Abstract
Rosiglitazone is in the thiazolidinedione class of drugs used in the treatment of type 2 diabetes mellitus. It works as an insulin sensitizer by binding to the peroxisome proliferator–activated receptor gamma. We investigated the effects of prenatally administered rosiglitazone on pyramidal cell numbers and morphologies in the hippocampus at postnatal period using histochemical and stereological techniques, congenital morphological properties and the number of offspring in rats. Eighteen female rats were grouped into control (C), low-dose rosiglitazone (LDR) and high-dose rosiglitazone (HDR). LDR pregnant rats received 2 mg/kg/day of rosiglitazone via oral gavage during the first 16 days of the pregnancy. HDR rats received 5 mg/kg/day. The infants were grouped into newborn (NB), 4 week (4 W) and 12 week (12 W). A side from histopathologic and congenital assessments, stereological analyses were performed using the optical fractionator method. Congenital anomaly was not detected in any of the rosiglitazone treatment groups, and their number of offspring was similar to that of the C group. Stereological counts revealed a significant reduction in the number of hippocampal pyramidal cells in the C and LDR groups but not in the HDR group until birth to 12th week. When NB groups were compared, the number of pyramidal cells in the HDRNB group was less than those in the LDRNB and CNB groups. HDR affected apoptosis or the proliferation and maturation of progenitor cells to the pyramidal neuron during neurodevelopment in the hippocampus, whereas LDR did not adversely affect neuronal development and did not cause congenital anomalies.
Introduction
Thiazolidinediones (TZDs) are insulin-sensitizing agents that are commonly studied and recently used in type 2 diabetes mellitus treatment. TZDs provide glycaemic control by decreasing insulin resistance. They can enter the nucleus because of their lipophilic structure and lead to activation by binding to the peroxisome proliferator–activated receptor gamma (PPAR-γ) through a peroxisome proliferator. 1
Rosiglitazone, which is a member of the TZD family, is reported by the Food and Drug Administration to be in the control (C) category in terms of its use during pregnancy. That is, no sufficient study and data have been found on its effects on the foetus. 2 Chan et al. stated that rosiglitazone could be transferred to the foetus through the placenta in the first trimester of pregnancy and that rosiglitazone exposure carries high risks for the foetus. 3 However, Klinkner et al. showed that rosiglitazone use did not lead to phenotypic disorders in the mouse foetus and did not damage blastocyst development under in vitro conditions. 4
Different studies have attempted to clarify the reliability of rosiglitazone use during pregnancy. Studies have also focused on the agonists of rosiglitazone, such as receptors (PPAR-γ) activated by a peroxisome proliferator, to examine the undefined mechanisms of rosiglitazone. The biology of PPARs in the physiological and pathophysiological processes has been studied particularly in peripheral organs and tissues. In recent years, PPARs have been indicated to play important roles in the pathogenesis of central nervous system (CNS) diseases. The activation of PPARs, particularly the PPAR-γ isoform, in the peripheral macrophages and human autoimmune disease models has been found to inhibit inflammation. These findings encourage researchers to examine these important effective activities in many CNS disorders with the same inflammatory component. 5 Many studies in this direction have indicated that PPAR-γ agonists prevented neurodegeneration in the hippocampal neurons. 6,7
Moreover, rosiglitazone affects different cell types at varying doses. PPAR-γ agonists were reported to induce apoptosis in different cell types, including endothelial, vascular smooth muscle and cancer cells. 8,9 Conversely, rosiglitazone was shown to protect cardiomyocytes, β-islet cells and neurons against apoptosis. 10 –12 These reports have contradictory findings, but these inconsistencies can be explained by the use of different concentrations, cell types and/or agonists. The biphasic mechanisms of TZDs related to their concentration-dependent cell and tissue protection have not yet been explained, and more studies should be performed on this topic.
In this study, rosiglitazone (PPAR-γ agonist), which is commonly used in type 2 diabetes mellitus treatment, was used. We aimed to examine the postnatal stereological and histological effects of the two different rosiglitazone doses that were administered in the prenatal period on the number of pyramidal cells located in the cornu ammonis (CA) area of the hippocampus.
