Abstract
Lindane is very commonly used organochlorine pesticide and has been reported to cause several toxic effects including respiratory insufficiency. However, effects of low concentration of lindane alone or in combination with microbial molecules on lungs are not fully understood. To understand the effects a preliminary study was designed on Swiss albino mouse. Male mice were divided into treatment and control group (20; each). Treatment mice were given lindane in ground nut oil orally at 0.25 mg kg−1 day−1 for 60 days. After treatment, 10 mice were challenged with intranasal Escherichia coli lipopolysaccharide (LPS; 80 μg per mice) and remaining 10 with normal saline. The mice were euthanized 16 h post-LPS exposure. Control mice (10 each) were given normal saline or the LPS alone. Mice exposed with lindane and in combination with LPS had increase in total cell counts and leukocyte counts in broncho-alveolar lavage. Histological examination showed lung injury in the lindane-treated mice. The histopathological changes were more pronounced in lindane along with LPS-exposed mice. Lindane alone and in combination with LPS showed expression of immunopositive Toll-like receptor (TLR)-4 and tumour necrosis factor-alpha (TNF-α) positive reaction in various cells of lungs. While LPS induced acute inflammation in the lungs, combination of lindane and LPS exacerbated histological signs of the inflammation. The data indicate that lindane alone or in combination with LPS caused changes in lung morphology and altered TLR-4 and TNF-α expression which may have led to altered response to LPS challenge.
Introduction
Lindane, the γ-isomer (>99% pure) of hexachlorocyclohexane, belongs to organochlorine family of pesticides with oral LD50 of 55–250 mg kg−1 body weight (BW) in mouse. 1 It is widely used in agriculture to eradicate insects and treat scabies. 2 However, the extensive use, chemical stability and bioaccumulation potential of lindane resulted its ubiquitous distribution in the ecosystem. Lindane accumulates in vivo to the highest percentage and is a major toxicant because of its lipophilicity which results in slow degradation and excretion. 3 Low-dose exposures to pesticides pose significant human health hazards such as respiratory dysfunction(s), immunosupression, reproductive abnormalities, hormonal disruption, diminished intelligence and increasing cancer incidences. 4,5
Occupational exposure to lindane occurs via ingestion, inhalation and dermal exposure, whereas dietary exposure is the main non-occupational exposure. 6 Chronic exposure to lindane induces infertility by decreasing gametogenic and steroidogenic activities in mammals 7,8 and affects the circulatory, excretory and immune systems. 9,10 However, there are very limited data available on the pulmonary exposure to environmental residual level of lindane.
Farmers get exposure of endotoxin in high concentration while working in the agricultural environments and other occupational settings. 11,12 Occupational exposure to organic dust was found to deliver 30–60 µg of endotoxin to the lung; over in 8 h work shift. 13 Endotoxin has been primarily implicated in number of occupational lung diseases like acute airway obstruction, chronic bronchitis, hypersensitivity pneumonitis and decreased lung function in humans. 14 Lipopolysaccharide (LPS) is a principal cell wall component of the gram-negative bacteria which stimulates macrophages leading to production of many pro-inflammatory factors specially tumour necrosis factor-alpha (TNF-α). The increased production of TNF-α results in migration and activation of inflammatory cells including neutrophils 15 and development of inflammatory responses that are hallmarks of many diseases including pulmonary diseases. 16 It is implicated in asthma, chronic bronchitis, chronic obstructive pulmonary disease, acute lung injury and acute respiratory distress syndrome. 17
Toll-like receptors (TLRs) respond to conserved structures within pathogens and activate macrophages, monocytes and neutrophils to produce cytokines and low molecular weight mediators. 18 TLR-4 is the best characterized signalling receptor for LPS 19 and plays vital role in the pro-resolution of lung inflammation and fibrosis in acute and chronic lung injury. 20 However, there is paucity of data on effects of lindane on non-target organs like lungs and pulmonary expression of TNF-α and TLR-4 after chronic exposure to lindane and followed by LPS challenge. Therefore, present study is being done to investigate the expression of inflammatory marker (TNF-α) and TLR-4 in pathophysiology of pulmonary dysfunctions putatively induced by environmental residual level of lindane and bacterial endotoxin.
