Abstract
Reactive oxygen species have a significant role in the pathogenesis of iron oxide nanorod (IONR) overload-induced organ toxicity in some organs such as the lungs. Green tea induces upregulation of phase II antioxidant enzymes that are transcriptionally organized by the nuclear factor (erythroid-derived 2)-like 2 (Nrf2) that when activated antagonize the oxidative stress induced by IONR overload that causes cardiotoxicity. The aim of the present study was to determine whether treatment of cardiotoxicity with iron chelators (deferiprone (DFP) or deferoxamine (DFO)) alone or in combination with phytochemical activation of Nrf2 (green tea) can protect cardiomyocytes from IONR overload-induced cardiotoxicity. One hundred five rats were distributed into seven groups: two control groups (non-IONR-overloaded and IONR-overloaded) and five IONR-overloaded groups such as a green tea group, DFP group, DFP combined with green tea group, DFO group, and DFO combined with green tea. Blood samples and cardiac tissues were obtained for estimation of total iron-binding capacity, ratio of myocardial 8-hydroxy-2′-deoxyguanosine/myocardial 2-deoxyguanosine, thiobarbituric acid reactive substances, glutathione (GSH) contents, and histopathological examination. The results showed mild histopathological changes in the heart and a significant decrease in all biochemical parameters, except for myocardial GSH, in the DFP group. The addition of green tea improved the biochemical and histopathological results compared with chelators alone.
Introduction
Nanoparticles generally have diameters ranging from 1 nm to 100 nm. Currently, they are receiving a great degree of attention due to their applications in medicine and industry. As a result, exposure to nanoparticles had increased through many routes such as the skin, lungs, and alimentary tract, and some studies have demonstrated the toxicity of nanoparticles. 1 Iron oxide nanoparticles (IONPs) have been used for many biomedical purposes, such as enhancement of magnetic resonance images, drug or gene delivery, and elevated body temperature. 2 The size and shape of nanoparticles plays a major role in their toxicity. Nanorods have many advantages over spherical nanoparticles such as offering longer blood circulation times, stronger interaction with tumors, enhanced retention at tumor sites, and improved targeting efficiency, making them excellent candidates for targeting carriers or magnetic resonance imaging contrast agents. However, rod-shaped IONPs induce extensive necrosis as a result of reactive oxygen species (ROS) production. 3
The deposition of iron in the heart can induce hypertrophy, degeneration, and fibrosis of the cardiomyocytes. 4
ROS can react with macromolecules and subsequently cause cellular injury. The antioxidant system protects against oxidative stress. 5 Antioxidants may upregulate endogenous antioxidants.
The activation of the endogenous antioxidant system, compared to treatment with exogenous antioxidants, enhances the capability of enzymatic antioxidants to control ROS. Some phytochemicals can enhance the activity of antioxidant enzymes via the enhancement of the nuclear factor (erythroid-derived 2)-like 2 (Nrf2). 6
Nrf2 is one of the agents in the basic leucine-zipper transcription factor family 7 and regulates the inducible and basal expression of about 200 genes. 8 It is termed the “master regulator” of the antioxidant defense mechanisms. 9 Cellular oxidative stress is known to stabilize Nrf2 and induce its translocation into the nucleus where it markedly upregulates expression of a battery of antioxidant response element (ARE)-associated target genes. In addition to its own expression, Nrf2 upregulates the expression of superoxide dismutase (SOD)—both copper zinc SOD and manganese SOD—as well as inducible hemeoxygenase 1, nicotinamide adenine dinucleotide phosphate (NAD(P)H) dehydrogenase (quinone) 1 (NQO1), numerous enzymes involved in the glutathione (GSH) system, and many others. 10,11
Deferiprone (DFP) and deferoxamine (DFO) are iron chelators and they ameliorate the oxidative stress on the heart by significantly decreasing total iron-binding capacity (TIBC), 8-hydroxy-2′-deoxyguanosine (8-OHdG), myocardial lipid peroxidation, and GSH content. 4
Many studies have addressed the molecular mechanisms of antioxidant enzymes induced by green tea polyphenols, for example, epigallocatechin-3-gallate (EGCG). Green tea polyphenols can induce antioxidant enzyme expression and subsequent activation of the Nrf2-ARE signaling pathway. Green tea extract also induces the upregulation of phase II detoxifying enzymes via Nrf2-ARE activation. 12 The green tea component, EGCG, has been shown to activate Nrf2. 6
The capability of phytochemicals such as green tea polyphenols to activate Nrf2 and protect the myocardium against oxidative stress is still not well studied. It is well known that an important factor for IONP cytotoxicity is the production of ROS. 13
Although IONPs have offered many therapeutic benefits, they are also associated with exposure risks. 14 Therefore, there is a significant need to study the IONP-induced cardiotoxicity.
The aim of the present experiment was to determine whether treatment of cardiotoxicity with iron chelators (DFP or DFO) alone or in combination with the phytochemical activation of Nrf2 (from green tea) could protect cardiomyocytes from iron oxide nanorod (IONR) overload-induced cardiotoxicity.
Materials and methods
Iron oxide nanorods
Synthesis
Rod-shaped IONPs were synthesized via a wet chemical method based on coprecipitation. This method is based on alkaline coprecipitation of ferric and ferrous salts in aqueous solution. 15,16 Briefly, the iron solution was prepared by dissolving 1.51 g of FeSO4 powder in 100 ml of distilled water and then heating for 10 min at 40°C. Then, 1.19 g of sodium hydroxide (NaOH) was dissolved in 100 ml of distilled water and heated to 50°C for 10 min, and 100 ml of the FeSO4 solution was mixed with 50 ml of the NaOH solution to balance the average solution pH, which is important for the formation of solid IONRs. After that, 100 ml of FeSO4 in NaOH was heated for 60 min to 25°C and the final solution was then heated in a microwave at 140°C for 2 min and dried in an oven overnight.
Preparation of water-nanoparticle suspension
A volume of 10-ml Milli-Q water was added to 0.1-g IONRs powder and ultrasonicated with a UP 200S processor (Stahnsdorf, Germany) at 40 A, 0.5 cycle on ice. After sonication, the suspension was centrifuged for 10 min at 5000 r/min and the obtained supernatant was filtered through a 0.45-µm polyvinylidene difluoride syringe filter (Millipore, Darmstadt, Germany).
Characterization
The crystallinity and grain size of the IONRs were characterized by X-ray diffraction (XRD-6000, Shimadzu Corp., Kyoto, Japan) with copper-Kα radiation (l1/41.54056 Å, 40 kV, 30 mA) in the 2-µ range of 1080 with a 2/min scanning rate (Figure 1(b)). The grain size (D) of a selected sample was estimated by Scherer’s equation, D = Kl/(B cos u), where k is a constant (0.89 <k< 1); l the wavelength of the X-ray; B the full-width at half-maximum width of the diffraction peak; and u the diffraction angle. The morphological features of the IONRs were examined using a high-resolution transmission electron microscope (TEM; JEM-2010, JEOL, Tokyo, Japan), for transmission examination by adding a dilute suspension of iron oxide nanostructures onto copper grids (Figure 1(a)). The optical properties of the prepared IONRs were analyzed via a UV (ultraviolet)–visible UV-2600 spectrophotometer (Shimadzu Corp, Kyoto, Japan; Figure 1(c)). The zeta potential of the IONRs in the plating bath was determined using a Zeta Plus™ zeta potential analyzer (LEZA-600; Otsuka Electronics, Japan) (Figure 1(d)).