Materials and methods
Animals
In this study, 18 male and 18 female Wistar albino rats weighing 210–250 g each were allowed to mate in the Experimental Animal Research Centre of Ondokuz Mayıs University. Pairs of male and female rats were mated overnight in standard plastic cages. Vaginal plaque formation was the accepted criterion for pregnancy determination, and the date when the vaginal plaque formation was observed was set as the first day of pregnancy. Pregnant animals were housed in standard sterile plastic cages in an air-conditioned room at 20°C under a 12-h light/12-h dark cycle and fed ad libitum.
Pregnant rats were transferred to individual cages during pregnancy. They were randomly divided into three groups: C, low-dose treated and high-dose treated. These main groups were further divided into three equal subgroups, namely, newborn (NB), 4-week-old (4W) and 12-week-old (12W) with each group containing two pregnant rats. The groups were labelled as follows: CNB, C4W, C12W, LDRNB, LDR4W, LDR12W, HDRNB, HDR4W and HDR12W. Drugs at 2 and 5 mg/kg/day were given to the rosiglitazone low-dose group (LDR) and rosiglitazone high-dose group (HDR), respectively, by oral gavage during the first 16 days of the pregnancy (GlaxoSmithKline (GSK) Brentford, London). The NB rats from each group were obtained upon spontaneous normal delivery. A total of 45 male NBs were included in the study at five NBs per group. Those in the NB group were sacrificed. The baby rats at 4 weeks old (4W) and 12 weeks old (12W) were maintained under the same conditions until they were killed. The presence or absence of malformations in the babies of the C and experimental groups was evaluated by the naked eye during the study.
Perfusion and fixation
After ketamine hydrochloride exposure (40–50 mg/kg; Alfazyne®, Egevet, Turkey), all rats were killed through the heart by 4% formaldehyde perfusion at room temperature. The brain tissues were removed and put in 10% neutral buffered formalin fixing solution. The tissues were exposed to routine histological procedures and then embedded in paraffin upon fixation. The 5- and 20-µm serial sections were obtained from paraffin blocks in accordance with the stereological sampling using a rotary microtome (Leica RM 2135; Leica Instruments, Nussloch, Germany). These sections were stained with Cresyl violet dye. This study was approved by the Experimental Animal Ethics Committee of Ondokuz Mayıs University, and all the experimental procedures were performed according to ethical rules.
Stereological analysis
The total number of pyramidal cells in the CA area of the hippocampus was calculated using the optical fractionator method. A pilot experiment was conducted before we initiated the study because of the principles of stereology. In accordance with the data obtained from the pilot study, sections were determined from the stereological analysis using the systematic random sampling method (i.e. one of every six sections for 4W and 12W rats and one of every four sections for NB rats). As a result of this sampling method, an average of 20 sections was obtained from each rat. Area sampling fractions of 4W and 12W rats as well as NB rats were found to be 900/40,000 and 625/122,500 µm 2 , respectively. The height of the dissector was arranged at 10 μm. We did not include the 2-μm distance in the upper part of the section in the calculations as we did for the upper guard zone analysis. Thus, we used the 20 μm/t sampling fraction thickness, with t as the mean thickness of the section. The stereological methods have been explained previously. 13,14
Statistical analysis
The Statistical Package for Social Sciences for Windows version 15.0 was used for the statistical analysis. The non-parametric Kruskal–Wallis test was used to compare the groups among each other. The results were regarded significant at a 95% confidence interval and when p < 0.05 and statistically insignificant when p ≥ 0.05. The non-parametric Mann–Whitney U-test with Bonferroni correction was used to compare the two groups (Bonferroni-corrected p value, p = 0.05 per group number).
Results
Neonatal examination and number of puppies
According to our observations, we did not determine any marked alteration during the pregnancy of the female rat groups. The number of infants in all groups was analysed by the Kruskal–Wallis test, and no significant difference among the groups was found (p = 0.892). No congenital morphology in NBs was found when they were observed with the naked eye.
Histological results
According to the results of the light microscopic rat brain sections in both the control and drug-treated all-age groups (Figures 1 to 3), the hippocampus was extended as a grey matter layer along the lower base of the horn of the lateral ventricle. The hippocampus side that facing the ventricles was convex and that facing the bottom of the hemisphere was concave. The CA in the hippocampus was divided into CA1, CA2, CA3 and CA4 because of the different cell types. The crescent-shaped gyrus dentatus surrounding the bottom of the CA4 region was in the form of a set of neurons, and it was clearly distinguished. The hippocampal neurons were pyramidal, with a basophilic cytoplasm and a euchromatic nucleus. We observed one or two nucleoli in each nucleus.