Materials and methods
The experiment protocols were approved by Institutional Animal Ethics Committee (IAEC), Guru Angad Dev Veterinary and Animal Sciences University, Ludhiana. Forty Swiss albino male mice (8–10 weeks) were procured from Department of Livestock Product Management of the University. Mice were housed in laboratory animal cages with room temperature of around 18–22°C and 12-light/12-h dark cycle and were acclimatized to these conditions for 2 weeks before entering the study. The animals were provided feed (Godrej Agrovet Limited, Khanna, Punjab, India) and drinking water ad libitum.
The lindane (Cat no. RM2651 – technical grade) was procured from HiMedia, Mumbai, Maharashtra, India. Lindane stock solution was prepared by dissolving 0.5 mg of lindane in 1 ml of groundnut oil. The solution was kept in refrigerator and was brought to room temperature. The dose was administered orally by gavage method. Escherichia coli LPS (0127: B8) was purchased from Sigma Aldrich Chemicals Pvt. Ltd (St Louis, Missouri, USA; cat no. L-3129). The solution of LPS was prepared by dissolving 2 mg of LPS in 2 ml of pyrogen free 0.9% normal saline solution (NSS). The xylazine–ketamine combination anaesthetic solution was prepared by mixing 0.5 ml (10 mg) xylazine (Indian Immunologicals Limited, India: Xylazine®) and 2 ml (100 mg) ketamine (Aneket®) in 7.5 ml of isotonic normal saline.
Experimental design
Mice were randomly divided into two broad groups, namely, treatment and control group (n = 20 each). Treatment group was administered lindane orally dissolved in groundnut oil at 0.25 mg kg−1 BW for 60 days and control group was administered groundnut oil. Ten animals from treatment and control group were challenged with LPS at 80 μg per mice and remaining with NSS at 80 μg per mice by intranasal route on day 61. For LPS/NSS challenge, mice were first anaesthetized by intraperitoneal administration of anaesthetic at 10 mg xylazine + 100 mg ketamine kg−1 BW (0.03 µl 10−1 gm BW for anaesthesia) and solution was instilled in one nostril of mouse by using blunt tuberculin syringe.
Mice from all groups were anaesthetized by administration of anaesthetic at 10 mg xylazine + 100 mg ketamine kg−1 BW (0.1 µl 10−1 gm BW of mice for euthanasia; intraperitoneally) after 16 h of LPS/NSS administration and humanely killed.
Collection of blood and BAL fluid
The mice were anaesthetized with actual dose (0.1 µl 10−1 gm BW) of xylazine ketamine combination after 16 h of LPS challenge. After anaesthesia blood was collected in ethylenediaminetetraacetic acid (EDTA)-coated vial by cardiac puncture using 1 ml tuberculin syringe. The blood was processed for total leukocyte count (TLC) and differential leukocyte count (DLC) analysis on the same day. For collection of broncheo-alveolar lavage (BAL) fluid, the thoracic cavity of mice was opened to expose the lungs. Trachea was separated from connective tissue up to its bifurcation. Right lung was ligated to prevent its flushing. A small nick was given on trachea. Blunt-ended scalp vein set of 26G was inserted through this nick and was fixed using cotton thread. BAL fluid was collected by inflating the left lung instilling 0.3 ml of phosphate buffer saline. The process was repeated three times to collect almost 0.9 ml of BAL fluid volume from left lung. The collected BAL fluid was subjected to TLC and DLC analysis on the same day.
TLC and DLC analyses
About 380 µl of the white blood cell (WBC) diluting fluid was taken in glass vial. About 20 µl of the blood was added and mixed properly. Cells were counted in four squares of chambers of haemocytometer under 40X magnification. TLC was calculated after putting the values in the formula given below:
For DLC, a drop of blood was taken on a slide and a clear blood smear was made with another slide, keeping it an angle of 45°. Smear was air dried and stained with Leishman stain. After 30–45 s, buffer was added to the slides and was mixed well. It was kept for 10–12 min. Slides were washed in running tap water and were kept for drying. About 100 cells per sample were counted under microscope at 40× and expressed in percentage.
H&E staining and immunohistochemistry of lung tissue
The tissue samples of lungs from all groups were collected in 10% neutral buffered formalin for fixation for 24 h at 4°C. Tissues were washed overnight in running tap water, dehydrated in ascending grades of alcohol and cleared in benzene. The 4–5 μm thick tissue sections were obtained on poly-L-lysine-coated clean glass slides.
