Abstract
The present study was designed to prepare a specific safe antiserum for Naja nigricollis using γ-irradiated (1.5KGy and3KGy) venoms. Rabbits were used for active immunization using irradiated venoms (1.5 and 3 kGy) as a toxoid, mice were used for determination of LD50 post immunization and the rats were used for neutralization of the cardiotoxic effect of venom. Results of the immunodiffusion test indicated that the sera of rabbits raised against non-irradiated, 1.5- and 3-kGy γ-irradiated venom, had the same results of precipitin bands. A significant inhibition of phospholipase A2 activities was obtained when neutralized with native, γ-irradiated (1.5KGy and3KGy) venoms. On the other hand, preincubation of the venom ½ LD50 (0.154 mg/kg i.p.) with each antiserum (non-irradiated or irradiated venom) at 37°C for 1 h in a ratio (1:4) produced a significant reduction in the values of creatine kinase and creatine kinase isoenzyme-MB. However, significant elevation in aspartate aminotransferase level and no change in lactate dehydrogenase level were observed. So the results of this study indicated that the irradiated venom treatment reduces the cardiotoxic effect of venom in immunized immunization animals for preparing vaccines.
Introduction
Snake venom is a complex mixture of many substances, such as toxins, enzymes, growth factors, activators and inhibitors with a wide spectrum of biological activities. 1,2 They are also known to cause different metabolic disorders by altering the cellular inclusions and enzymatic activities of different organs. Although the majority of snake species is non-venomous rather than venomous, snakebite remains an important medical problem in both developing and developed countries. 3 Snake bite poses a main health risk in many countries, with the international snake bites exceeding 5,000,000 per year. 4 Snake bite envenomations are frequently treated with parenteral administration of horse- or sheep-derived antivenoms aiming at the neutralization of toxins. 5
Naja nigricollis or the black-necked spitting cobra is a venomous snake species belonging to the family Elapidae, which is the most venomous and widely distributed snake family. 6 It is one of the most famous snakes distributed in the southern part of Egypt. 7 The venom of N. nigricollis is somewhat unique among elapids in that it consists primarily of cytotoxins 8 but with other components also. It retains the typical elapid neurotoxic properties while combining these with highly potent cytotoxins (necrotic agents) 9 and cardiotoxins. 10 Bite symptoms include severe external haemorrhaging and tissue necrosis around the bite area and difficulty breathing. Although mortality rate in untreated cases is low (approximately 5–10%), when death occurs, it is usually due to asphyxiation by paralysis of the diaphragm. 11
Snake venom is one of the most abundant sources of secretory phospholipases A2 (PLA2s), which are one of the potent molecules in snake venoms. 12,13 All snake venoms so far investigated contain PLA2 activities. 14 Elapid and crotalid snake venoms also are known as rich sources of multiple forms of PLA2 enzymes. 15
Various biological activities have been found in snake venom PLA2s such as neurotoxicity, myotoxicity and anticoagulant activity. Some snake venom PLA2 enzymes inhibit blood coagulation. Based on their anticoagulant properties, they are classified into strong, weak and non-anticoagulant enzymes. 12,16
Many of the venom biological activities could be neutralized by the homologous and heterologous antivenins; the lethality of elapids could only be neutralized by the corresponding antivenins. 17 –19
Ionizing radiation has been successfully employed to modify the immunological properties of biomolecules. Very promising results were obtained when crude animal venoms, as well as isolated toxins, were exposed to cobalt (Co)-60 γ-rays, producing toxoids with greatest immunogenicity. The achievement of modified antigens with lower toxicity and preserved or improved immunogenicity can be very beneficial. Ionizing radiation has already been confirmed to be a potent tool to reduce snake venom toxicity without affecting, and even increasing, their immunogenic properties. However, little is known about the alteration that irradiated molecules undergo and even less about the immunological response that such antigens elicit. 20
In order to improve antisera production and extend the lifespan of immunized animals to cut down its production cost, several trials have been used to detoxify venoms. One of the most important methods used to prepare an effective and safe toxoid is exposed to γ-radiation 21 –25 .
So, the present work aims to prepare an effective and safe toxoid for N. nigricollis venom by γ-irradiation (1.5 and 3 kGy) regarding the cardio toxicity of the venom in the immunized animal.
