Abstract
The aim of this study is to assess cardiotoxic effect of testosterone (TES) and dehydroepiandrosterone (DHEA) in Sprague Dawley rats. We compared the impact of subacute (14 days) and subchronic (90 days) administration of suprapharmacologic doses of TES and DHEA on body weight, locomotor activity, muscle strength, echocardiographic parameters, heart histopathology, and oxidative stress markers with the control group. Testosterone (10, 30, and 100 mg/100 g body weight) and DHEA (10 mg/100 g body weight) administration decreased the body weights and locomotor activity (p < 0.05), and the combination of both increased muscle strength (p < 0.05) in rats. In our histopathological evaluation, misshapen cell nuclei, disorganized myocardial fibers, and leukocytic infiltrates were observed in high-dose TES (100 mg/100 g)-treated rats, especially on day 14. On day 90, mild changes such as misshapen cell nuclei, disorganized myocardial fibers, and leukocytic infiltrates were observed in TES and DHEA-treated groups. According to our echocardiographic study on day 14 and day 90, TES, especially at high doses, induced increase in left ventricular posterior wall diameter and ejection fraction (p < 0.05). In this study, blood oxidative stress marker malondialdehyde was increased slightly but not significantly in TES and DHEA groups. On the other hand, antioxidant enzymes such as SOD and glutathione peroxidase (GSH-Px) levels were slightly but not significantly increased in TES and DHEA groups. These data demonstrate that the potential risk to cardiac health due to exogenous androgen use may be related to oxidative stress in rats.
Introduction
Anabolic androgenic steroids (AAS) are synthetic derivatives of testosterone (TES) approved for the treatment of wasting syndrome in human immunodeficiency virus infection, hypogonadism, anemia accompanying renal and bone marrow failure, endometriosis, and cancer. Not infrequently, powerlifters and bodybuilders tend to abuse AAS to enhance their sportive performance. Abuse of AAS has been linked to a variety of different cardiovascular side effects. In case reports, acute myocardial infarction is the most common event presented, but other adverse side effects include left ventricular hypertrophy, reduced left ventricular contractile function, hypertension, arterial thrombosis, pulmonary embolism, endothelial dysfunction, alterations of lipid metabolism, life-threatening arrhythmia, and several cases of sudden cardiac death. 1 –5
Dehydroepiandrosterone (DHEA) is a naturally occurring, cholesterol-derived steroid hormone secreted mainly from the adrenal cortex and serves in humans as a precursor of both estrogens and androgens. 6 DHEA has a wide variety of beneficial biological and physiological actions. 7 Recently, it has been shown that DHEA treatment slows down the progression of severe pulmonary arterial hypertension in vascular endothelial growth factor receptor blocker Sugen 5416/hypoxia/normoxia-exposed rats and protects the right ventricle against apoptosis and fibrosis, thus preserving right ventricular contractile function. 8
Oxidative stress is the result of imbalance between the production of reactive oxygen species (ROS) and antioxidant defense mechanisms. ROS are unstable and very reactive by-products of normal metabolism, causing damaging effects on the principal biomolecules. 9 As a consequence of these activities, physiological levels of ROS are low. However, with increased levels of ROS, defense systems can be overwhelmed resulting in cellular damage since AAS usually enhances the metabolic rate. 10 –12 Accordingly, high TES levels produce oxidation in rat and rabbit testicular tissues, 13 –15 rat muscles, 16 and human placenta. 17 On the other hand, antioxidant properties of TES have been shown in human prostate 18 and rat nervous systems. 19 However, to the best of our knowledge, little is known on association between AAS-induced cardiotoxicity and oxidative stress. Germanakis et al. have recently shown that short-term effects of repeated administration of turinabol and methanabol induce cardiotoxic effects due to their antioxidant actions in young rabbits. 20
Previously, histologic cardiac lesions including necrosis, misshapen cell nuclei, interstitial and endocardial fibrosis, lymphocytic infiltrates, and vascular dystrophies were observed in TES-treated rabbits. 21 Rats are more easily accessible than rabbits, and they are also more suitable for chronic studies with high number of experimental animals. In addition, smaller animals are much more used in experimental studies due to ethical considerations. To our knowledge, there is no study on cardiac lesions in TES and DHEA-treated rats.