The paraffin sections of the lungs from all the animals were stained with haematoxylin and eosin (H&E) staining for routine histopathology. The protocol for immunohistochemistry was same as described earlier by Sethi et al. 21 Briefly, tissues were deparaffinized, antigen retrieval was performed by keeping slides in boiling Tris-EDTA buffer for 5 min and endogenous peroxidase was blocked by 3% hydrogen peroxide in methanol and blocking of nonspecific sites with 1% bovine serum albumin. The sections were treated with primary antibody (TLR-4 – 1:200 and TNF-α – 1:2000; Santa Cruz Biotechnology, Santa Cruz, California, USA) followed by specific biotinylated secondary antibody (1:100; DAKO Labs, USA) at room temperature to immunolocalize TLR-4 and TNF-α. The reaction was visualized using a colour development kit (Vector Laboratories, Inc. Burlingame, California, USA). The sections were also counterstained with haematoxylin. Immunohistochemical controls included incubation with normal goat immunoglobulin G or omission of primary antibodies. The sections were also treated with primary antibody for rabbit anti-Von Willebrand factor (VWF; 1:6000; Sigma) to visualize the endothelial linings of blood vessels.
The TLC and DLC data from control and treatment group was compared by one-way analysis of variance followed by Tukey’s test. The value of p < 0.05 was considered significant.
All applicable international, national and/or institutional guidelines for the care and use of animals were followed. All procedures performed in studies involving animals were in accordance with the ethical standards of the IAEC, Guru Angad Dev Veterinary and Animal Sciences University, Ludhiana, at which the studies were conducted.
Results
Blood and BAL fluid analysis
Lindane and LPS alone or in combination resulted increase in TLC of blood compared to control but was not significant statistically (Figure 1(a)). However, there was significant increase (p < 0.05) in the TLC of BAL fluid following exposure to lindane and/or LPS compared to control group (Figure 2(a)). LPS resulted more significant increase (p < 0.05) in TLC compared to lindane, however, lindane combined with LPS showed significantly higher TLC (p < 0.05) than lindane or LPS alone.