Materials and methods
Animals
White male Swiss albino mice, male Newzealand white rabbits and adult male Wistar albino rats were used. Animals were obtained from the Institute of Ophthalmology (Giza, Egypt). The animals were kept under appropriate conditions; for mice and rats, temperature was being 20–25°C, the humidity was 40–70% and light was 12/12-h light/dark cycle, while the rabbit’s temperature was 21 ± 2°C and the same humidity and light of mice and rats throughout the period of investigation. They were allowed free access to food consisting of typical pellets obtained from El-Nasr Chemical Company (Cairo, Egypt) and water ad libitum. The study was carried out at the National Center for Radiation Research and Technology, according to the approval of the Ethics Committee for Animal Experimentation at Faculty of Pharmacy, Cairo University and in accordance with the guidelines set by the EEC regulations (revised directive 86/609/EEC).
Venom
Naja nigricollis venom was obtained from laboratory unit of Medical Research Center, Faculty of Medicine, Ain Shams University. We prepared the venom from milking healthy snake and dried and kept in desiccator at 4°C till used.The geographic origin of this venom is the southern part of Egypt. The sexuality of the snake does not affect the venom production.
Irradiation of the venom
The venoms were exposed to γ-irradiation at a dose of 1.5 and 3 kGy in the National Center for Research and Radiation Technology, Cairo, Egypt, using Co-60 γ-cell. The radiation dose rate was 1.26 Gy/s. In this study, a certain weight of N. nigricollis snake venom dissolved in saline solution by the ratio 1:1, then exposed to γ-radiation at dose levels of 1.5 and 3 kGy.
Experimental design
Six male Newzealand white rabbits weighing 2–3 kg were divided into three groups (two rabbits in each stainless bank style cage) treated with native venom, venom irradiated at 1.5 kGy γ-irradiated and venom irradiated at 3 kGy. Rabbits were injected subcutaneously with 200 μl of venom emulsified in 0.5 ml complete Freund’s adjuvant at the 1st, 7th and 21st. A booster dose without adjuvant was given 1 week later; after 6 days, the blood samples were drawn from ear rabbits and then the pooled sera of two immunized rabbits for each group of immunization were used. 26
The mice used for neutralization of lethality of the venom were divided into three sets, and each was subdivided into five groups and each group included four animals, according to Keegan et al., 27 as follows: set I: potency of N. nigricollis antiserum was prepared with native venom; set II: potency of N. nigricollis antiserum was prepared with 1.5-kGy γ-irradiated venom; and set III: potency of N. nigricollis antiserum was prepared with 3-kGy γ-irradiated venom.
Thirty male albino rats weighing 120–150 g were used for neutralization of the cardiotoxic effect of venom and were classified into five groups (six rats in each cage) as follows: group 1: rats injected intraperitoneally with saline and served as normal; group 2: rats injected intraperitoneally with native venom ½ LD50 (0.154 mg/kg); group 3: rats injected intraperitoneally with mixture of (native venom – antiserum raised against non-irradiated 1:4 according to Ismail et al.) incubated at 37°C for 1 h, in a dose equivalent to the dose of native venom ½ LD50 (0.154 mg/kg); group 4: as in group 3, but using antiserum raised against 1.5-kGy γ-irradiated venom instead of non-irradiated venom; and group 5: as in group 3, but using antiserum raised against 3-kGy γ-irradiated venom instead of non-irradiated venom. Rats of all groups were killed after 4 h of injection by decapitation (4-h interval was chosen because the peak of the venom toxicity is 2–4 h). Blood was collected from each rat into plain centrifuge tubes and left for 1 h at room temperature (25°C ± 2), and serum was separated by centrifugation at 3000 r/min for 20 min, for evaluation of creatine kinase (CPK) activity, creatine kinase isoenzyme-MB (CK-MB) activity, lactate dehydrogenase (LDH) activity and aspartate aminotransferase (AST) activity.
