Abstract
The incidence of nonalcoholic fatty liver disease (NAFLD) is considered a risk factor for hepatic fibrosis. Therefore, there is critical need to develop novel cheap and effective therapeutic approaches to prevent and reverse NAFLD. Caffeine is commonly consumed beverage and has antioxidant and anti-inflammatory activities. This study examined whether caffeine can ameliorate liver injury induced by high-fat diet (HFD) feeding. Four groups of rats were used and treated for 16 weeks as follows: control group, rats were fed a standard diet; HFD group, rats were fed HFD; and caffeine 20 and caffeine 30 groups, rats were fed HFD for 16 weeks in addition to different doses of caffeine (20 or 30 mg/kg, respectively) for last 8 weeks. The HFD-induced liver injury is determined biochemically by evaluating serum alanine aminotransferase (ALT), aspartate aminotransferase (AST), albumin, bilirubin, triglycerides, cholesterol, and high-density lipoprotein-cholesterol and by histopathological examination. Tissue malondialdehyde, total nitrate/nitrite, and glutathione concentration were also measured. Real-time reverse transcription polymerase chain reaction technique was used to determine the expression of lipogenic enzyme genes. Caffeine treatment significantly decreased the elevated serum ALT, AST, and bilirubin and increased the reduced albumin level. Interestingly, the hepatic mRNA expression of Fatty acid synthase and acetyl CoA carboxylase was decreased by caffeine, while the protein expression of hepatic carnitine palmitoyltransferase 1 and proliferation-activated receptor α was increased. Furthermore, caffeine reduced tissue lipid peroxidation and oxidative stress. These effects suggest that caffeine could improve HFD-induced hepatic injury by suppressing inflammatory response and oxidative stress and regulating hepatic de novo lipogenesis and β-oxidation.
Introduction
Nonalcoholic fatty liver disease (NAFLD) is worldwide liver disease. It has a wide range of manifestations starting from fat accumulation in liver (steatosis) to nonalcoholic steatohepatitis and cirrhosis. 1 The hallmark of NAFLD is disruption of synthesis, and metabolism of fatty acids and triglycerides (TG) with increasing predisposition of the liver to injury mediated by release of inflammatory cytokines and oxidative stress, which lead to steatohepatitis. 2
Treatment of NAFLD is recently directed toward several strategies that manage the body weight, reduce oxidative stress and insulin resistance, and optimize lipid profile. Caffeine has drawn great attention for its ability to target NAFLD treatment strategies through the hepatoprotective effect of caffeine on chronic liver diseases. 3 Recent study has shown that intrahepatic lipid content is reduced and hepatic β-oxidation is induced by caffeine, and also it reduces TG and cholesterol levels by stimulation of lipolysis and suppression of lipogenesis. 4
This study was presented to estimate the effect of caffeine consumption in different doses (20 or 30 mg/kg) on the development of NAFLD prompted by high-fat diet (HFD) and investigate the underlying mechanism. Serum biochemical parameters, liver oxidative parameters, histopathological changes, and mRNA expression levels of lipogenic enzymes were determined.
Materials and methods
Drugs and chemicals
Caffeine was purchased from Sigma-Aldrich (St Louis, Missouri, USA). The HFD components were purchased from local commercial sources. All other chemicals and reagents used were of analytical grade.
Animals
Forty male Wistar rats, weighing 50–95 g, were obtained from “Egyptian Organization for Biological Products and Vaccines,” Agouza, Giza, Egypt. The animals were kept at room temperature with 12-h light/12-h dark cycle. They were given constant conditions during the experimental period in accordance with the procedure. The experiments comply with the guiding principles for the care and the use of laboratory animals adopted by the “Research Ethics Committee,” Faculty of Pharmacy, Mansoura University.
