Abstract
The present study investigated the probable protective effect of vanillin (VLN) against kidney injury induced by cisplatin (CSN) and methotrexate (MTX) in rats. The rats received a single injection of either CSN (7.5 mg/kg, i.p.) or MTX (20 mg/kg, i.p.). VLN treatment (150 mg/kg/day, i.p.) was started 1 day before administration of the nephrotoxic agents and continued for 7 days. Both CSN and MTX significantly increased serum creatinine, cystatin C, and neutrophil gelatinase–associated lipocalin and kidney tissue renal malondialdehyde, inducible nitric oxide synthase, tumor necrosis factor-α, interleukin-18, nuclear factor-κB p65, cytosolic cytochrome C, and caspase-3 and significantly decreased renal total antioxidant capacity and Bcl-2/Bax ratio in rats. VLN significantly ameliorated the changes of biochemical parameters induced by CSN and MTX. VLN also significantly reduced CSN- and MTX-induced histopathological injury and the expression of Fas ligand in rat kidneys. In conclusion, VLN significantly protected against CSN- and MTX-induced kidney injury in rats by inhibiting oxidative/nitrosative stress, inflammation, and apoptosis.
Introduction
Cisplatin (CSN), the alkylating agent, and methotrexate (MTX), the antifolate agent, are commonly used cancer chemotherapeutics. CSN is used for treatment of solid malignancies, including testicular, ovarian, cervical, breast, bladder, lung, and head and neck cancers. 1 MTX is used for leukemia, lymphoma, osteosarcoma, lung, and breast cancers. Additionally, MTX is used as an immunosuppressant in rheumatoid arthritis, psoriasis, and inflammatory bowel diseases. 2 Despite wide clinical uses and high efficacy, nephrotoxicity associated with high morbidity and mortality remains a major and dose-limiting problem of both agents. Acute kidney injury (AKI) and dysfunction occur in about 25–30% of patients receiving CSN therapy and in about 2–12% of patients with hematological malignancies treated with high-dose MTX. 3,4 The pathophysiologic mechanisms of CSN- and MTX-induced nephrotoxicity are complex; however, increased generation of reactive oxygen species (ROS) and reactive nitrogen species (RNS) seems to play a pivotal role. 5,6 Oxidative/nitrative stress increases the production of inflammatory cytokines, as tumor necrosis factor-α (TNF-α), via activation of nuclear factor-κB (NF-κB) pathway. Subsequent upregulation of the apoptotic pathways finally leads to AKI and renal failure. 7,8 In addition, several antioxidants and antiapoptotic agents were used successfully against AKI induced by CSN and MTX. 9 –12
Vanillin (VLN), 4-hydroxy-3-methoxybenzaldehyde, is the major active component isolated from vanilla bean. It is commonly used as a flavoring agent in food, beverage, cosmetic, and pharmaceutical industries. VLN exhibits many biological activities, including anti-oxidative, anti-inflammatory, anticarcinogenic, and antimutagenic properties. 13 Previous reports showed that VLN significantly mitigated CCl4-induced nephrotoxicity and DNA damage in rats and significantly ameliorated molecular, biochemical, and histopathological injuries induced by potassium bromate in mice kidneys. 14,15 A recent study demonstrated that CSN-induced renal injury was attenuated by VLN treatment in rats. 16 However, in the present study, the effect of VLN on novel indicators of kidney injury and additional oxidative, nitrative, inflammatory, and apoptotic biomarkers were investigated in CSN-treated rats. In addition, the effect of VLN against MTX nephrotoxicity was not yet investigated, as per the best of our knowledge. Thereby, the present work was done to investigate the probable protective effect of VLN against nephrotoxicity induced by CSN and MTX in rats.