Determination of PLA2 activity of the native and irradiated venoms
Venom phospholipase activity was determined according to the method of Marinetti 28 with slight modifications. 29 One egg yolk was separated and diluted with 0.9% sodium chloride to get a final volume of 100 ml (egg yolk stock suspension); 1 ml of the suspension was added to 4.9 ml of isotonic saline and mixed well, then stood for 5 min at 41°C. To the suspension, 0.1 ml of the test solution in saline, native, 1.5 and 3-kGy γ-irradiated N. Nigricollis venoms was added and mixed. The absorbency was recorded each 5 min for 30 min at 925 nm. The absorbency per 10 min was taken as the relative amount of enzyme activity (this test depends on the clearance of the venom to the egg yolk suspension due to PLA2 acting on the lipoproteins to produce lysolecithin and this lysolecithin is capable of solubilizing the egg yolk suspension).
Coagulant assay (plasma recalcification time) of the native and irradiated venoms
This test was estimated according to Biggs and Macfarlene, 30 4.5 ml of human blood was added to a clean tube containing 0.5-ml sodium citrate (3.8%) and mixed gently. The citrated blood was centrifuged for 20 min at 3000 r/min to obtain the plasma; 0.1 ml of plasma was added into a clotting glass tube and incubated at 37°C, then 100 µl (1-mg/ml concentration) of native or 1.5- or 3-kGy γ-irradiated venom was added and incubated at 37°C in water bath with gradual shaking. A control tube was prepared by adding 0.1 ml saline instead of the venom. Finally, 0.1 ml of 25 mM calcium chloride was added and the clotting time was immediately recorded with a stop watch.
Venom neutralization tests
The lethality in mice
An amount of 1-mg/ml saline of thenative venom was used, then serial dilutions of the venom were prepared in concentration of 1:2, 1:4, 1:8, 1:16 and 1:32 (i.e. 0.5, 0.25, 0.125, 0.062 and 0.031 mg). Fixed volume (1 ml) of antiserum raised against the non-irradiated or irradiated venom (1.5 and 3 kGy γ-irradiated) was added to the different dilutions of venom. Following incubation of the mixtures at 37°C for 1 h, the content of each tube (2 ml) was injected intraperitoneally into four mice. Mice which received equal amounts of venom were caged together. Test animals were observed for a period of 24 h. Survival at this time was the criterion for determination of neutralization. LD50 was calculated from the following equation. 27
Immunological properties (double immunodiffusion technique)
The immunodiffusion technique was carried out as described by Ouchterlony. 31 Immunodiffusion test was used for detection of the presence of antibodies in the immunized sera of rabbits.
PLA2 activity
Neutralization of venom phospholipase activity was determined according to the method 28 with slight modifications. 29 Venom was incubated with the specific antivenom raised against the non-irradiated or irradiated venom (1.5 and 3 kGy γ-irradiated) for 1 h at 37°C, in the proportion of 1-mg venom to 4-ml serum. 32 After incubation of the mixtures for 1 h, the enzyme activity of the mixtures was assayed as mentioned before.
The cardiotoxic effect of venom in rats
Serum CPK activity was estimated according to Szasz et al., 21,32,33 CK-MB activity was evaluated by the immunoinhibition methodology, 34 LDH activity was measured according to Wacker et al. 35 and AST activity was determined according to the method of Reitman and Frankel. 36
Statistical analysis
Values were calculated as mean ± standard error of the mean. Comparisons between different groups were obtained by one-way analysis of variance followed by Tukey–Kramer multiple comparison tests, using ‘Instant software, v. 3 (GraphPad Inc., San Diego, California, USA)’. The p value was set at ≤0.05.
Results
PLA2 activity
Data from Table 1 compared to the native venom showed a significant decline in phospholipase activity of irradiated venom at 1.5 kGy as well as 3 kGy.
PLA2 activity of native, 1.5- and 3-kGy γ-irradiated Naja nigricollis venoms.a,b

PLA2: phospholipase A2; SEM: standard error of the mean.
a100 µg of venom used.
bThe tabulated results represent the mean of six values ± SEM.
cSignificant difference from the native venom group (p ≤ 0.05).
Coagulation activity
The 1.5- and 3-kGy γ-irradiated venoms were recorded delay in coagulation time as compared to native venom, since the plasma did not form clot up to 90 min (Table 2).
Anticoagulant activity of native, 1.5- and 3-kGy γ-irradiated Naja nigricollis venoms.a,b

ND: not detected; SEM: standard error of the mean.
aThe tabulated results represent the mean of six values ± SEM.
bThere was not any clotting activity in the irradiated venom test.