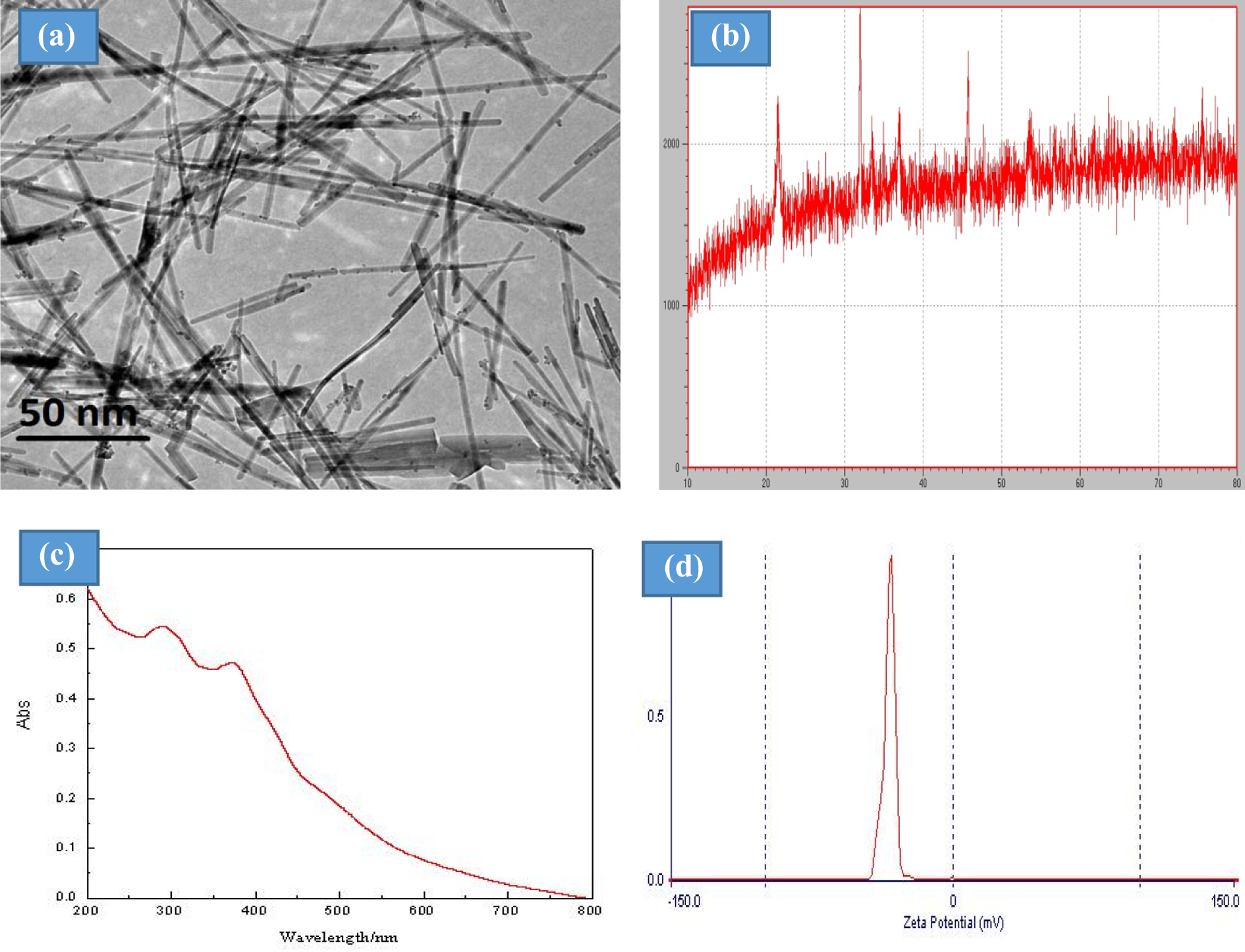
(a) TEM images of the iron oxide nanostructures. (b) XRD patterns of the IONRs. (c) UV absorption spectra of the IONRs. (d) Surface charges of the IONRs measured using zeta potentiometry.
Analysis of green tea extract
Green tea powder extract (40 mg) was dissolved in 30 ml of distilled water in a volumetric flask; then 0.4 ml of the green tea powder extract solution was mixed with 2 ml of Fe2(C4H4O6)3. Next, 2 ml of 1/15 M H3PO4 buffer solution (pH 7.5) was added and the mixture was incubated for 5 min at room temperature and then centrifuged at 3000 r/min for 10 min. Then, the solutions were assayed using high-performance liquid chromatography (HPLC). Catechins and (–) epigallocatechin-3-gallate (EGCG) were quantified against (+) catechin, (–) epicatechin (EC), (–) epicatechingallate (ECG), (–) epigallocatechin (EGC), and GCG as external standards. The green tea powder extract was assayed using a model 2690 HPLC system equipped with a model 996 photodiode array detector (Nihon Waters Co., Ltd, Osaka, Japan) at 278 nm and a Capcell Pak C18 UG120 column (4.6 mm i.d. × 250 mm, particle size 5 μm, Shiseido Co. Ltd, Tokyo, Japan). The mobile phase was composed of two solutions: distilled water and N,N-dimethylformamide: methanol: acetic acid, 40:2:1.5, and used in a gradient elution. It was confirmed that the polyphenol content was more than 98%, and each 1 g of extract contained EGCG, 55%; EGC, 22%; and EC, 5.5%.
Experimental animals and diet
One hundred five male Wistar rats weighing 135–170 g (7–10 weeks old) were obtained from the National Centre of Research (Cairo, Egypt). The rats were housed individually in conventional solid-bottom polycarbonate cages with standard stainless-steel lids and hardwood chip bedding on a normal light–dark cycle at approximately 22°C and 55 ± 5% humidity. The rats were handled according to the guidelines for the care and use of laboratory animals from the National Institutes of Health, USA. All procedures were approved by the research ethics committee of the Faculty of Pharmacy, Tanta University, Egypt.
Experimental design
The animals were divided into the following groups: Control group (15 rats): non-IONR-overloaded control group (group I). IONR-overloaded group (intoxicated group, group I) had been intoxicated by giving oral IONR 500 mg/kg/day via oral gavage for 6 months. The iron-overloaded group using the same dose and duration as the IONR-overloaded cohort and then divided into five equal groups: Green tea group (group III): Fifteen IONR-overloaded rats were given green tea extract at a dose of 70 mg/kg by gavage daily for 2 weeks.
17
DFO group (group IV): Fifteen IONR-overloaded rats were given DFO subcutaneously at a dose of 200 mg/kg for 2 weeks. The DFO was dissolved in normal saline at a concentration of 1%.
4
DFO combined with green tea group (group V): Fifteen IONR-overloaded rats were given DFO as for the previous group and also given green tea extract at a dose of 70 mg/kg by gavage daily for 2weeks.
17
DFP group (group VI): Fifteen IONR-overloaded rats were given DFP orally at a total daily dose of 75 mg/kg in three divided doses for 2weeks.
4
DFP was dissolved in normal saline at a concentration of 1%. DFP combined with green tea group (group VII): Fifteen IONR-overloaded rats were given DFP as for the previous group in combination with green tea extract in the same dose and duration as above. The duration of treatment with green tea extract, DFO, and DFP was 2 weeks after IONR-overload.