The present study was undertaken to test the hypothesis that oxidative stress is involved in cardiotoxicity induced by high doses of TES and/or DHEA in rats. We also aimed to demonstrate whether the subacute and subchronic administration of suprapharmacological doses of TES esters and DHEA to rats could induce cardiac lesions similar to rabbits as AAS cardiotoxicity has seldom been investigated in laboratory animals. In addition, echocardiographic assessment was performed. Oxidative stress parameters were also measured to make clear the possible mechanism of AAS-induced cardiotoxicity in rats. Additionally, we aimed to evaluate the cardiotoxic effects of TES and DHEA in our rat model, which may be a beneficial model in future drug-induced cardiotoxicity studies.
Materials and methods
Animals
Male Spraque Dawley rats aged 6 months and weighing approximately 450 g at the beginning of the study were used for the experiment. They were housed in a cage with five rats in each in an air-conditioned animal room (22 ± 2°C) with controlled humidity (55 ± 15%) and 12-h light/12-h dark cycles, and they were given rat chow and tap water ad libitum. Their health status was checked daily, and their weight was measured once a week. The experimental study plan was approved by the Animal Ethics Committee of Gülhane School of Medicine (No: 13/46, dated 8 March 2013, Ankara, Turkey).
Treatments
Fifty male Spraque Dawley albino rats have been randomly assigned to 10 groups with 5 animals in each group. TES (Ustanon®, containing testosterone propionate, testosterone phenylpropionate, testosterone isocaproate, and testosterone decanoate) has been given by intramuscular injection once a week for 14 days at doses of 10 mg/100 g body weight (TES 10 mg, T10 group, group I), 30 mg/100 g body weight (TES 30 mg, T30 group, group II), and 100 mg/100 g body weight (TES 100 mg, T100 group, group III), respectively. Group IV received DHEA at dose of 10 mg/100 g body weight (DHEA group) by oral gavage daily for 14 days. Group V (control group) was given saline, in equal volumes with the treated animals, once a day by oral gavage and once a week by intramuscular injection for 14 days. Groups VI, VII, and VIII received intramuscular injections of TES 10 mg/100 g (body weight, T10 group), 30 mg/100g (T30 group), and 100 mg/100 g (T100 group) for 90 days, respectively. Group IX received DHEA at dose of 10 mg/100 g body weight by oral gavage daily for 90 days. Group X (control group) has been given saline once a day by oral gavage, and they also received intramuscular injections of saline, in equal volumes with the treated animals, once a week for 90 days.
Body weight and locomotor activity measurements
Body weights of animals were measured and recorded weekly. On day 0, +14, and +90, locomotor activity of the animals were measured. Locomotor activity was defined as the distance walked by the animals in a certain period and measured for 30 min in lightened cages with 40 × 40 cm2 black-colored basements. Movements of the rats were transferred to a computer by a charge-coupled device camera (Safer SF-2222) placed 57 cm above the locomotor activity area. This system was programmed to process images from eight cameras at the same time. The images were digitalized by a video tracking system (Ethovision v3.1.16, Noldus Information Technology, Wageningen, the Netherlands). This software used difference in contrast option to recognize white rats on black area. An integrated filter to decrease 29 frames/s (fps) video velocity to 6 fps was used, and the sensitivity for locomotor activity change was 1 cm at minimum. To remove tails from video recording, the erosion and deletion filters were used respectively. Both filters were set at 1 pixel per each animal.
Muscle strength measurements
To measure the effect of treatments on muscle strength, a special set up was used on day 0, +14, and +90. A soft polyurethane basement was placed under the animals, and a 0.5 cm nylon rope was placed tightly 70 cm above the basement. The rats were let to hold the rope with their front extremity paws. The time period until animals fall was measured, recorded, and scored (0 = 0–2 s, 1 = 3–4 s, 2 = holding for 5 s without placing third limb, and 3 = holding with third limb for 5 s.
Echocardiographic assessments
On days 14 and 90, 5 animals from each group were anesthetized intramuscularly with 400 mg/kg ketamine (Alfamine %10, Ege Vet, İzmir, Turkey) and 32 mg/kg xylazine (Alphazyne, %2 Ege Vet, İzmir, Turkey). Then, echocardiographic images were obtained using the Vivid S (General Electric, Fairfield, Connecticut, USA, 10 MHz, frequency transducer 10 × 13 mm2 footprint) at an image depth of 2 cm. Left parasternal and left apical echocardiographic images of anesthetized rats lying in the dorsal recumbency position have been obtained. Left parasternal M-mode measurements at the level of papillary muscles were used to define wall thicknesses and internal diameters at systole and diastole. The measured parameters were intraventricular septum in diastole (IVSD), intraventricular septum in systole (IVSS), left ventricular internal diameter in systole (LVIDS), left ventricular internal diameter in diastole (LVIDD), left ventricular posterior wall thickness in diastole (LVPWD), left ventricular posterior wall thickness in systole, ejection fraction (EF), fractional shortening (FS), aorta diameter (AO), and left atrium (LA).