Experimental design
After 1 week of acclimatization to experimental conditions, the rats were randomly assigned into four groups of 10 rats/group and treated as follows for 16 weeks: (1) the control (CTRL) group, rats were fed a standard diet and received the vehicle; (2) HFD group or group 2, rats were fed HFD and received the vehicle; and groups 3 and 4, animals were fed HFD and received caffeine at 20 or 30 mg/kg/day orally, respectively, for last 8 weeks. The HFD consisted of 45% lipid, 30% carbohydrate, and 25% protein. 5
Serum and tissue sampling
Rats were anesthetized by thiopental sodium. Cardiac puncture was made to collect blood samples using syringe and put immediately into Weatherman tubes. Centrifugation of these samples was carried out at 3000 rpm for 15 min; then, serum was obtained. Liver tissues were collected and separated into three parts. The first part was kept in RNA later for RNA extraction and real-time reverse transcription polymerase chain reaction (RT-PCR) analysis. Second part was used for preparation of liver homogenate and kept at −80°C for analysis of oxidative stress markers. The last part was put in 10% formalin and processed for histopathological evaluation and preparing of paraffin blocks.
Assessment of serum biochemical parameters
There are important biochemical parameters that were used to estimate the liver function. These parameters include alanine aminotransferase (ALT), aspartate aminotransferase (AST), bilirubin, albumin, and serum lipids, that is, TG, cholesterol, and high-density lipoprotein-cholesterol (HDL-C). These parameters were estimated in serum by using diagnostic kits purchased from Biomed Diagnostics, Egy-Chem (Badr City, Egypt).
Measurement of oxidative stress parameters
Liver tissues were suspended in a physiological saline containing heparin to prevent blood clots. Homogenization of liver tissues (10% w/v) in 20 mmol/L Tris-HCl was carried out and after that the homogenates were centrifuged at 3000 × g for 20 min at 4°C. The resultant supernatants were collected for assaying some liver homogenate biomarkers such as malondialdehyde (MDA), total nitrate/nitrite (NOx), and reduced glutathione. The MDA was assessed as a tissue lipid peroxidation marker according to the method described by Ohkawa et al. 6 Total NOx products were assessed according to the technique of Miranda et al. 7 Liver reduced glutathione (GSH) content was analyzed according to the method of Moron et al. 8
RNA extraction and RT-PCR
Fatty acid synthase (FAS), acetyl CoA carboxylase (ACC), carnitine palmitoyltransferase 1 (CPT1), and peroxisome proliferation-activated receptor α (PPARα) mRNA expression were analyzed by using RT-PCR. Trizol reagent was used to isolate total RNA from the frozen tissues according to the manufacturer’s protocol (Promega, Madison, Wisconsin, USA). mRNAs were used to generate cDNA and were incubated in the thermal cycler instrument (Arktik Thermal Cycler, Foster City, California, USA) which is programmed to conduct 40 cycles. The primers were designed and purchased (Vivantis Technologies, Malaysia) in the following sequences:

Histological examination
Standard histopathological techniques were followed for processing the tissue and preparation of paraffin blocks. Steatosis changes were evaluated in sections (5 μm thick) stained with Masson’s trichrome and hematoxylin–eosin, respectively, in a blind manner using the histological scoring system of Brunt and associates in 1999. 9
Statistical analysis
The experimental data are expressed as mean ± SEM. Significant difference was calculated at p < 0.05. Data were analyzed by one-way analysis of variance followed by followed by Tukey’s range test using Prism V 5.00 (GraphPad Software Inc, San Diego, California, USA).
Results
Effect of HFD-induced hepatic steatosis on biochemical parameters
The administration of HFD to rats for 16 weeks produced significant increase in serum ALT, AST, and total bilirubin activities and significant reduction in serum albumin concentration (p < 0.05), in comparison with the CTRL group. A significant decrease in serum ALT, AST, and total bilirubin was observed in all groups treated with caffeine in comparison with HFD-fed group with the caffeine dose of 30 mg/kg caused significant increase in level of serum albumin, compared with HFD group (Table 1).