Materials and methods
Drugs
VLN, CSN, and MTX were purchased from Sigma-Aldrich, St. Louis, MO, USA. All the drugs were dissolved in physiological saline. The doses of VLN, CSN, and MTX were selected based on previous investigations. 7,14,17
Animals
The Animal House, College of Medicine, King Faisal University, provided 60 male Sprague-Dawley rats, weighing 220–250 g. The rats were kept in standard housing conditions (24°C, 45% humidity, and 12-h light/dark cycle) and supplied with commercial chow and tap water ad libitum. The experiments were done according to the international guidelines for care and use of laboratory animals.
Study plan
The rats were randomly allocated into six equal groups (n = 10) as follows: Group I (control) received a daily i.p. injection of physiological saline for 7 days. Group II received a single i.p. injection of CSN (7.5 mg/kg) and a daily i.p. physiological saline for 7 days starting 1 day before administration of CSN. Group III received CSN and treated with VLN (150 mg/kg/day, i.p.) for 7 days starting 1 day before CSN administration. Group IV received a single i.p. injection of MTX (20 mg/kg) and a daily i.p. physiological saline for 7 days starting 1 day before administration of MTX. Group V received MTX and treated with VLN (150 mg/kg/day, i.p.) for 7 days starting 1 day before MTX administration. Group VI received only VLN for 7 days.
Sampling and biochemical processes
Rats were killed by thiopental (70 mg/kg, i.p.) at the end of the experiments. Blood was collected via left ventricular puncture. Serum creatinine was measured by an enzymatic procedure using an automatic analyzer (Hitachi 902, Hitachi Ltd., Tokyo, Japan), and serum cystatin C and neutrophil gelatinase–associated lipocalin (NGAL) were measured by ELISA kits (R&D Systems, Minneapolis, MN, USA). The kidneys were dissected, the right kidney was homogenized in cold potassium phosphate buffer (pH 7.4, 0.05 M), and the homogenate was centrifuged at 4000 r/min for 10 min at 4°C. The supernatant was used to assess malondialdehyde (MDA), and total antioxidant capacity (TAC) by colorimetric kits (Biovision, Milpitas, CA, USA). ELISA kits were used to measure TNF-α, interleukin-18 (IL-18; R&D Systems, Minneapolis, MN, USA), inducible nitric oxide synthase (iNOS; Cusabio, Wuhan, China), Bcl-2, and Bax (Lifespan Biosciences, Seattle, WA, USA).
A colorimetric kit (R&D Systems, Minneapolis, MN, USA) was used to determine caspase-3 activity. Degradation of a specific enzyme substrate releases p-nitroaniline (pNA). Absorbance of pNA of different groups was measured by spectrophotometry at 405 nm and compared to the control.
A portion of the kidney homogenate was recentrifuged at 15,000 r/min for 30 min at 4°C. The supernatant (cytosolic fraction) was used to measure cytochrome C by an ELISA kit (R&D Systems, Minneapolis, MN, USA), and the pellet (nuclear fraction) was used to measure NF-κB p65 unit by an ELISA kit (Novus Biologicals, Littleton, Colorado, USA).
Histopathology processes
The left kidney was fixed in formalin 10% solution and embedded in paraffin wax. Sections were cut at 5 µm, stained with hematoxylin and eosin, and examined under a light microscope by a pathologist unaware of the slide identity. In addition, the degree of renal tubular necrosis was assessed by a semiquantitative scale in which 0 is no necrosis, 1 is <10%, 2 is 10–25%, 3 is 25–75%, and 4 is >75%. 18
Immunohistochemistry processes
The sections were deparaffinized and rehydrated, and hydrogen peroxide (3%) in methanol was used to inhibit endogenous peroxidase. Sections were pretreated in citrate buffer (pH 6.0, 10 mM) in a microwave and incubated with rabbit polyclonal antibody against rat Fas ligand (FasL; Thermo Scientific, Fremont, CA, USA; 1:100). Sections were incubated with biotinylated goat anti-polyvalent, streptavidin peroxidase, and lastly with DAB as chromogen. Counterstaining by hematoxylin was done, and immunostaining was detected by a light microscope using a digital imaging software program (cellSens; Olympus Corporation, Miami, FL, USA) to evaluate the immunostained area (μm2) in five different microscopic fields. The mean ± SEM of each group was calculated.