Histopathological examinations
Blood samples of 5 ml from each animal were taken by intracardiac route. Then, the hearts were immediately cut from apex to the base to obtain full transverse slices of the midsection of the biventricular mass. One 5 mm slice from each heart was fixed in formalin and stained using hematoxylin–eosin for histopathological examination. A trained pathologist (Dr Kurt) assessed histopathological changes (misshapen cell nuclei, disorganized myocardial fibers, vascular dysplasia, interstitial fibrosis, endocardial fibrosis, and leukocyctic infiltrates) using a semiquantitive scale: 0 = no change, x = mild changes, xx = moderate changes, xxx = marked changes, and xxxx = severe changes.
Blood oxidative stress parameters
Erythrocytes were isolated from blood collected in ethylenediaminetetraacetic acid by centrifugation. The erythrocytes were washed thrice with ice-cold normal saline and then lysed by adding ice-cold distilled water. The hemolysate was used for the entire assays. The protein content was first measured according to the method of Lowry et al. 22 with bovine serum albumin as the standard. Lipid peroxidation levels were measured using the thiobarbituric acid reaction by the method of Ohkawa et al. 23 This method was used to obtain a spectrophotometric (Helios Epsilon, Thermo Electron Scientific Instruments LLC, Madison, Wisconsin, USA) measurement of the color produced during the reaction of thiobarbituric acid with malondialdehyde at 535 nm. The carbonylated protein content (PCC) of tissues was determined using the method described by Levine et al. 24 Final calculated thiobarbituric acid reactive substances (TBARS) and PCC levels were expressed as millimoles per gram protein. The activity of the antioxidant enzyme SOD was assayed using the nitro blue tetrazolium (NBT) method of Sun et al. 25 In brief, NBT was reduced to blue formazan by the superoxide anion radical, which has strong absorbance at 560 nm. One unit (U) of SOD is defined as the amount of protein that inhibits the rate of NBT reduction by 50%. Finally, GSH-Px activity was measured using the method described by Paglia and Valentine. 26 The estimated SOD and GSH-Px activities were expressed as units per gram protein.
Chemicals
All chemicals used for the above-described assays were obtained from Sigma-Aldrich Chemie GmbH (Taufkirchen, Germany), and all organic solvents were obtained from Merck KGaA (Darmstadt, Germany). All reagents were of analytical grade, were prepared each day (except the phosphate buffer), and were stored in a refrigerator at +4°C. The reagents were equilibrated at room temperature for 0.5 h before use when the analysis was initiated or reagent containers were refilled. Phosphate buffers were stable at +4°C for 1 month.
Statistical analyses
Values shown are means ± SEM. Analyses of variance with Scheffé post hoc test was used for comparisons among the experimental groups. Histopathological changes were compared across groups for each parameter using the nonparametric Kruskall and Wallis test. Differences were considered significant at p < 0.05.
Results
No adverse clinical effects were observed during the 90 days of the study, except for two animals of DHEA and T100 groups (groups I and IV) that died during the injection of anesthetics drugs on day 14, and two animals from group VIII were found dead in their cages at the end of the first and last weeks of the study.
Body weight gains
We observed no statistically significant difference in body weights of the study groups during the first 5 weeks of study. However, body weight of T10 group was significantly lower than DHEA and control groups at 6th week and was lower than DHEA group at 7th week (p < 0.05). No statistically significant difference in body weights of the study was observed at the 8th week. Body weight of T10 group was significantly lower than DHEA and control groups at weeks 9, 10, 11, and 12 (p < 0.05; data not shown).
Locomotor activity assessments
No statistically significant difference among the locomotor activity measurements of the study groups were observed at the beginning, day 14, and day 90 of the study. However, locomotor activity of T10, T30, and DHEA groups decreased at days 14 and 90 when compared with the values of day 1. Although only values of day 14 were significantly different from those seen at day 1 (p < 0.05). Additionally, locomotor activity of T100 group at both day 14 and day 90 were slightly but not significantly lower than the value of day 1 (p > 0.05; data not shown).