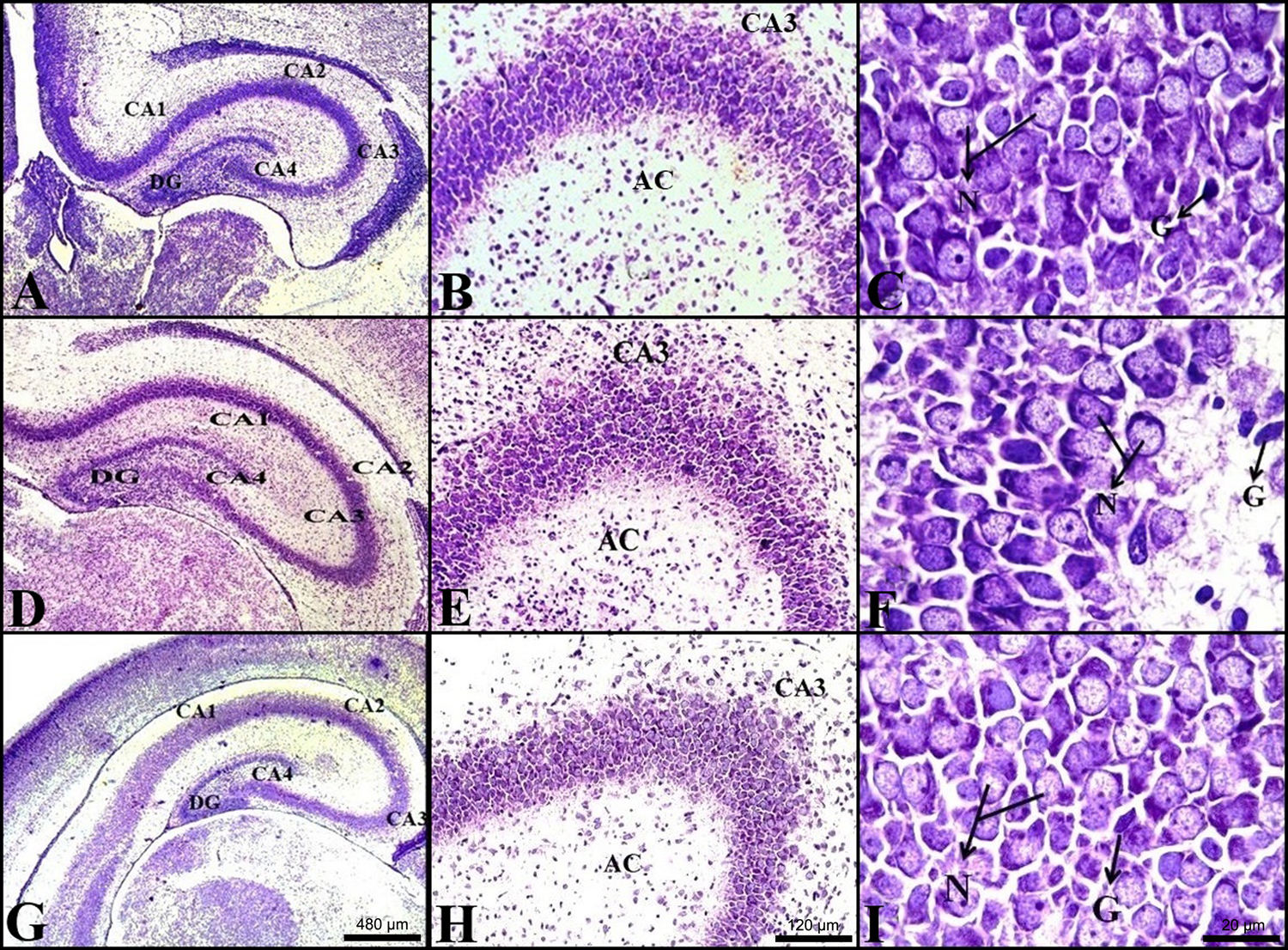
Light microscope sections obtained from NB rats.

Light microscope sections obtained from 4-week-old rats.

Light microscope sections obtained from 12-week-old rats.