Effect of caffeine consumption on liver functions in HFD-induced hepatic steatosis.a

CTRL: control group; HFD: high-fat diet-fed group; Caff (20 mg/kg): caffeine (20 mg/kg)-treated group; Caff (30 mg/kg): caffeine (30 mg/kg)-treated group; ALT: aminotransferase; AST: aspartate aminotransferase.
aValues represent the mean ± SEM of 5 animals. Rats were fed HFD, which consists of 45% lipid, 30% carbohydrate, and 25% protein, for 16 weeks. Caffeine-treated groups were fed HFD for 16 weeks in addition to caffeine in different doses (20 or 30 mg/kg/day, respectively, oral) for last 8 weeks. Data were statistically analyzed using one-way analysis of variance followed by Tukey–Kramer multiple comparisons test.
b p < 0.05 compared to CTRL group.
c p < 0.05 compared to HFD group.
Also, HFD feeding to rats for 16 weeks caused significant increase in serum cholesterol and triglyceride level and a significant decrease in serum HDL-C level of group 2 rats, compared with the control group. Treatment with caffeine decreased serum cholesterol and triglyceride level and increased serum HDL-C level in groups 3 and 4 animals (Table 2).
Effect of caffeine consumption on lipid profile in HFD-induced hepatic steatosis.a

CTRL: control group; HFD: high-fat diet-fed group; Caff (20 mg/kg): caffeine (20 mg/kg)-treated group; Caff (30 mg/kg): caffeine (30 mg/kg)-treated group; HDL-C: high-density lipoprotein-cholesterol.
aValues represent the mean ± SEM of 5 animals. Rats were fed HFD, which consists of 45% lipid, 30% carbohydrate, and 25% protein, for 16 weeks. Caffeine-treated groups were fed HFD for 16 weeks in addition to caffeine in different doses (20 or 30 mg/kg/day, respectively, oral) for last 8 weeks. Data were statistically analyzed using one-way analysis of variance followed by Tukey–Kramer multiple comparisons test.
b p < 0.05 compared to CTRL group.
c p < 0.05 compared to HFD group.
Effect on oxidative stress parameters in HFD-induced hepatic steatosis
The HFD feeding promoted lipid peroxidation because the MDA and NOx levels in liver were significantly increased in HFD only-treated group (p < 0.05). A significant reduction in hepatic MDA and NOx content was observed in all groups treated with caffeine, compared with the HFD only-treated group. Hepatic GSH content was significantly reduced in rats fed with HFD for 16 weeks. All caffeine-treated groups showed significant increase in hepatic GSH content, when compared to HFD-fed group (Table 3).
Effect of caffeine consumption on oxidative stress parameters in HFD-induced hepatic steatosis.a

CTRL: control group; HFD: high-fat diet-fed group; Caff (20 mg/kg): caffeine (20 mg/kg)-treated group; Caff (30 mg/kg): caffeine (30 mg/kg)-treated group; MDA: malondialdehyde; NOx: nitrate/nitrite; GSH: glutathione.
aValues represent the mean ± SEM of 5 animals. Rats were fed HFD, which consists of 45% lipid, 30% carbohydrate, and 25% protein, for 16 weeks. Caffeine-treated groups were fed HFD for 16 weeks in addition to caffeine in different doses (20 or 30 mg/kg/day, respectively, oral) for last 8 weeks. Data were statistically analyzed using one-way analysis of variance followed by Tukey–Kramer multiple comparisons test.
b p < 0.05 compared to CTRL group.
c p < 0.05 compared to HFD group.
Effect on hepatic expression of ACC, FAS, PPARα, and CPT genes in HFD-induced hepatic steatosis
As shown in Figures 1 and 2, the mRNA expression of ACC and FAS was significantly increased, whereas CPT and PPARα levels were significantly reduced in the HFD group in comparison with the CRTL group. All groups treated with caffeine significantly increased hepatic CPT1 and PPARα expression, compared with the HFD group. The expression of ACC was significantly lower in groups treated with caffeine in doses of 20 and 30 mg/kg than in HFD group. Moreover, the expression of FAS was significantly decreased in caffeine (30 mg/kg)-treated group than in the HFD group.