All chemicals used were reagent grade and were purchased from the Sigma Chemical Company, except the DFP, which was obtained from Orphan Australia Ltd (Berwick, Victoria, Australia), and the green tea from the Royal Company (Egypt) and prepared as aqueous extract. Each 1 g of extract contained EGCG, 55% EGC, 22% ECG, and 5.5% EC as determined by HPLC. Green tea extract HPLC standards were obtained from the Cayman Chemical Co. (Ann Arbor, Michigan, USA).
On the last day of the experiment, the animals were anesthetized by intraperitoneal coadministration of ketamine and xylazine at a dose of 45 and 5 mg/kg, respectively, and blood samples were drawn by cardiac puncture. The serum was obtained by centrifugation at 1300g at 25°C for 15 min. The rats were sacrificed by decapitation, and the ventricles of hearts were excised, rinsed with cold saline, dried with a filter paper, and divided into two halves. Half of each heart was homogenized using a Bio-Gen pro 250 homogenizer (Pro Scientific, Oxford, UK) in phosphate buffer (pH 7). The homogenates were centrifuged at 1300g at 4°C for 15 min. The supernatant of the homogenate and serum were used for biochemical assays. The second half of the heart was used for light and electron microscopic examination.
Determination of serum iron and TIBC
The samples were analyzed using a Hitachi 736-50 colorimeter (Boehringer Mannheim Corp, Somerville, NY, USA). 18
Determination of serum LDH activity
The lactate dehydrogenase (LDH) activity was determined using a commercially available LDH kit (Linear Chemicals, S.L., Madrid, Spain) according to the method of Whitaker. 19 The reaction was monitored kinetically at 340 nm using a UV–visible spectrophotometer (Shimadzu).
Determination of serum CK-MB activity
The creatine kinase (CK-MB) serum activity was determined with a commercially available CK-MB assay kit (Bioassay Systems, Hayward, California, USA) that used the method of Bishop et al. 20 The rate of NADH formation was measured at 340 nm using a UV–visible spectrophotometer (Shimadzu).
Determination of the cTn-T level
The troponin-T (cTn-T) level was measured in whole blood by point-of-care test using the Roche Cobas® h 232 immunoassay analyzer (Roche Diagnostics, Mannheim, Germany). The range of detection was 0.1–3 μg/l. 21
Determination of myocardial 8-OHdG and 2-dG
The 8-hydroxy-2′-deoxyguanosine (8-OHdG) and 2-deoxyguanosine (2-dG) levels of the hydrolyzed DNA samples were measured by electrochemical HPLC (HPLC-ECD) and a variable wavelength detector HPLC (HPLC-UV), respectively, as previously described. 22 The 2-dG and 8-OHdG were quantified using the standard; the level of 8-OHdG was expressed as the number of 8-OHdG molecules per 106dG.
Determination of myocardial lipid peroxidation
The rate of production of thiobarbituric acid reactive substances (TBARS) was estimated as an indicator of lipid peroxidation and expressed as nanomole per milligram of protein according to the method of Buege and Aust. 23 The absorbance was determined spectrophotometrically at 535 nm after cooling using a UV–visible spectrophotometer (Shimadzu).
Determination of the myocardial nitrotyrosine content
The nitrotyrosine levels of myocardial tissues were quantified by a modified enzyme-linked immunosorbent assay procedure. 24 The amount of nitrotyrosine in these samples was estimated using standard curves generated from nitrated bovine serum albumin (BSA) containing known amounts of nitrotyrosine and expressed as picomole per milligram of protein.
NAD(P)H oxidase activity
The myocardial tissue nicotinamide adenine dinucleotide phosphate (NAD(P)H) oxidase activity was estimated according to the method of Souza et al. 25 The NAD(P)H oxidase activity was measured in the presence of 100-mmol/l NAD(P)H and expressed as counts per minute per milligram of dry weight.
Determination of myocardial SOD activity
The total cellular SOD activity was determined by the method of Spitz and Oberley. 26 SOD standard curve was used in the estimation of the SOD activity, which was expressed as units per milligram of cellular protein.
Determination of myocardial catalase activity
The method of Aebi 27 was used to measure the catalase activity. The catalase activity was expressed as the micromole of hydrogen peroxide (H2O2) consumed/min/mg of cellular protein.
Determination of the myocardial GSH content
The cellular GSH content was determined according to previously described procedures by Cao and Li. 28 GSH standard curve was used to estimate the myocardial GSH content that was expressed as nanomole of GSH per milligram of cellular protein.
Determination of myocardial GPx activity
The cellular GSH peroxidase (GPx) activity was estimated by the method of Flohe and Gunzler. 29 The GPx activity was estimated using an extinction coefficient of 6.22/mM/cm and was expressed as nanomole of NAD(P)H consumed/min/mg of cellular protein.
Determination of myocardial GR
The myocardial glutathione reductase (GR) activity was assayed by the method of Carlberg and Mannervik. 30 The reaction was started by adding the enzyme source, and the oxidation of NAD(P)H was monitored at 340 nm against a reagent blank. The enzyme activity was expressed in micromole μmol of NAD(P)H oxidized/mg protein/min.
Determination of the protein content
A 0.2-ml volume of cardiac homogenate was taken in triplicate, centrifuged, and washed with distilled water, and the pellets then frozen. The total protein content was determined according to a modified Lowry procedure. 31 The OD750 was measured against a BSA standard curve with a Lambda 25 UV/VIS spectrometer (Perkin Elmer, Midrand, South Africa).
Histopathological examination of the heart tissue
Ventricles from the second half of each heart were fixed in a 10% neutral formalin solution, then embedded in paraffin, sectioned at a thickness of 5 µm, stained with hematoxylin and eosin (H&E), and examined under the light microscope. The ventricular muscle specimens were evaluated for typical histopathological features associated with IONR-overload-induced cardiotoxicity (e.g. myocyte vacuolar degradation, inflammation, interstitial fibrosis, and necrosis of the myofibers). Patho-histological evidence of inflammation of the heart was classified in terms of the degree of cellular infiltration and graded on a five-point scale ranging from 0 to 4+ (with zero score indicating no lesions or the questionable presence of lesions in each category; 1+ score described a limited focal distribution of myocardial lesions; 2+ to 3+ score described intermediate severity with multiple lesions; and 4+ score described the presence of coalescent and extensive lesions overall the heart tissue). 32
Immunohistochemistry
Formalin-fixed tissues were treated with anti-Nrf2 antibody (H-300; Santa Cruz Biotechnology Santa Cruz, CA, USA, catalog number sc-13032) at a dilution of 1:250 for 1 h and developed using horseradish peroxidase (Dako, Glostrup, Denmark). Nonimmune rabbit immunoglobulin G (Jackson ImmunoResearch Laboratories, West Grove, Pennsylvania, USA) was used as a negative control. To demonstrate the specificity of antibody staining, we preincubated the anti-Nrf2 antibody with Nrf2 and luciferase in vitro transcribed and translated protein, respectively, for 30 min and then immunohistochemical staining was carried out.
Statistical analysis
The results were expressed as the mean ± standard deviation. A statistical comparison between the different groups was performed using one-way analysis of variance, followed by Tukey–Kramer’s multiple comparison tests to compare the differences between the various groups. Significant differences were designated at p < 0.05.
Results
Characterization of IONRs
Particle size and surface charge of IONR nanoparticles were measured by TEM and the zeta potentiometer, respectively. The results showed that the average size of IONRs was 50 ± 5 nm (length) and 15 ± 5 nm (width) with surface charge of −28.1 mV.