Statistical processes
GraphPad Prism software program, version 6.01, was used for analysis of data (mean ± SEM) by applying one-way analysis of variance test followed by Tukey test for post hoc comparisons, and significance was at p < 0.05.
Results
Figure 1 shows that administration of a single dose of CSN (7.5 mg/kg, i.p.) and MTX (20 mg/kg, i.p.) resulted in significant increases of serum creatinine, cystatin C, and NGAL and kidney tissue of MDA, iNOS, TNF-α, IL-18, and NF-κB p65 and significantly decreased TAC in rat kidneys as compared to the control (p < 0.05). Treatment with VLN (150 mg/kg/day, i.p.) for 7 days starting 1 day prior to CSN and MTX injections significantly decreased serum creatinine, cystatin C, and NGAL and kidney tissue MDA, iNOS, TNF-α, IL-18, and NF-κB p65 and significantly increased TAC in kidneys of rats challenged with CSN and MTX (p < 0.05; Figure 1). Additionally, VLN significantly prevented the elevations of cytochrome C and caspase-3 and blocked the decrease of Bcl-2/Bax ratio induced by CSN and MTX in rat kidneys (p < 0.05; Figure 2).

Effects of VLN on (a) serum creatinine, cystatin C, and NGAL; (b) kidney MDA, TAC, and iNOS; and (c) NF-κB p65, TNF-α, and IL-18 in rats received CSN or MTX. Results are mean ± SEM, *p < 0.05 versus control, °p < 0.05 versus CSN, and • p < 0.05 versus MTX. VLN: vanillin; NGAL: neutrophil gelatinase–associated