Effect of caffeine consumption in hepatic expression of (a) FAS and (b) ACC in HFD-induced hepatic steatosis. Values represent the mean ± SEM of 5 animals. Rats were fed HFD, which consists of 45% lipid, 30% carbohydrate, and 25% protein, for 16 weeks. Caffeine-treated groups were fed HFD for 16 weeks in addition to caffeine in different doses (20 or 30 mg/kg/day, respectively, oral) for last 8 weeks. CTRL group: control group, HFD: high-fat diet-fed group, Caff (20 mg/kg): caffeine (20 mg/kg)-treated group, Caff (30 mg/kg): caffeine (30 mg/kg)-treated group. Data were statistically analyzed using one-way analysis of variance followed by Tukey–Kramer multiple comparisons test. *p < 0.05 compared to CTRL group; #p < 0.05 compared to HFD group. FAS: fatty acid synthase; ACC: acetyl CoA carboxylase.
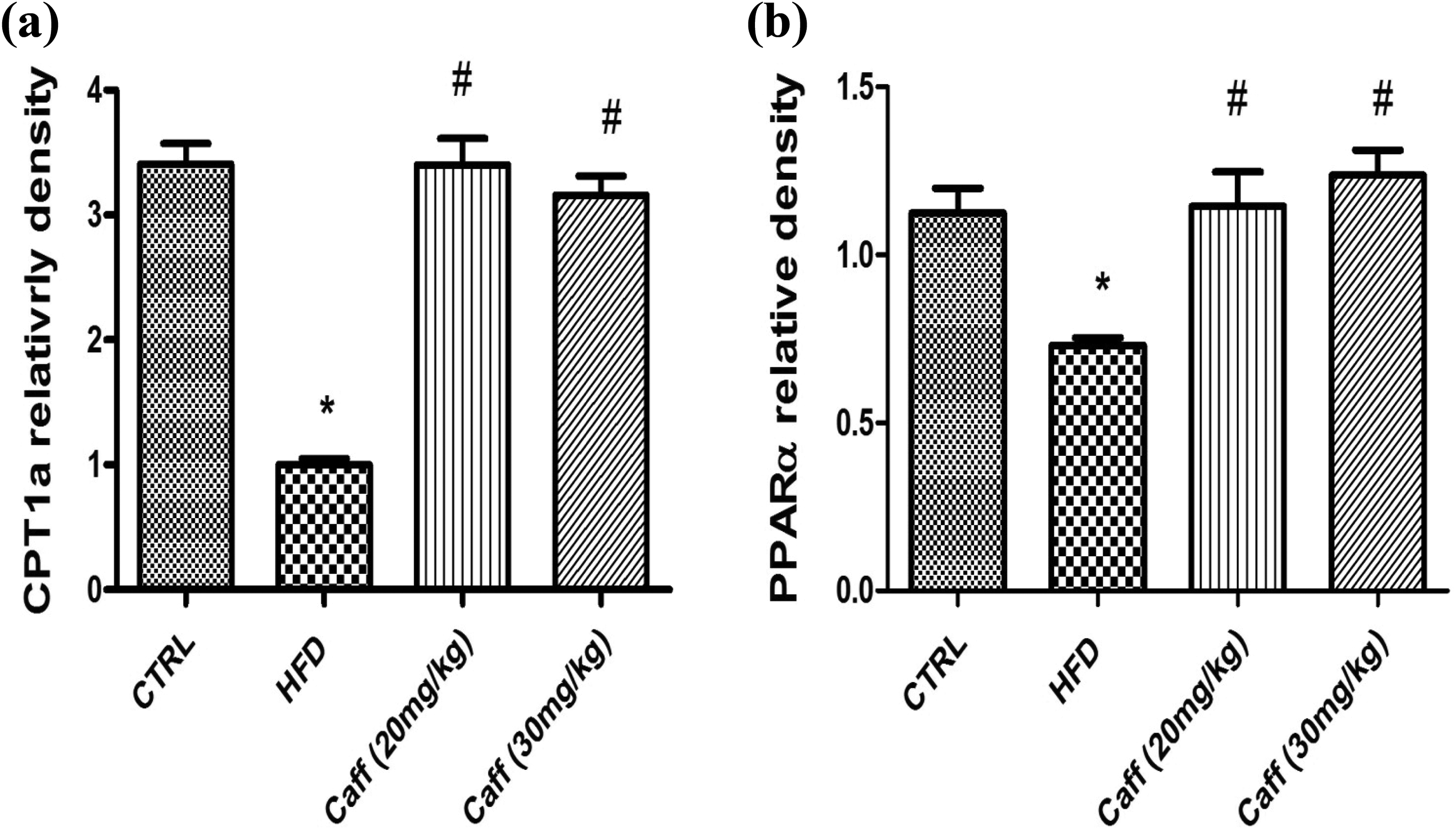
Effect of caffeine consumption in hepatic expression of (a) CPT1 and (b) PPARα in HFD-induced hepatic steatosis. Values represent the mean ± SEM of 5 animals. Rats were fed HFD, which consists of 45% lipid, 30% carbohydrate, and 25% protein, for 16 weeks. Caffeine-treated groups were fed HFD for 16 weeks in addition to caffeine in different doses (20 or 30 mg/kg/day, respectively, oral) for last 8 weeks. CTRL group: control group, HFD: high-fat diet-fed group, Caff (20 mg/kg): caffeine (20 mg/kg)-treated group, Caff (30 mg/kg): caffeine (30 mg/kg)-treated group. Data were statistically analyzed using one-way analysis of variance followed by Tukey–Kramer multiple comparisons test. *p < 0.05 compared to CTRL group; #p < 0.05 compared to HFD group. CPT1: carnitine palmitoyltransferase 1; PPARα: peroxisome proliferation-activated receptor α.
Effect on histological changes of the liver tissues in HFD-induced hepatic steatosis
The HFD groups showed marked macrovesicular steatosis with or without foci of mild lobular or portal inflammation, hepatocyte injuries with hepatocellular ballooning, and inflammation with minimum fibrosis. Caffeine administration to HFD-fed rats in different doses decreased steatosis, ballooning, and inflammation when compared to the HFD group (Figure 3).
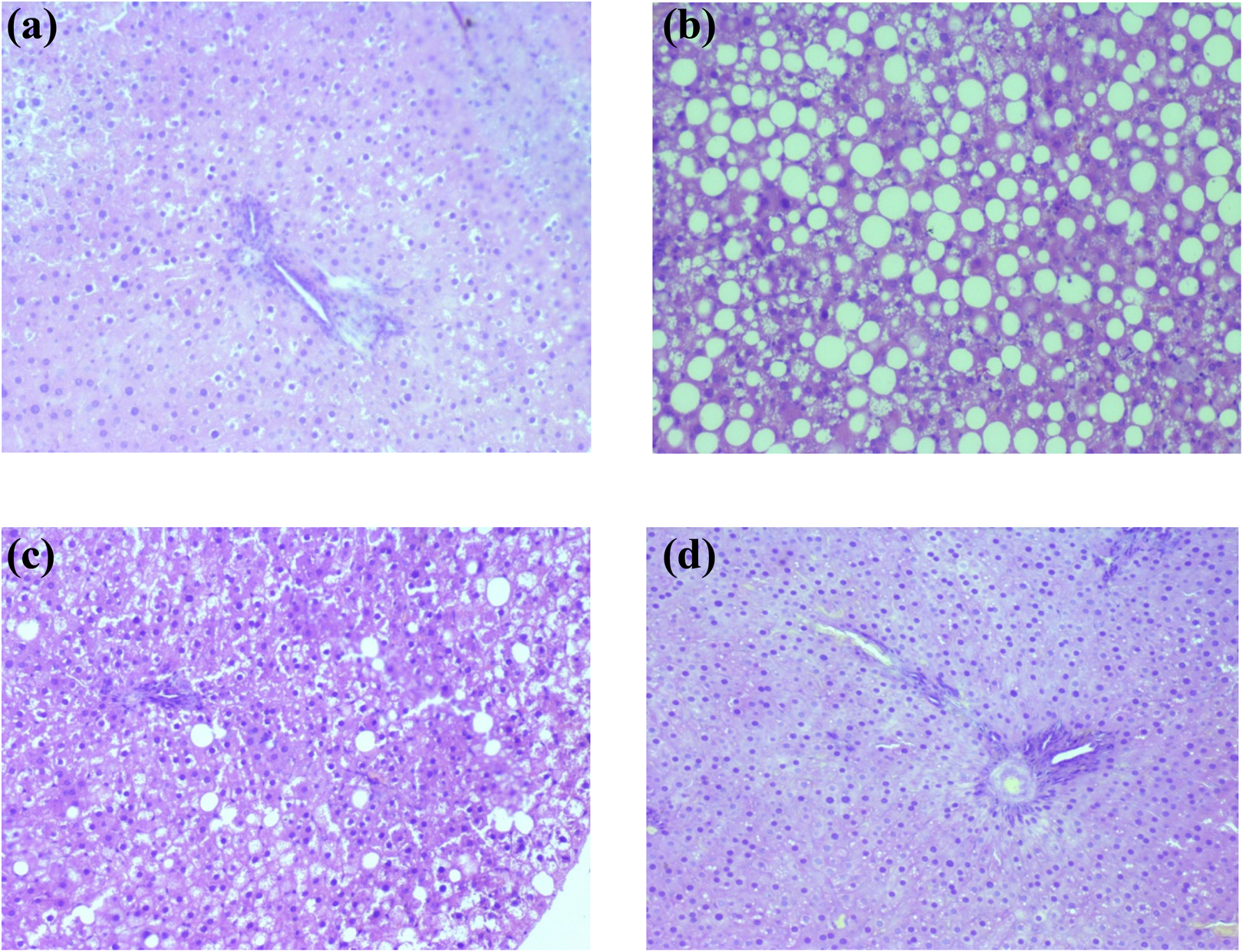