Biochemical results
This study showed a significant increase in the serum iron level in IONR-overloaded rats compared with the controls, as shown in Table 1. All IONR-overloaded rats that were given green tea, DFO alone or in combination with green tea, and DFP as chelators showed significant increases in the serum iron level compared with the controls. On the other hand, the serum iron level was significantly decreased in the DFO and DFP alone or in combination with green tea groups compared with the control (IONR-overloaded) group. No significant changes in serum iron levels were observed with supplementation of green tea compared with the control IONR-overloaded group (Table 1). These results demonstrated that DFP decreased the serum iron level more than DFO (182% and 130.7%, respectively) compared with the control non-IONR-overloaded group. In this study, supplementation with green tea in addition to DFP showed no significant changes in the serum iron level compared with the control non-IONR-overloaded group (Table 1).
Serum iron, the TIBC, and the myocardial 8-OHdG/2-dG levels in the control and IONRs-overloaded groups.a
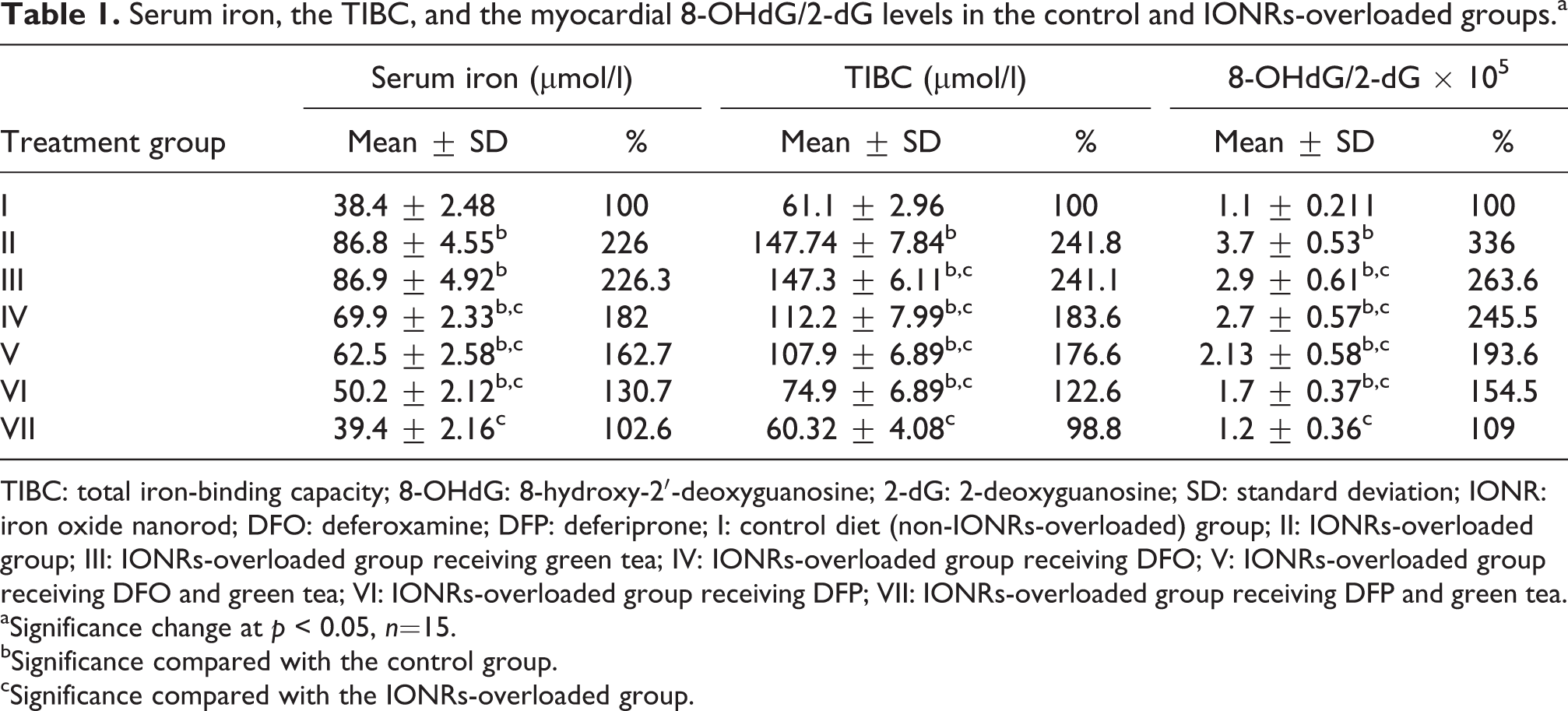
TIBC: total iron-binding capacity; 8-OHdG: 8-hydroxy-2′-deoxyguanosine; 2-dG: 2-deoxyguanosine; SD: standard deviation; IONR: iron oxide nanorod; DFO: deferoxamine; DFP: deferiprone; I: control diet (non-IONRs-overloaded) group; II: IONRs-overloaded group; III: IONRs-overloaded group receiving green tea; IV: IONRs-overloaded group receiving DFO; V: IONRs-overloaded group receiving DFO and green tea; VI: IONRs-overloaded group receiving DFP; VII: IONRs-overloaded group receiving DFP and green tea.
aSignificance change at p < 0.05, n=15.
bSignificance compared with the control group.
cSignificance compared with the IONRs-overloaded group.
The present study demonstrated that the IONR-overloaded group showed a significant increase in TIBC compared with the control non-IONR-overloaded group (Table 1). Rats treated with green tea, DFO alone or in combination with green tea, and DFP showed a significant increase in TIBC compared with the control. However, rats treated with DFO and DFP alone or in combination with green tea showed a significant decrease in TIBC level compared with the IONR-overloaded group (Table 1). The addition of green tea showed no significant change in the TIBC level compared with the IONR-overloaded group (Table 1). DFP also decreased TIBC levels more than DFO (122.6% and 183.6%, respectively). No significant change in the serum iron level occurred with supplementation of green tea in addition to DFP compared with the control non-IONR-overloaded group (Table 1).
The ratio between the myocardial 8-OHdG and myocardial 2-dG was significantly increased in the IONR-overloaded and DFO groups compared with the controls. Treatment with green tea, DFO, and DFP alone or in combination with green tea was associated with a significant decrease of the ratio of myocardial 8-OHdG and myocardial 2-dG compared with the IONR-overloaded group. However, DFP was more powerful in decreasing the ratio of myocardial 8-OHdG and myocardial 2-dG than DFO (154.5% and 245.5%, respectively) compared with the control IONR-overloaded group (Table 1).
The serum CK-MB activity was significantly increased in the IONR-overloaded group compared with the control non-IONR-overloaded group, as shown in Table 2. All of the IONR-overloaded groups administered green tea, DFO, and DFP as chelators alone or in combination with green tea showed a significant increase in CK-MB activity compared with the control non-IONR-overloaded group. On the other hand, the CK-MB activity was significantly decreased in the green tea, DFO, and DFP groups compared with the control IONR-overloaded group. The CK-MB activity was lower but did not reach the control non-IONR-overloaded level (Table 2). However, DFP was more powerful in decreasing the CK-MB activity than DFO (144% and 170%, respectively) compared with the control non-IONR-overloaded group. A significant elevation in the CK-MB activity was noted with supplementation with green tea in addition to DFO and DFP compared with the control non-IONR-overloaded group. On the other hand, these groups showed a significant decrease in the CK-MB activity compared with the control IONR-overloaded group; however, treatment with DFP was more powerful in decreasing the CK-MB activity than treatment with DFO (Table 2).