(a) Total leukocyte count of blood of control and lindane-exposed mice. (b) Lymphocyte count. (c) Neutrophil count of BAL fluid of control and lindane-exposed mice. *Means with at least one common superscript do not differ significantly as per the one-way ANOVA (p < 0.05). ANOVA: analysis of variance; BAL: broncho-alveolar lavage.
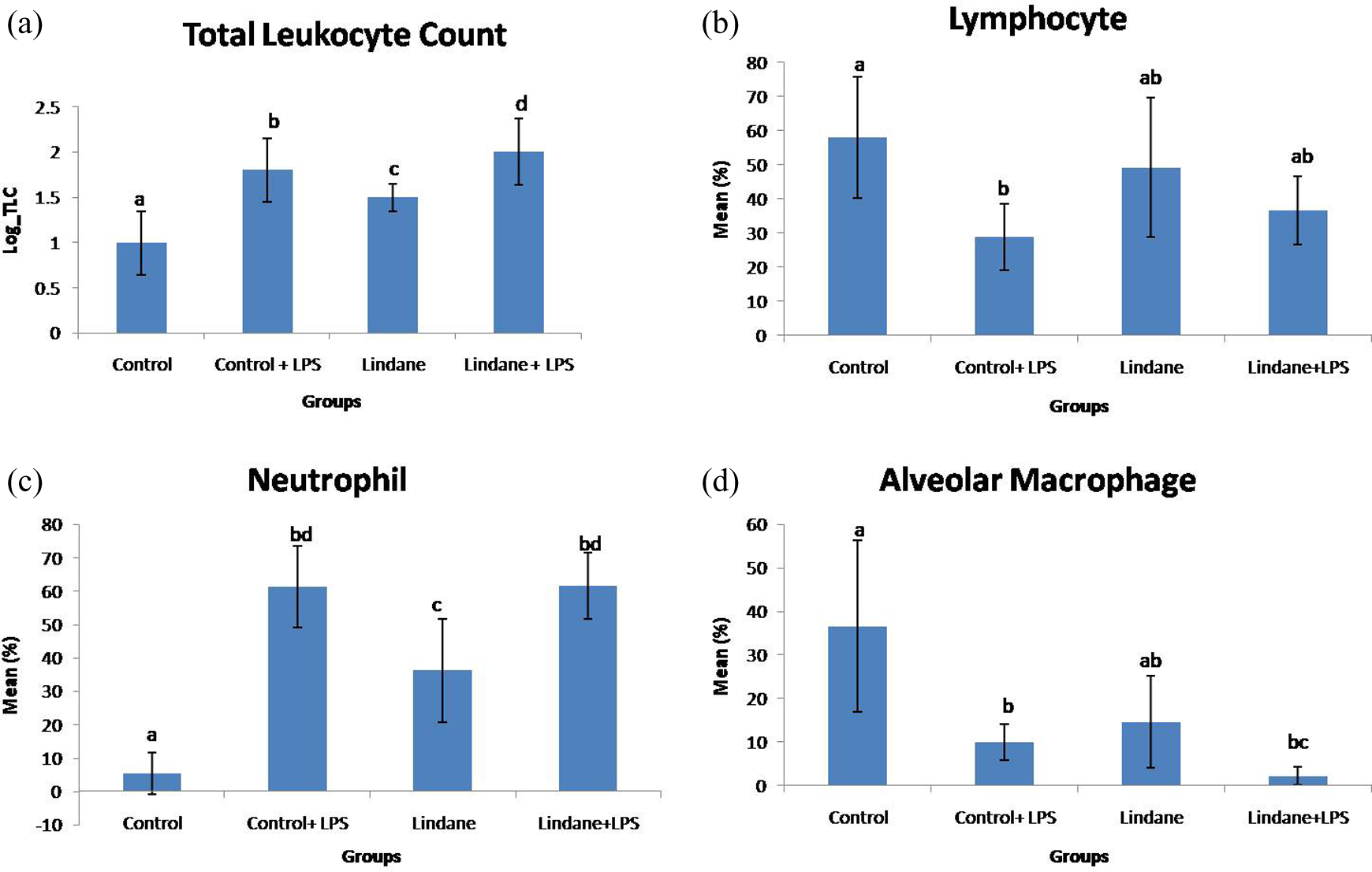
(a) Total leukocyte count of BAL fluid of control and lindane-exposed mice. (b) Lymphocyte count. (c) Neutrophil count and (d) alveolar macrophage count of BAL fluid of control and lindane-exposed mice. *Means with at least one common superscript do not differ significantly as per the one-way ANOVA (p < 0.05). ANOVA: analysis of variance; BAL: broncho-alveolar lavage.
DLC analysis of BAL fluid revealed that LPS resulted significantly reduction (p < 0.05) in the number of lymphocyte compared to control group (Figure 2(b)). Further there was no significant difference in the number of lymphocytes following exposure to lindane alone or in combination with LPS compared to control. There was a significant increase (p < 0.05) in the number of neutrophils following exposure to lindane alone, LPS or both lindane and LPS compared to control (Figure 2(c)). However, the increase in number of neutrophils following exposure to lindane was significantly less (p < 0.05) than LPS. There was significant reduction in alveolar macrophage following LPS challenge compared to control group and lindane exposure did not alter the number of alveolar macrophages (Figure 2(d)). However, when combined with LPS, lindane resulted significant decrease (p < 0.05) in the alveolar macrophages compared to control.
Histopathological observations
The control group showed normal histo-architecture of lung with residential alveolar macrophages (Figure 3(a) and (b)). LPS exposure caused aggregation of mononuclear cells in perivascular and peribronchial area (Figure 3(c)). Lindane also showed peribronchial and perivascular infiltration of mononuclear cells (Figure 3(d)). However, peribronchial and perivascular infiltration became more intense in the lindane combined with LPS group (Figure 3(e) and (f)). There was occasional congestion of blood vessels and presence of inflammatory cell-like neutrophils in the perivascular space (Figure 3(f)).