Effects of caffeine consumption on histopathological changes in HFD-induced hepatic steatosis in rats histopathology images representing liver sections stained with hematoxylin–eosin isolated from (a) control group (200×) and (b) HFD-fed group showed marked macrovesicular steatosis with or without foci of mild lobular or portal inflammation, hepatocyte injuries with hepatocellular ballooning, and inflammation (100×), (c) caffeine (20 mg/kg)-treated group showed minimal inflammatory reactions and steatosis (200×), and (d) caffeine (20 mg/kg)-treated group showed minimal portal inflammatory reactions (200×). HFD: high-fat diet.
Discussion
Consumption of HFD by rats for 16 weeks in the present study induced hepatocellular injury and hepatic steatosis, and this was confirmed by increased serum bilirubin and albumin levels, AST and ALT activities, serum lipid disorders, liver histopathological changes, and increased level of oxidative stress parameters. These findings are in line with that obtained by Gauthier et al., who has reported that consumption of diet containing 45% fat for 16 weeks causes hepatic steatosis, liver damage, hypertriglyceridemia, and oxidative stress similar to NAFLD. 10
Results of the present study revealed that caffeine administration to HFD-fed rats improved the signs of hepatic injury and hepatic steatosis, attenuated the production of hepatic FAS and ACC expression, and stimulated the hepatic expression of PPARα and CPT1. FAS appear to participate in hepatic lipid metabolism both by stimulation of de novo lipogenic activity in the liver in case of feeding with HFD, by promoting β-oxidation of fatty acids through activation of PPARα, and in case of starvation. 11
PPARα is activated in case of energy deprivation under prolonged fasting conditions and is necessary for completion of lipid B-oxidation process. 