Neutralization tests
The lethality of the neutralized venoms
Incubation of N. nigricollis venom with the specific native antivenom protected 100% of the mice against the lethal action of a dose of the venom equivalent to threefold the LD50. It also protected 75% of the mice against the lethal action of the dose of the venom equivalent to six times the LD50. Also, the incubation of N. nigricollis venom with the specific 1.5-kGy γ-irradiated antivenom protected 100% of the mice against the lethal action of the dose of the venom equivalent to three times the LD50. It also protected 25% of the mice against the lethal action of the dose of the venom equivalent to six times the LD50. However, incubation of N. nigricollis venom with the specific 3-kGy irradiated antivenom protected 75% of the mice against the lethal action of the dose of the venom equivalent to three times the LD50 (Table 3). Therefore, the two antisera raised against irradiated venom provided greater protection at higher doses of venom than did antiserum raised against native venom.
Determination of the potency of Naja nigricollis antivenoms prepared with native or irradiated venom at the two dose levels (1.5 and 3 kGy).

Immunodiffusion technique
In this test, the antisera raised against non-irradiated venom, 1.5- and 3-kGy γ-irradiated venom, showed similar results. Faint precipitin bands were observed in Figure 1. The lines were identical and were joined smoothly at the corners, indicating the presence of antibodies in the immunized sera of rabbits.
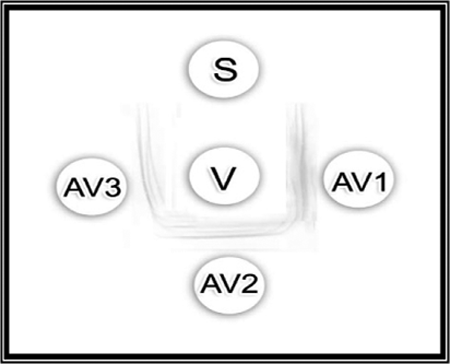
Double immunodiffusion reaction of native Naja nigricollis venom (20 mg/ml; central well) with rabbit antisera from non-irradiated, 1.5- and 3-kGy γ-irradiated N. nigricollis venom. S: saline; V: native (non-irradiated) venom (20 mg/ml); AV1: antiserum raised against non-irradiated venom; AV2: antiserum raised against 1.5-kGy γ-irradiated venom; AV3: antiserum raised against 3-kGy γ-irradiated venom.
PLA2 activity assay
As shown in Figure 2, a significant inhibition in PLA2 activities had been occurring when the venoms were neutralized by antisera raised against native, 1.5- and 3-kGy γ-irradiated venoms. Sera of rabbits immunized with 1.5- and 3-kGy γ-irradiated venom neutralized PLA2 activity with approximately the same degree as well as the antiserum raised against non-irradiated venom.

Neutralization of PLA2 activity with native, 1.5- and 3-kGy γ-irradiated Naja nigricollis antisera. Each value represents mean ± SEM of six results. *Significant difference from native venom group (p ≤ 0.05). PLA2: phospholipase A2; SEM: standard error of the mean.
Neutralization of the cardiotoxic effect of N. nigricollis venom in rats using the peak time after 4-h post injection
Table 4 exhibited a significant decrease in the activities of CPK and CK-MB. On the other hand, a significant elevation in AST activity and no change in LDH level were observed.
Effect of native Naja nigricollis venom (0.154 mg/kg i.p.) ½ LD50 and mixtures of antivenoms raised against non-irradiated, 1.5- and 3-kGy γ-irradiated venoms on heart enzymes in adult male albino rats (N = 6) after 4 h.a,b

CPK: creatine kinase; CK-MB: creatine kinase isoenzyme-MB; LDH: lactate dehydrogenase; AST: aspartate aminotransferase; ANOVA: analysis of variance; SEM: standard error of the mean.
aStatistical analysis was carried out by one-way ANOVA followed by Tukey–Kramer multiple comparison test.
bEach value represents mean ± SEM of six values.
cSignificant difference from control group (p ≤ 0.05).
dSignificant difference from venom-treated group (p ≤ 0.05).
Discussion
Referring to Abdou et al., 37 the LD50 for native and γ-irradiated (1.5 and 3 kGy) N. nigricollis venom was 0.440, 4.79 and 5.38 mg/kg. Irradiation of the whole venom with 1.5 and 3 kGy reduced its lethality 10.8 and 12.22 times as compared to its native venom, respectively. Moreover, there was no change in the immunogenicity of the venom pre and post irradiation, native venom caused a significant elevation in the heart enzymes, while irradiated venom did not affect the activity of heart enzymes, and also the histological examination of the heart of the native and irradiated venoms confirmed these results. Depending on these results, the present study was performed.