Muscle strength measurements
At day 14, the time period until fall (latency) of T100 group was significantly longer than the control (p < 0.01), DHEA, and T10 groups (p < 0.05). Accordingly, the muscle strength score of T100 group was significantly higher than the control, DHEA, and T10 groups (p < 0.01; data not shown). At day 90, latency value of DHEA and T10 and T30 groups were significantly longer than the control (p < 0.05), whereas latency value of T100 group was the longest among the groups and it was also longer compared with the control group (p < 0.01). In addition, muscle strength scores of DHEA, T30, and T100 groups were significantly higher than the control (p < 0.05 and p < 0.01, respectively; data not shown).
Echocardiographic assessments
According to our echocardiographic study at days 14 and 90, TES, especially at high doses, induced increase in left ventricular posterior wall diameter and EF (p < 0.05).
On day 14, TES increased EF and FS slightly but not significantly. Additionally, DHEA slightly but not significantly increased LWPWD, EF, and FS (Table 1).
Echocardiographic measurements on day 14.a

T: testosterone; EF: ejection fraction; FS: fractional shortening; DHEA: dehydroepiandrosterone; LVIDD: left venrticular internal diameter in diastole; LVPWD: left ventricular posterior wall thickness in diastole; AO: aorta diameter.
aValues are represented as mean ± SEM.
bp < 0.01: versus control group.
cp < 0.05: versus control group.
On day 90, high doses of TES (30 mg/100 g and 100 mg/100 g) increased LWPWD significantly when compared with the control group (p < 0.05). Additionally, TES (30 mg/100 g and 100 mg/100 g) increased EF and FS slightly but not significantly. TES (10 mg/100 g) increased FS significantly when compared with the control group (p < 0.01) (Table 2). On the other hand, DHEA increased LWPWD, EF, and FS slightly but not significantly. Echocardiographic parameters (apical four chamber views and M-mode measurements) are shown in Figures 1 and 2.
Echocardiographic measurements on day 90.a

T: testosterone; EF: ejection fraction; FS: fractional shortening; DHEA: dehydroepiandrosterone; LVIDD: left venrticular internal diameter in diastole; LVPWD: left ventricular posterior wall thickness in diastole; LVIDS: left ventricular intrenal diameter in systole; AO: aorta diameter.
aValues are represented as mean ± SEM.
bp < 0.05: versus DHEA group.
cp < 0.01: versus DHEA group.
dp < 0.01: versus control group.
ep < 0.05: versus control group.

Apical four-chamber view of heart structure in T100 group on day 90. LA: left atrium; LV: left ventricle; RA: right atrium; RV: right ventricle.

Typical M-mode measurements of left ventricular structures over several cycles in T100 group on day 90. IVSD: interventricular septum in diastole; IVSS: interventricular septum in systole; LVEDD: left ventricular end diastolic diameter; LVEDS: left ventricular end systolic diameter; PWD: posterior wall in diastole; PWS: posterior wall in systole.
Histopathological findings
In control group, no accordant cardiac lesions were observed. However, high doses of TES induced myocardial lesions ranked as moderate (xx) and marked (xxx) histopathological changes. Highest dose of TES (100 mg/100 g) induced more frequent histological lesions (p < 0.05) including misshapen cell nuclei, disorganized myocardial fibers, and leukocytic infiltrates when compared with the control group at day 14 (Table 3). At day 90, high doses of TES (30 mg/100 g and 100 mg/100 g) induced more frequent and marked histological lesions including misshapen cell nuclei, disorganized myocardial fibers, and leukocytic infiltrates as compared to the control group (p < 0.05; Table 4, Figures 3 and 4).
Incidence and severity of histologic cardiac lesions observed in rats on day 14.a

T: testosterone; DHEA: dehydroepiandrosterone.
aThe severity of changes was assessed semiquantitatively and is denoted as follows: 0 = no changes; x = mild changes; xx = moderate changes; xxx = marked changes.
Incidence and severity of histologic cardiac lesions observed in rats on day 90.a

T: testosterone; DHEA: dehydroepiandrosterone.
aThe severity of changes was assessed semiquantitatively and is denoted as follows: 0 = no changes; x = mild changes; xx = moderate changes; xxx = marked changes.