12 Accordingly, it can be said that caffeine administration to HFD-fed rats had a double impact on hepatic lipid metabolism. Firstly, it directly inhibited FAS activity leading to attenuation of the de novo lipogenic activity of the liver under the conditions of concurrent HFD feeding and Caffeine administration. Secondly, the novel finding of this study is the promoting effect of caffeine on hepatic expression of PPARα and its downstream effectors. It directly stimulated PPARα activity leading to stimulation of the process of lipid B-oxidation. The adaption of the idea of a double antisteatotic effect of caffeine could be supported by our finding that its concurrent administration with HFD feeding had led to inhibition of hepatic ACC activity and stimulation of hepatic CPT1 activity that will lead to decrease in the lipogenic activity of the liver and increase in the rate of consumption of fat as a source of energy, under the conditions of continuous HFD consumption. Supporting these findings, Caffeine was also reported effective in improving liver functions and ameliorating dyslipidemia and metabolic syndrome in previous animal experiments. 13 –15
On the other hand, Sinha et al. find that caffeine stimulated the hepatic lipid autophagy pathway in mice thus stimulates hepatic lipid B-oxidation. 16 Lipid autophagy will afford a huge amount of fatty acids that are ready for B-oxidation to be consumed as an energy source, here comes the role of caffeine-stimulated PPARα and CPT1 activity. Accordingly, it can be said that caffeine has a multifactorial intervention with the process of lipid accumulation in the liver in case of concurrent consumption of HFD.
Accumulated evidence showed a close relationship between PPARα and NAFLD. PPARα is transcription factor which modulates the expression of genes involved in hepatic lipid metabolism and an important downstream targets of AMP-activated protein kinase (AMPK). AMPK is considered as the main regulator of hepatic β-oxidation through activation of every enzymatic step involved in the β-oxidation. Particularly, PPARα activates the expression of CPT1. 17 PPARα also induces the lipolysis through activation of lipoprotein lipase. Upregulation of PPARα not only can prevent accumulation of hepatic fats but also upregulate anti-inflammatory genes that inhibit inflammatory NFκB pathway. 18 Moreover, PPARα directly stimulates the expression of catalase enzyme which neutralizes ROS and protects the liver from oxidative injury. 