Serum CK-MB activity, LDH, and cTn-T levels in the control and IONRs-overloaded groups.a
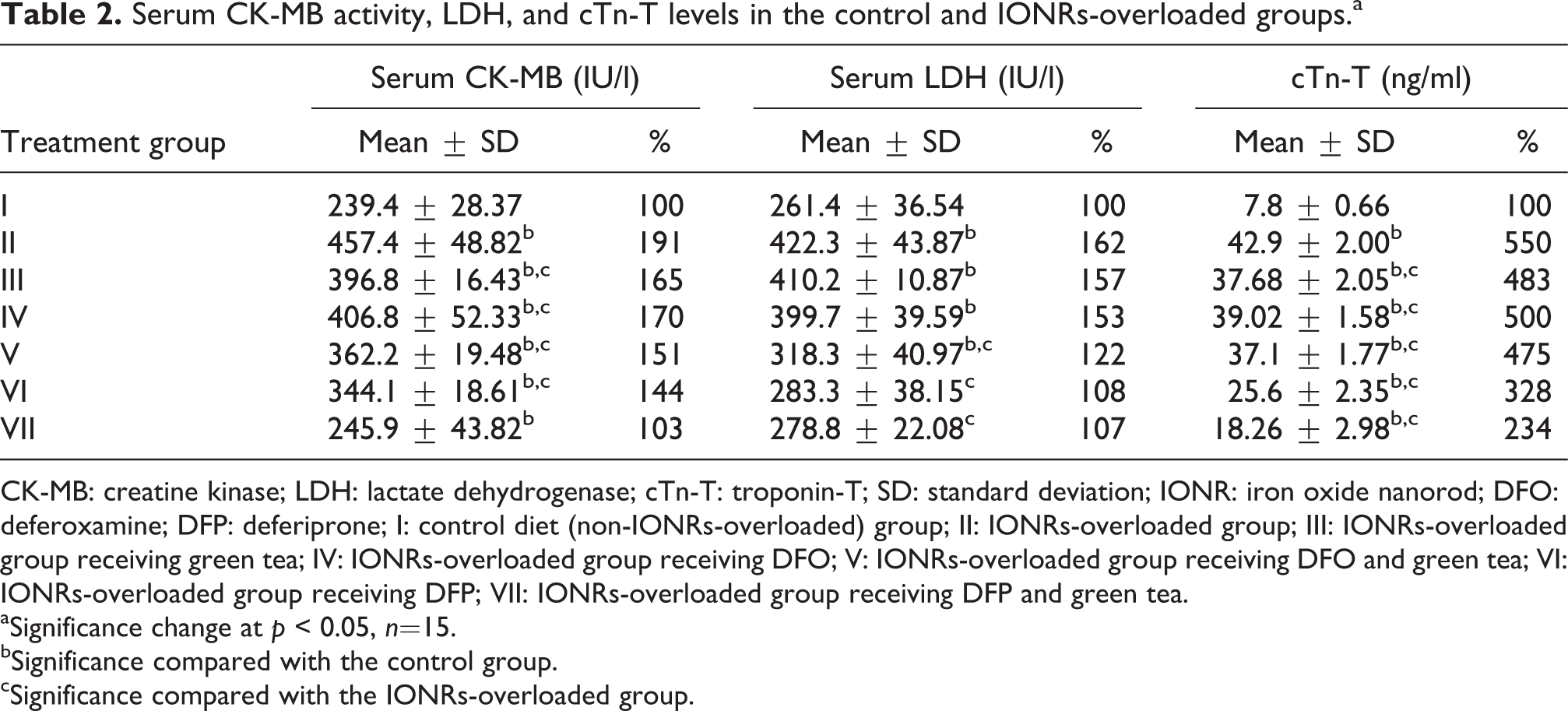
CK-MB: creatine kinase; LDH: lactate dehydrogenase; cTn-T: troponin-T; SD: standard deviation; IONR: iron oxide nanorod; DFO: deferoxamine; DFP: deferiprone; I: control diet (non-IONRs-overloaded) group; II: IONRs-overloaded group; III: IONRs-overloaded group receiving green tea; IV: IONRs-overloaded group receiving DFO; V: IONRs-overloaded group receiving DFO and green tea; VI: IONRs-overloaded group receiving DFP; VII: IONRs-overloaded group receiving DFP and green tea.
aSignificance change at p < 0.05, n=15.
bSignificance compared with the control group.
cSignificance compared with the IONRs-overloaded group.
LDH was significantly elevated in the IONR-overloaded group compared with the control non-IONR-overloaded group (Table 2). Rats treated with green tea, DFO alone, or DFO in combination with green tea showed a significant elevation in LDH compared with the control non-IONR-overloaded group. However, rats treated with DFO in combination with green tea, and DFP alone or in combination with green tea, showed a significant decrease in the LDH level compared with the IONR-overloaded group (Table 2). The present study also showed that DFP was more powerful in decreasing the LDH level than DFO (108% and 153%, respectively; Table 2).
The level of cTn-T was significantly elevated in the IONR-overloaded group compared with the control non-IONRs-overloaded group (Table 2). Rats treated with green tea, DFO, and DFP alone or in combination with green tea showed a significant elevation in the cTn-T level compared with the control non-IONR-overloaded group. However, they showed a significant decrease in the cTn-T level compared with the IONR-overloaded group (Table 2). DFP alone or in combination with green tea was more powerful in decreasing the cTn-T level than DFO (328% and 234%, respectively; Table 2).
The TBARS levels were significantly increased in the IONR-overloaded group compared with the control non-IONR-overloaded group. Rats treated with green tea, DFO, and DFP alone or in combination with green tea extract had significantly higher TBARS levels compared with the control non-IONR-overloaded group. However, they showed a significant decrease in the TBARS level compared with the control IONR-overloaded group. DFP alone or in combination with green tea were more powerful in decreasing the TBARS levels than DFO (Figure 2(a)).

(a) TBARS (nanomole per milligram of protein), (b) GR (micromole of NAD(P)H/min/mg of protein), (c) SOD (unit per milligram protein), (d) GPx (unit per milligram protein), (e) glutathione, (f) NAD(P)H oxidase activity (counts per minute per milligram), (h) nitrotyrosine (pmol/mg protein), and (j) CAT (unit per milligram of protein) of male rats receiving (I) control diet (non-IONRs-overloaded) group; (II) IONRs-overloaded group; (III) IONRs-overloaded group receiving green tea; (IV) IONRs-overloaded group receiving DFO; (V) IONRs-overloaded group receiving DFO and green tea; (VI) IONRs-overloaded group receiving DFP; and (VII) IONRs-overloaded group receiving DFP and green tea. Significance change at p < 0.05, n = 15. aSignificance compared with control group. bSignificance compared with IONRs-overloaded group.
The myocardial SOD, GPx, and GSH levels were significantly decreased in the IONR-overloaded group compared with the control non-IONR-overloaded group. However, rats treated with green tea, DFO, DFP, DFO plus green tea, and DFP plus green tea showed a significant elevation in myocardial SOD, GPx, and GSH levels compared with the IONR-overloaded group. Rats treated with DFP alone or in combination with green tea had higher myocardial SOD, GPx, and GSH levels than the DFO-treated rats (Figure 2(c) to (e)).
