramount role in molecular medicine. 55 Although the measurement of gene expressions in brains with neurodegenerative diseases is a major area of brain research and different publications have described a good correlation between TLRs mRNA levels quantified by RT-PCR and protein levels quantified, 56,57 posttranscriptional or posttranslational modifications may play a role, and thus, discrepancy may exist between mRNA and protein levels. Since activation of TLR-2 and TLR-4 triggers a cascade of signaling events resulting in the activation of NF-κB and subsequent expression of pro-inflammatory cytokines such as TNF-α and IL-1β, 58,59 we further investigated the effect of TQ treatment on TNF-α and IL-1β in protein level to strengthen our results.
According to our results, TQ at all levels of doses succeeded to reduce both TNF-α and IL-1β levels compared to AD group. As such, TQ (20 mg/kg) middle dose caused a significant reduction in pro-inflammatory cytokines levels as compared to the other two doses. On the other hand, the effect of TQ administration on CTQ groups is only evident at TQ high dose. Unlike its anti-inflammatory effect on AD rat model, TQ high dose administration to the CTQ group resulted in a significant increase in the pro-inflammatory cytokines levels.
In this study, the gene expression of TLR-2, TLR-4, MyD88, TRIF, IRF-3, and NF-κB in mRNA levels aligns with what we have demonstrated regarding the protein levels of the pro-inflammatory cytokines TNF-α and IL-1β. Additionally, the mRNA expression of TLRs signaling molecule was consistent with what we have detected functionally in behavioral test. Thus, we can hypothesize that TQ could improve cognition and the brain morphological changes by attenuating the detrimental inflammatory effect of the pro-inflammatory cytokines release. TQ anti-inflammatory effect may be mediated through its inhibitory effect on TLR-2 and TLR-4 signaling pathways (Figure 9).
Conclusions
TQ improved the AD-induced cognitive decline and brain morphological changes through its anti-inflammatory effect. The anti-inflammatory effect exerted by TQ may depend not only on the inhibition TLR-2/TLR-4 signaling pathways to produce its toxicity through releasing of pro-inflammatory cytokines, but also on the mRNA expression of TLR-2/TLR-4. Both TLR-2 and TLR-4 are targets for TQ; TQ also inhibits MyD88-dependent and TRIF-dependent pathways. The dual anti- and pro-inflammatory effects of TQ require further investigation, specifically the protein level of TLR-2, TLR-4, MyD88, TRIF, IRF-3, and NF-κB.

Potential mechanism of TQ on TLRs signaling in the brain. TLR: toll-like receptor; TQ: thymoquinone.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