19 Caffeine has been reported as antagonist of adenosine A1 and A2 receptors and phosphodiesterase inhibitor in moderate to heavy consumers, thereby increasing intracellular cAMP level and, hence, activating AMPK. 20 Caffeine dose used in this study is reported to be approximately corresponding to the amount of caffeine present in 3 to 4 cups of caffeine used daily as a beverage in humans.
Considering oxidative stress role in the NAFLD pathogenesis, it initiates the production of inflammatory cytokines leading to hepatocyteinjury due to lipid peroxidation and inflammation. ROS generation leads to the release of tumor necrosis factor-alpha through hepatocytes, adipose tissue, and Kupffer cells that can upregulate pro-inflammatory pathways. 21 MDA is defined as cytotoxic product of lipid peroxidation that may impair cellular functions and may show hepatic fibrogenesis. 22 Also, in the presence of injury, the hepatocytes is induced to progress to irreversible cell death and necrosis through increased NOx production. 23 GSH shows an integral role in preserving the intracellular redox equilibrium and protects tissues from oxidative stress. 24
In the present study, hepatic MDA levels and hepatic NOx levels were markedly augmented and hepatic GSH level decreased in HFD group, representing increased oxidative stress. In addition, caffeine consumption increased antioxidant enzymes activity and increased GSH level. Also, caffeine administration decreased hepatic MDA and NOx levels. In line with these findings, it has been reported that caffeine protects membranes against damage and reduces peroxidation of the lipid in rat liver. 25 It was reported that Adenosine enhances NOx production through adenosine A2A receptors. Thus, caffeine could antagonize A2A receptors that reduce the generation of NOx. 26
In conclusion, this study revealed that caffeine treatment could attenuate hepatic steatosis and hepatocellular injury induced by HFD through upregulation of β-oxidation, downregulation of de novo lipogenesis, and suppression of oxidative stress.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflict of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