lipocalin; MDA: malondialdehyde; TAC: total antioxidant capacity; iNOS: inducible nitric oxide synthase; NF-κB p65: nuclear factor-κB p65; TNF-α: tumor necrosis factor-α; IL-18: interleukin-18; CSN: cisplatin; MTX: methotrexate.
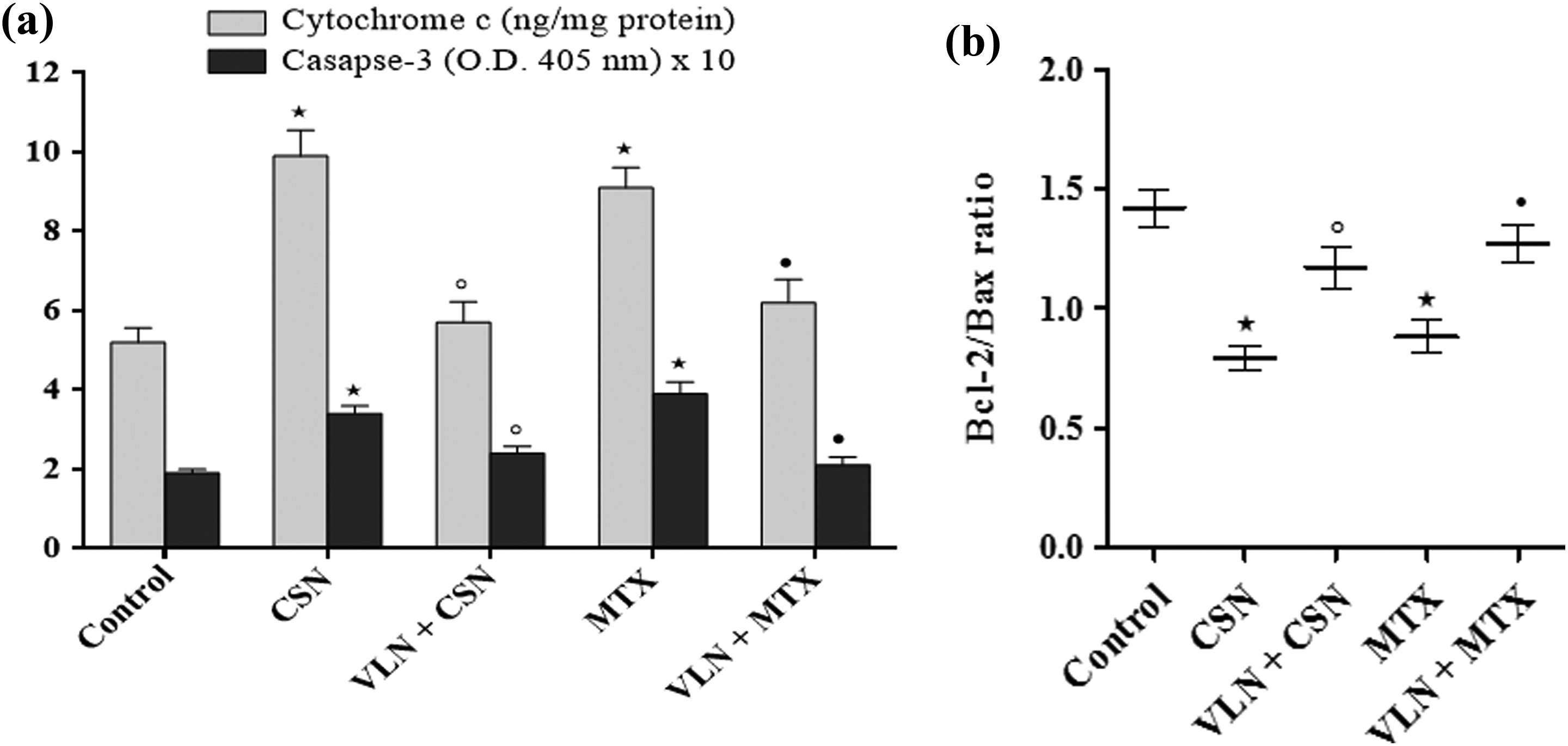
Effects of VLN on (a) cytosolic cytochrome C and caspase-3; and (b) Bcl-2/Bax ratio in kidneys of rats received CSN or MTX. Results are mean ± SEM, *p < 0.05 versus control, °p < 0.05 versus CSN, and • p < 0.05 versus MTX. VLN: vanillin; CSN: cisplatin; MTX: methotrexate.
Figure 3 shows widespread renal tubular necrosis, degeneration and desquamation of tubular epithelium, vacuolization, interstitial edema, and leukocytic infiltration in rat kidneys challenged with CSN and MTX. Treatment with VLN markedly minimized renal injury and significantly decreased the CSN- and MTX-induced elevations of tubular necrosis score (p < 0.05). In addition, CSN and MTX significantly increased the immunoreactivity of FasL in the kidney tissues of rats in comparison with the control (p < 0.05). On the other hand, VLN significantly reduced FasL expression induced by both nephrotoxic agents in rat kidneys (p < 0.05; Figure 4). Negative immunostaining was observed in kidneys of rats challenged CSN and MTX in which the primary antibody was replaced by normal rabbit serum denoting the antibody specificity (figures not included).