PLA2 is one of the major constituents in the elapid venom, which confers multiple toxicity to the prey or victim such as membrane damaging, neurotoxicity, oedema and prolongation of coagulation time. 38 Moreover, Sallau et al. 39 confirmed that PLA2 has a wide range of myotoxicity and cardiotoxicity.
Our results indicated that the irradiation decreases the PLA2 activity of N. nigricollis venom. These results are in agreement with the results of Shaaban 40 who showed that γ-radiation of Naja haje as well as Cerastus cerastus snake venom leads to decrease in PLA2 activity of both venom.
In addition, Bennacef-Heffar and Laraba-Djebari 41 studied the effect of γ-irradiation on the venom of Vipera lebetina and recorded that the PLA2 activity was abolished in the irradiated venom with a dose of 2 kGy.
Many snake venoms are known to cause pathological properties associated with haematological disturbances leading to incoagulability of blood. Some local tissue necrosis always accompany envenomation from this snake species. Spontaneous bleeding and coagulation disturbances are some of the haematological effects of N. nigricollis in patients. 42
Snake venom proteins affect the haemostasis process of victim/prey either by prolonging or shortening the clotting time. Elapid venoms are anticoagulant in nature due to the presence of large amounts of strong and weak anticoagulant PLA2 enzymes. Moreover, non-enzymatic protein from the elapid venom like cardiotoxins from Naja nigricollis crawshawii and hemextin A and hemextin AB complex from Hemachatus haemachatus venom are also reported to be anticoagulant in nature. 43 –45 So, the present results recorded that the irradiation of the venom preserves the anticoagulant effect.
In addition, Das et al. 45 reported that the Naja kaouthia venom significantly delayed the recalcificaion time, PT and APTT of plasma under in vitro condition due to the presence of strong anticoagulant proteins in the venom. Venom PLA2 enzymes prevent activation of FX to FXa that leads to disruption in the formation of prothrombinase complex, which is required for blood coagulation. 46 –48 The venom proteins, especially the PLA2 enzymes, hydrolyze the phospholipids that are required for the prothrombinase complex formation. Thus, the strongly anticoagulant enzyme CM-IV constrains both the extrinsic tenase and prothrombinase complexes, creating two bottlenecks in the coagulation cascade, whereas the weakly anticoagulant enzymes CM-I and CM-II only prevent the extrinsic tenase complex and get only a single bottleneck. 49
The data of the current work demonstrated that antiserum raised against 1.5-kGy irradiated N. nigricollis venom was found to neutralize venom lethality to the same degree (threefold LD50), as well as the antiserum raised against non-irradiated venom (crude venom). Thus, the antiserum raised against the 1.5-kGy γ-irradiated venom is quite as effective as the antiserum raised against the native venom in neutralizing the lethality induced by native venom. This agrees with Flowers 50 who showed that crotalied snake venom exposed to X-rays, revealed a decrease in its local reactivity (50–70%) and an increase in the LD50, in mice (sixfolds). This inactivation was accompanied with little change in its antigenic character. At the same time, the antivenin produced when using X-irradiated venom, neutralized both the local and the lethal effects of the venom. This effect was more or less, equal to that obtained with control antivenin produced when using non-irradiated venom.
In addition, Yang 51 reported that antigen antibody complex was hardly affected, even at the high dose level of irradiation; therefore, irradiation of venom solutions could provide an effective and safe toxoid without change in its antigenic activities.
However, Netto et al. 52 showed that the use of irradiated Cortalus durissus terrificus venom in sheep immunization induces a powerful and lasting humoral immune response shown by both in vitro neutralization and potency tests and by indirect ELISA antibody level detection technique. Sera from the irradiated group were five times more potent than the sera from the natural group.
Moreover, Shaaban and Hafez 53 reported that irradiation did not decrease the antilethal power of antivenin where equal amounts of irradiated or non-irradiated antivenin were capable of neutralizing 2.5 LD50 of the Egyptian cobra (Naja haje) venom. No increment in the dose of irradiated antivenin was needed to neutralize an identical quantity of the venom. These data therefore showed that γ-irradiation did not affect the antivenin immunoglobulin power.