Stereological results
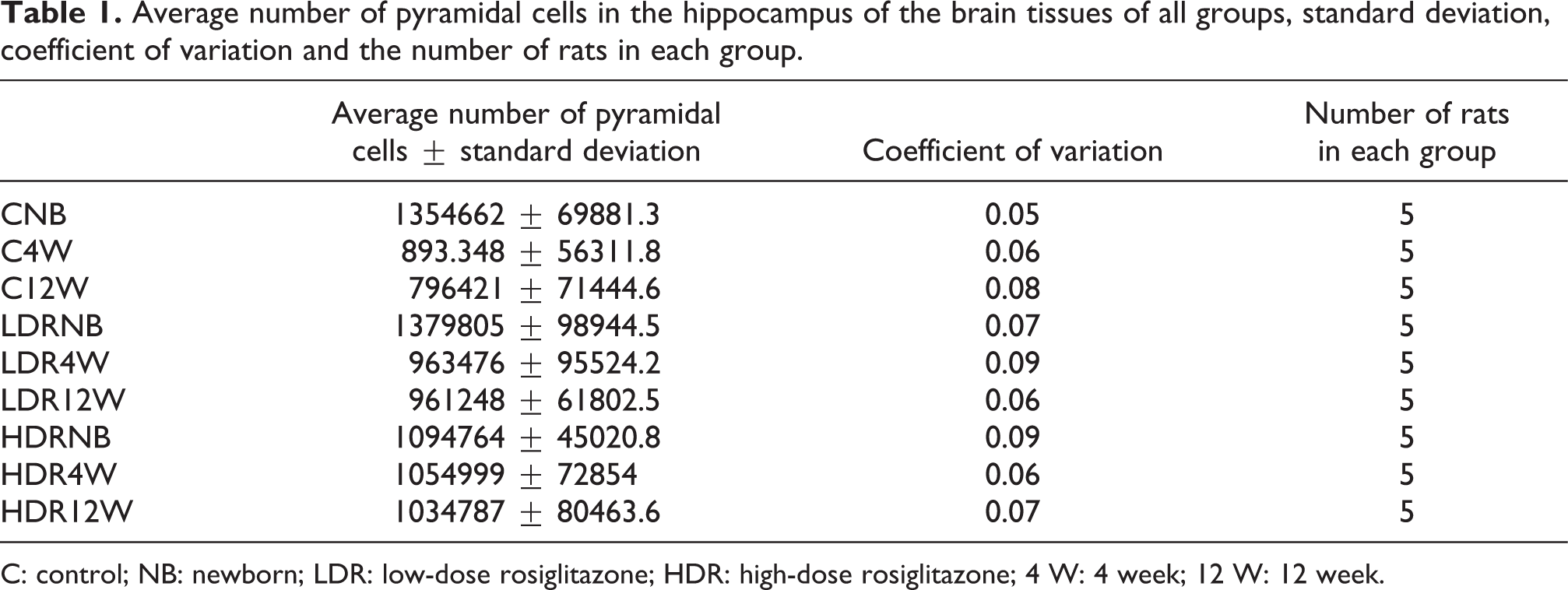
The total number of pyramidal cell counts and the coefficients of variations are given in Figure 4 and Table 1, respectively. These findings were obtained from the stereological calculations of the CA area in the hippocampus of all groups.

Comparison of the total number of pyramidal cell counts of the NB, 4-week-old and 12-week-old age groups between the control and the experimental groups.
Average number of pyramidal cells in the hippocampus of the brain tissues of all groups, standard deviation, coefficient of variation and the number of rats in each group.
C: control; NB: newborn; LDR: low-dose rosiglitazone; HDR: high-dose rosiglitazone; 4 W: 4 week; 12 W: 12 week.
Comparison of the neonatal group
No significant difference was found between the CNB and LDRNB groups (p = 0.75). In evaluating the neuron numbers of the CNB and HDRNB groups, a statistically significant decrease in the neuron numbers of the LDRNB group (p = 0.009*) was observed. In comparing between the LDRNB and HDRNB groups, a statistically significant decrease was found in the neuron numbers of the HDRNB group (p = 0.009*).
Comparison of the 4-week-old groups
No significant difference was found between the C4W and LDR4W groups (p = 0.251) and between the LDR4W and HDR4W groups (p = 0.076). A statistically significant increase was observed in the neuron numbers of the HDR4W group compared with the C4W group (p = 0.009*).