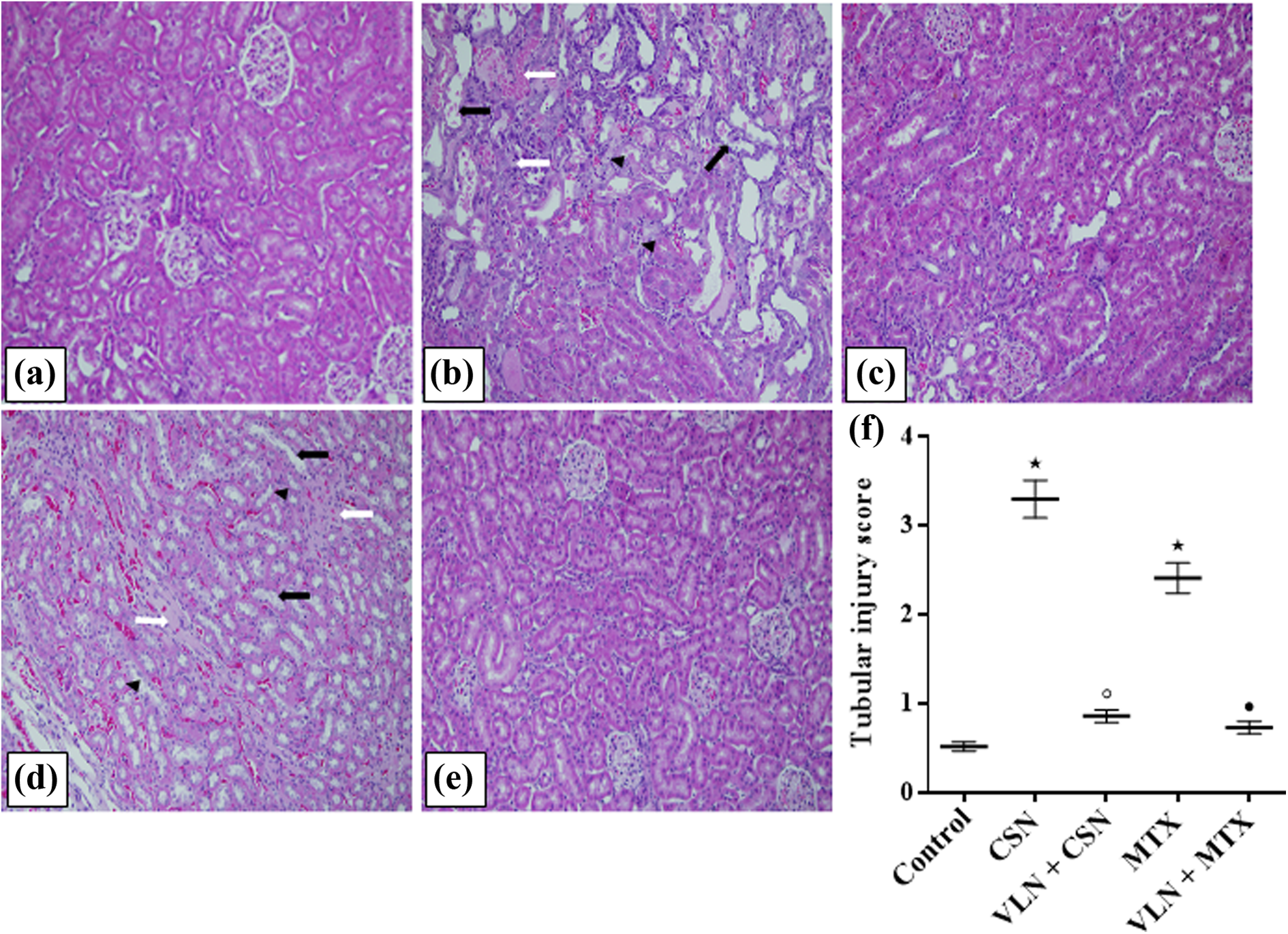
H&E (200×) of rat kidneys from (a) control group showing normal renal histology; (b) and (d) CSN and MTX groups, respectively, showing marked distortion of kidney architecture, widespread necrosis of renal tubules, tubular dilatation, epithelial desquamation (black arrows), vacuolization (black heads), and coagulative necrosis (white arrows); (c) and (e) VLN + CSN and VLN + MTX, respectively, showing that normal kidney architecture is preserved; and (f) tubular injury score. Results are mean ± SEM, *p < 0.05 versus control, °p < 0.05 versus CSN, and • p < 0.05 versus MTX. H&E: hematoxylin and eosin; VLN: vanillin; CSN: cisplatin; MTX: methotrexate.

FasL immunohistochemistry (200×) of rat kidneys from (a) control group showing NS; (b) and (d) CSN and MTX groups, respectively, showing significant elevations of FasL immunostaining in brown color; (c) and (e) VLN + CSN and VLN + MTX, respectively, showing significant decreases in FasL positivity; and (f) immunoreactive area (µm2). Results are mean ± SEM, *p < 0.05 versus control, °p < 0.05 versus CSN, and • p < 0.05 versus MTX. FasL: Fas ligand; NS: negative staining; VLN: vanillin; CSN: cisplatin; MTX: methotrexate.