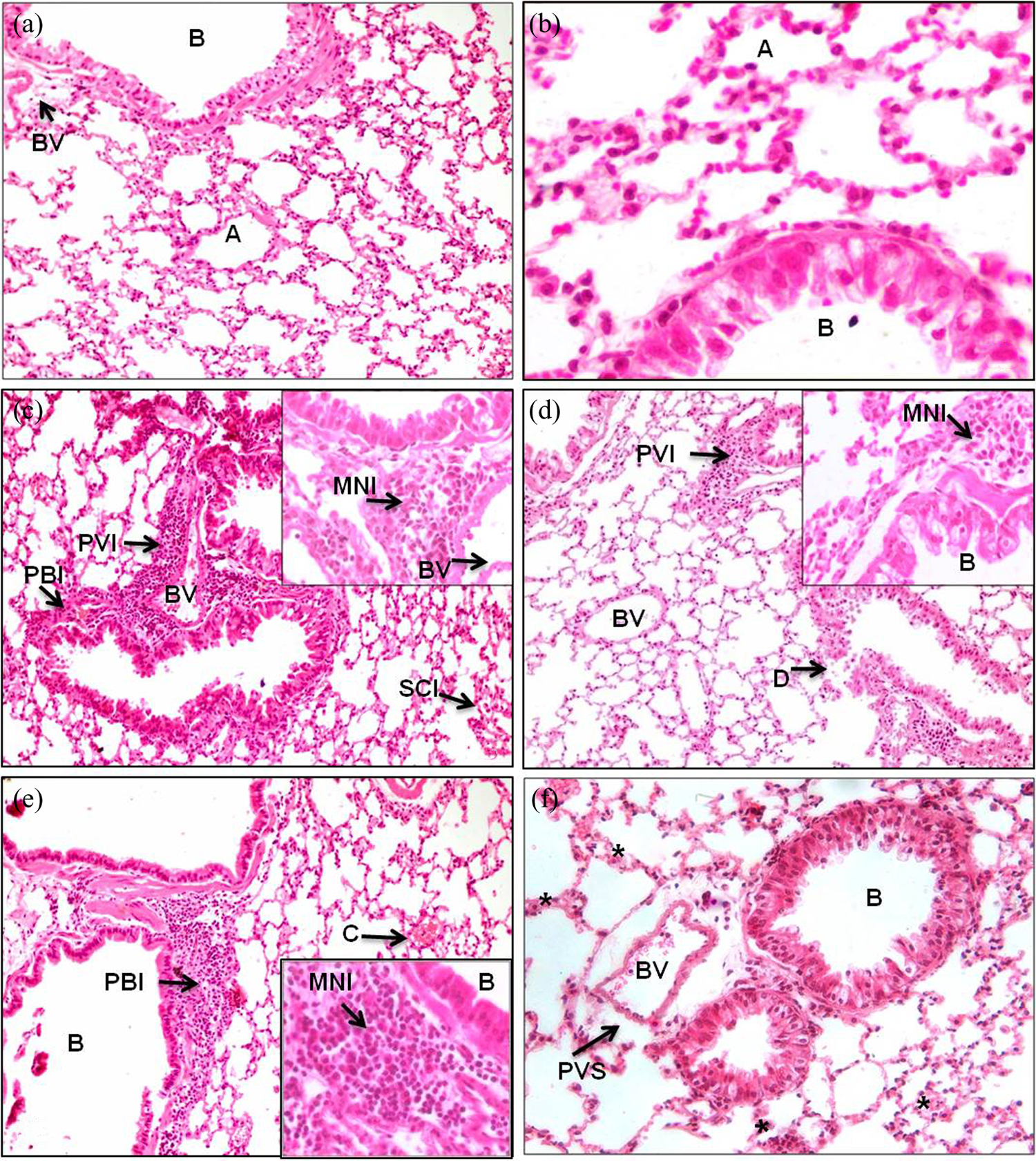
(a) and (b) Lung of control mouse challenged with NSS showing normal architecture (B: bronchiole; A: alveoli; BV: blood vessel). (c) Lung of control mouse challenged with LPS showing PVI, PBI and SCI. (d) Lung of lindane-treated mouse challenged with NSS showing PVI and damaged bronchial epithelium (d). (e) Lung of lindane-treated mouse challenged with LPS showing infiltration of PMNs around perivascular and peribronchial area along with congestion (c). (f) Lindane-treated mouse challenged with LPS showing thickening of alveolar septa (*) and widening of perivascular space. PVI: perivascular infiltration; PBI: peribronchial infiltration; SCI: septal cell infiltration; PMN: polymorphnuclear cell. (All figures are at 20X and insets are at 40X; H&E staining.)