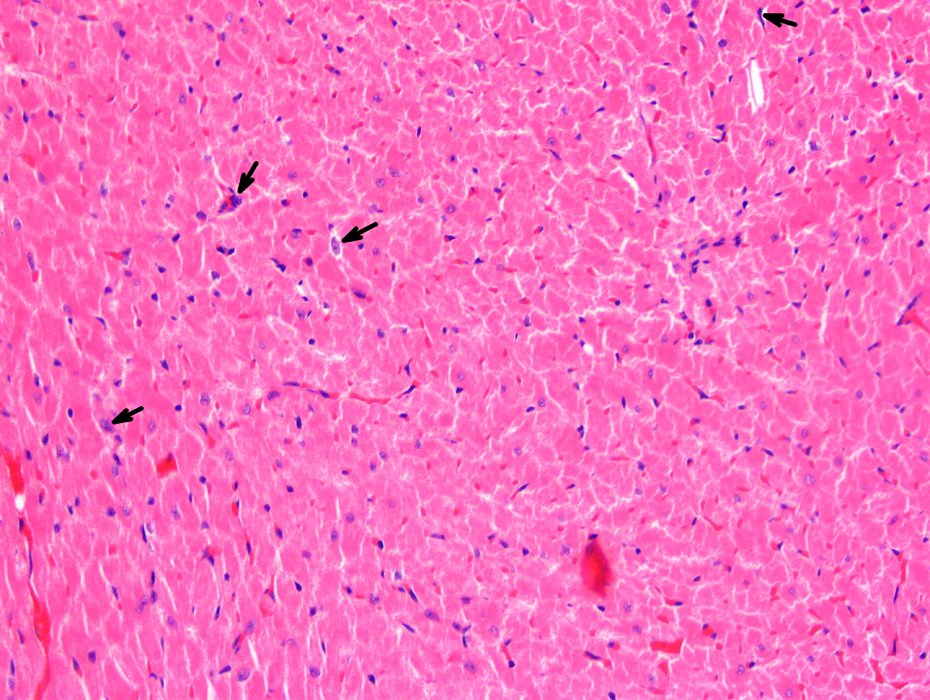
An example of histologic cardiac lesions observed in rats that received testosterone (100 mg/100 g body weight, once a week, T100 group) by the intramuscular route for 90 days. Arrows indicate some of the misshapen nuclei (×200, HE). HE: hematoxylin and eosin.

An example of histologic cardiac lesions observed in rats that received testosterone (100 mg/100 g body weight, once a week, T100 group) for 90 days. Patchy necrosis with inflammatory cell infiltration in myocardium. Arrows indicate border between viable myocardium and the lesion (×200, HE). HE: hematoxylin and eosin.
Oxidative stress parameters
Levels of systemic oxidative stress parameters monitored in the present study are presented in Figures 5 to 7. In particular, oxidative stress marker TBARS increased slighty but significantly in TES and DHEA groups. On the other hand, SOD and GSHPx levels were slighty but not significantly increased in TES and DHEA groups (Figures 5 to 7).

Levels of TBARS in experimental groups of rats. TBARS: thiobarbituric acid reactive substances.

Superoxide dismutase levels in experimental groups of rats.

Glutathione peroxidase levels in experimental groups of rats.
Discussion
In this study, the administration of TES and DHEA was found to induce various histologic cardiac lesions in rats including misshapen cell nuclei, disorganized myocardial fibers, and leukocytic infiltrates. Previously, Belhani et al. reported for the first time that TES might induce similar cardiac lesions in rabbits. 21 In our study, TES, especially at high doses, induced increase in LVPWD and EF, which indicates there is increased cardiac contractility in the hearts of these animals. Conclusively, high-dose TES is associated with consistent increase in left ventricular wall, but not chamber, dimensions, indicating hypertrophy without ventricular dilatation. DHEA increased left ventricular posterior wall thickness, EF, and FS. We observed that AO was increased in TES groups compared with the control group. The reason for this increase may be related to increase in afterload in TES-treated groups. In DHEA group, LVIDs was increased with no concomitant increase in LVIDD. In accordance with these findings, stroke volume (diastolic volume − systolic volume) and cardiac output may be decreased in DHEA group. Previously, Brown et al., assessed echocardiographic parameters in triiodothyronine-treated hyperthyroid rats, and they found an increase in LVPWD as a marker of hypertrophy. 27
Cardiac and metabolic effects of AAS remain inadequately characterized. 3 The heart has elevated intrinsic oxidative metabolic activity and fragile antioxidant resistance, ın comparison with other parts of the body. Therefore, it is susceptible to free radical damage. Our findings indicate that administration of TES and DHEA may induce oxidative stress. More specifically, DHEA and TES (dose dependently) increased TBARS levels. At the same time, the increase in SOD and GSH-Px levels that we observed could correspond to a probable rescue mechanism to counteract oxidative stress induction. Recently, Germanakis et al. have demonstrated that repeated low level administration of turinabol and methanabol may induce localized cardiotoxic effects based on their echocardiographic findings in young rabbits, Furthermore, the oxidative stress levels, as depicted by glutatione and total antioxidant capacity levels in serum, have been increased in methanabol-administered rabbits. 20 Although a decrease in GSH or catalase activity is expected under oxidative stress, this is not always the case. In several studies, 28,29 there is an increase either in GSH or in other antioxidant factors under oxidative stress. The explanation for these contradictory findings is that sometimes under oxidative stress conditions the organism increases antioxidant molecules as a compensatory mechanism against free radicals. High TES levels produce oxidation in rat and rabbit testicular tissues, 13 –15 rat muscles, 16 and human placenta. 17 Accordingly, oxidative stress is suggested to be one of the apoptotic pathways leading to cardiotoxic effect. 30 The present data together with the studies stated above demonstrate that the potential risk to cardiac health due to exogenous androgen use may be related to oxidative stress.