The results obtained with the group of rats received only VLN were comparable to the corresponding values of the control group without significant differences (results not shown).
Discussion
The molecular mechanisms underlying nephrotoxicity induced by CSN and MTX are multifactorial. Growing evidence indicates the major role of oxidative stress, increased ROS generation, depletion of endogenous antioxidants, and lipid peroxidation of biological membranes with increased MDA production in the pathogenesis of AKI induced by both agents. 19,20 Excess ROS upstreams the inflammatory cascades, including TNT-α, NF-κB, and iNOS pathways. The ROS and TNT-α surge boost nuclear translocation of NF-κB p65, the main unit of cytoplasmic NF-κB sequestration, which augments gene transcription of TNT-α and iNOS. Therefore, the inflammatory responses, RNS yield, and nitrosative stress of cellular macromolecules are reinforced. 21,22 This is in accordance with the current investigation, in which CSN and MTX upregulated the oxidative, nitrative, and inflammatory markers in rat kidneys. In addition, the present study illustrated that VLN significantly impeded AKI induced by CSN and MTX as demonstrated by mitigation of lipid peroxidation, maintenance of antioxidant defenses, suppression of inflammatory responses, and inhibition of NF-κB and iNOS pathways. The renoprotective effect of VLN was reflected by downregulation of the novel sensitive biomarkers of AKI, namely, serum cystatin C and NGAL and kidney IL-18, which have diagnostic and prognostic importance. Interestingly, as to the best of our knowledge, the effect of VLN on these new indices of AKI was not studied before. Similarly, earlier reports related the nephroprotective effect of VLN to the inhibition of oxidative/nitrative stress and inflammation. 14 –16
Along the same line of the current work, previous investigations revealed that both mitochondrial-dependent and mitochondrial-independent apoptotic pathways are involved in AKI induced by CSN. 23 Although apoptosis was reported in the literature to be implicated in MTX nephrotoxicity, 24,25 to the best of our knowledge, this is the first study which demonstrated that mitochondrial-dependent and mitochondrial-independent apoptotic pathways are integrated in this issue. The present study also disclosed that VLN significantly increased Bcl-2, antiapoptotic protein, and significantly decreased Bax, proapoptotic protein, in kidneys of rats received CSN and MTX. This points to the inhibitory effect of VLN of the mitochondrial-dependent pathway of cell apoptosis which is controlled by the Bcl-2 family proteins, mainly Bcl-2 and Bax. The increase in Bcl-2/Bax ratio, observed with VLN treatment, maintained the integrity of mitochondrial membranes and prevented the mitochondrial release of cytochrome C into the cytosol, thereby halted caspase-3 activation. Regarding the mitochondrial-independent apoptotic pathway, the present investigation showed that VLN downregulated kidney tissue levels of TNF-α and FasL, which were eventually responsible for caspase-3 activation. Therefore, it can be concluded that VLN significantly inhibited both the intrinsic and extrinsic apoptotic pathways and cut off the activity of caspase-3, the main executioner of cell apoptosis. 26
In agreement with the present work, past studies showed that CSN and MTX caused significant histopathological kidney tissue injuries. 19,27 The present study also denoted that kidney architecture was significantly kept intact in CSN- and MTX-challenged rats and treated with VLN.
From the current results, we concluded that VLN furnished a significant shield against CSN- and MTX-induced AKI by combating oxidative/nitrative stresses, inflammation, and apoptosis. Further studies are recommended to detect the dose–response efficacy and safety of VLN in AKI resulting from CSN and MTX.
Footnotes
Acknowledgements
The research teamwork is greatly thankful to Mr. Ibrahim Khaled Al-Jassim, the Pharmacology lab technician, College of Medicine, for his sincere help and cooperation.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