GR, nitrotyrosine, NAD(P)H oxidase, and catalase activity were significantly increased in the IONR-overloaded group compared with the control non-IONR-overloaded group. Rats treated with green tea, DFO, and DFP alone or in combination with green tea showed a significant decrease in GR, nitrotyrosine, NAD(P)H oxidase, and catalase activity compared with the control IONR-overloaded group. Rats treated with DFP alone or in combination with green tea had lower GR, nitrotyrosine, NAD(P)H oxidase, and catalase activity (Figure 2(b), (f), (h), and (j)).
Histopathological results
Examination of the heart tissues under light microscopic and stained with H&E produced the following results: Group I (non-IONR-overloaded) showed normal myocardial syncytial fibers (Figure 3). Group II (IONR-overloaded group) showed focal vacuolar changes in the myocardial muscle fibers (V) with interstitial edema and the dilatation of interstitial blood vessels (D; Figure 3). Group III (IONR-overloaded rats treated with green tea) showed apparently normal syncytial myocardial fibers with mild interstitial edema between the bundles with some wavy arrangements of fibers (Figure 3). Group IV (IONR-overloaded rats treated with DFO) showed focal areas of vacuolar changes (V) with a loss of striation of the myocardial fibers and moderate interstitial edema (Figure 3). Group V (IONR-overloaded rats treated with DFO and green tea) showed apparently normal syncytial myocardial fibers with mild to moderate interstitial edema and wavy arrangements of the fibers (Figure 3). Group VI (IONR-overloaded rats treated with DFP) showed focal areas of vacuolar changes (V) with moderate interstitial edema (Figure 3). Group VII (IONR-overloaded rats treated with DFP and green tea) showed apparently normal syncytial myocardial fibers with focal areas of mild interstitial edema (Figure 3).
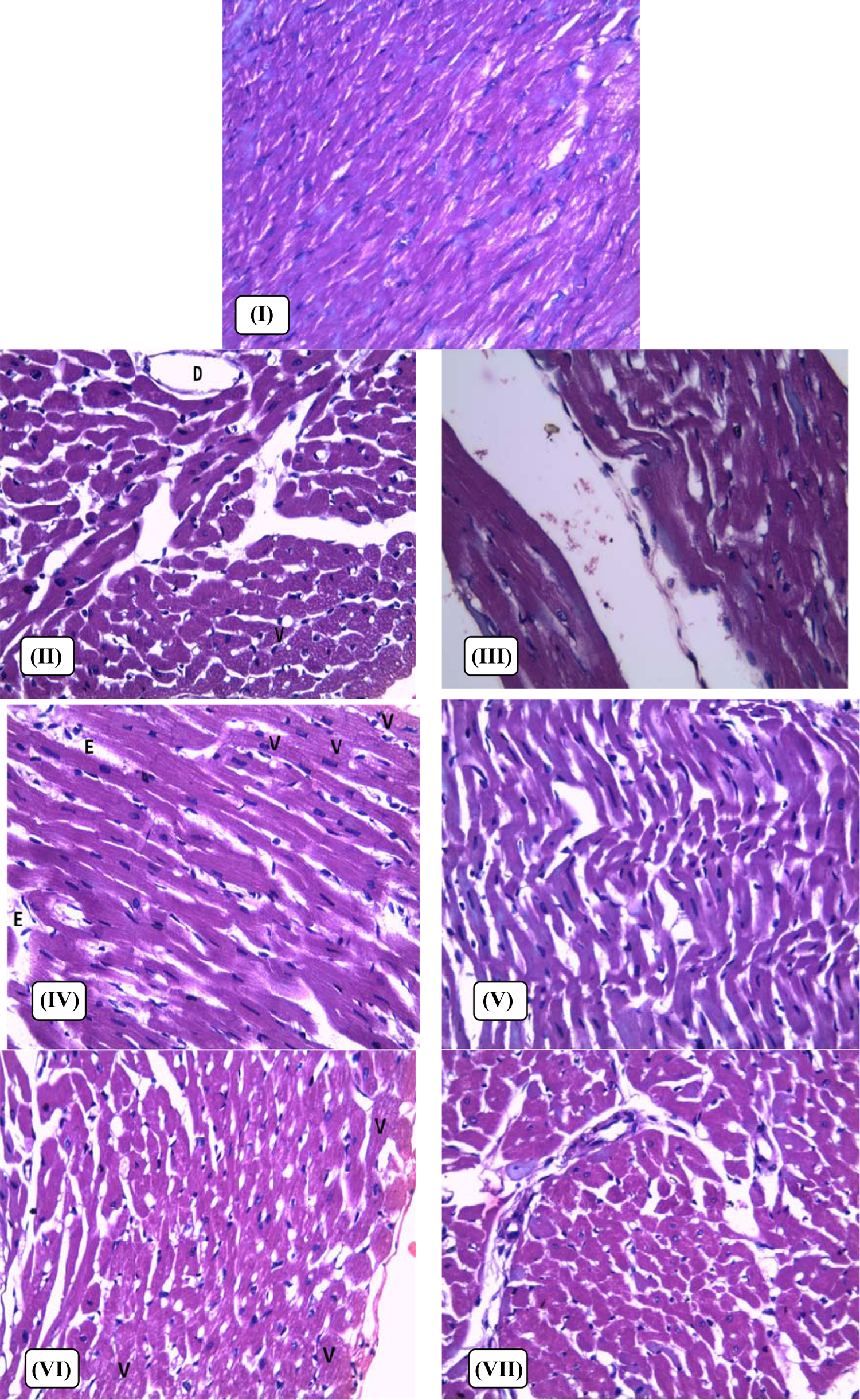
Light micrograph of the heart of male rats; (I) control diet (non-IONRs-overloaded) group; (II) IONRs-overloaded group; (III) IONRs-overloaded group receiving green tea; (IV) IONRs-overloaded group receiving DFO; (V) IONRs-overloaded group receiving DFO and green tea; (VI) IONRs-overloaded group receiving DFP; and (VII) IONRs-overloaded group receiving DFP and green tea. The specimens were fixed in formalin; sections were cut on amicrotome and stained H&E. Photograph was taken using a ×200 objective.
Immunohistochemical examination of heart sections revealed the following: Group I (non-IONR-overloaded) showed no cytoplasmic staining for Nrf2 (Figure 4). Group II (IONR-overloaded group) showing extensive diffuse highly positive cytoplasmic staining (++++) for Nrf2 (Figure 4). Group III (IONR-overloaded rats treated with green tea) showed no cytoplasmic staining for Nrf2 (Figure 4). Group III (IONR-overloaded rats treated with DFO) showed severe focal or patchy positive cytoplasmic staining (+++) for Nrf2 (Figure 4). Group IV (IONR-overloaded rats treated with DFO and green tea) showed mild to moderate positive cytoplasmic staining (++) for Nrf2 (Figure 4). Group V (IONR-overloaded rats treated with DFP) showed moderate positive cytoplasmic staining (++) for Nrf2 (Figure 4). Group VI (IONR-overloaded rats treated with DFP and green tea) showed mild cytoplasmic staining (+) for Nrf2 (Figure 4).