Immunohistochemical observations
The omission of primary and/or secondary antibody (Figure 4(a)) resulted in lack of colour reaction in the lung tissues. Intact endothelial lining of blood vessels of control group was visualized by VWF marker in Figure 4(b).
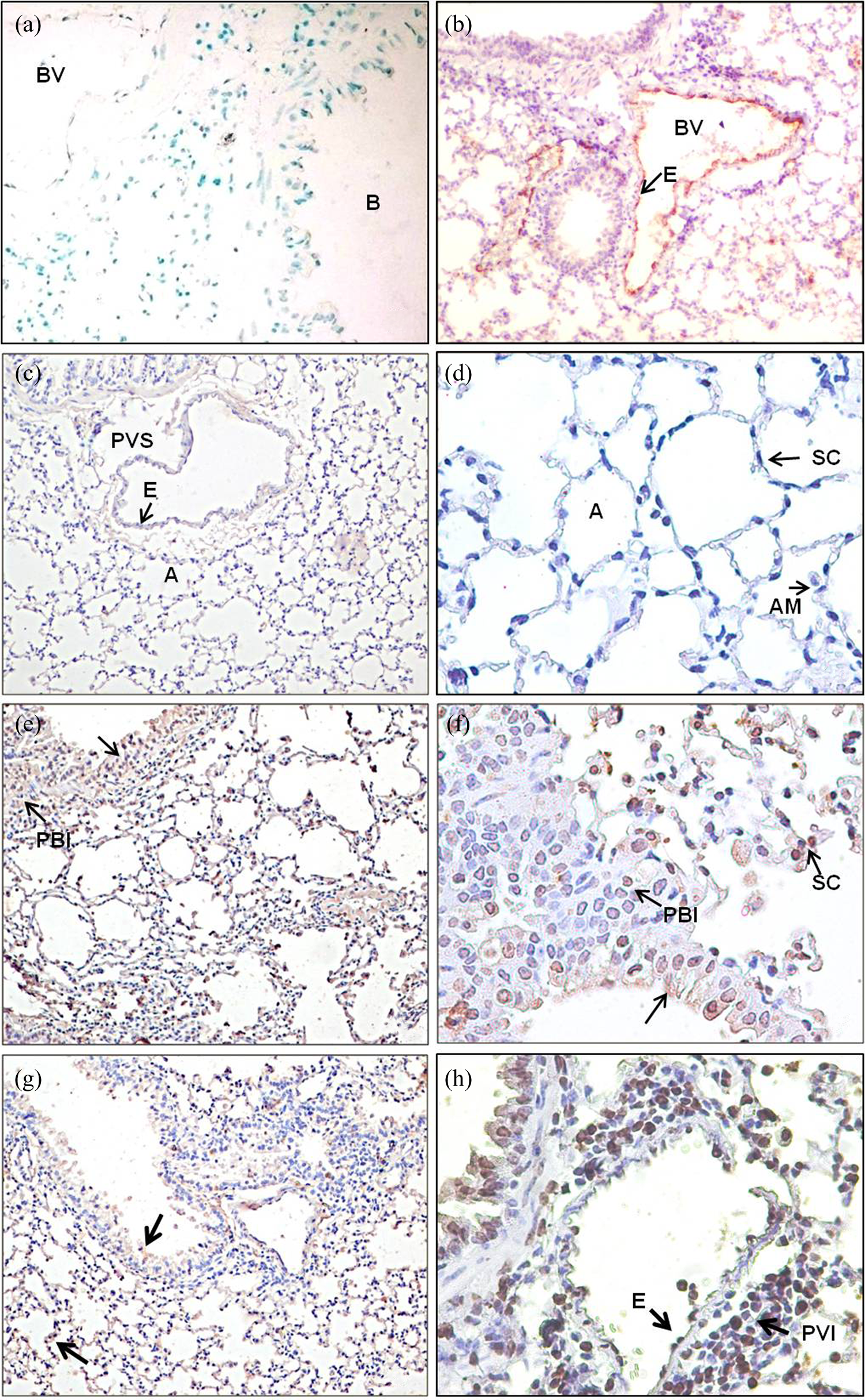
(a) Negative control. (b) Endothelial lining (E) of blood vessel (BV) of control group mice stained with VWF marker. (c) and (d) Lung of control mouse challenged with NSS showing minimal or no reaction. (e) and (f) Lung of control mouse challenged with LPS showing expression of TLR-4 on epithelial airways and septal cells (black arrow). There was presence of PBI cells showing positive reaction LPS challenge resulted in increased expression of TLR-4. (g) and (h) Lung of lindane-treated mouse challenged with NSS showing expression of TLR-4 in luminal side of airways epithelium (black arrow) and SCs (Figure (g)-lower arrow); PBI cells and endothelium of blood vessel showing positive reaction for TLR-4, inset in (g) showing infiltration of cells towards luminal side. (i) and (j) Lung of lindane-treated mouse challenged with LPS showing intense expression of TLR-4 in luminal side of airways epithelium (black arrow) and SCs; Presence of PBI and PVI cells showing positive reaction for TLR-4. PBI: peribronchial infiltration; A: alveolus; PVI: perivascular infiltrative; PVS: perivascular space; AM: alveolar macrophage; TLR: Toll-like receptor; SC: septal cells; LPS: lipopolysaccharide; VWF: Von Willebrand factor. (All figures are at 20× and 40×.)