Comparison of the 12-week-old groups
No significant difference was found between the C12W and LDR12W groups (p = 0.016) and between the LDR12W and HDR12W groups (p = 0.251). A statistically significant increase was observed in the neuron numbers of the HDR12W group compared with the C12W group (p = 0.009*).
Comparison of the control age groups among themselves
The neuron numbers of the CNB group were significantly higher than those of the C4W group (p = 0.009*). Similarly, the neuron numbers of the CBN group were significantly higher than those of the C12W group (p = 0.009*). No significant difference was found between the C4W and C12W groups (p = 0.047).
Comparison of the low-dose rosiglitazone age groups among themselves
A significant difference was found between the LDRNB and LDR4W groups (p = 0.009*) and between the LDRNB and LDR12W groups (p = 0.009*). No significant difference was observed between the LDR4W and LDR12W groups (p = 0.754).
Comparison of the high-dose rosiglitazone age groups among themselves
No significant difference was observed among the groups according to the analysis of the Kruskal–Wallis tests (p = 0.512).
Discussion
The results from the present study showed that no significant difference was found between the infants of rosiglitazone-given rats and the control rats in terms of number and morphology. Klinkner et al. evaluated the rosiglitazone use in pregnancy, blastocyst development, number and weight of offspring and morphological changes. 4 They indicated that the rosiglitazone use in pregnancy did not lead to a phenotypic disorder of the foetus and did not damage the in vitro blastocyst development. Two case reports stated that rosiglitazone use in the early periods of pregnancy did not cause any malformation in the baby. 15,16 In this study, we also compared the numerical calculations obtained from pyramidal cell counts performed by stereological counting methods between the C and experimental groups in terms of age group. Three interesting results were determined in the evaluation of the total pyramidal cell number in the hippocampus according to age and treatment group.
The first finding is related to the number of pyramidal cells in the hippocampal brain tissue in different control age groups. According to our results, cell numbers of the CNB rats decreased in the C4W and C12W rats. The formation of all neurons in the brain is completed in rats after they are separated from their mothers. The speed and the termination time of the cell division in the brain can be altered in different periods. For example, the cell division in the hippocampus occurs in the earlier period and ends 6 days after birth. 17 According to the results of Dobbing, neurogenesis is completed on the 2nd day of birth in rats and on the 25th week of pregnancy in humans. 18 Later, the growing period begins, and this period starts on the 25th day after birth in rats and at the end of the second trimester in humans. This growing period continues until the age of two. Rat brain or the different parts of rat brain are known to exhibit weight increase continuously after the 90th day of birth. 19 –21 This weight increment of the brains is due to the increase in glial cells and amounts of myelin, cholesterol, phospholipids and glycolipids but not to increase in neuron number. 12 On the basis of the literature, we considered the possible reasons for the pyramidal cell number reduction with age. Neurogenesis is completed in rats in the first postnatal week, and then the brain undergoes a rapid growth period. During this period, cells die through apoptosis after the first week and the increment of the neuron number ends.
The second interesting finding is that no difference was found in the cell numbers between LDRNB rats and CNB rats. However, the HDRNB rats had a lesser cell number. In the development of the hippocampus, cell numbers of pyramidal neurons are limited by apoptosis at the level of progenitor or stem cells and postmitotic neurons. Conversely, the pyramidal neuron cell number increases in the ventricular and subventricular regions through the differentiation of the progenitor or stem cells and mitosis. In our study, rosiglitazone treatment, which was applied up to gestational day E17, did not affect the pyramidal neuron cell numbers in the hippocampus of offspring in the neonatal period. However, a high-dose application decreased the pyramidal neuron numbers. This finding can be explained by the high-dose triggering the apoptosis in neuronal stem cells or postmitotic neurons in the prenatal period or inhibiting the mitosis in progenitor cells. In the literature, different experimental models have shown that rosiglitazone has protective and antiapoptotic effects on both neural stem cells (NSCs) and mature neurons by preventing the glial-induced inflammation or regulating the intercellular pathways. In ischemia–reperfusion-induced and traumatic brain injury animal models, 22 –28 rosiglitazone has been reported to suppress the glial inflammatory cytokine production (TNFα and IL-1β) through the over-activation of glial cells (astrocyte or microglia). This finding correlates with the reduction in neuronal death rates in the hippocampus or other brain areas. Various studies have shown that rosiglitazone treatment decreases apoptotic cell death in various ways. For example, it regulates different intracellular pathways, such as decreasing NMDA-induced excitotoxicity in a temporal lobe epilepsy model. 