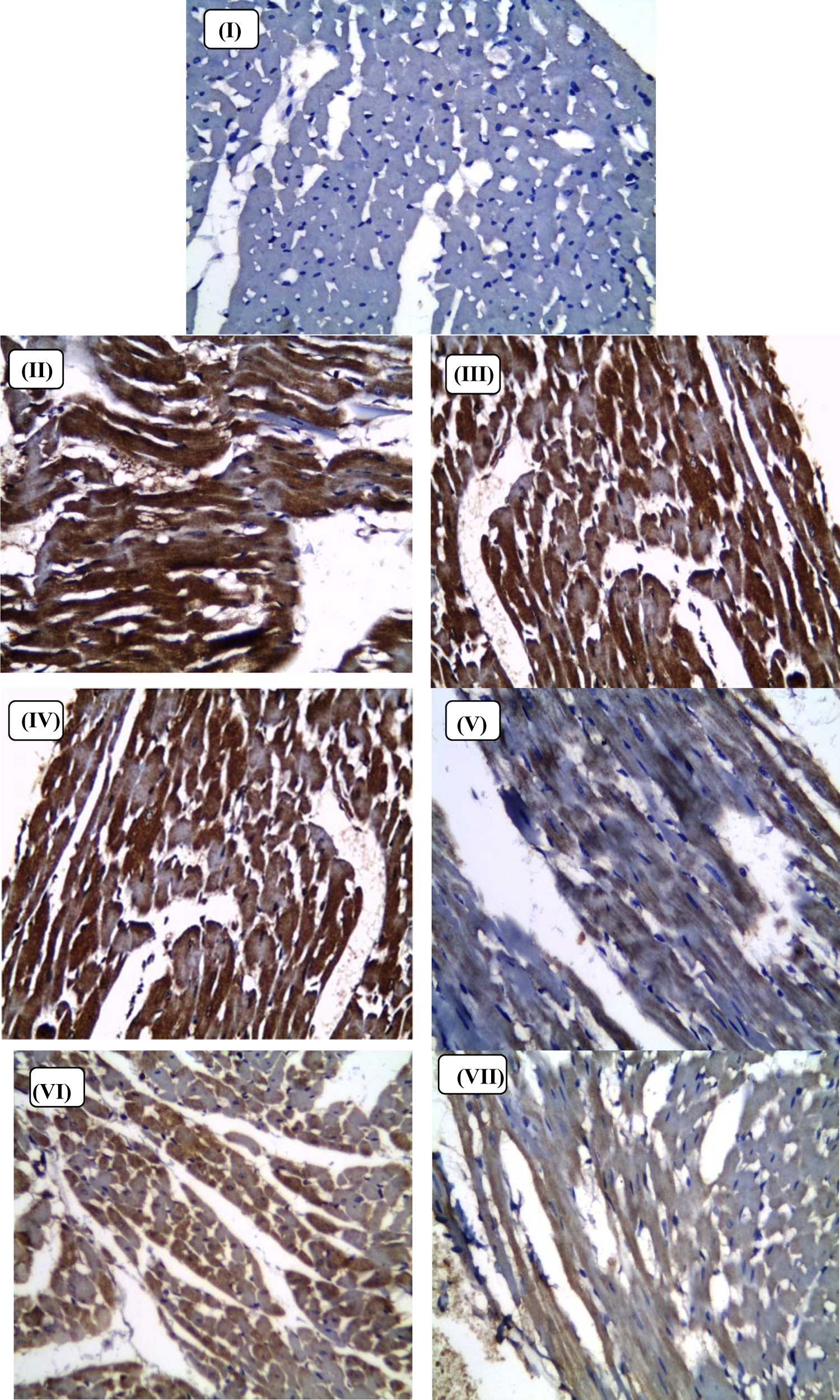
Light micrograph of the heart of male rats; (I) control diet (non-IONRs-overloaded) group; (II) IONRs-overloaded group; (III) IONRs-overloaded group receiving green tea;(IV) IONRs-overloaded group receiving DFO; (V) IONRs-overloaded group receiving DFO and green tea; (VI) IONRs-overloaded group receiving DFP; and (VII) IONRs-overloaded group receiving DFP and green tea. The specimens were fixed in formalin; sections were cut on a microtome and stained for Nrf2 (PAP. ×400).
Discussion
In this study, we demonstrated that treatment with iron chelators (DFP or DFO) alone or in combination with the phytochemical activation of Nrf2 (by green tea extracts) can protect cardiomyocytes from IONR overload-induced cardiotoxicity.
The utilization of nanoparticles in medicine is an important modality in many therapeutic areas. 33 Physicalproperties of IONRs can be modified and they can be fabricated to be used in many nanomedical applications. 34
Zhu et al. 35 demonstrated that IONRs can damage endothelial cells and lead to subsequent cardiovascular pathology, such as myocardial infarctions, hypertension, and atherosclerosis. The size and shape of IONPs can cause multiple tissue responses including the production of ROS and cell death. The deleterious effects of IONPs are linked to oxidative stress. 36
The accumulation of iron in the heart, liver, bone marrow, spleen, pancreas pituitary, and the central nervous system can induce dysfunction to these organs. The accumulation of iron in the cardiomyocytes results in iron overload cardiomyopathy (IOC). IOC characterized by systolic or diastolic cardiac dysfunction. 37
Gujja et al. 37 and Pucheu et al. 38 found that DFO and DFP prevent iron overload. These chelating agents are approved by the US FDA for clinical use in humans.
In the present study, IONR-overloaded rats showed a significant increase in serum iron levels compared with those of the control non-IONR-overloaded group. Galleano et al. 39 found that iron supplementation can induce a significant elevation (up to 12-fold) in plasma iron concentration. The DFO and DFP alone groups or in combination with green tea groups showed a significant decrease in the serum iron level compared with the IONRs-overloaded group. However, DFP was more powerful in decreasing the serum iron level than DFO. These results are in agreement with those of Franchini et al., 40 who demonstrated that DFP can dramatically decrease high iron concentrations in iron-overloaded patients, indicating high efficacy of DFP. Breno et al. 41 found that DFP decreases iron levels and is more effective in the treatment of iron overload than DFO. On the other hand, Chaim et al. 42 demonstrated that DFP was less effective than DFO and that its use was accompanied by increase in agranulocytes. Maggio et al. 43 reported that DFP has similar effect in decreasing iron levels as DFO during the treatment of iron overload patients.
The present study demonstrated that the IONR-overloaded group showed a significant increase in the TIBC compared with the control non-IONR-overloaded group. Rats treated with DFO and DFP alone or in combination with green tea showed a significant decrease in TIBC compared with the IONR-overloaded. This study also showed that DFP was more powerful in decreasing TIBC than DFO.
Oxidative stress can be prevented by iron chelators through induction of glycolytic enzyme gene expression and aerobic glycolysis. It was found that changes in energy production resulting from mitochondrial glucose utilization for aerobic glycolysis are accompanied by resistance to oxidative stress. 44 The decline in ROS production from aerobic glycolysis resulted from the production of the antioxidant pyruvate and reduction of mitochondrial glucose oxidation. 45
The production of hydroxyl radicals (OH−) from the iron-catalyzed reaction of superoxide (O2 –) and H2O2 can be prevented by DFO and DFP. 46
One of the indicators of oxidative stress is the TBARS. Zhang 47 reported that accumulation of iron in cardiac tissues elevates free radical levels and causes lipid peroxidation.
Ritter et al. 48 demonstrated that DFO significantly declined in TBARS production in all tissues. On the other hand, Cighetti et al. 49 reported that iron chelators have no effect on production of TBARS in iron-overloaded patients.