In immunodiffusion test, antisera raised against non-irradiated, 1.5 and 3-kGy γ-irradiated venom showed similar results. The lines were identical and were joined smoothly at the corners, indicating the presence of antibodies in the immunized sera of rabbits. This result is in accordance with Keegan et al. 27 who reported that there were the same precipitin bands when tested the control antiserum (produced against non-irradiated venom) and the antisera raised against irradiated Naja haje venom (15 and 25 kGy).
In the present study, in vitro neutralization of PLA2 activity was applied. The assay has several advantages, which include easy, fast and many samples can be studied simultaneously. The data in the present study revealed that a significant inhibition of PLA2 activities was obtained when neutralized with native, 1.5- and 3-kGy irradiated antiserum. Sera of rabbits immunized with 1.5- and 3-kGy irradiated venom neutralized PLA2 activity with the same degree as well as the antiserum raised against non-irradiated venom. This could be attributed that antibodies generated against the 1.5-kGy irradiated venom were reactive towards the non-irradiated venom and vice-versa, showing no change in the immunogenecity. These data are in agreement with results of Keegan et al. 27 who mentioned that a significant inhibition of both PLA2 and protease activities was obtained when neutralized with 15-kGy irradiated venom toxoid as 15-kGy toxoid antiserum neutralized venom lethality, 95% of protease activity and 50% of PLA2.
Incubation of the same venom dose ½ LD50 (0.154 mg/kg i.p.) together with 4 ml of each antiserum (non-irradiated or irradiated) at 37°C for 1 h before i.p. injection produced a significant reduction in the values of CPK and CK-MB compared to venom-treated group. However, still significant elevation in AST level was observed that might be due to hepatocytes injury or other tissue injury. This indicates that there was a protective action of the formed antivenom against toxic ingredients of the venom mainly cardiotoxicity.
It was observed that the neutralizing capacities of non-irradiated and (1.5 or 3 kGy) irradiated antiserum were approximately the same. They neutralize both the lethal and the biological effects of the venom almost to the same extent. Thus, it neutralized the significant increase in CPK and CK-MB serum levels. This finding agrees with the finding of Gutiérrez et al. 54 who reported that the antivenin contains antibodies capable of preventing and neutralizing the toxic and enzymatic activities of the venom.
Neutralization studies gave different results depending upon the way antivenom is administered. When venom and antivenom are incubated before injection, neutralization was occurred, indicating that antivenom has antibodies capable of counteracting tissue-damaging toxins. These results agree with previous observations with Bothrops asper 55 and other venoms, 54,56 since both tissue damage and haemorrhage are adequately neutralized by this antivenom in preincubation experiments. In contrast, when antivenin is administered intravenously after venom injection, neutralization is only partial. These observations can be due to the fact that tissue damage, particularly local myonecrosis and haemorrhage, develops very fast post venom injection. Thus, when antibodies extent the different tissues, damage has already happened. This interpretation has been previously proposed to explain the neutralization of myonecrosis and haemorrhage induced by several Central American crotaline venoms. 54 –57
According to Ferreira et al., 58 the irradiation causes chemical and physicochemical changes in the secondary and tertiary structures of proteins but keeps their immunogenic properties. This detoxification may be an effective method to reduce the venom toxic effects in immunized animals and to improve antigens for toxoid and vaccine preparation.
Finally, researchers have been seeking alternatives to prepare toxoids via venom biological detoxification, which would preserve its immunogenicity and reduce damages caused to antiserum producer animals. 59
Conclusion
The present study supported that γ-irradiation of N. nigricollis snake venom with 1.5-kGy dose offers an effective method for reducing the chronic toxic effect of venom in immunized animals for preparing the best toxoids and vaccines via increasing LD50 of the venom and decreasing the cardiotoxic effect of the venom.
Footnotes
Acknowledgements
The authors are kindly appreciative to the staff members of γ-irradiation unit (NCRRT) for their generous support in carrying out the experimental irradiation and Dr Ahmed Abdel Basset gave us the venom in the alypholized form as a gift (who prepared the venom by milking), who worked at laboratory unit of Medical Research Center, Faculty of Medicine, Ain Shams University.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