5 Moreover, it enhances the increased mitochondrial function (reflected by the levels of adenosine triphosphate and Mit mass) and increases the PPAR-γ-dependent mRNA upregulation of PGC1α and mitochondrial genes (nuclear respiratory factor-1 and TFAM) and oxidative defence genes (superoxide dismutase 1, superoxide dismutase 2 and glutathione peroxidase 1) in human NSCs against amyloid-β-induced mitochondrial dysfunction and oxidative stress. 29 Furthermore, rosiglitazone treatment normalizes the endoplasmic reticulum stress sensors, such as Bip, CHOP and ASK1, in mutant huntingtin-expressing neuroblastoma cells. 30 It also increases NF-α1 and BCL-2 expressions in H2O2-induced cytotoxicity in Neuro2a cells and primary hippocampal neurons. 31 It decreases the glutamate and malondialdehyde levels in neurons in an experimental rat model of subarachnoid haemorrhage. 21 It reduces the ROS generation and enhances the HO-1 expression in chlorpyrifos-exposed SH-SY5Y cells (21); increases the UCP2 expression; reduces protein oxidation, O(·2)(-) overproduction and dysfunction of mitochondrial complex I; hinders the translocation of Bax and cytochrome c; and reduces the DNA fragmentation in the CA3 subfield in the hippocampus following experimental status epilepticus. 32 According to findings of this literature review, high doses of rosiglitazone can lead to decreased neuron cell amount in the neonatal group, and rosiglitazone plays a role in the neonatal differentiation and the inhibition of mitosis rather than in increasing the neuronal apoptosis. Similarly, various studies 22,23,29 have shown that rosiglitazone treatment also causes a biphasic effect on the neuronal differentiation or mitosis. Wada et al. found that physiological concentrations of rosiglitazone induced the NSC growth. 33 Conversely, high-dose rosiglitazone (HDR) inhibited the differentiation of NSCs into neurons with the activation of PPAR-γ. 29 Consequently, rosiglitazone inhibited the in vitro growth of seven human neuroblastoma cell lines (Kelly, LAN-1, LAN-5, LS, IMR-32, SK-N-SH and SH-SY5Y) in a dose-dependent manner (only at high concentrations; 10 and 100 µM). According to the results of Ren et al., a high dose of rosiglitazone (20 µM) significantly inhibited the SK-N-AS cell adhesion. 22
This study interestingly showed a significant decrease in cell numbers of the C and the LDRNB, LDR4W and LDR12W rats with age; only the HDR-treated group did not show any difference in cell numbers with age. The decrease in neuron cell numbers in the case of low doses was parallel to that in the healthy control group cell numbers. This result is related to the control of the neuron cell numbers by apoptosis from the first week of the postnatal period. A decrease according to the weeks was observed when high doses were applied. This occurrence can be explained by the fact that the organism or the organ provides a sufficient number of neurons to the hippocampus in the postnatal 4th and 12th weeks because of the low numbers of foetal neurons. This control mechanism can be explained by apoptotic deaths, which were observed from the first week in the C group, being inhibited in this group.
Recent literature has shown a growing interest in the use of TZDs in complicated pregnancies, including those with intrauterine growth restriction, preterm birth, pre-eclampsia and gestational diabetes. Data on rosiglitazone use in pregnancy are limited, and this study provides the first report on the effects of rosiglitazone use in pregnancy on hippocampal development. Although this work demonstrates that the low-dose rosiglitazone used in pregnancy did not lead to any morphological abnormality and hippocampal neuron number in the neonatal and postnatal periods, additional studies must be undertaken before conducting human studies.
Conclusions
Our findings suggest that HDR affects apoptosis or the proliferation and maturation of progenitor cells to the pyramidal neuron during neurodevelopment in the hippocampus (CA1, CA2, CA3 and CA4). This situation may cause short- and long-term memory deficits in the offspring. However, a low-dose intake makes up a significant data related to usage for pregnancy as it did not both affect neuronal development negatively and cause congenital anomaly. This study will illuminate the other studies on this subject, as no sufficient experimental study on rosiglitazone on the foetus has been made.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the project number PYO.FEN.1904.09.023 of 19 May University