TLR-4: In present study, control group mice showed mild expression of TLR-4 in alveolar epithelium and large septal cells (Figure 4(c) and(d)). LPS treatment resulted into increased expression of TLR-4 on epithelial airways and septal cells (Figure 4(e) and(f)). Administration of lindane resulted intense immunopositive TLR-4 expression in airways epithelium especially towards luminal side and in infiltrating peribronchiolar cells and large septal cells (Figure 4(g) and(h)). Lindane and LPS treatments together also caused intense TLR-4 staining in airways, alveolar septa and the vascular endothelial cells (Figure 4(i) and(j)).
TNF-α: There was occasional TNF-α immunopositive reaction in the airway epithelium and alveolar septal cells in lungs of control mice (Figure 5(a) and(b)). However, LPS challenge resulted comparatively strong reaction in epithelial cells of airways and septal cells (Figure 5(c) and(d)). Further, the reaction was more towards luminal side of airways. There was more marked expression of TNF-α in airways epithelium and large septal cells in lindane-treated mice (Figure 5(e) and(f)). Lindane and LPS treatments together show significant difference for expression of immunopositive TNF-α reaction in large septal cells (Figure 5(g) and(h)).

(a) and (b) Lung of control mouse challenged with NSS showing minimal reaction; few septal cells showed expression of TNF-α in control group mice whereas alveolar epithelium did not show any staining. (c) and (d) Lung of control mouse challenged with LPS showing expression of TNF-α on epithelial airways (black arrow) and SCs; presence of PBI cells and AM showing positive reaction. (e) and (f) Lung of lindane-treated mouse challenged with NSS showing expression of TNF-α in luminal side of airways epithelium (black arrow) and large SCs; PVI and PBI cells. (g) and (h) Lung of lindane-treated mouse challenged with LPS showing intense expression of TNF-α in luminal side of airways epithelium (black arrow) and SCs. Presence of PBI cells showing positive reaction for TNF-α. PBI: peribronchial infiltration; AM: alveolar macrophage; PVI: perivascular infiltrative; AM: alveolar macrophage; TLR: Toll-like receptor; SC: septal cells; LPS: lipopolysaccharide; TNF-α: tumour necrosis factor-alpha. (All figures are at ×20 and ×40.)
Discussion
Lindane is extensively used in agriculture sector to control pests. 8 While there is significant information on the biomedical effects of dermal or oral exposure to lindane, considerably fewer data are available on pulmonary exposure to lindane alone or in conjunction with pathogen-derived molecules such as LPS. Therefore, we used a mouse model to obtain first data on the pulmonary effects of lindane alone or co-exposure with LPS. The data show that lindane alone or in conjunction with LPS induced pulmonary inflammation along with increased expression of TLR-4 and TNF-α.