Another important finding that should be indicated is that some TES-treated animals show leukocyte infiltrates misshapen nuclei and disorganized myocardial fibers in the myocardium by day 90. Previously, similar lesions of the myocardium have been reported in athletes with a history of anabolic steroid abuse 1 and in rabbits administered with TES. 21 TES has been reported to exert pro-inflammatory effects with the release of tumor necrosis factor α and interleukin 1β, which in turn resulted in apoptosis of cardiac cells. 31 Our results, together with previous reports, suggest that inflammatory pathways may account for the pathological alterations in TES-treated animals.
In this study, we have found that subacute and subchronic TES and DHEA administration decrease the body weights and locomotor activity, and they increase muscle strength in rats. Previously, Perrot-Sinal et al. and Mead et al. showed that plasma TES levels were correlated with various aspects of locomotor activity in wild-caught and laboratory-bred male meadow voles. 32,33 Additionally, TES replacement increased locomotor activity in castrated rodents. 34 On the other hand, Clark and Barber reported that TES replacement increased aggressiveness, but it did not change locomotor activity in castrated male Long-Evans rats. 35 In this study, the observed decrease in locomotor activity may be related to TES and DHEA-induced cardiomyopaty and associated decrease in activity of rats. In our study, we observed that body weights of TES-treated rats decreased after week 8 of the experiment. In accordance with our findings, several studies showed that TES may induce hypophagia and decrease of body weight in rats. 36 –42 To our knowledge, there is no study related to the effects of DHEA on food intake and body weight in rats. In our study, the observed decrease in body weights in DHEA-treated rats may be related to irritation with gavage needle and related decrease in food intake. In our study, the observed increase in muscle strength is similar to previous studies which reported muscle strength increase due to AAS treatment. 43 –46
Echocardiography is a noninvasive method of choice to define cardiac parameters serially and to monitor drug-induced cardiotoxicity progression and treatment. Parameters include left ventricular mass and dimensions, as well as the characterization of blood flow through the heart and great vessels. 47 This technique is less frequently applied in studies on rat, the most commonly used model of human cardiovascular disease. Previously, Brown et al. showed that this technique provides useful information on cardiac changes occurring in thyroid dysfunction as well as on cardiac abnormalities. 27 Our study showed that cardiotoxic effects of TES and DHEA may be evaluated with echocardiography in rats which may be a beneficial model in future drug-induced cardiotoxicity studies.
The main findings of our study were assessed though within study limitations. We were not able to measure oxidative stress parameters in heart tissue because we did not have a validated method and experience for measurement of tissue concentrations of TBARS, SOD, and GSH-Px, and we plan to measure oxidative stress parameters in heart tissue in our future studies. In the present study, we used only one high dose of DHEA, but we plan to try higher doses in our future experiments. However, the consistent patterns of observed differences, the concordance pathological and echocardiographic findings with oxidative stress marker assessment, argue for a potential hazardous effect to anabolics when applied in the heart.
In summary, based on echocardiographic and pathological findings of this study, cardiotoxic effects of anabolic androgenic steroids and, in particular, TES have been identified resulting in heart lesions similar to those seen in toxic myocarditis. Furthermore, oxidative stress, as depicted by TBARS, SOD, and GSH-Px levels in serum, may be related to mechanism of this cardiotoxic effect. Therefore, anabolic administrations may create possible deleterious long-term effects on the heart and should be strongly discouraged in human subjects.
Footnotes
Acknowledgment
We would like to thank Dr Hakan Kayir for his assistance in locomotor activity measurements.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