Natural compounds can modulate ROS to reduce its cytotoxicity in pathological lesions in heart diseases. Green tea extract has been reported to decrease formation of aortic lesions and lipid oxidation. Green tea compounds have been reported to suppress lipid peroxidation and scavenge ROS in many biological systems, such as mitochondria or microsomes. Green tea is considered by many to be a health-promoting beverage, whose components are mainly catechins and catechin derivatives with antioxidant properties. 50,51
The present study showed that myocardial SOD, GPx, GR, and GSH levels were significantly decreased in the IONR-overloaded group compared with those in the control non-IONR-overloaded group. However, rats treated with green tea, DFO, DFP, DFO plus green tea, and DFP plus green tea showed a significant elevation in myocardial SOD, GPx, and GSH levels compared with the IONR-overloaded group. Rats treated with DFP alone or in combination with green tea had increased levels of myocardial SOD, GPx, and GSH compared with those treated with DFO.
Pucheu et al. 38 found that the activity of the antioxidant enzyme, GPX, was significantly decreased in IONR-overloaded group, whereas the catalase activity was increased. Additionally, O2 –, H2O2, or OH−may be involved. Increased production of ROS has been involved in the pathogenesis of cardiovascular disease (CVD) and enhanced endogenous antioxidants have been proposed as a mechanism for regulating the redox balance.
The ratio of myocardial 8-OHdG and myocardial 2-dG showed a significant increase in the IONR-overloaded and DFO groups compared with the control non-IONR-overloaded group. Treatment with green tea, DFO, and DFP alone or in combination with green tea showed a significant decrease in the ratio of myocardial 8-OHdG and myocardial 2-dG compared with the IONR-overloaded group. DFP was shown to be more powerful in decreasing myocardial 8-OHdG than DFO.
In agreement with these results, Zhang 47 found that the myocardial effects of iron overload can be assessed by determining myocardial TBARS, myocardial GSH, and 8-OHdG. Classical antioxidant enzymes, including SOD, catalase, and GPx directly inactivate ROS and prevent ROS-initiated reactions. 52 Phase II detoxifying enzymes, including glutathione-S-transferase (GSTs), NAD(P)H, quinine oxidoreductase (NQO1), and glutamate-cysteine ligase catalytic, also indirectly act as antioxidant enzymes by regulating the biosynthesis/recycling of thiols or facilitating the excretion of oxidized reactive secondary metabolites. 47
This study showed that NAD(P)H oxidase, nitrotyrosine, and catalase activity were significantly increased in the IONR-overloaded group compared with the control non-IONR-overloaded group. Rats treated with green tea, DFO, and DFP alone or in combination with green tea showed a significant decrease in NAD(P)H oxidase, nitrotyrosine, and catalase activity compared with the control IONRs-overloaded group.
Cabrera et al. 53 found that green tea exerts antioxidant effects by direct action as a radical scavenger or metalchelator and indirectly via the modulation of transcription factors or enzymes. Rice-Evans et al. 54 revealed that green tea extract has multiple protective effects against oxidative stress through decreasing lipid peroxidation and normalizing SOD activity and GSH in tissues as well as protecting against DNA fragmentation and histopathological changes.
Nrf2 is a transcriptional regulator of phase II antioxidant enzymes, and the activation of Nrf2 has been suggested to be an important step in attenuating oxidative stress associated with CVD. Nrf2 is activated by green tea and subsequent induction of phase II enzymes via the ARE. 55 In this study, we explored whether supplementation with green tea, and activation of Nrf2, causes induction of phase II detoxification enzymes and protects cardiomyocytes from oxidant-induced histopathological and biochemical changes. Reuland et al. 6 and Zhang 47 found that phytochemical treatment of cardiomyocytes was accompanied with an accumulation of Nrf2 in the nucleus, significant induction of phase II antioxidant enzymes, and protection against hydrogen peroxide-induced apoptosis. They also found that phytochemical treatment is a more powerful activator of Nrf2 than isoxidant treatment, and augmenting the use of the phytochemicals is a potential treatment to elevate the antioxidant defenses and protect cardiomyocytes against an oxidative challenge.
EGCG is an important green tea polyphenol. It eliminates and inactivates ROS by inducing expression of GPx, glutamate cysteine ligase, glutathione S-transferase, hemeoxygenase-1, and so on. Phase II detoxifying or antioxidant enzymes are regulated by Nrf2 (a redox-sensitive transcription factor). Nrf2 bound a cytoskeleton-binding protein, named the Kelch-like erythroid CNC homologue-associated protein 1 (Keap1), in normal physiological conditions, subsequently repressing its translocation to the nucleus or its activation. 56
Kobayashi et al. 57 hypothesized that covalent binding or oxidizing of cysteine residues found in Keap1 may potentiate the dissociation of the Keap1–Nrf2 complex or elevate the stability of Nrf2. Dinkova-Kostova et al. 58 suggested that the cysteine residues that are present in Keap1 act as a molecular sensor for identifying altered intracellular redox status triggered by electrophiles or ROS. Thus, activation of Nrf2 acts as a molecular target of many chemoprotective substances.
EGCG has chemopreventive effects through many mechanisms. One of the important mechanisms of EGCG is to exert powerful effects on cellular defense by the induction of phase II antioxidant enzymes. EGCG initiates the cellular defense against oxidative stress through Nrf2-mediated effects to synthesize phase II detoxifying or antioxidant enzymes. In addition, Nrf2 has been shown to directly or indirectly affect the expression of several genes that modulate cell growth, inflammation, apoptosis, and adhesion. 47
LDH was significantly increased in the IONR-overloaded group compared with the control non-IONR-overloaded group. Treatment with DFO and DFP alone or in combination with green tea compounds showed a significant decrease in LDH than the IONR-overloaded group. This study also showed that DFP was more powerful in decreasing LDH than DFO. In agreement with these results, Pucheu et al. 38 found that an iron nanoparticle overload was associated with a slight decrease in cardiac function and an increase in LDH level.
Additionally, Goda et al. 59 found that all cardiac markers including serum LDH, CK-MB, cardiac cTn-I, and cardiac TBARS were increased in cardiotoxicity. The mechanism of cardiotoxicity may be attributed to increased oxidative stress, possibly through the induction of NAD(P)H oxidase, the major enzyme responsible for the formation of ROS in the cardiovascular system.
The serum CK-MB activity and cTn-T levels were significantly increased in IONRs-overloaded rats compared with those in the control non-IONRs-overloaded group. The serum CK-MB activity and cTn-T were significantly decreased in the green tea, DFO, and DFP groups compared with the control IONRs-overloaded group. The present study also showed that DFP alone or in combination with green tea was more powerful in decreasing the cTn-T level and CK-MB activity than DFO.
Conclusions
The present study demonstrated that supplementation with green tea extracts and iron chelators decreases iron-mediated myocardial oxidative stress. Therefore, green tea is a potential therapeutic agent that could decrease the cardiovascular toxicity induced by IONR-overload. We can regulate IONR-overload to a tolerable level with DFP. Enhancement of iron excretion can be obtained by combining DFP or DFO with green tea therapy. Early treatment with chelators provides the best chelation and prevents cardiotoxicity.
Footnotes
Acknowledgement
The authors would like to express their gratitude to Professor Karima El-Desuky, Professor of Pathology, Faculty of Medicine, Tanta University, Egypt, for her devoted help in carrying out the histopathological study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