Lung inflammation is characterized by influx of inflammatory cells such as neutrophils, monocytes and lymphocytes. 22 BAL fluid is an important condition to determine the changes in cellular composition of alveoali. Lindane and LPS alone or both significantly increased the number of total cells and neutrophils but lindane with LPS significantly decreased the number of alveolar macrophages in BAL fluid as reported earlier following exposure to diazinon 23 and LPS. 24,25 Inhalation of LPS results prompt and sustained neutrophilic airway inflammation causing increase in the numbers of neutrophils and total cells in BAL fluid. 26 LPS exposure also replaces alveolar macrophage and induces vigorous recruitment and activation of inflammatory cells in the alveolar space as an effective host defence mechanism against invasion of bacteria and other pathogens. 27 Lindane exposure along with LPS resulted significantly higher TLC compared to lindane or LPS alone suggests their synergistic effect.
Lindane or LPS treatment showed peribronchial, periepithelial and perivascular infiltration of mononuclear cells. Pesticide-induced histopathological alterations in lungs are attributed to decrease in the antioxidant levels. 28 Thiacloprid at 10 mg kg−1 day−1 orally for 28 days result mild congestion and haemorrhage in the lungs in Gallus domesticus. 29 Sub lethal doses of diazinon induce dose-dependent infiltration of the macrophages and mononuclear cells, haemorrhage, congested blood vessels, oedema, pyknosis and necrosis in the lung of guinea pig. 23 High residual amount of deltamethrin in lungs results inflammation leading to progressive emphysema. 30
Lindane combined with LPS showed more intense peribronchial, periepithelial and perivascular infiltration along with congestion of blood vessels and migration of inflammatory cell-like neutrophils from blood vessels to epithelium. Pesticides and bacterial interaction mimic over-expression of certain genes resulting host tissues injury and higher morbidity and mortality rates. 31
TLR-4 plays important role for early and effective clearance of the injured or dead cells, unfolded/misfolded proteins or debris to resolve chronic inflammation and fibrosis following lung injury. 32 LPS challenge resulted increased expression of immunopositive TLR-4 reactivity in airways epithelial and septal cells. LPS induces localization of TLR-4 in alveolar septal cells, bronchial epithelium, macrophages and endothelium of large and peribronchial blood vessels. 33 Lindane alone or in combination with LPS resulted intense immunopositive TLR-4 reaction in airways epithelium and septal cells along with peribronchiolar infiltrative cells. Triggered TLR-4 response results following exposure to dioxin 34 and pentachlorophenol and other organic substances (e.g. atrazine, bisphenol A). 35 Toluene significantly upregulates mRNA production of TLR-4 and pro-inflammatory cytokines. 36 TLR-4 activation promotes the self-defense of epithelium and protects from lung injury through TLR-4-dependent basal activation of NF-B. 37,38
Lindane or LPS increased TNF-α immunopositive reaction in epithelial cells of airways and septal cells of lungs. High level of TNF-α is reported among insecticides-exposed workers 39 and following administration of bacterial endotoxin. 40 Xenobiotic including insecticides and endotoxin induces oxidative stress by production of reactive oxygen species (ROS), 41 and TNF-α stimulates generation of ROS from pulmonary and non-pulmonary tissues. 17
Lindane exposure followed by LPS challenge resulted more TNF-α immunopositive reaction in large septal cells. Chlorpyrifos or its metabolites alter the production of cytokine like IFN-γ in the presence LPS-like immunogens. 42 Chronic exposure to low levels of the organothiophophate insecticide (acephate) enhances responses to LPS-induced induction of pro-inflammatory cytokines, namely, IL-1β, TNF-α and IFN-γ in rats. 43
We conclude that lindane exposure resulted lung inflammation and increased immunopositive expression of TLR-4 and TNF-α. Further combination with LPS marked the lung injury and expression of immunopositive TNF-α activity in large septal cells. The present study do not reveal mechanism behind such increase in the expression of TLR-4 and TNF-α but suggests that lindane exposure may also modulate pulmonary inflammatory responses to other farm exposures such as endotoxin. The data also indicate more studies should be directed specially at transcriptional level to arrive at some logistic conclusion.
Footnotes
Acknowledgements
The authors are thankful to Director of research and Department of Veterinary Medicine, Guru Angad Dev Veterinary and Animal Sciences, University for providing facilities for the research. The first author is thankful to Department of Science and Technology Government of India, for providing INSPIRE scholarship during the PhD.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This work was financially supported by School of Public Health and Zoonoses, GADVASU, Ludhiana, Punjab.

